Analysis of Gene Regulation in Rabbit Corneal Epithelial Cells Induced by Ultraviolet Radiation
Abstract
:Introduction
Materials and Methods
Cell Culture
Corneal Epithelial Cells
UVB Irradiation
RNA Isolation
Fluorescent Differential Display-Reverse Transcription-Polymerase Chain Reaction (FDDRT-PCR)
6% Denaturing Polyacrylamide Electrophoresis
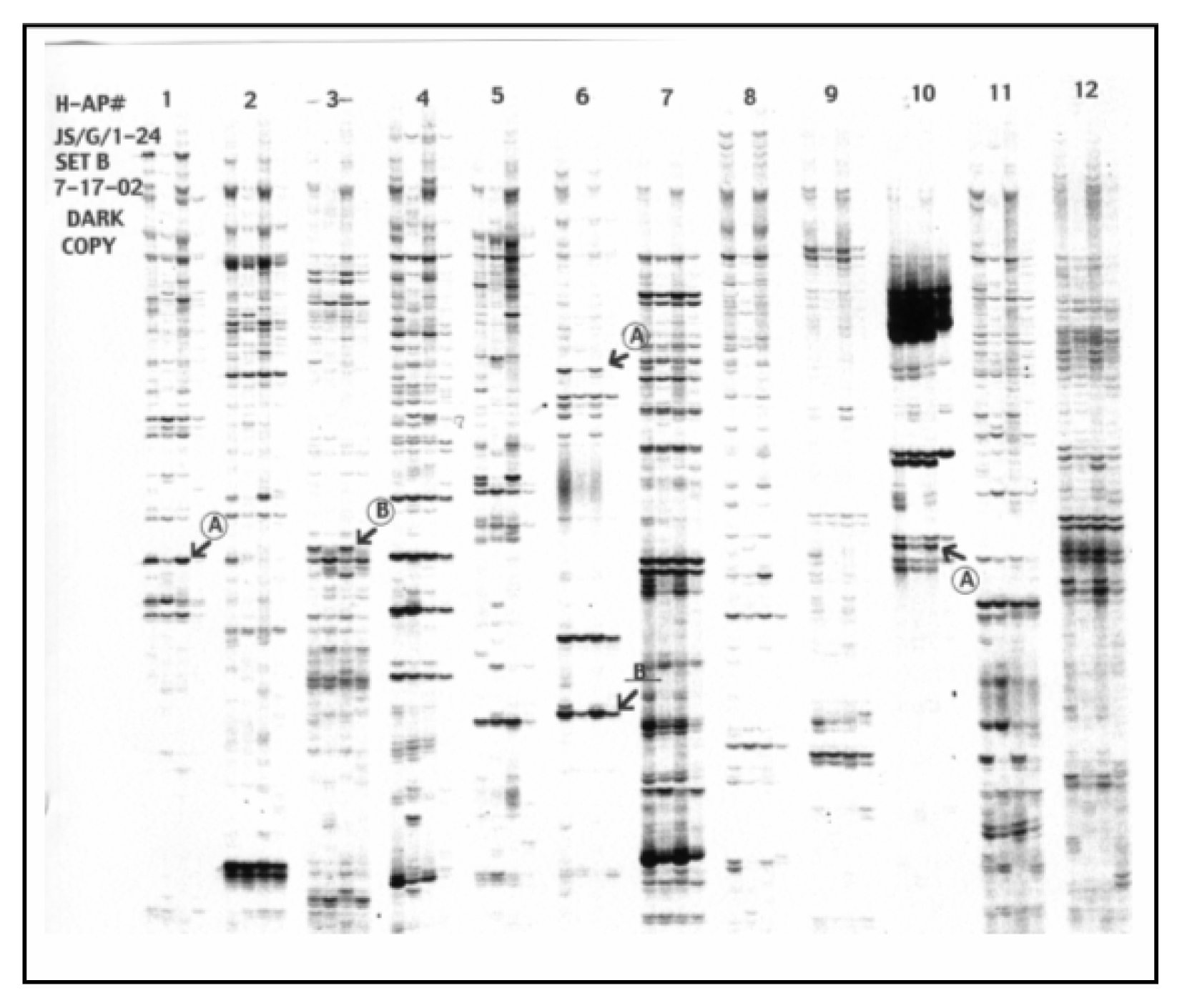
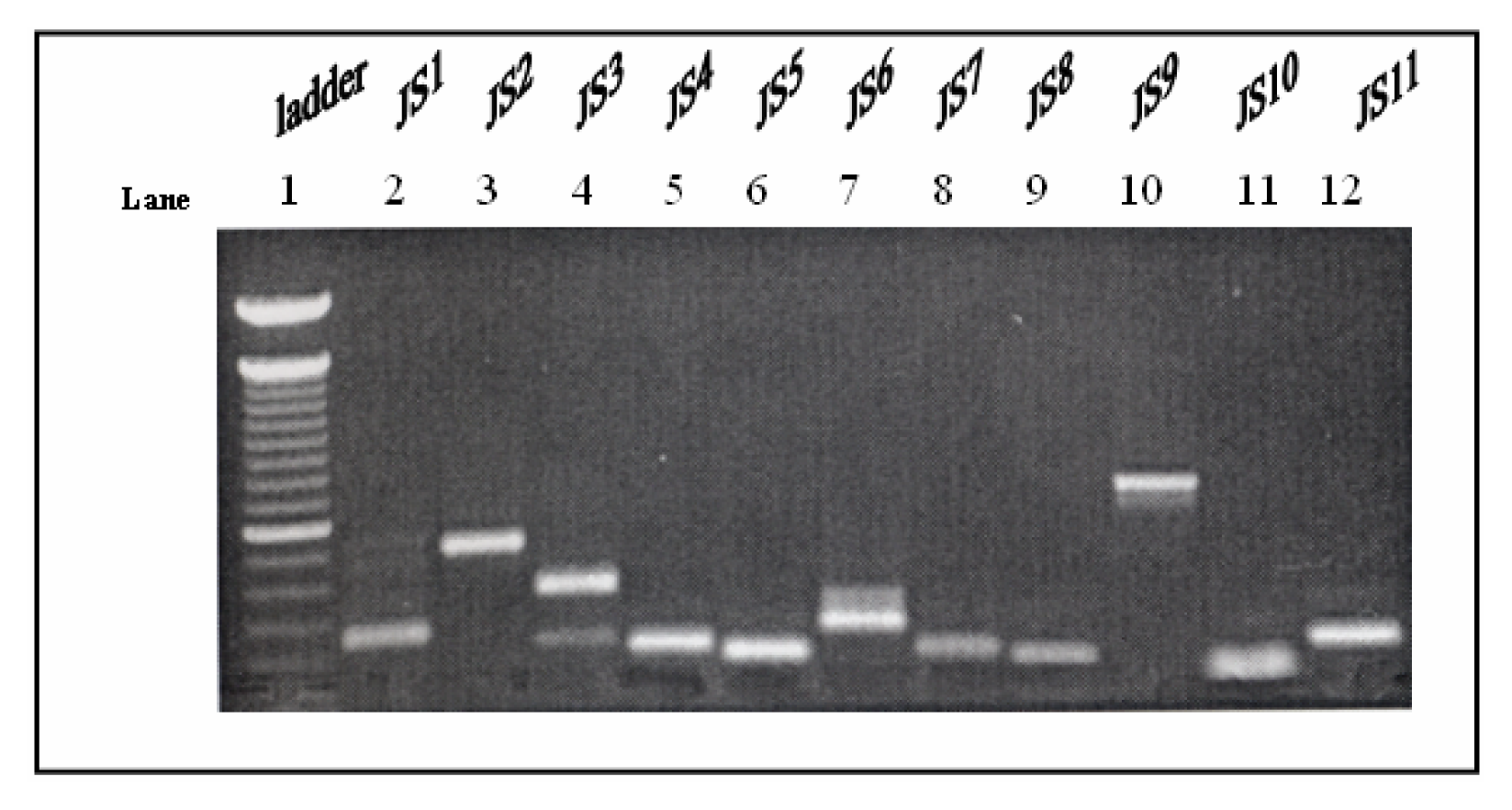
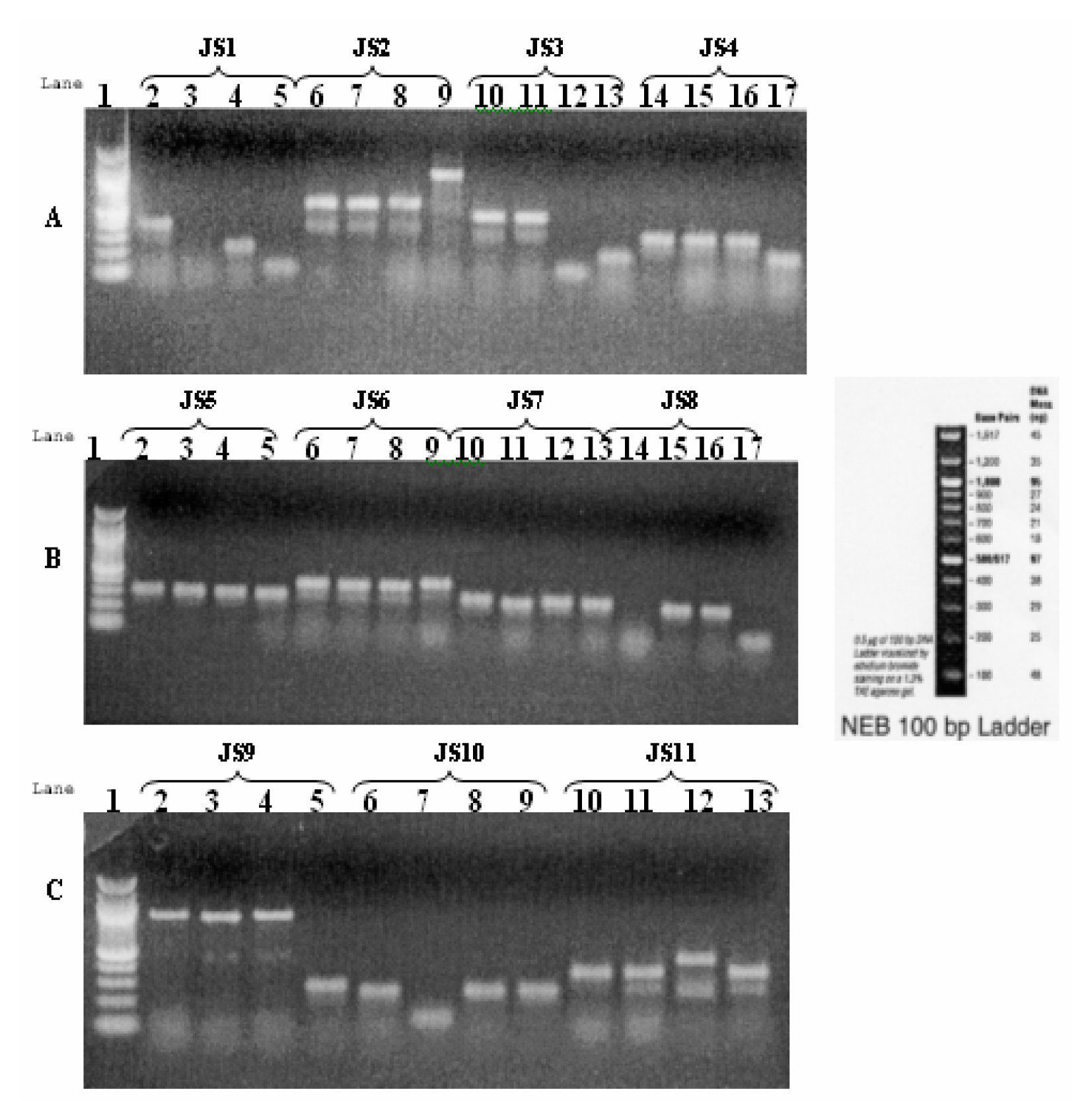
Selection of Differentially Expressed Bands, Re-amplification of cDNAs and Cloning System
Northern Blot Analysis
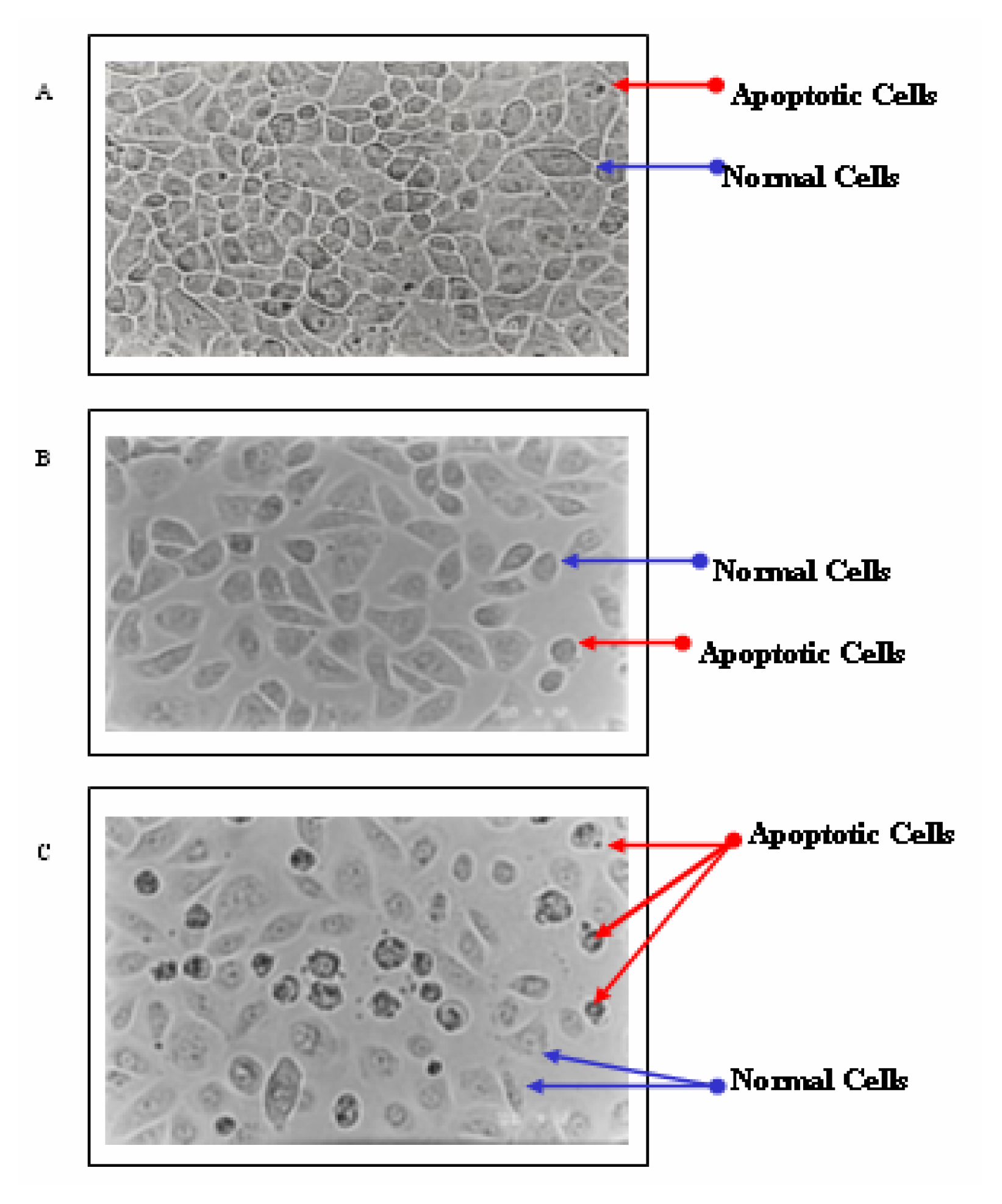
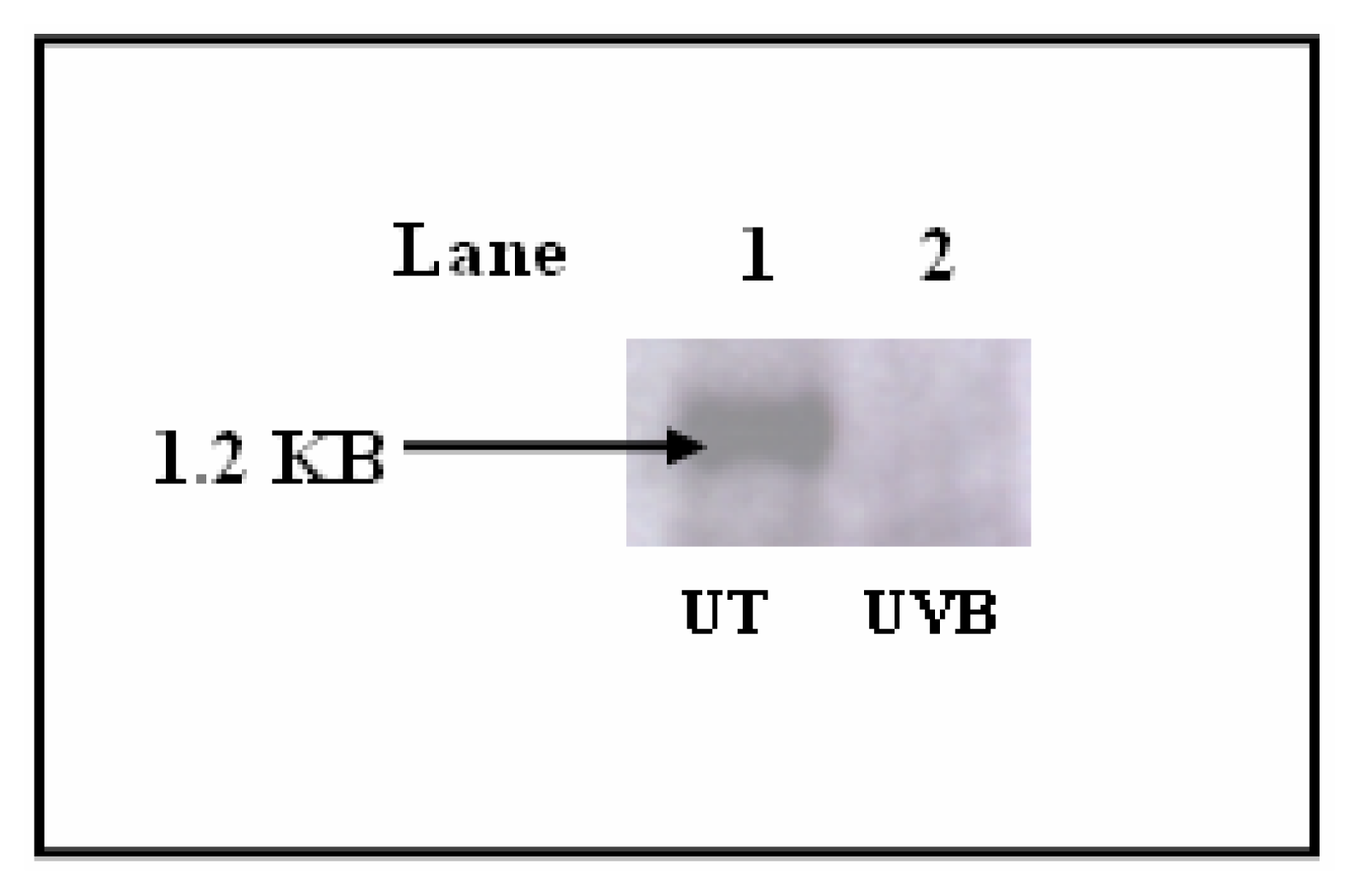
Results
Discussion
Acknowledgements
References
- Horneck, G. Quantification of biologically effective environmental UV irradiance. Adv Space Res 2000, 26, 1983–1994. [Google Scholar]
- Dixon, A. J.; Dixon, B. F. Ultraviolet radiation from welding and possible risk of skin and ocular malignancy. Med J. Aust 2004, 181(3), 155–157. [Google Scholar]
- Coldiron, B. M. Thinning of the ozone layer: facts and consequences. J. Am Acad Dermatol 1992, 27 5 Pt 1, 653–662. [Google Scholar]
- Dickerson, R. R.; Kondragunta, S.; Stenchikov, G.; Civerolo, K. L.; Doddridge, B. G.; Holben, B. N. The Impact of aerosols on solar ultraviolet radiation and photochemical smog. Science 1997, 278(5339), 827–830. [Google Scholar]
- Babu, V.; Misra, R. B.; Joshi, P. C. Ultraviolet-B effects on Ocular tissues. Biochem. Biophys. Res. Comn 1995, 210, 417–423. [Google Scholar]
- Wicket, H.; Zaar, K.; Grauer, A.; Zimmerman, M.; Gillardon, F. Differential induction of proto-oncogene expression and cell death in ocular tissues following ultraviolet irradiation of the rat eye. Br. J. Ophthalmol 1999, 83, 225–230. [Google Scholar]
- Freeman, S. E.; Hacham, H.; Gange, R. W.; Maytum, D. J.; Sutherland, J. C.; Sutherland, B. M. Wavelength dependence of pyrimidine dimmer formation in DNA of human skin irradiated in situ with ultraviolet light. Proc. Natl. Acad. Sci 1989, 86(14), 5605–5609. [Google Scholar]
- Fishman, G. A. Ocular Phototoxicity: guidelines for selecting sunglasses. Surv. Opthalmology 1986, 31(2), 119–124. [Google Scholar]
- Roberts, J. E. Ocular Phototoxicity. J. Photochem. Photobiol 2001, 64(2–3), 136–143. [Google Scholar]
- Longstreth, J. Anticipated Public Health Consequences of Global Climatic Change. Environ. Health Persp 1991, 96, 139–144. [Google Scholar]
- Hodge, W. G.; Whitcher, J. P.; Satariano, W. Risk factors for age-related cataracts. Epidemiol. Rev 1995, 17(2), 336–346. [Google Scholar]
- Spector, A.; Wang, G. M.; Wang, R. R. A brief photochemically induced oxidative insult causes irreversible lens damage and cataract II mechanism of action. Exp. Eye Res 1995, 60, 483–493. [Google Scholar]
- Zigman, S.; Paxhia, T.; McDaniel, T.; Schultz, J. New mechanisms for UV-induced cataract. Holick, M. F., Kligman, A. M., Eds.; In Biologic Effects of Light; Walter de Gruyter, 1992; pp. 245–252. [Google Scholar]
- Katiyar, S. K.; Perez, A.; Mukhtar, H. Green Tea Polyphenol Treatment to Human Skin Prevents Formation of Ultraviolet Light B-induced Pyrimidine Dimers in DNA. Clinical Cancer Research 2000, 6, 3864–3869. [Google Scholar]
- Podskochy, A.; Gan, L.; Fagerholm, P. Apoptosis in UV-exposed rabbit corneas. Cornea 2000, 19(1), 99–103. [Google Scholar]
- Sinha, R. P.; Hader, D. P. UV-induced DNA damage and repair: A review. Photochem. Photobiol. Sci 2002, 1, 225–236. [Google Scholar]
- Sato, H.; Suzuki, J.; Tanaka, M.; Ogiso, M.; Tohyama, C.; Kobayashi, S. Gene Expression in Skin Tumors Induced in hairless mice by chronic exposure to Ultraviolet B Irradiation. Photochemistry and Photobiology 1997, 65(5), 908–914. [Google Scholar]
- Quigyi, W.; Lee, J.; Gershenwald, J.; Ross, M.; Mansfield, P.; Strom, S.; Wang, L.; Guo, Z.; Qiao, Y.; Amos, C.; Spitz, M.; Duvic, M. Repair of UV Light-Induced Damage and Risk of Cutaneous Malignant Melanoma. J. of the National Cancer Institute 2003, 95(4), 308–315. [Google Scholar]
- Zhung, L.; Wang, B.; Sauder, D. N. Molecular mechanism of ultraviolet-induced keratinocyte apoptosis. Interferon Cytokine Res 2000, 20, 445–454. [Google Scholar]
- Sidjanin, D.; Grdina, D.; Woloschak, G. UV-Induced changes in cell cycle and gene expression within rabbit lens epithelial cells. Photochemistry and Photobiology 1995, 63(1), 79–85. [Google Scholar]
- Liang, P.; Pardee, A. Differential display of eukaryotic messenger RNA by means of the polymerase chain reaction. Science 1992, 257, 967–971. [Google Scholar]
- Abts, H. F.; Breuhahn, K.; Michel, G.; Kohrer, K.; Esser, P.; Ruzicka, T. Analysis of UVB-modulated Gene Expression in Human Keratinocytes by mRNA Differential Display Polymerase Chain Reaction. Photochemistry and Photobiology 1997, 66(3), 363–367. [Google Scholar]
- Choi, D. K.; Ito, T.; Mitsui, Y.; Sakaki, Y. Fluorescent differential display analysis of gene expression in apoptotic neuroblastoma cells. Gene 1998, 26, 223(1–2), 21–31. [Google Scholar]
- Kantorow, M.; Kays, T.; Horwitz, J.; Hwang, Q.; Sun, J.; Piatigorsky, J.; Carper, D. Differential Display Detects Altered Gene Expression between Cataractous and Normal Human Lenses. Invest Ophthalmol Vis. Sci 1998, 39, 2344–2354. [Google Scholar]
- Poirier, G. M.; Pyati, J.; Wan, J. S.; Erlander, M. G. Screening differentially expressed cDNA clones obtained by differential display using amplified RNA. Nucleic Acid Research 1997, 25(4), 913–914. [Google Scholar]
- Cubitt, C. L.; Tang, Q.; Monteriro, C. A.; Lausch, R. N.; Oakes, J. E. IL-8 gene expression in cultures of human corneal epithelial cells and kerotocytes. Invest Ophthalmol. Vis. Sci 1993, 34, 3199–3206. [Google Scholar]
- Sambrook, J.; Fritsch, E. F.; Maniatis, T. In Molecular Cloning: A Laboratory Manual, 2nd Edition; Sambrook, J., Fritsch, E. F., Maniatis, T., Eds.; Cold Springs Harbor Laboratory Press: New York, 1989. [Google Scholar]
- McGabon, A. J.; Martin, S. J.; Bissonnette, R. P.; Mabboubi, A.; Shi, Y.; Mogil, R. J.; Nishioka, W. K.; Green, D. R. The end of the (cell) line: methods for the study of apoptosis in vivo. Methods Cell. Biol 1995, 46, 153–185. [Google Scholar]
- Rogers, C.; Chan, L.; Sims, Y.; Byrd, K.; Hinton, D.; Twining, S. The effects of subsolar levels of UV-A and UV-B on rabbit corneal and lens epithelial cells. Experimental Eye Research 2004, 1007–1014. [Google Scholar]
- Gratsch, T. E. Differential Display. Methods Mol Biol 2002, 198, 213–221. [Google Scholar]
- Carper, D. A.; Sun, J. K.; Iwata, T.; Zigler, S.; Ibaraki, N.; Lin, L.; Reddy, V. Oxidative stress induces differential gene expression in a human lens epithelial cell line. Invest Ophthalmol Vis Sci 1999, 40, 400–406. [Google Scholar]
- Corton, J.; Gustafsson, J. Increased Efficiency in Screening Large Numbers of cDNA Fragments Generated by Differential Display. Biotechniques 1997, 22, 802–810. [Google Scholar]
- Liang, P.; Pardee, A. Differential display of eukaryotic messenger RNA by means of the polymerase chain reaction. Science 1992, 257, 967–971. [Google Scholar]
- Sehgal, A.; Keener, C.; Boynton, A. L.; Young, R. F.; Vermeulen, S. S.; Yonemura, K. S.; Kohler, E. P.; Aldape, H. C.; Simrell, C. R.; Murphy, G. P. Isolation and characterization of a novel gene from human glioblastoma multiforme tumor tissue. International J. of Cancer 1997, 71(4), 565–572. [Google Scholar]
- Chang, G. T.; Tapsi, N.; Steenbeek, M.; Blok; vam Weerden, W. M.; van Alewijk, D. C.; Eussen, B. H.; van Steenbrugge, G. J.; Brinkmann, A. O. Identification of gene on human chromosome 8q11 that is differentially expressed during prostate-cancer progression. International J. of Cancer 1999, 83(4), 506–511. [Google Scholar]
- Stevens, J. J. The Determination of a Light Activated Genes in the Basidiomycete, Corprinus congregatus. Ph.D. Dissertation, University of California, Santa Barbara, 1995. [Google Scholar]
- Luehrsen, K. R.; Marr, L. L.; van der Knaap, E.; Cumberledge, S. Analysis of differential display RT-PCR products using fluorescent primers and GENESCAN software. Biotechniques 1997, 1, 168–74. [Google Scholar]
© 2005 MDPI. All rights reserved.
Share and Cite
Stevens, J.J.; Rogers, C.; Howard, C.B.; Moore, C.; Chan, L.-M. Analysis of Gene Regulation in Rabbit Corneal Epithelial Cells Induced by Ultraviolet Radiation. Int. J. Environ. Res. Public Health 2005, 2, 51-57. https://doi.org/10.3390/ijerph2005010051
Stevens JJ, Rogers C, Howard CB, Moore C, Chan L-M. Analysis of Gene Regulation in Rabbit Corneal Epithelial Cells Induced by Ultraviolet Radiation. International Journal of Environmental Research and Public Health. 2005; 2(1):51-57. https://doi.org/10.3390/ijerph2005010051
Chicago/Turabian StyleStevens, Jacqueline J., Christian Rogers, Carolyn B. Howard, Caronda Moore, and Lai-Man Chan. 2005. "Analysis of Gene Regulation in Rabbit Corneal Epithelial Cells Induced by Ultraviolet Radiation" International Journal of Environmental Research and Public Health 2, no. 1: 51-57. https://doi.org/10.3390/ijerph2005010051



