Photodecomposition and Phototoxicity of Natural Retinoids
Abstract
:Introduction
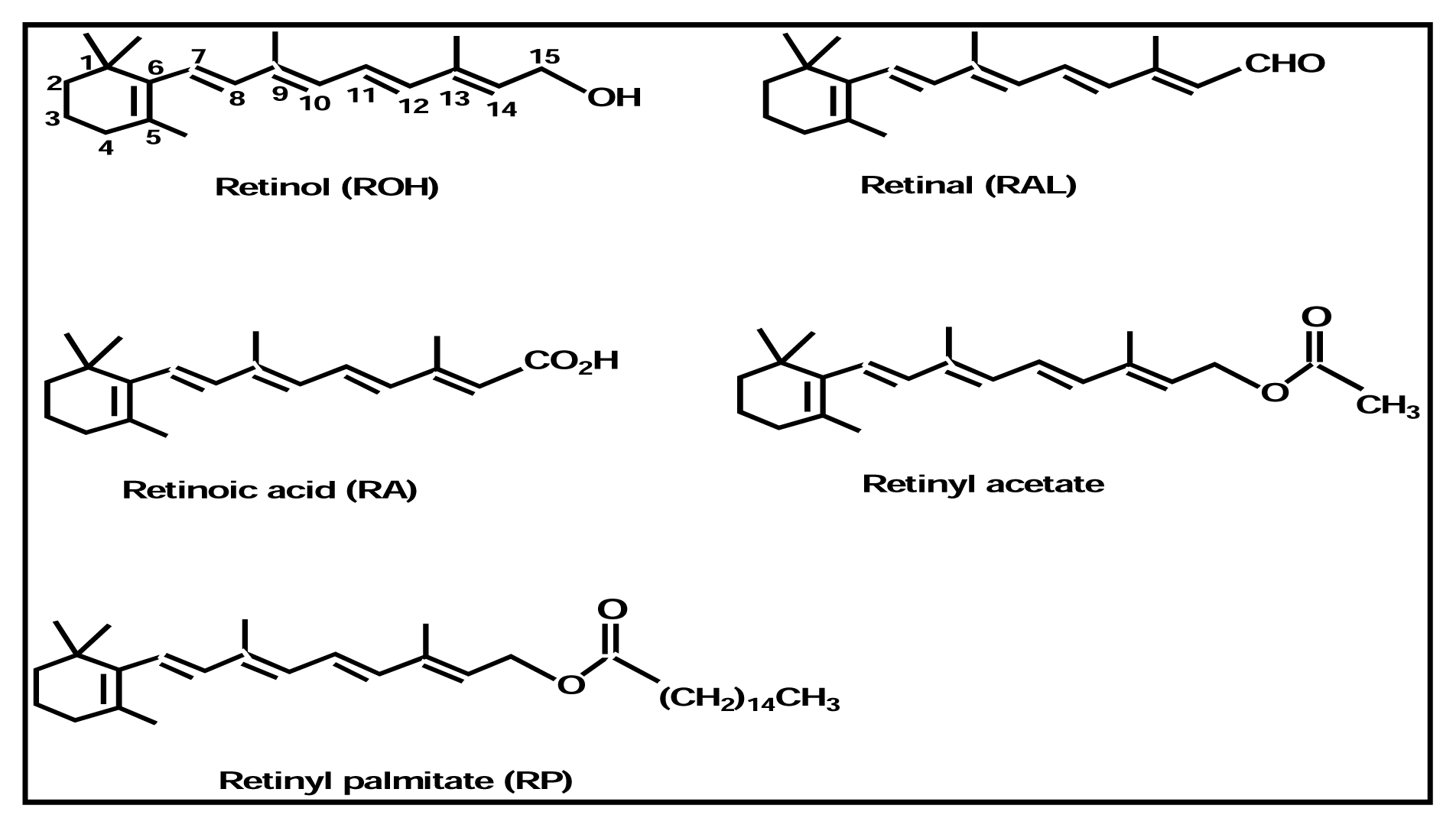
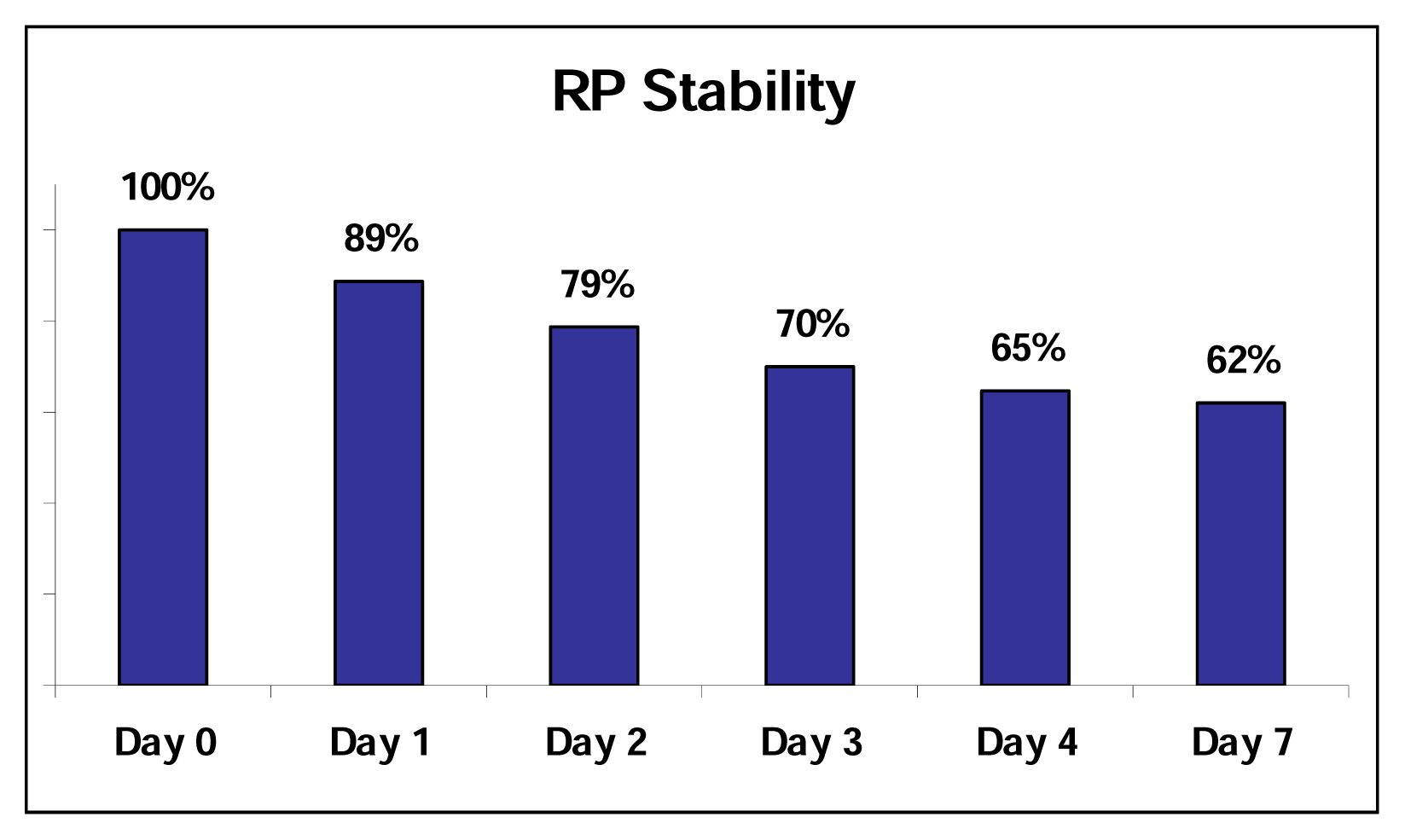
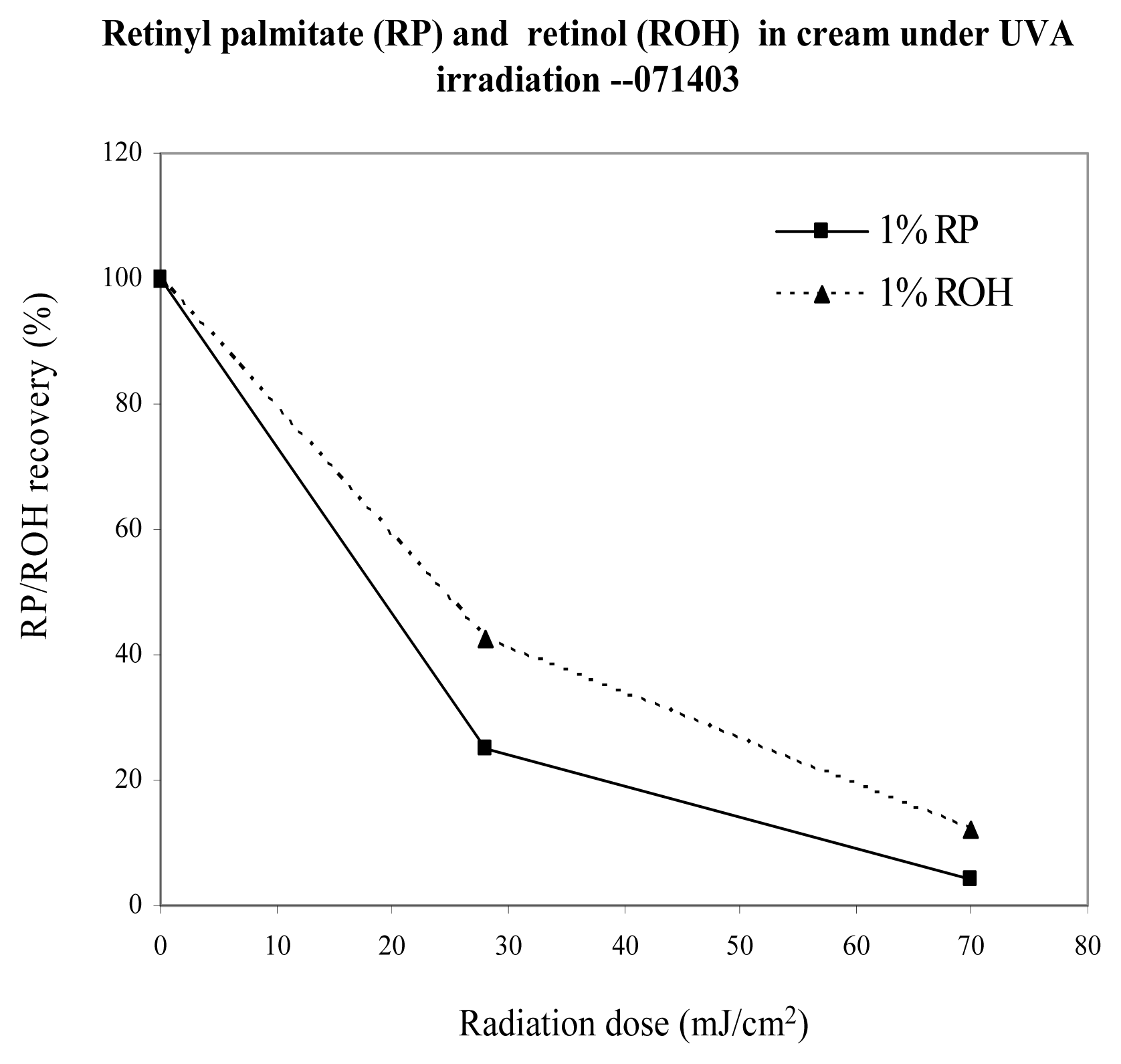
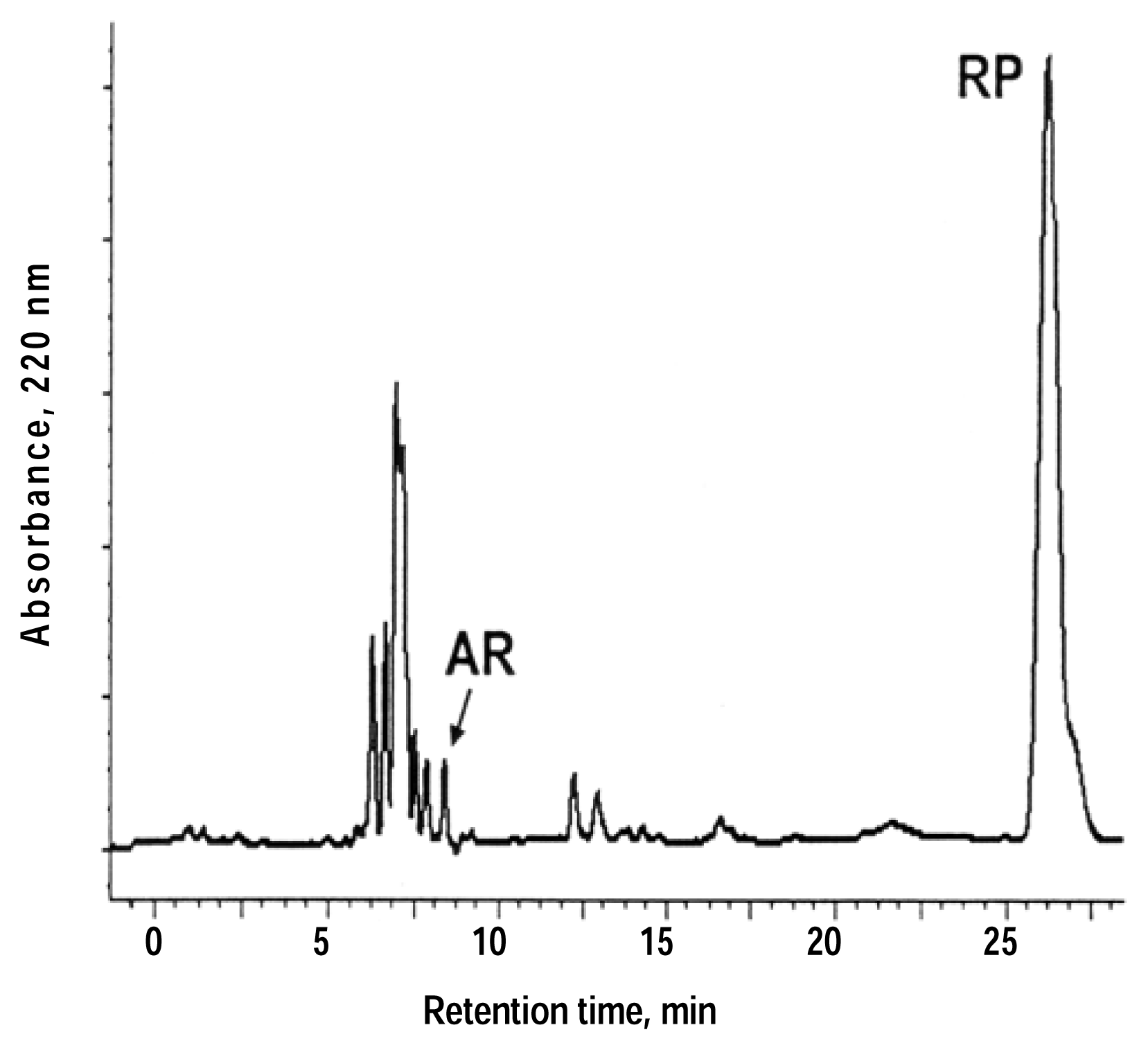
Thermal and Photo Stability
Photoreactions
Isomerization
Photoreaction of Retinoids
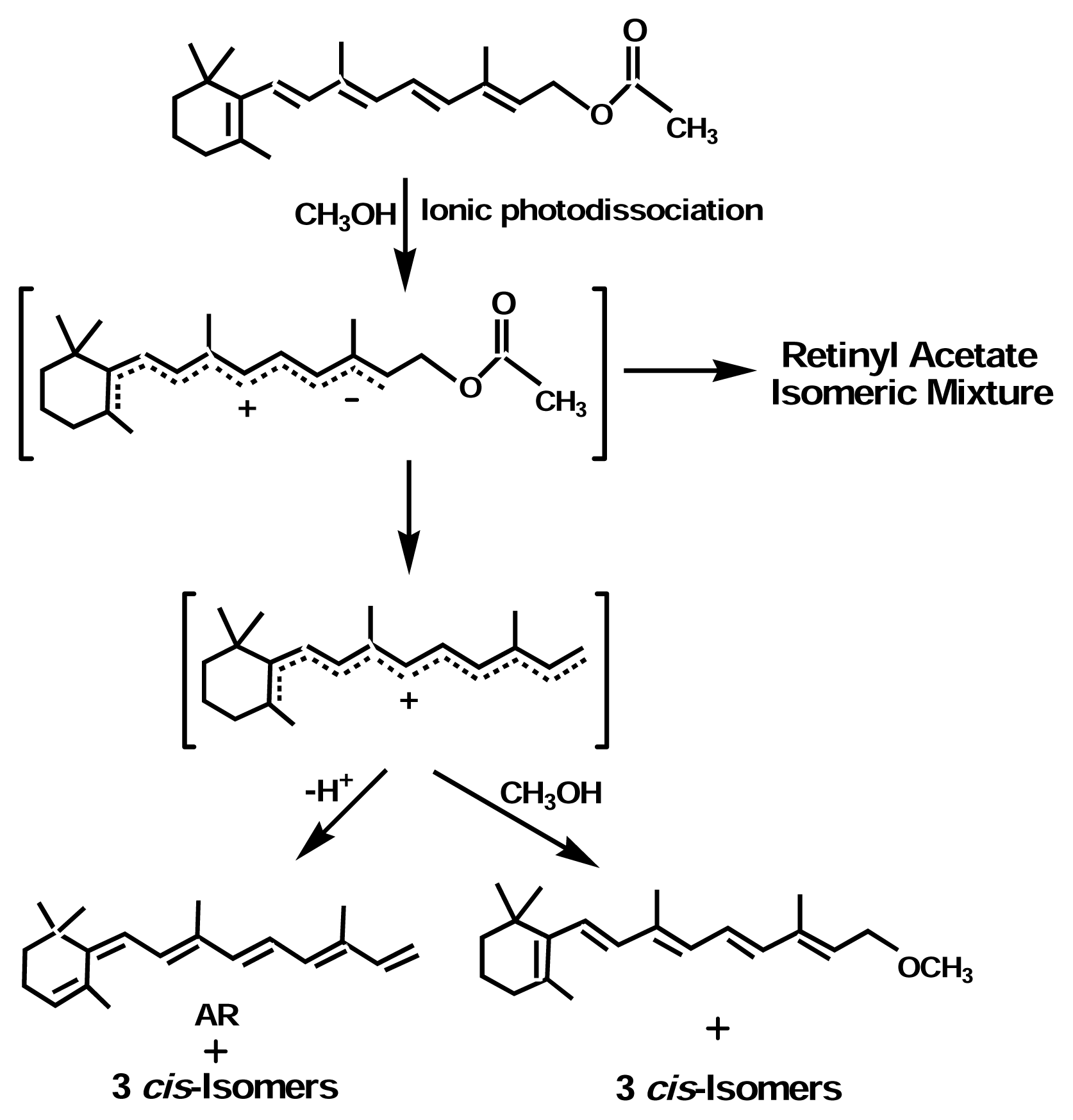
Mechanisms of Photoreaction
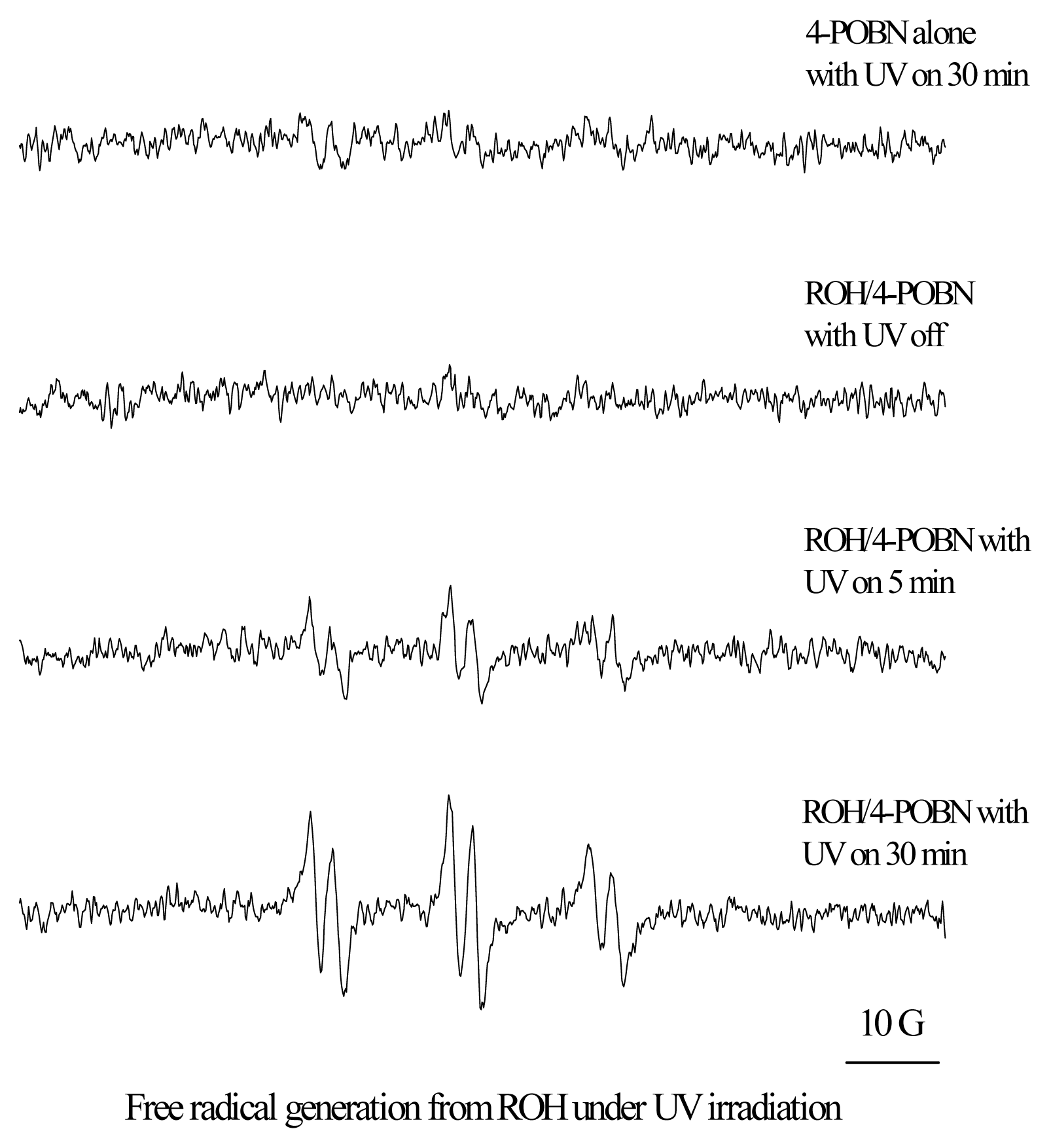
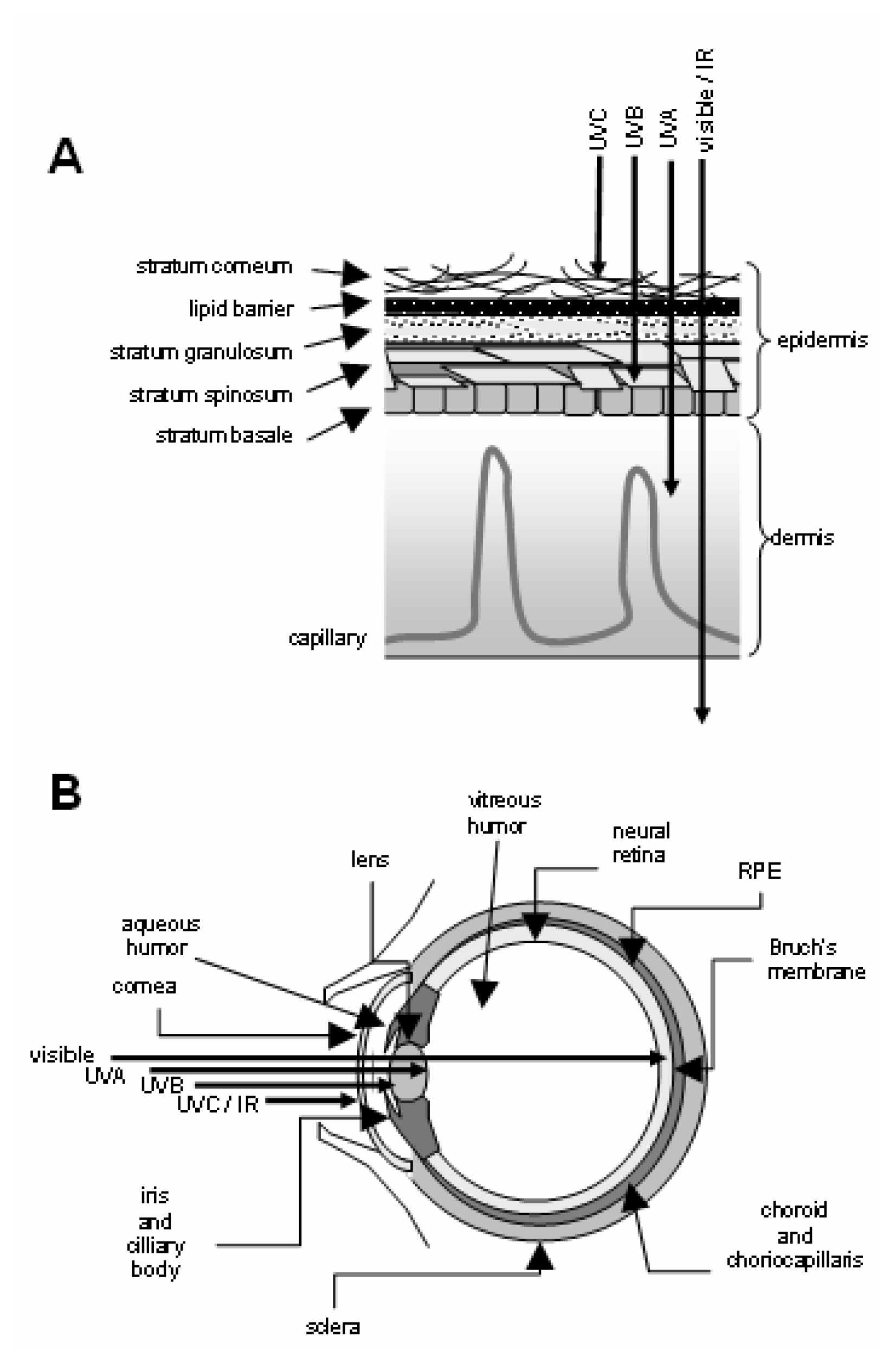
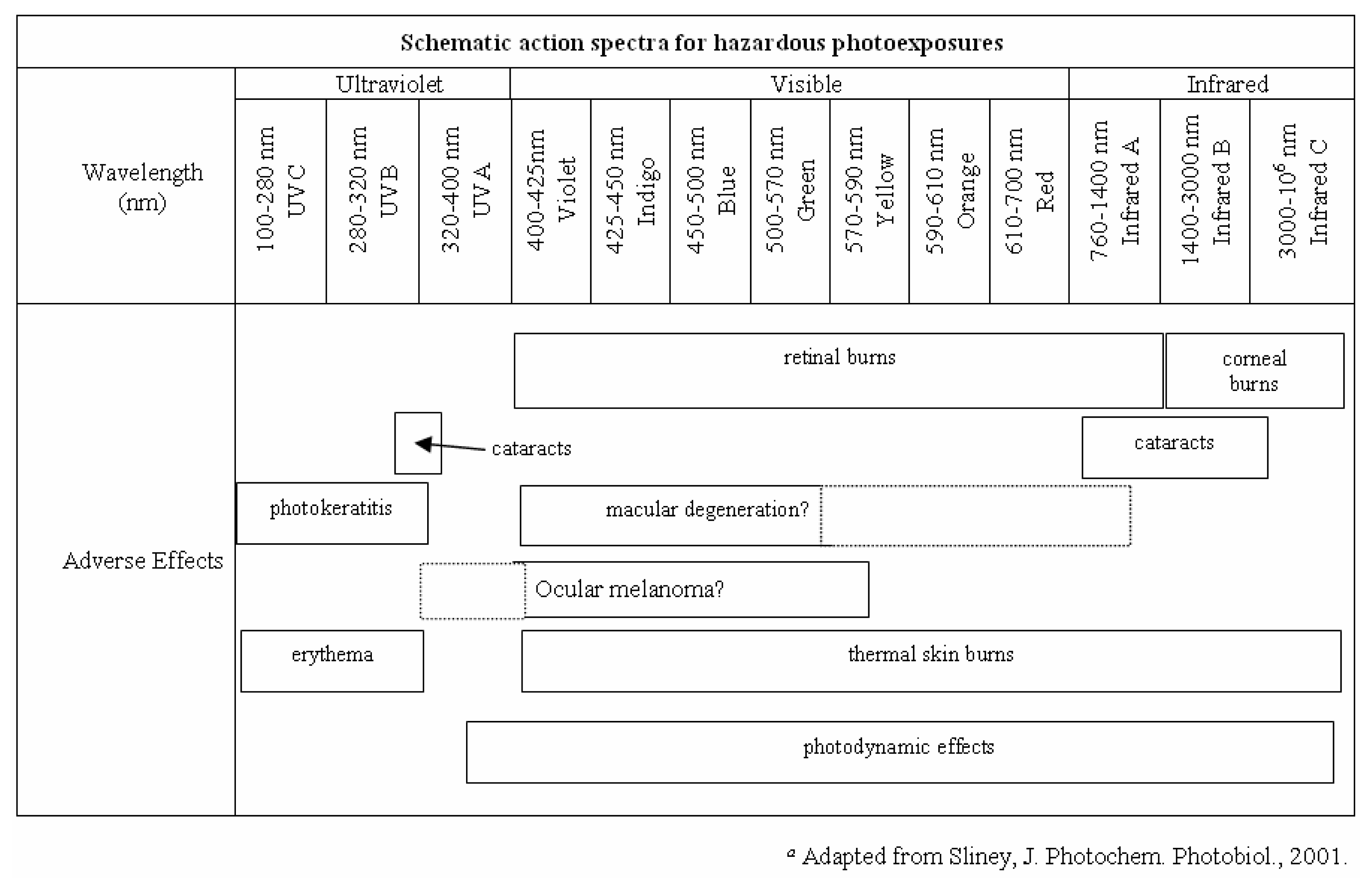
Phototoxicity
Cytotoxicity and Genotoxicity
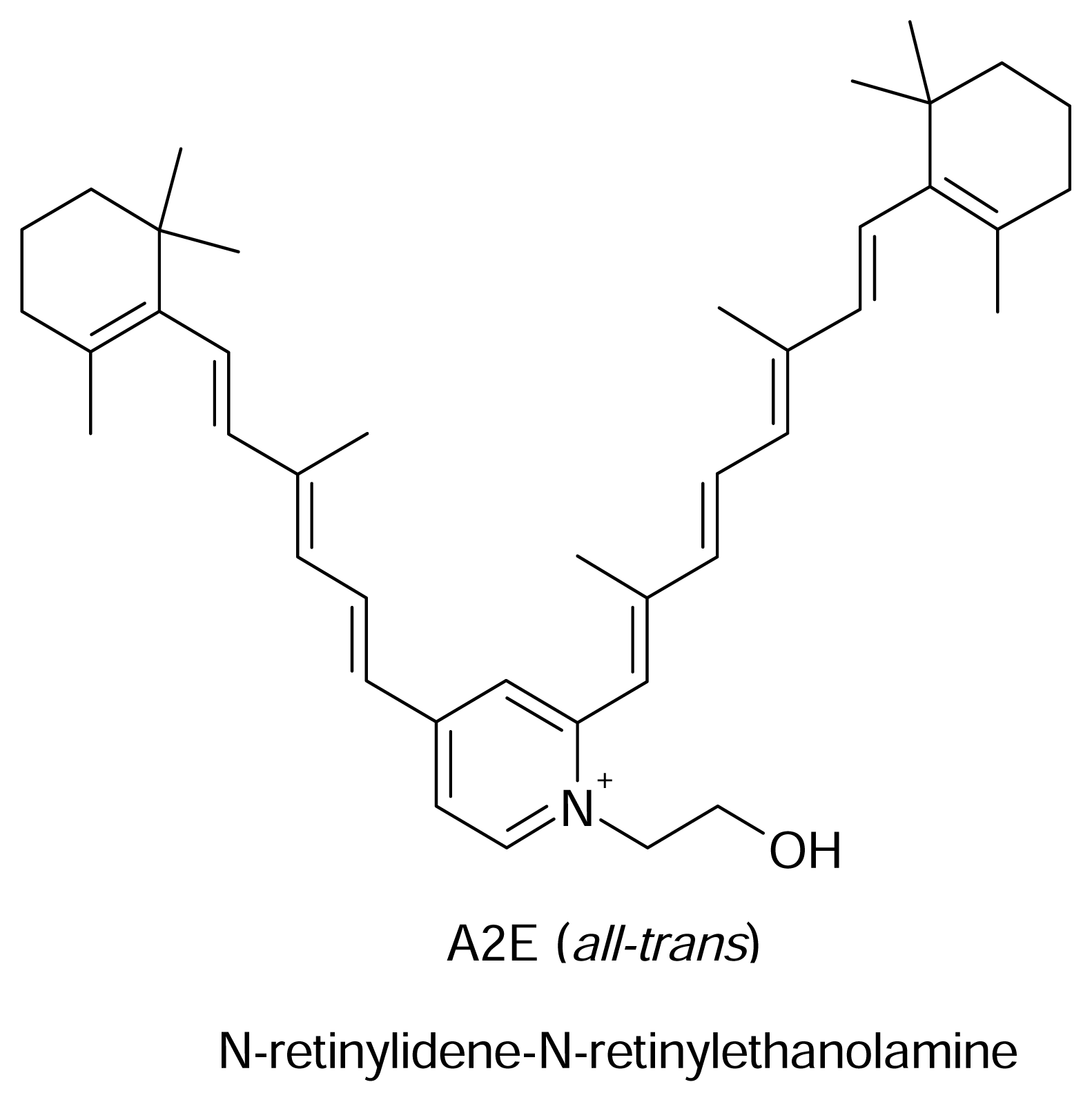
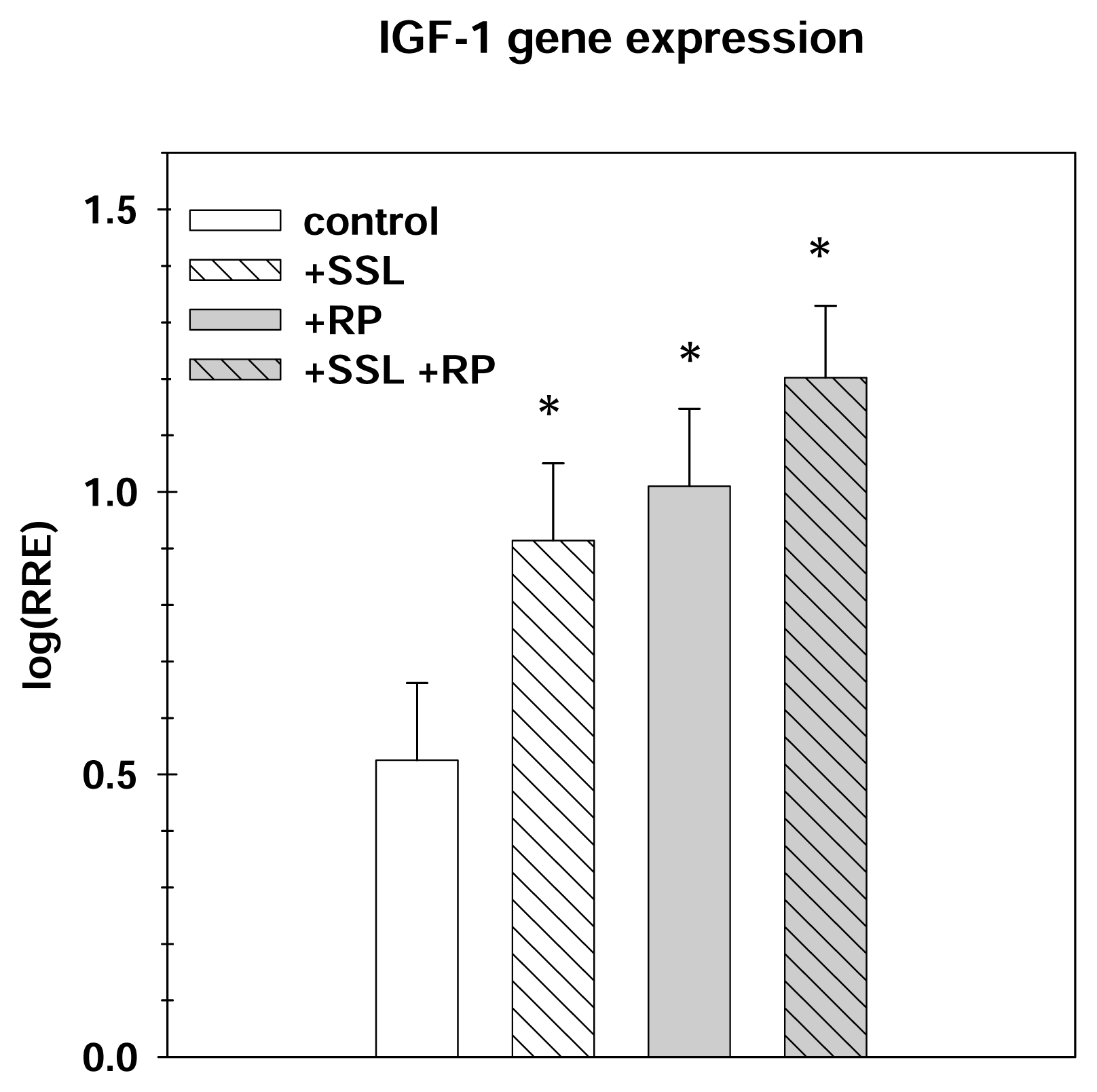
Effect on Gene Expression
Tumorigenicity
Perspective


Acknowledgement
References
- (IARC), Solar and ultraviolet radiation. In IARC Monographs on the Evaluation of Carcinogenic Risks to Humans; International Agency for Research on Cancer, 1992; Volume 55.
- Idson, B. Vitamins in cosmetics, an update I. Overview and Vitamin A. Drug Cosmet. Ind 1990, 146, 26–91. [Google Scholar]
- (IARC), Vitamin A. International Agency for Research on Cancer; Handbooks of Cancer Prevention: Lyon, French, 1998; Volume 3.
- Code of Federal Regulations; Washington, D.C, 2000; Title 21, Part 720.4 (d)(1).
- The Cosmetic, Toiletry and Fragrance Association, International Cosmetic Ingredient Dictionary and Handbook, Eighth Edition; 2000; Wenninger, J.; Canterbery, R.C.; McEwen, G.N., Jr (Eds.) CTFA: Washington, D.C, 1999; p. 1279.
- Ihara, H.; Hashizume, N.; Hirase, N.; Suzue, R. Esterification makes retinol more labile to photolysis. J. Nutr. Sci. Vitaminol 1999, 45, 353–358. [Google Scholar]
- Ji, H.-G.; Seo, B. Retinyl palmitate at 5% in a cream: its stability, efficacy and effect. Cosmet. and Toilet 1999, 114, 61–68. [Google Scholar]
- McBee, J. K.; Kuksa, V.; Alvarez, R.; de Lera, A. R.; Prezhdo, O.; Haeseleer, F.; Sokal, I.; Palczewski, K. Isomerization of all-trans-retinol to cis-retinols in bovine retinal pigment epithelial cells: dependence on the specificity of retinoid-binding proteins. Biochemistry 2000, 39, 11370–11380. [Google Scholar]
- Boehnlein, J.; Sakr, A.; Lichtin, J. L.; Bronaugh, R. L. Characterization of esterase and alcohol dehydrogenase activity in skin. Metabolism of retinyl palmitate to retinol (vitamin A) during percutaneous absorption. Pharm. Res 1994, 11, 1155–1159. [Google Scholar]
- Roos, T. C.; Jugert, F. K.; Merk, H. F.; Bickers, D. B. Retinold metabolism in the skin. Pharm. Rev 1998, 50, 315–333. [Google Scholar]
- Mousseron-Canet, M.; Mani, J-C.; Favie, C.; Lerner, D. On the Photochemical Isomerization of Vitamin A. Compt. Rend 1966, 262, 153–155. [Google Scholar]
- Mousseron-Canet, M. Photochemical Transformation of Vitamin A. Vitamins and Coenzymes. McCormic, D. B., Wright, L. D., Eds.; In Methods in Enzymology; 1971; pp. 591–615. [Google Scholar]
- Dillon, J.; Gaillard, E. R.; Bilski, P.; Chignell, C. F.; Reszka, K. J. The photochemistry of the retinoids as studied by steady-state and pulsed methods. Photochem. Photobiol 1996, 63, 680–685. [Google Scholar]
- Fu, P. P.; Cherng, S.-H.; Coop, L.; Xia, Q.; Culp, S. J.; Tolleson, W. H.; Wamer, W. G.; Howard, P. C. Photoreaction, phototoxicity, and photocarcinogenicity of retinoids. Environ. Carcinogen. Ecotoxicol. Rev 2003, 21(2), 165–197. [Google Scholar]
- Landers, G. M.; Olson, J. A. Absence of isomerizaiton of retinyl palmitate, retinol, and retinal in chlorinated and nonchlorinated solvents under gold light. J. AOAC. Int 1986, 69, 50–55. [Google Scholar]
- Teraoka, R.; Konishi, Y.; Matsuda, Y. Photochemical and oxidative degradation of the solid-state tretinoin tocoferil. Chem. Pharm. Bull 2001, 49, 368–372. [Google Scholar]
- Murayama, A.; Suzuki, T.; Matsui, M. Photoisomerization of retinoic acids under room light: a warning for cell biological study of geometrical isomers of retinoids. J. Nutr. Sci. Vitaminol 1997, 43, 167–176. [Google Scholar]
- Reddy, A.M.; Rao, V. J. Ionic photodissociation of polyenes via a highly polarized singlet excited state. J. Org. Chem 1992, 57, 6727–6731. [Google Scholar]
- Crank, G.; Pardijanto, M. S. Photooxidations and photosensitized oxidations of vitamin A and its palmitate ester. J. Photochem. Photobiol. A 1995, 85, 93–100. [Google Scholar]
- Lamb, L.E.; Zareba, M.; Plakoudas, S. N.; Sarna, T.; Simon, J. D. Retinyl palmitate and the blue-light-induced phototoxicity of human ocular lipofuscin. Arch. Biochem. Biophys 2001, 393, 316–320. [Google Scholar]
- Tatariunas, A.; Matsumoto, S. A retinyl palmitate model of the phenomenon of the intrinsic fluorescence increase in ceroid-lipofuscin cytosomes. Exp. Gerontol 2000, 35, 1327–1341. [Google Scholar]
- Fu, P. P.; Xia, Q.; Blankenship, L. G.; Webb, P. J.; Wamer, W. G.; Howard, P. C. Photostability and photogenotoxicity of retinyl palmitate. 42nd SOT Annual Meeting; Abstract #1007. 2003. [Google Scholar]
- Isoe, S.; Hyeon, S. B.; Katsumura, S.; Sakan, T. Photooxygenation of carotenoids. II. The absolute configuation of loliolide and dihydroactinidiolide. Tetrahedron. Lett 1972, 25, 2517–2520. [Google Scholar]
- Boulton, M. E.; Rozanowska, M.; Rozanowska, B. Retinal photodamage. J. Photochem. Photobiol. B 2001, 64, 144–161. [Google Scholar]
- Ferguson, J.; Johnson, B. E. Retinoid assoicated phototoxicity and photosensitivity. Pharmac. Ther 1989, 40, 123–135. [Google Scholar]
- Delmelle, M. Retinal damage by light: possible implication of singlet oxygen. Biophys. Struct. Mech 1977, 3, 195–198. [Google Scholar]
- Delmelle, M. Possible implication of photooxidation reactions in retinal photo-damage. Photochem. Photobiol 1979, 29, 713–716. [Google Scholar]
- El Ghissassi, F.; Barbin, A.; Nair, J.; Bartsch, H. Formation of 1,N6-ethenodeoxyadenosine and 3,N4-ethenodeoxycytidine by lipid peroxidation products and nucleic acid bases. Chem. Res. Toxicol 1995, 8, 278–283. [Google Scholar]
- Wang, M.-Y.; Liehr, J. G. Induction by estrogens of lipid peroxidation and lipid peroxide-derived malondialdehyde-DNA adducts in male Syrian hamsters: role of lipid peroxidation in estrogen-induced kidney carcinogenesis. Carcinogenesis 1995, 16, 1941–1945. [Google Scholar]
- Threshold limit values for chemical substances and physical agents and biological exposure indices. American Conference of Governmental Industrial Hygenists (ACGIH) 1996.
- Sliney, D. H. Photoprotection of the eye - UV radiation and sunglasses. J. Photochem. Photobiol 2001, 64, 166–175. [Google Scholar]
- Xia, Q.; Cherng, S.-H.; Blankenship, L. R.; Wamer, W. G.; Bucher, J. R.; Howard, P. C.; Fu, P. P. Formation of reactive oxygen species and lipid peroxides from photoirradiation of retinyl palmitate and its photodecomposition products. The 32nd Annual Meeting of the American Society for Photobiology; Abstract #164. 2004. [Google Scholar]
- Yan, J.; Cherng, S.-H.; Xia, Q.; Wamer, W. G.; Bucher, J. R.; Howard, P. C.; Yu, H.; Fu, P. P. Photoclastogenicity of retinyl palmitate and its photodecomposition products. The 32nd Annual Meeting of the American Society for Photobiology; Abstract #165. 2004. [Google Scholar]
- Sparrow, J. R.; Vollmer-Snarr, H. R.; Zhou, J.; Jang, Y. P.; Jockusch, S.; Itagaki, Y.; Nakanishi, K. A2E-epoxides damage DNA in retinal pigment epithelial cells. Vitamin E and other antioxidants inhibit A2E-epoxide formation. J. Biol. Chem 2003, 278, 18207–18213. [Google Scholar]
- Heck, D. E.; Gerecke, D. R.; Vetrano, A. M.; Laskin, J. D. Solar ultraviolet radiation as a trigger of cell signal transduction. Toxicol. Appl. Pharmacol 2004, 195, 288–297. [Google Scholar]
- Abe, T.; Oue, N.; Yasui, W.; Ryoji, M. Rapid and preferential induction of ATF3 transcription in response to low doses of UVA light. Biochem. Biophys. Res. Commun 2003, 310, 1168–1174. [Google Scholar]
- Dazard, J. E.; Gal, H.; Amariglio, N.; Rechavi, G.; Domany, E.; Givol, D. Genome-wide comparison of human keratinocyte and squamous cell carcinoma responses to UVB irradiation: implications for skin and epithelial cancer. Oncogene 2003, 22, 2993–3006. [Google Scholar]
- Sesto, A.; Navarro, M.; Burslem, F.; Jorcano, J.L. Analysis of the ultraviolet B response in primary human keratinocytes using oligonucleotide microarrays. Proc. Natl. Acad. Sci. USA 2002, 99, 2965–9670. [Google Scholar]
- Lee, K. H.; Chang, M. Y.; Ahn, J. I.; Yu, D. H.; Jung, S. S.; Choi, J. H.; Noh, Y. H.; Lee, Y. S.; Ahn, M. J. Differential gene expression in retinoic acid-induced differentiation of acute promyelocytic leukemia cells, NB4 and HL-60 cells. Biochem. Biophy. Res. Commun 2002, 296, 1125–1133. [Google Scholar]
- Pfaffl, M. W. A new mathematical model for relative quantification in real-time RT-PCR. Nucleic. Acis Res 2001, 29, e45. [Google Scholar]
- Hasegawa, R.; Takahashi, M.; Furukawa, F.; Toyoda, K.; Sato, H.; Hayashi, Y. Co-carcinogenic effect of retinyl acetate on forestomach carcinogenesis of male F344 rats induced with butylated hydroxyanisole. Jpn. J. Cancer Res. (Gann) 1985, 79, 320–328. [Google Scholar]
- Kligman, L. H. Retinoic acid and photocarcinogenesis - a controversy. Photodermatology 1987, 4, 88–101. [Google Scholar]
- Forbes, P. D.; Urbach, F.; Davies, R. E. Enhancement of experimental photocarcinogenesis by topical retinoic acid. Cancer Lett 1979, 7, 85–90. [Google Scholar]
- Halliday, G. M.; Robertson, B. O.; Barnetson, R. St. C. Topical retinoic acid enhances, and a dark tan protects, from subedemal solar-simulated photocarcinogenesis. J. Invest. Dermatol 2000, 114, 923–927. [Google Scholar]
- Forbes, P. D. Photocarcinogenesis: an overview. J. Invest. Dermatol 1981, 77, 139–143. [Google Scholar]
- Epstein, J. H.; Grekin, D. A. Inhibition of ultraviolet-induced carcinogenesis by all-trans retinoic acid. J. Invest. Dermatol 1981, 76, 178–180. [Google Scholar]
- Kligman, L. H.; Kligman, A. M. Inhibition of ultraviolet-induced carcinogenesis by all-trans retinoic acid. Arch Dermatol Res 1981, 270, 453–462. [Google Scholar]
© 2005 MDPI. All rights reserved.
Share and Cite
Tolleson, W.H.; Cherng, S.-H.; Xia, Q.; Boudreau, M.; Yin, J.J.; Wamer, W.G.; Howard, P.C.; Yu, H.; Fu, P.P. Photodecomposition and Phototoxicity of Natural Retinoids. Int. J. Environ. Res. Public Health 2005, 2, 147-155. https://doi.org/10.3390/ijerph2005010147
Tolleson WH, Cherng S-H, Xia Q, Boudreau M, Yin JJ, Wamer WG, Howard PC, Yu H, Fu PP. Photodecomposition and Phototoxicity of Natural Retinoids. International Journal of Environmental Research and Public Health. 2005; 2(1):147-155. https://doi.org/10.3390/ijerph2005010147
Chicago/Turabian StyleTolleson, William H., Shui-Hui Cherng, Qingsu Xia, Mary Boudreau, Jun Jie Yin, Wayne G. Wamer, Paul C. Howard, Hongtao Yu, and Peter P. Fu. 2005. "Photodecomposition and Phototoxicity of Natural Retinoids" International Journal of Environmental Research and Public Health 2, no. 1: 147-155. https://doi.org/10.3390/ijerph2005010147
APA StyleTolleson, W. H., Cherng, S.-H., Xia, Q., Boudreau, M., Yin, J. J., Wamer, W. G., Howard, P. C., Yu, H., & Fu, P. P. (2005). Photodecomposition and Phototoxicity of Natural Retinoids. International Journal of Environmental Research and Public Health, 2(1), 147-155. https://doi.org/10.3390/ijerph2005010147



