Effects of α-Tocopherol on Oxidative Status and Metabolic Profile in Overweight Women
Abstract
:Introduction
Materials and Methods
Subjects
Reagents and Blood Sample Collection
Clinical Chemistry
Oxidative Stress
Statistical Analysis
Results
Clinical Characteristics
Metabolic Profile
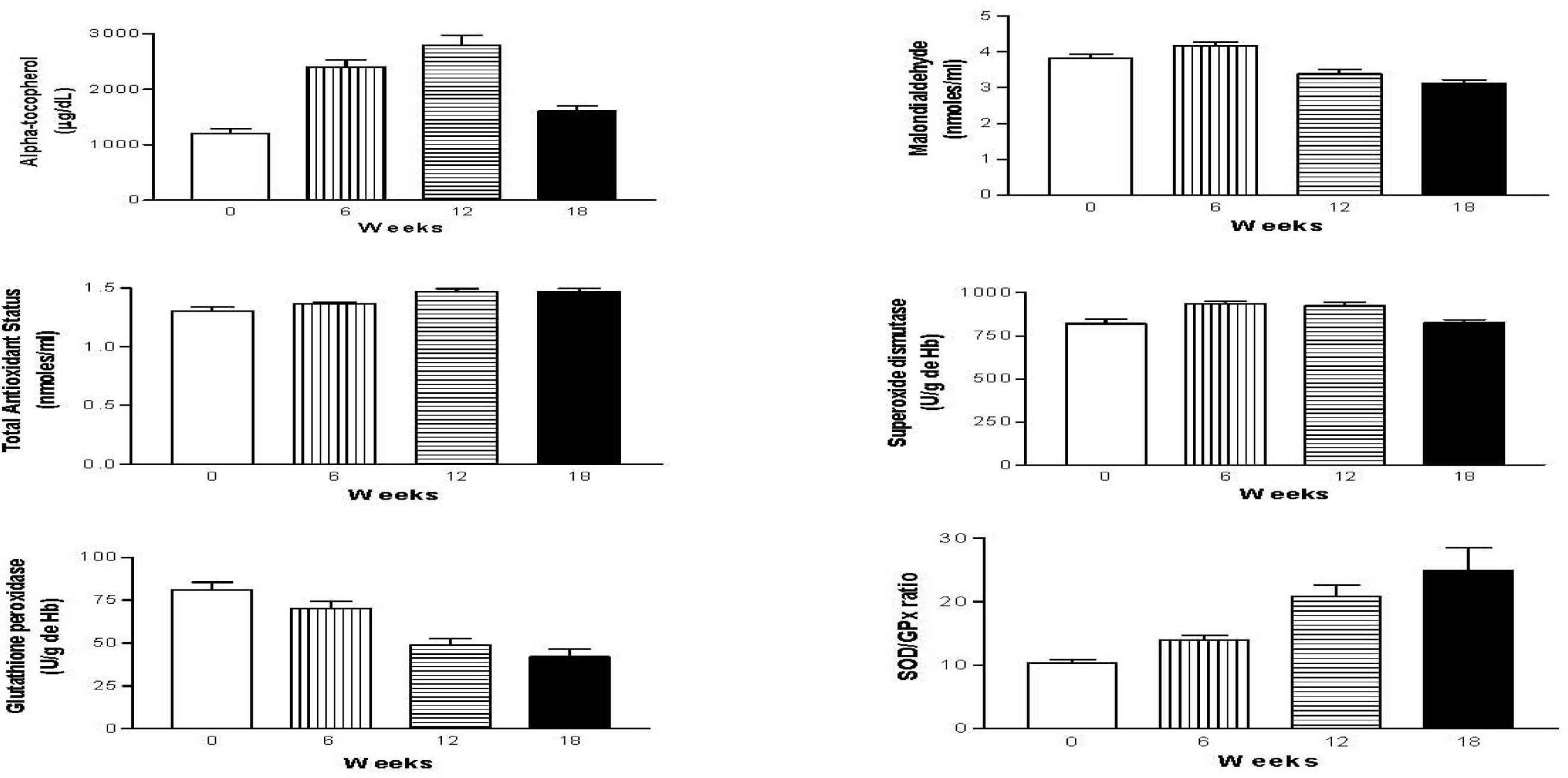
Oxidative Stress Markers
Discussion
Conclusions
| Characteristic | Mean ± SD |
|---|---|
| Age (years) | 51.8 ± 4.47 |
| Body weight (Kg) | 61.59 ± 6.77 |
| Height (m) | 1.53 ± 0.04 |
| BMI (Kg/m2) | 26.18 ± 2.85 |
| Systolic blood pressure* (mmHg) (mmHg) | 112.5 ± 8.56 |
| Diastolic blood pressure* (mmHg) | 71.13 ± 8.56 |
| FSH (mIU/ml) | 57.90±32.65 |
| Parameter | Week 0 | Week 6 | Week 12 | Week 18 (Washout) | p |
|---|---|---|---|---|---|
| Glucose | 96.88±6.722 | 98.38±6.479 | 97.13±5.427 | 93.81±8.183 | NS |
| Glycated hemoglobin (%) | 3.894±0.2175 | 3.625±0.274 | 3.131±0.2414 | 3.244±0.1590 | 0 vs 6, p <0.001 0 vs 12, p <0.001 0 vs 18, p <0.001 6 vs 12, p <0.001 6 vs 18, p <0.001 12 vs 18, p >0.05 |
| Uric Acid | 4.669±0.6416 | 4.325±0.591 | 4.281±0.7556 | 4.594±0.8410 | NS |
| Cholesterol | 196.9±33.75 | 192.2±36.60 | 196.7±35.11 | 194.0±42.52 | NS |
| Low density lipoproteins | 108±33.28 | 107.2±34.96 | 110.6±34.90 | 117.2±41.38 | NS |
| High density lipoproteins | 56.50±13.36 | 53.06±9.19 | 53.19±9.955 | 48.75±11.76 | NS |
| Cholesterol/High density lipoproteins | 3.614±0.768 | 3.718±0.5099 | 3.823±0.9976 | 4.129±0.970 | NS |
| Triglycerides | 162.1±66.18 | 159.80±72.71 | 164.7±66.98 | 140.2±48.73 | NS |
| Phosphates | 3.650±0.547 | 3.494±0.763 | 3.869±0.490 | 3.40±0.4351 | NS |
| Magnesium | 2.089±0.2157 | 2.104±0.1120 | 2.305±0.1423 | 2.024±0.2055 | 0 vs 12, p <0.01 6 vs 12, p <0.05 12 vs 18, p <0.001 |
| Retinol | 45.86±7.966 | 44.60±7.682 | 47.6±8.1 | 43.5±6.9 | NS |
Acknowledgments
- Author’s contributions: JDM, JCDZ, GCV and RMS participated in the study design. JLBC coordinated the trial, performed the analytic assays and wrote the initial draft of the manuscript. GCV, JDM, HRA and JCDZ assisted in the interpretation of the results and contributed to the editing and review of the manuscript.
References
- González-Correa, JA; Arrebola, MM; Guerrero, A; Cañada, MJ; Muñoz-Marín, J; Sánchez de la Cuesta, F; De la Cruz, JP. Antioxidant and antiplatelet effects of the alpha-tocopherol-aspirin combination in type 1-like diabetic rats. Life Sciences 2006, 79, 1405–1412. [Google Scholar]
- Mustacich, DJ; Leonard, SW; Devereaux, MW; Sokol, RJ; Traber, MG. α-tocopherol regulation oh hepatic cytochrome P450s and ABC transporters in rats. Free Rad Biol Med 2006, 41, 1069–1078. [Google Scholar]
- Brown, KM; Morrice, PC; Duthie, GG. Erythrocyte vitamin E and plasma ascorbate concentrations in relation to erythrocyte peroxidation in smokers and nonsmokers. Dose response to vitamin E supplementation. Am J Clin Nutr 1997, 65, 496–502. [Google Scholar]
- Winterbone, MS; Sampson, MJ; Saha, S; Hughes, JC; Hughes, DA. Pro-oxidant effect of α-tocopherol in patients with Type 2 Diabetes after an oral glucose tolerance test-a randomized controlled trial. Cardiovasc Diabetol 2007, 6, 8. [Google Scholar]
- Hatchcock, JN; Azzi, A; Blumberg, J; Bray, T; Dickinson, A; Frei, B; Jialal, I; Johnston, CS; Kelly, FJ; Kraemer, K; Packer, L. Vitamin E and C are safe across a broad range of intakes. Am J Clin Nutr 2005, 81, 736–45. [Google Scholar]
- Bjelakovic, G; Nikolova, D; Simonetti, RG; Gluud, C. Antioxidant supplements for prevention of gastrointestinal cancers: a systematic review and meta-analysis. Lancet 2004, 364, 1219–1228. [Google Scholar]
- Miller, ER; Pastor-Barriuso, R; Dalal, D; Riemersma, RA; Appel, LJ; Guallar, E. Meta-analysis: high-dosage vitamin E supplementation may increase all-cause mortality. Ann Intern Med 2005, 142, 37–46. [Google Scholar]
- Skrha, J; Sindelka, G; Kvasnicka, J; Hilgertova, J. Insulin action and fibrinolysis influenced by vitamin E in obese type 2 diabetes mellitus. Diabetes Res Clin Pract 1999, 44, 27–33. [Google Scholar]
- Paolisso, G; D’Amore, A; Galzerano, D; Balbi, V; Giugliano, D; Varricchio, M; D’Onofrio, F. Daily vitamin E supplements improve metabolic control but not insulin secretion in elderly type II diabetic patients. Diabetes Care 1993, 16, 1433–1437. [Google Scholar]
- Jacob, S; Ruus, P; Hermann, R; Tritschler, HJ; Maerker, E; Renn, W; Augustin, HJ; Dietze, GJ; Rett, K. Oral administration of RAC-α-lipoic acid modulates insulin sensitivity in patients with type 2 diabetes mellitus: a placebo-controlled pilot trial. Free Rad Biol Med 1999, 27, 309–314. [Google Scholar]
- Ford, ES; Mokdad, AH; Giles, WH; Brown, DW. The metabolic syndrome and antioxidant concentrations: findings from the Third National Health and Nutrition Examination Survey. Diabetes 2003, 52, 2346–2352. [Google Scholar]
- Keaney, JF, Jr; Larson, MG; Vasan, RS; Wilson, PW; Lipinska, I; Corey, D; Massaro, JM; Sutherland, P; Vita, JA; Benjamin, EJ. Obesity and systemic oxidative stress: clinical correlates of oxidative stress in the Framingham Study. Arterioscler Thromb Vasc Biol 2003, 23, 434–439. [Google Scholar]
- Olusi, SO. Obesity is an independent risk factor for plasma lipid peroxidation and depletion of erythrocyte cytoprotectic enzymes in humans. Int J Obes Relat Metab Disord 2002, 26, 1159–1164. [Google Scholar]
- Furukawa, S; Fujita, T; Shimabukuro, M; Iwaki, M; Yamada, Y; Nakajima, Y; Nakayama, O; Makishima, M; Matsuda, M; Shimomura, I. Increased oxidative stress in obesity and its impact on metabolic syndrome. J Clin Invest 2004, 114, 1752–1761. [Google Scholar]
- Manning, PJ; Sutherland, WHF; Walker, RJ; Williams, SM; de Jong, SA; Ryalls, AR. Effect of high-dose vitamin E on insulin resistance and associated parameters in overweight subjects. Diabetes Care 2004, 27, 2166–2171. [Google Scholar]
- McGavin, JK; Mann, JI; Skeaff, CM; Chisholm, A. Comparison of a vitamin E-rich diet and supplemental vitamin E on measures of vitamin E status and lipoprotein profile. Eur J Clin Nutr 2001, 55, 555–561. [Google Scholar]
- National Nutrition Survey. Nutritional status of children and women in Mexico. Mexico Department of Health. Nutrition. (ENN-99). 1999. Internet: http://www.salud.gob.mx/unidades/cdi/documentos/nutrición.pdf (accessed March 10, 2006).
- Bucolo, G; David, H. Quantitative determination of serum triglycerides by the use of enzymes. Clin Chem 1973, 19, 476–482. [Google Scholar]
- Allain, CC; Poon, LS; Chanm, CS; Richmond, W; Fu, PC. Enzymatic determination of total serum cholesterol. Clin Chem 1974, 20, 470–475. [Google Scholar]
- Friedewald, WT; Levy, RI; Fredrickson, DS. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem 1972, 18, 499–502. [Google Scholar]
- Paglia, DE; Valentine, WN. Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J Lab Clin Med 1967, 70, 158–169. [Google Scholar]
- McCord, JM; Fridovich, I. Superoxide dismutase. An enzymatic function for erythrocuprein (hemocuprein). J Biol Chem 1969, 244, 6049–6055. [Google Scholar]
- Sowell, AL; Huff, DL; Yeager, PR; Caudill, SP; Gunter, EW. Retinol, alpha-tocopherol, lutein/zeaxanthin, beta-cryptoxanthin, lycopene, alpha-carotene, trans-beta-carotene and four retinyl esters in serum, determined simultaneously by reversed-phase HPLC with multiwavelength detection. Clin Chem 1994, 40, 411–416. [Google Scholar]
- Wasowicz, W; Neve, J; Peretz, A. Optimized steps in fluorometric determination of thiobarbituric acid-reactive substances in serum: importance of extraction pH and influence of sample preservation and storage. Clin Chem 1993, 39, 2522–2526. [Google Scholar]
- Brigelius-Flohé, R; Kelly, FJ; Salonen, JT; Neuzil, J; Sing, JM; Azzi, A. The European perspective on vitamin E: current knowledge and future research. Am J Clin Nutr 2002, 76, 703–716. [Google Scholar]
- St-Onge, MP; Janssen, I; Heymsfield, SB. Metabolic syndrome in normal-weight Americans. New definition of the metabolically obese, normal-weight individual. Diabetes Care 2004, 27, 2222–2228. [Google Scholar]
- McNeill, AM; Rosamond, WD; Girman, CJ; Golden, SH; Schmidt, MI; East, HE; Ballantyne, CM; Heiss, G. The metabolic syndrome and 11-year risk of incident cardiovascular disease in the atherosclerosis risk in communities study. Diabetes Care 2005, 28, 385–390. [Google Scholar]
- Wellen, KE; Hotamisligil, GS. Inflammation, stress, and diabetes. J Clin Invest 2005, 115, 1111–1119. [Google Scholar]
- Palmieri, VO; Grattagliano, I; Portincasa, P; Palasciano, G. Systemic oxidative alterations are associated with visceral adiposity and liver steatosis in patients with metabolic syndrome. J Nutr 2006, 136, 3022–3026. [Google Scholar]
- Upritchard, JE; Schuurman, CR; Wiersma, A; Tijburg, LB; Coolen, SA; Rijken, PJ; Wiseman, SA. Spread supplemented with moderate doses of vitamin E and carotenoids reduces lipid peroxidation in healthy, nonsmoking adults. Am J Clin Nutr 2003, 78, 985–992. [Google Scholar]
- Sutherland, WHF; Manning, PJ; Walker, RJ; de Jong, SA; Ryalls, AR; Bery, EA. Vitamin E supplementation and plasma 8-isoprostane and adiponectin in overweight subjects. Obesity 2007, 15, 386–391. [Google Scholar]
- Meagher, EA; Barry, OP; Lawson, JA; Rokach, JR; FitzGerald, GA. Effects of vitamin E on lipid peroxidation in healthy persons. JAMA 2001, 285, 1178–1182. [Google Scholar]
- Ble-Castillo, JL; Carmona-Díaz, E; Mendez, JD; Larios-Medina, FJ; Medina-Santillán, R; Cleva-Villanueva, GG; Diaz-Zagoya, JC. Effect of alpha-tocopherol on the metabolic control and oxidative stress in female type 2 diabetics. Biomed Pharmacother 2005, 59, 290–295. [Google Scholar]
- Ceriello, A; Giugliano, D; Quatraro, A; Donzella, C; Dipalo, G; Lefebvre, PJ. Vitamin E reduction of protein glycosylation in diabetes. New prospect for prevention of diabetes complications? Diabetes Care 1991, 14, 68–72. [Google Scholar]
- Lonn, E; Yusuf, S; Hoogwerf, B; Pogue, J; Yi, Q; Zinman, B; Bosch, J; Dagenais, G; Mann, JF; Gerstein, HC. Effects of vitamin E on cardiovascular and microvascular outcomes in high-risk patients with diabetes: results of the HOPE study and MICRO-HOPE substudy. Diabetes Care 2002, 25, 1919–1927. [Google Scholar]
- Rodríguez-Morán, M; Guerrero-Romero, F. Oral magnesium supplementation improves insulin sensitivity and metabolic control in type 2 diabetic subjects. A randomized double-blind controlled trial. Diabetes Care 2003, 26, 1147–1152. [Google Scholar]
- McLean, RM. Magnesium and its therapeutic uses. Am J Med 1994, 96, 63–76. [Google Scholar]
- Manuel y Keenoy, B; Moorkens, G; Vertommen, J; Noe, M; Nève, J; De Leeuw, I. Magnesium status and parameters of the oxidant-antioxidant balance in patients with chronic fatigue: Effects of supplementation with magnesium. J Am Coll Nutr 2000, 19, 374–382. [Google Scholar]
- Paolisso, G; Tagliamonte, MR; Barbieri, M; Zito, GA; Gambardella, A; Varricchio, G; Ragno, E; Varricchio, M. Chronic vitamin E administration improves brachial reactivity and increases intracellular magnesium concentration in Type II diabetic patients. J Clin Endocrinol Metab 2000, 85, 109–115. [Google Scholar]
- Michiels, C; Raes, M; Toussaint, O; Remacle, J. Importance of Se-glutathione peroxidase, catalase, and Cu/Zn-SOD for cell survival against oxidative stress. Free Radic Biol Med 1994, 17, 235–248. [Google Scholar]
- de Haan, JB; Cristiano, F; Iannello, R; Bladier, C; Kelner, MJ; Kola, I. Elevation in the ratio of Cu/Zn-superoxide dismutase to glutathione peroxidase activity induces features of cellular senescence and this effect is mediated by hydrogen peroxide. Hum Mol Genet 1996, 5, 283–292. [Google Scholar]
- Brown, KM; Morrice, PC; Arthur, JR; Duthie, GG. Effects of vitamin E supplementation on erythrocyte antioxidant defense mechanisms of smoking and non-smoking men. Clin Sci 1996, 91, 107–111. [Google Scholar]
- Chiarelli, F; Santilli, F; Sabatino, G; Blasetti, A; Tumini, S; Cipollone, F; Mezzetti, A; Verrotti, A. Effects of vitamin E supplementation on intracellular antioxidant enzyme production in adolescents with type 1 diabetes and early microangiopathy. Ped Res 2004, 56, 720–725. [Google Scholar]
- Eder, K; Flader, D; Hirche, F; Brandsch, C. Excess dietary vitamin E lowers the activities of antioxidative enzymes in erythrocytes of rats fed salmon oil. J Nutr 2002, 132, 3400–3404. [Google Scholar]
- Olaiz-Fernández, G; Rivera-Dommasco, J; Shamah-Levy, T; Rojas, R; Villalpando-Hernández, S; Hernández-Avila, M. Encuesta Nacional de Salud y Nutrición; Cuernavaca, México; Instituto Nacional de Salud Pública, 2006. [Google Scholar]
© 2007 MDPI All rights reserved.
Share and Cite
Ble-Castillo, J.L.; Cleva-Villanueva, G.; Díaz-Zagoya, J.C.; Medina-Santillán, R.; Rubio-Arias, H.O.; Méndez, J.D. Effects of α-Tocopherol on Oxidative Status and Metabolic Profile in Overweight Women. Int. J. Environ. Res. Public Health 2007, 4, 260-267. https://doi.org/10.3390/ijerph200704040001
Ble-Castillo JL, Cleva-Villanueva G, Díaz-Zagoya JC, Medina-Santillán R, Rubio-Arias HO, Méndez JD. Effects of α-Tocopherol on Oxidative Status and Metabolic Profile in Overweight Women. International Journal of Environmental Research and Public Health. 2007; 4(4):260-267. https://doi.org/10.3390/ijerph200704040001
Chicago/Turabian StyleBle-Castillo, J. L., G. Cleva-Villanueva, J. C. Díaz-Zagoya, R. Medina-Santillán, H. O. Rubio-Arias, and J. D. Méndez. 2007. "Effects of α-Tocopherol on Oxidative Status and Metabolic Profile in Overweight Women" International Journal of Environmental Research and Public Health 4, no. 4: 260-267. https://doi.org/10.3390/ijerph200704040001




