Vernonia amygdalina: Anticancer Activity, Authentication, and Adulteration Detection
Abstract
:Introduction
Materials and Methods
Cells and Chemicals
Sample Collection and Preparation of Aqueous Extracts
Cell Proliferation Determination
[3H] Thymidine Incorporation Studies
DNA Extraction Methods
Endonuclease Digestion
Gel Electrophoresis
Statistical Analysis
Results
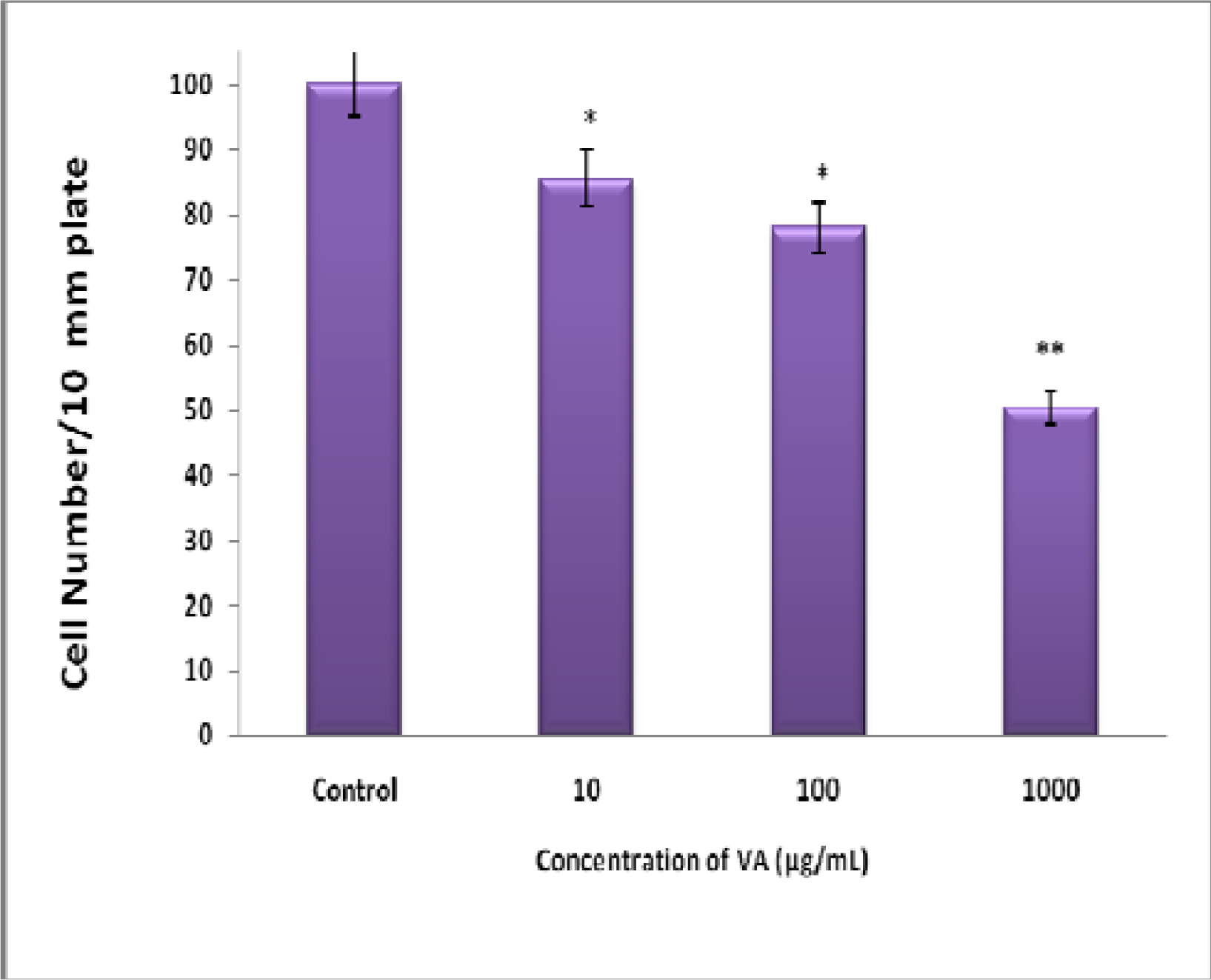
Inhibition of BT-549 Cell Proliferation by Aqueous VA Extracts
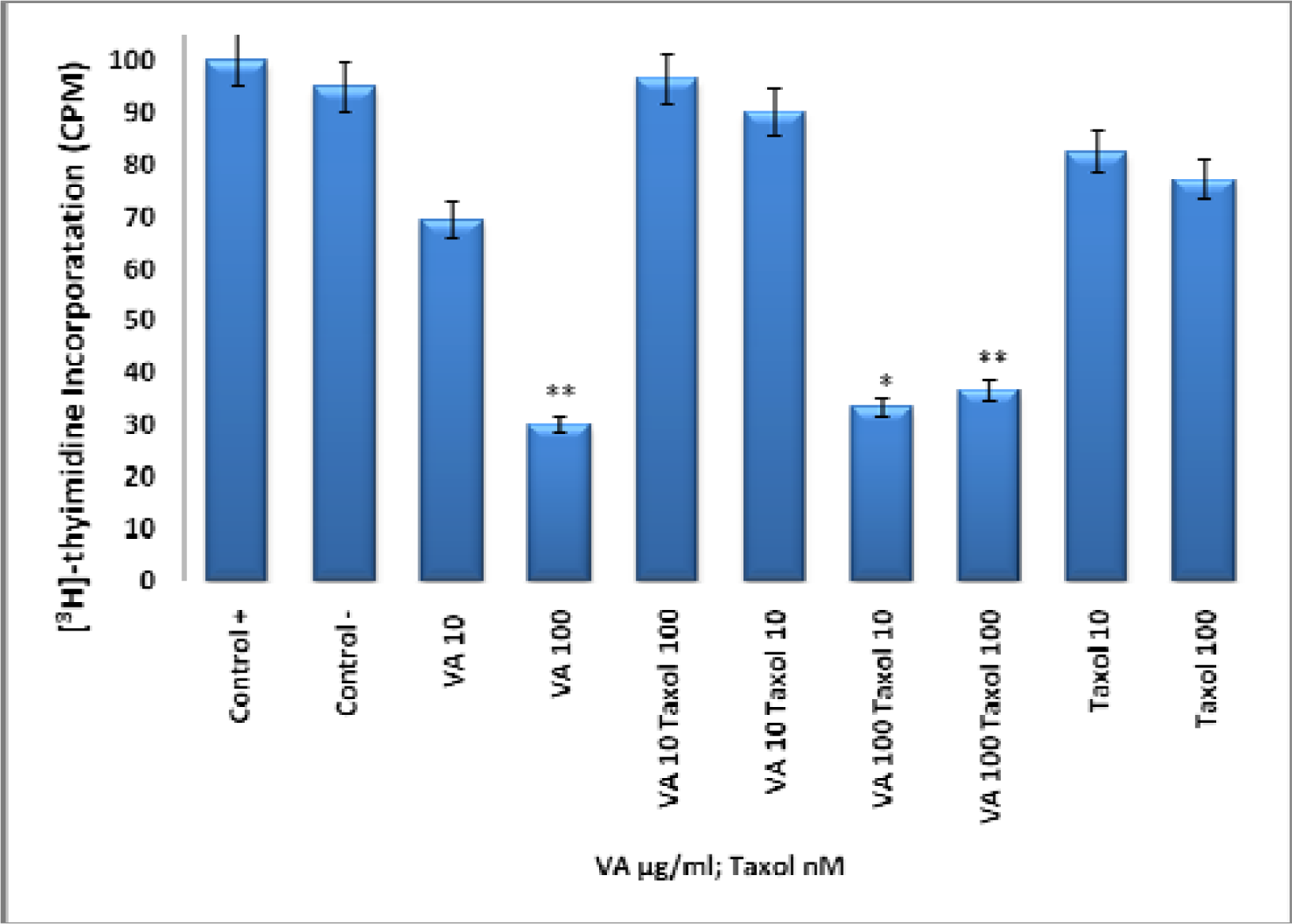
Inhibition of DNA synthesis by VA Aqueous extracts
Effect of Taxol, VA, and TAX/VA Combination on DNA Synthesis
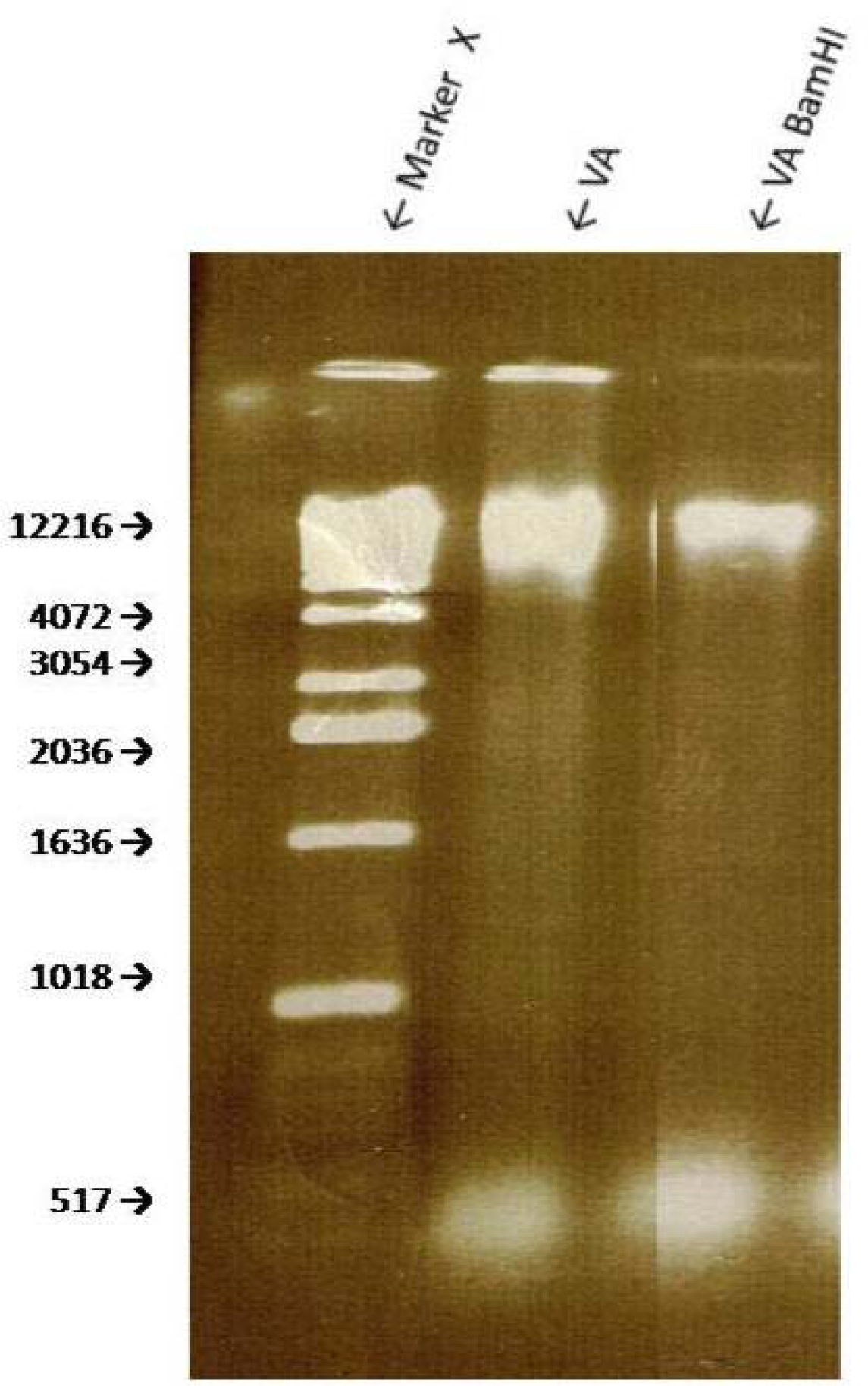
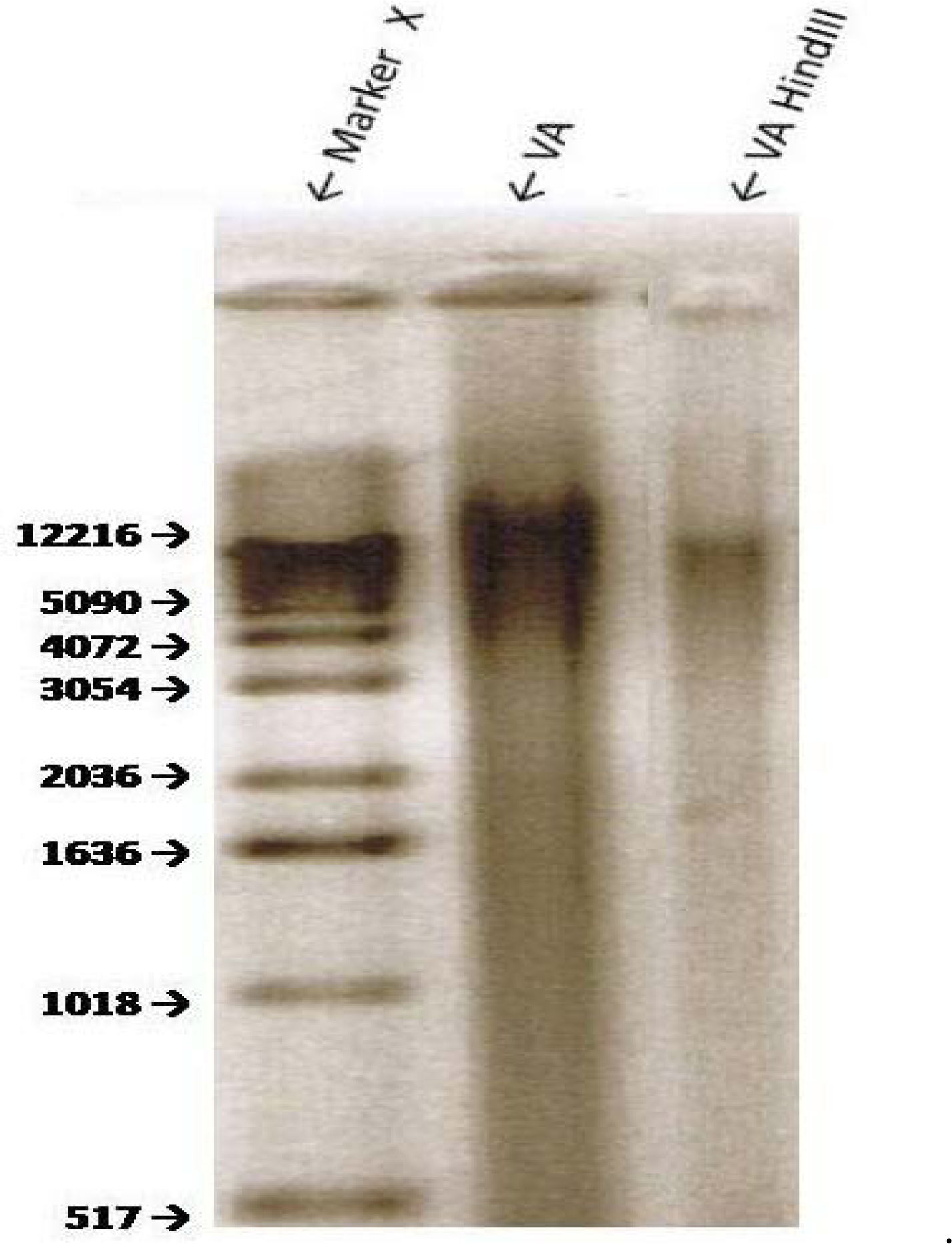
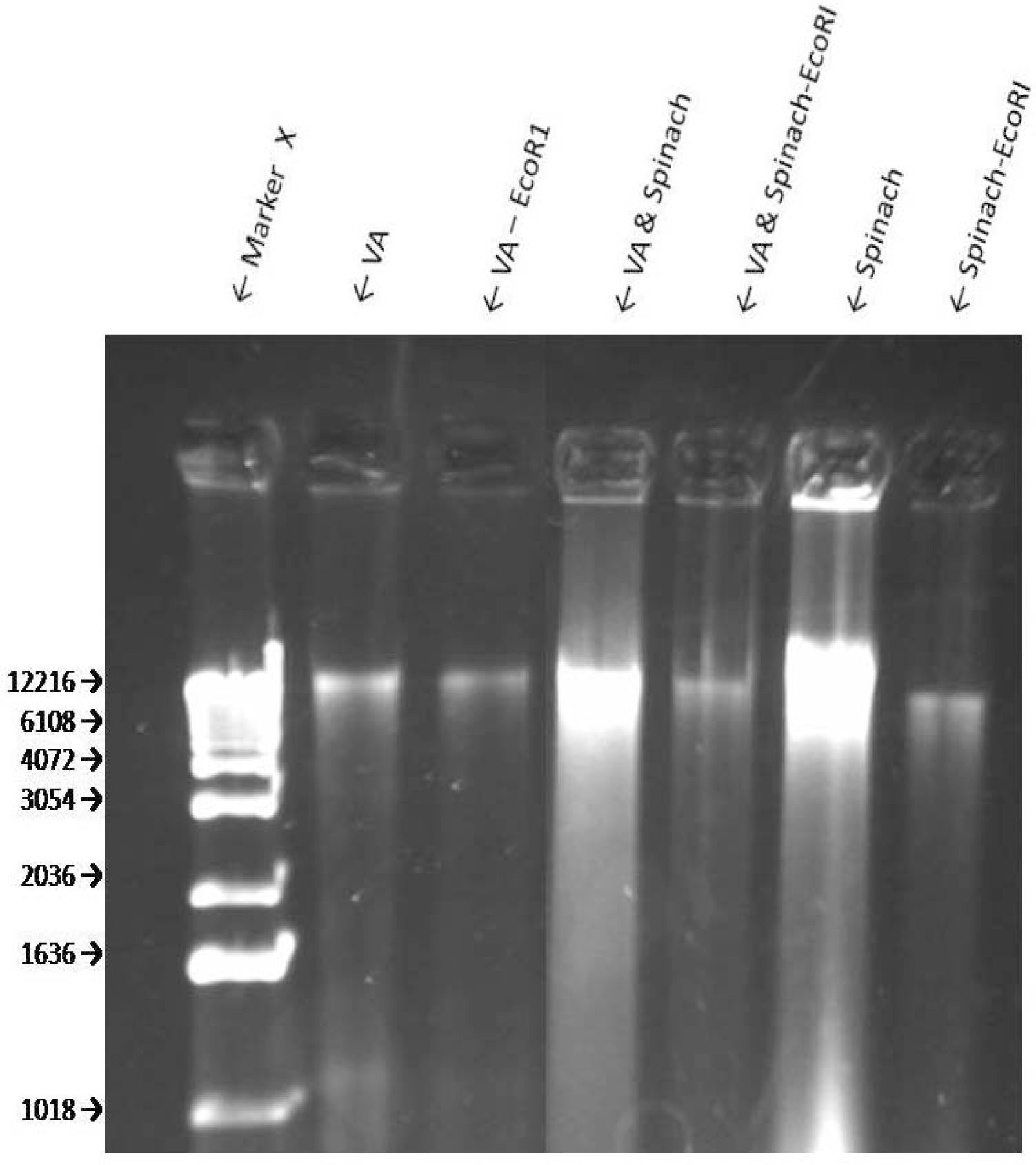
Sensitivity of VA Genomic DNA to Endoclease Digestion
Discussion

Acknowledgments
References and Notes
- American Cancer Society, Cancer Facts & Letters; 2007; Atlanta, GA.
- American Cancer Society, Cancer Statistics; 2008.
- Dougherty, MK; Schumaker, LM; Jordan, VC; Welshons, WV; Curran, EM; Ellis, MJ; El-Ashry, D. Estrogen Receptor Expression and Sensitivity to Paclitaxel in Breast Cancer. Cancer Biology & Therapy, 2004, 3(5), 460–467. [Google Scholar]
- Sui, M; Huang, Y; Park, BH; Davidson, NE; Fan, W. Estrogen Receptor A Mediates Breast Cancer Cell Resistance to Paclitaxel through Inhibition of Apoptotic Cell Death. Cancer Research 2007, 67(11). [Google Scholar]
- Cunningham, JE; Butler, WM. Racial disparities in female breast cancer in South Carolina: clinical evidence for a biological basis. Breast Cancer Research and Treatment 2004, 88(2), 161–176. [Google Scholar]
- Ziv, E; Tice, J; Smith-Bindman, R; Shepherd, J; Cummings, S; Kerlikowske, K. Mammographic density and estrogen receptor status of breast cancer. Cancer Epidemiology Biomarkers and Prevention 2004, 13(12), 2090–2905. [Google Scholar]
- Woodward, WA; Hwang, EH; McNeese, M.D; Perkins, GH; Tucker, SL; Strom, E.A; Middleton, L; Hahn, K; Hortobagyi, GN; Buchholz, TA. African-American race is associated with a poorer overall survival rate for breast cancer patients treated with mastectomy and doxorubicin-based chemotherapy. Cancer 2006, 107(11), 2662–8. [Google Scholar]
- Oleszek, W; Igile, G; Burda, S; Jurzysta, M. Nutritional assessment of Vernonia amygdalina leaves in growing mice. Journal of Agricultural and Food Chemistry 1995, 4, 2162–2166. [Google Scholar]
- Farombi, EO. African indigenous plants with chemotherapeutic potentials and biotechnological approach to the production of bioactive prophylactic agents. African Journal of Biotechnology 2003, 2(12), 662–671. [Google Scholar]
- Erasto, P; Grierson, DS; Afolayan, AJ. Bioactive sesquiterpene lactones from the leaves of Vernonia amygdalina. J Ethnopharmacology 2006, 106, 117–120. [Google Scholar]
- Moundipa, PF; Kamini, G; Flore, M; Bilong Bilong, CF; Bruchhaus, I. In vitro amoebicidal activity of some medicinal plants of the Bamun region (Cameroon). African J Trad CAM, 2005, 2(2), 113–121. [Google Scholar]
- Akah, PA; Ekekwe, RK. Ethnopharmacology of some of the asteraceae family used in the Nigerian traditional medicine. Fitoterapia 1995, 66, 352–355. [Google Scholar]
- Akah, PA; Okafor, CL. Blood sugar lowering effects of V. amygdalina Del in an experimental rabbit model. Phytother Res 1992, 6, 171–173. [Google Scholar]
- Akinpelu, DA. Antimicrobial activity of Vernonia amygdalina leaves. Fitoterapi 1999, 70, 432–234. [Google Scholar]
- Krief, S; Hladik, C; Haxaire, C. Ethnomedicinal and bioactive properties of plants ingested by wild chimpanzees in Uganda. Journal of Ethnopharmacology 2005, 101, 1–5. [Google Scholar]
- Iwalokun, BA; Efedede, BU; Alabi-Sofunde, JA; Oduala, T; Magbagbeola, OA; Akinwande, AI. Hepatoprotective and antioxidant activities of Vernonia amygdalina on acetaminophen-induced hepatic damage on mice. Journal of Medicinal Food 2006, 9(4), 524–30. [Google Scholar]
- Kupchan, SM; Hemingway, RJ; Karim, A; Werner, D. Tumor inhibitors. XLVII. Vernodalin and Vernomygdin, two new cytotoxic sesquiterpene lactones from Vernonia amygdalina Del. Journal of Organic chemistry 1969, 34, 3908–3911. [Google Scholar]
- Jasaka, M; Ohigashi, H; Takagaki, T; Nozaki, H; Tada, T; Hiroto, M; Irie, R; Huffman, MA; Nishida, T; Kaji, M; Koshimizu, K. Bitter steroid glucosides, Vernoniosides A1, A2,A3 and related B1 from possible medicinal plant-Vernonia amygdalina used by wild chimpanzees. Tetrahedron 1992, 48(4), 625–632. [Google Scholar]
- Izevbigie, EB. Discovery of water soluble anticancer agents (Edotides) from vegetables found in Benin City, Nigeria. Exp.Biol.Med 2003, 228, 293–298. [Google Scholar]
- Howard, CB; Stevens, J; Izevbigie, EB; Walker, A; McDaniel, O. Time and dose-dependent modulation of phase 1 and phase 2 gene expression in response to treatment of MCF-7 cells with a natural anti-cancer agent. Cell. Mol. Biol 2003, 49(7), 1057–1065. [Google Scholar]
- Izevbigie, EB; Bryant, JL; Walker, A. A novel natural inhibitor of extracellular signal-regulated kinases human breast cancer cell growth. Exp.Bio.Med 2004, 229, 163–169. [Google Scholar]
- Opata, M; Izevbigie, EB. Aqueous V. amygdalina Extracts Alter MCF-7 Cell Membrane Permeability and Efflux. Int. J. Environ. Res. Public Health, 2006, 3(2), 174–179. [Google Scholar]
- Sayed, MD; Zaki, AY; El-Marzabai, MM; Doss, SL. Medicinal Plants. Phytochemistry 1987, 21, 944. [Google Scholar]
- Tona, L; Cimanga, RK; Mesia, K; Musuamba, CT; De Bruyne, T; Apers, S; Hermans, N; Van Miret, S; Pieters, L; Totté, J; Vlietink, AJ. In Vitro Antiplasmodial activity of extracts and fractions of seven medicinal plants used in the Democratic Republic of Congo. Journal of Ethnophamacology 2004, 93(1), 27–32. [Google Scholar]
- Harborne, JB. Phytochemical Methods; London, Chapman and Hall: New York, USA, 1973; p. 278. [Google Scholar]
- Wall, ME; Wani, MC; Manikumar, G; Abraham, P; Taylor, H; Huges, TJ; Warner, J; MacGivney, R. Plant antimutagenic agents, flavonoids. Journal of Natural Products 1998, 51(6), 1084–109. [Google Scholar]
- GraphPad Software, GraphPad Software, Inc: Sand Diego, CA, USA.
- Aparajita, G; Akech, J; Mukherjee, S; Das, SK. Differential expression of cholinephosphotransferase in normal and cancerous human mammary epithelial cells. Biochem. Biophys. Res 2002, Comm.. 297, 1043–1048. [Google Scholar]
- Fonagy, A; Swiderski, C; Ostrovsky, AM; Bolton, W.E; Freeman, J.E. Effect of nuclear P120 expression level on the proliferation capacity of breast cancer cells. Cancer Research 1994, 54, 1859–1864. [Google Scholar]
- Greenwald, P; Clifford, CK; Milner, JA. Diet and cancer prevention. European Journal of Cancer 2001, 37(8), 948–965. [Google Scholar]
- Hennekens, C; Buring, JE; Mayrent, SL. Epidemiology in Medicine; Lippincott Williams and Wilkins, 1987; Volume 3, p. 36. [Google Scholar]
- Peto, R; Doll, R; Buckley, JD; Sporn, MB. Can dietary beta-carotene materially reduce human cancer rates? Nature 1981, 290, 201–208. [Google Scholar]
- Cragg, GM; Newman, DJ. International collaboration in drug discovery and development from natural sources. Pure Appl. Chem., 2005, 77(11), 1923–1942. [Google Scholar]
- Vladusic, EA; Hornby, AE; Guerra-Vladusic, FK; Lakins, J; Lupu, R. Expression and regulation of estrogen receptor beta in human breast tumors and cell lines. Oncol. Rep 2000, 7(1), 157–167. [Google Scholar]
© 2008 MDPI All rights reserved.
Share and Cite
Gresham, L.J.; Ross, J.; Izevbigie, E.B. Vernonia amygdalina: Anticancer Activity, Authentication, and Adulteration Detection. Int. J. Environ. Res. Public Health 2008, 5, 342-348. https://doi.org/10.3390/ijerph5050342
Gresham LJ, Ross J, Izevbigie EB. Vernonia amygdalina: Anticancer Activity, Authentication, and Adulteration Detection. International Journal of Environmental Research and Public Health. 2008; 5(5):342-348. https://doi.org/10.3390/ijerph5050342
Chicago/Turabian StyleGresham, Lecia J., Jetaime Ross, and Ernest B. Izevbigie. 2008. "Vernonia amygdalina: Anticancer Activity, Authentication, and Adulteration Detection" International Journal of Environmental Research and Public Health 5, no. 5: 342-348. https://doi.org/10.3390/ijerph5050342



