Abstract
The uncertainty in the risk assessment of trace metal elements in soils when total metal contents are used can be decreased by assessing their availability and/or extractability when the soils have a high background value or different sources of trace metal elements. In this study, the added water-soluble nickel (Ni) toxicity to barley root elongation was studied in 17 representative Chinese soil samples with and without artificial rainwater leaching. The extractability of added Ni in soils was estimated by three sequential extractions with ethylenediaminetetraacetic acid (EDTA). The results showed that the effective concentration of EDTA extractable Ni (EC50), which caused 50% inhibition of barley root elongation, ranged from 46 to 1019 mg/kg in unleached soils and 24 to 1563 mg/kg in leached soils. Regression models for EDTA extractable Ni and total Ni added to soils against soil properties indicated that EDTA extractable Ni was significantly correlated with the total Ni added to soils and that pH was the most important control factor. Regression models for toxicity thresholds based on EDTA extractable Ni against soil properties showed that soil citrate dithionate extractable Fe was more important than soil pH in predicting Ni toxicity. These results can be used to accurately assess the risk of contaminated soils with high background values and/or different Ni sources.
1. Introduction
Most current soil risk assessment and soil quality criteria for trace metal elements are based on their total concentrations in soil [1]. However, the biological toxicity of trace metal elements is hard to identify if total metal concentrations are used to assess ecological risk [2]. Therefore traditional soil quality criteria for trace metal elements may not be suitable when attempting to assess the ecological risk of soils with high background values and/or different input sources of trace metal elements. These inaccuracies could give rise to underestimations or overestimations of trace metal pollution [3] because total concentrations of trace metal elements could not represent metal toxicity well [4]. Previous results suggested that it was more accurate to estimate the toxicity of trace metal elements in soils by measuring extractable fractions rather than total concentrations [5,6].
The concentrations, forms and extractability of trace metal elements generally determined their mobility, availability and toxicity. There are many different methods are used to assess the different chemical forms and fractions of trace metal elements in soils [7]. Generally, trace metal elements in soils are fractionated operationally by sequential extraction procedures into water-soluble plus exchangeable, specifically adsorbed on soil particle surfaces, and fractions bound to carbonates, Fe(Al)-Mn oxides, organic matter, and primary and layer silicates [8]. In the sequential extraction procedures, a chelating reagent, ethylenediaminetetraacetic acid (EDTA), was used to remove the specifically adsorbed (inner-sphere surface complexes) fraction [8,9,10]. Also, an EDTA extraction protocol for trace metal elements in soils and certified reference soil materials were developed by the Measurements and Testing Programme (formerly BCR) of the European Commission [11,12]. When comparing EDTA extractability of Zn and Mn with two sequential extraction procedures (modified Tessier [13] and proposed by the Community Bureau of Reference (BCR) [14]), it was found that in the term of phytoavailability predictions the sequential extractions are not better than EDTA single extraction [15]. For different fractions of trace metal elements in soils, the water-soluble plus exchangeable fractions are considered to be readily available; the specific adsorbed fraction, which can be extracted by powerful chelating reagents, can be potentially available; and fractions bound to carbonates, Fe(Al)-Mn oxides, organic matter and minerals are generally hard to be taken up by plants [10,16]. Therefore, the fractions of water-soluble, exchangeable and specifically adsorbed trace metal elements in soils may be important to their availability/toxicity, especially for risk assessment in contaminated soils.
Single chemical extraction methods are most commonly used to evaluate the availability of trace metal elements [10,17,18]. The major extractants for estimating availability/toxicity presently in use are unbuffered and buffered salt solutions, dilute acidic solutions, and solutions containing chelating agents [18]. Diluted acidic solutions, such as 0.43 mol/L HNO3 (ISO 17586-2016) [19] has been used, which partially dissolve trace metal elements associated with different fractions such as exchangeable, carbonates, iron and manganese oxides and organic matter [20]. However, there are some limitations for diluted acidic solutions in high calcareous soils. The salt solutions, such as 0.01 mol/L CaCl2, 1 mol/L NH4NO3, were also used to extract exchangeable trace metal elements in soils. The exchangeable fraction includes weakly adsorbed elements retained on soil solid surfaces by relatively weak electrostatic interactions, elements that can be released by ion-exchange processes and elements that can be coprecipitated with carbonates [18]. The powerful chelating agents, such as diethylenetriamine pentaacetic acid (DTPA) and EDTA, have been used widely in soil extractions for predicting the availability/toxicity of trace metal elements in soils. Quevauviller [21,22] reported that 0.005 mol/L DTPA (pH 7.3) was less suitable for Cr and Ni than other metals and EDTA was found to be the method of preference. Echevarria et al. [23] found that Ni in the plant of red clover came from the pool of the isotopically exchangeable Ni in soil, and in their experimental conditions, DTPA extracted mainly the isotopically exchangeable Ni. However, some of the Ni chelated by EDTA was not fully isotopically exchangeable [24]. Gupta and Sinha [25] studied the heavy metal accumulation in sesame using different single extractants such as EDTA, DTPA, NH4NO3, CaCl2 and NaNO3 in soil amended with sludge. Accordingly, among this group of reagents, EDTA extraction is widely used to quantify the labile pool or to predict the available pool [26]. However, because of the competition with soil constituents (e.g., organic matter), a single-step EDTA extraction procedure may underestimate the potentially toxicity of trace metal elements in soil, multiple sequential EDTA extractions may be a suitable method in the assessment of their potential ecological risk.
In the present study, the toxicity of added water-soluble nickel (Ni) to barley root elongation was studied in 17 representative Chinese soil samples with and without leaching by artificial rainwater. The extractability of Ni in soil samples were analyzed with three sequential EDTA extractions. The aims of the study were to develop a method for soil risk assessment based on the extractability of trace metal elements in soils instead of total metal concentrations. In particular, we sought to (1) determine the relationships between EDTA extractable Ni, total added Ni and soil properties; and (2) develop quantitative relationships between soil properties and toxicity thresholds based on EDTA extractable Ni in soils.
2. Materials and Methods
2.1. Soil Samples and Treaments
Seventeen soils (0–20 cm depth) from multiple locations in China were chosen as representatives of major Chinese soil types. The soil pH and organic matter content distributions were consistent with agricultural soils in China. The soil property ranges (Table 1) were as follows: pH 4.93 to 8.90; organic carbon content (OC) 0.60% to 4.28%; cation exchange capacity (CEC) 6.36 cmol+/kg to 33.59 cmol+/kg, and clay content 10% to 66%.

Table 1.
Main properties of the soils used in this study.
Air-dried soil samples (<2 mm) were spiked with NiCl2 solution and each soil sample set consisted of eight added Ni concentration treatments. Soils with pHs < 5, from 5 to 7, and >7 were spiked at the rates of 12.8–800, 25–1600, and 37.5–2400 mg/kg Ni, respectively. After equilibrating at 100% maximum water-holding capacity for 2 d, the spiked soils were left to air-dry and then were sieved again through a 2-mm plastic mesh.
Half of the spiked soil samples were leached by artificial rainwater [27] to overcome potential salinity effects and to simulate natural precipitation. This treatment reduced the difference in Ni speciation between laboratory-treated and field-aged soils. The artificial rainwater used for leaching consisted of 5 × 10−4 M calcium chloride, 5 × 10−4 M calcium nitrate, 5 × 10−4 M magnesium chloride, 10−4 M sodium sulfate, and 10−4 M potassium chloride at pH 5.9. Details about the leaching treatment can be found in Li et al. [28]. The leached soil samples were also air-dried and sieved through a 2-mm plastic mesh before use. The total Ni concentrations in the unleached and leached soils were taken from Li et al. [28], and were obtained using atomic absorption spectroscopy (ZEEnit 700, Analytik Jena, Jena, Germany) according to Zarcinas et al. [29].
2.2. Extraction of Added Ni in Soils
Soil extraction was carried out using EDTA, which is one of the chemical reagents widely used in fractionation and single extraction procedures, 0.05 mol/L EDTA extraction is widely used to quantify the labile pool or to predict the available pool [7,25,26] The soil samples (5 g each, air-dried, <2 mm) were combined with 25 mL of 0.05 mol/L EDTA in 50 mL polypropylene centrifuge tubes and shaken for 2 h at room temperature (~20 °C) on a reciprocating shaker. The soil suspension was centrifuged at about 4000 r/min for 20 min, after which the supernatants were passed through a syringe filter with a pore size of 0.45 μm. Another 25 mL EDTA was then added to the residue to continue the extraction. The extraction step was repeated three times over 6 h which resulted in stable Ni levels in the samples. The EDTA extractable Ni concentrations in each extraction were measured by using atomic absorption spectroscopy (ZEEnit 700; Analytik Jena, Jena, Germany). There were two replicates for each treatment.
2.3. Barley Root Elongation Assay
The barley root elongation assay was performed according to ISO 11269-1 [30]. Detailed procedures for the barley root elongation assay and barley root elongation data acquisition can be found in Li et al. [28]. The barley root elongation percentage with respect to the controls (RE, %) in a test medium was calculated using the equation:
where REt is root length in the test medium and REc is root length in the control, which had not been spiked with Ni.
2.4. Statistical Analysis
The dose-response data were fitted by a log-logistic curve [31]
where Y is relative barley root elongation (%), X is the measured EDTA extractable Ni (mg/kg) after log10 transformation, M is ECx (effective concentration of EDTA extractable Ni that decreases barley root elongation at EC10, EC20, or EC50 after log10 transformation, and Y0 and b are curve fitting parameters.
The EC10, EC20, and EC50 values were based on the measured EDTA extractable Ni concentrations in soils and were estimated from the respective log-logistic dose-response curves. The 95% confidence intervals (95% CI) for the EC10, EC20, and EC50 values were calculated according to Haanstra et al. [31], and were obtained from the fitted curve parameters data.
A stimulation response that occurs at low doses, but is inhibited at higher doses is defined as hormesis, and was modeled according to Schabenberger et al. [39] using Tablecurve 2D V5.01 (Systst Software Inc, San Jose, CA, USA). The EC10, EC20, and EC50 values, with their respective 95% CI, were determined as follows:
where Y is barley root elongation; X is the measured EDTA extractable Ni concentration; a, b, c, and d, are curve fitting parameters; and k is a variable that is related to the effective concentrations (e.g., EC10 and EC50). Further details about the hormesis analysis can be found in Guo et al. [40] and Li et al. [28]. When k equals 10, 20, or 50, the parameter c is defined as the EC10, EC20, or EC50 value, respectively, and its confidence interval can be calculated by Tablecurve 2D V5.01. The significance level for hormesis is detected by parameter b and the hormesis response is considered to be significant when the 95% confidence intervals of parameter b are above zero.
Stepwise multiple linear regressions were calculated using SPSS 19.0 for Windows (SPSS, Chicago, IL, USA) and were used to examine the relationship between toxicity thresholds (EC10, EC20, and EC50). They were based on the measured EDTA extractable Ni concentrations and soil property parameters, which were only log transformed if the normality and homogeneity of variance tests suggested they should be. Relationships were deemed significant at p ≤ 0.05.
3. Results and Discussion
3.1. Toxicity Thresholds for Nickel in Soils Based on EDTA Extractions
The EC10, EC20 and EC50 values for the unleached soils varied from 3 (Haikou soil) to 426 mg/kg (Hangzhou soil), from 6 (Haikou soil) to 510 mg/kg (Hangzhou soil); and from 22 (Haikou soil) to 722 mg/kg (Lingshan soil), respectively, which represents 142-, 85-, and 33-fold variations among the soils when one single EDTA extraction was used. The EC10, EC20 and EC50 values for the leached soils varied from 2 to 789 mg/kg, from 4 to 839 mg/kg, and from 13 to 1013 mg/kg, respectively, which represents 395-, 210-, and 78-fold variations, respectively, among the soils when one single EDTA extraction was used. The wide variations in EC10, EC20 and EC50 values indicated that the EDTA extraction could not accurately predict Ni toxicity in soils. Therefore, the soil properties have to be taken into account when the EDTA extraction method is used to predict Ni toxicity in soils.
Hormesis was observed in non-leached Chongqing soil and leached Guangzhou soil. One of the EC10 values, three of the EC20 values, and six of the EC50 values in leached calcareous soils (pH ≥ 8.2) could not be calculated because the leaching reduced or eliminated the toxicity caused by the added Ni in the soils (Table 2). The concentrations of one single EDTA extractable Ni in these calcareous soils ranged from 2 to 732 mg/kg. However, there was no Ni toxicity in the soils after leaching. Similar results were also found by Li et al. [28] when the ECx values were expressed as the total Ni added to these calcareous soils.

Table 2.
Toxicity thresholds based on the concentrations of a single EDTA extractable Ni and barley root elongation in unleached and leached soils.
The toxicity thresholds at the three inhibition levels for barley root elongation estimated by three sequential EDTA extractions in unleached and leached soils are listed in Table 3. The EC10, EC20, and EC50 values increased by 83%, 103%, and 46%, respectively, in leached soils compared with the results with one single EDTA extraction, and by 136%, 151%, and 185% in unleached soils, respectively. Similarly wide variations in ECx values were also found in other studies when three sequential EDTA extractions were used. The orders for the ECx values in this study were EC10 (103-fold) > EC20 (60-fold) > EC50 (22-fold) for unleached soils and EC10 (335-fold) > EC20 (142-fold) > EC50 (65-fold) for leached soils.

Table 3.
Toxicity thresholds based on the Ni concentrations of three EDTA extractions and barley root elongation in unleached and leached soils.
3.2. The Influence of Leaching on Ni Toxicity
In leached soils, there was a significant (p ≤ 0.05) decrease in toxicity of a single EDTA extractable Ni in six soils (35%) at the EC10 level, 10 soils (59%) at the EC20 level, and 13 soils (76%) at the EC50 level; when three sequential EDTA extractions were used, there was also a decrease in toxicity of the three EDTA extractable Ni in five soils (29%) at the EC10 level, eight soils (47%) at the EC20 level, and 10 soils (59%) at the EC50 level (Table 2 and Table 3). The leaching effects on decrease in Ni toxicity depended on the soil properties. Leaching had a comparatively stronger influence on alkaline soils than on acidic and neutral soils (Table 2 and Table 3).
The ECx values based on the single and three EDTA extractions, the leaching treatment decreased the toxicity of soluble metal salts in soils. Oorts et al. [27] showed the Ni toxicity effects on soil microbial processes were decreased by leaching to a greater extent in an alkaline soil (pH 7.6) than that in acidic and neutral soils (pH 4.5–6.1) when the ECx values are expressed as total added Ni in soils. Furthermore, Li et al. [28] also found that the greatest decrease in Ni toxicity to barley root elongation induced by leaching occurred in soils where the pH ≥ 8.2, which was consistent with the results produced in this study. It has been suggested that leaching alleviates or eliminates Ni toxicity in soils by reducing the pH and salinity [28]. Jiang et al. [41] reported that when the pH increased by 1, Ni phytotoxicity decreased 2.60-fold (1.86–3.72) and that this was due to the formation of Ni precipitates on solid surfaces. Furthermore, the maximum added Ni concentration (3200 mg/kg) in field soils from Dezhou, Shandong did not lead to a significant decrease in wheat growth. However, leaching decreased Ni toxicity when the EC values were expressed as EDTA extractable Ni in soils. That is to say, EDTA extractable Ni represents potential toxicity. It is not an indicator of toxicity. When EDTA extractable Ni is selected as a risk assessment criterion, the effects of soil properties cannot be ignored.
3.3. The Relationships between EDTA Extractable and Total Ni as Affected by Soil Properties
The extractability of Ni added to soils by EDTA was expressed as the proportion (%) extracted relative to the total Ni added to the soils. One single EDTA extraction can extract about 46% of the total Ni added to the leached and unleached soils; and three EDTA extractions can extract about 70–74% on average.
Linear regressions were used to study the relationships between EDTA extractable Ni and total Ni added to soils as affected by soil properties. The total Ni added to soils segregated into two ranges: 0–300 mg/kg and 0–2000 mg/kg (Table 4). The relationships between EDTA extractable Ni and soil properties, such as pH, OC, and CEC, could be good predictors of total Ni added to soils. When the total Ni concentration added to soils was under 300 mg/kg, Ni and pH and one EDTA extraction explained 48% to 65% of the variance in total Ni added to the unleached and leached soils. Also, Extractable Ni with three EDTA extractions and pH could predict the total Ni in soils with an R2 = 0.73 for unleached soils and an R2 = 0.78 for leached soils. When the total Ni concentration added to soils was 2000 mg/kg, extractable Ni with three EDTA extractions and pH could predict total Ni in soils with an R2 = 0.83 for unleached soils and an R2 = 0.77 for leached soils. Incorporating OC or CEC into the regression models for unleached and leached soils led to a small or no improvement in predictability for total Ni added to soils when one and three EDTA extractions were used. The predictive ability of the linear regression equations based on three EDTA extractions is better than that based on one single EDTA extraction for unleached and leached soils. The measured total Ni and the predicted total Ni are shown in Figure 1. The high correlations found between total Ni added to soils, EDTA extractable Ni, and soil properties indicates that EDTA extractable Ni can be used as an alternative to the total Ni added to soils, which means that the disadvantages associated with using total Ni as a risk assessment criterion can be overcome.

Table 4.
Linear regressions between EDTA extractable Ni and total Ni added to soils as affected by soil properties.
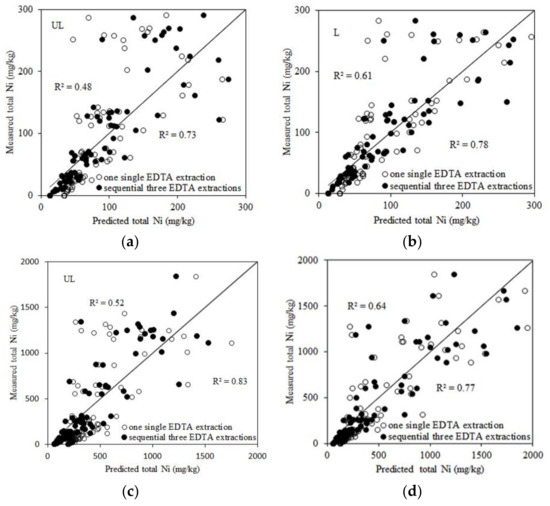
Figure 1.
The relationships between the measured and predicted total Ni. (a) calculated from Equations (1) and (3) in Table 4; (b) from Equations (5) and (7) in Table 4; (c) from Equations (9) and (11) in Table 4; and (d) from Equations (13) and (15) in Table 4. UL and L represent unleached and leached soils, respectively.
The toxicity of metals in soils depends on many factors, including their speciation, pH, organic matter content, cation exchange capacity, and soil texture [42]. In this study, soil pH was the most important factor affecting the relationships between EDTA extractable Ni and the amounts that were actually added to the soils. Cui et al. [43] also reported that pH was negatively correlated with the concentrations of leachable, available, and bioaccessible copper (Cu) and cadmium (Cd). This was probably because soil pH is one of the most important factors controlling the sorption and mobility of trace metal elements in soils [44,45]. The results from this study showed that three EDTA extractions improved the accuracy and stability of the prediction models for EDTA extractable Ni in soils. Furthermore, EDTA extractable Ni in soils better expresses the trace metal elements toxicity than total Ni.
3.4. Toxicity Thresholds Based on EDTA Extractable Ni in Soils as a Function of Soil Properties
Simple and multiple stepwise regressions were carried out between Ni toxicity thresholds (EC10, EC20, and EC50 values-based EDTA extractable Ni) and soil properties (Table 5 and Table 6). Although there is generally less statistical uncertainty for EC50 than EC10 and EC20 [46], the effects of soil properties on EC10 and EC20 values are listed in Table 5 and Table 6 because they are important when deciding risk assessment and ecological criteria for Ni in soils.

Table 5.
Simple and multiple linear regressions between Ni toxicity thresholds based on the Ni concentrations (mg/kg) of one EDTA extraction and selected soil properties.

Table 6.
Simple and multiple linear regressions between Ni toxicity thresholds based on extractable Ni concentrations (mg/kg) of three EDTA extractions and selected soil properties.
The results with one EDTA extraction in unleached soils showed that when CEC or OC were used as the single factor in the regression models, CEC was found to explain 33% of the variance in the EC10 values, and OC could explain 32% to 36% of the variance in the EC20 and EC50 values. However, when two factors were used, such as CEC and citrate dithionate extractable (CD)Mn or OC and CDAl in the regression models, the predictability of the regression models improved significantly, with R2 = 0.54 for EC10 and R2 = 0.66 for EC50. Furthermore, when OC, CDAl, and OXFe (oxalate extractable Fe) were incorporated into the regression models, the multiple linear regressions were further improved, with R2 = 0.72 for EC20. For leached soils, CDFe was found to be the best single factor for predicting Ni toxicity at the EC10 (R2 = 0.42), EC20 (R2 = 0.64), and EC50 (R2 = 0.73) levels. Fe, Al, and Mn oxides, CaCO3, and pH factors could also improve the models. Furthermore, when four factors (Fe, Al and Mn oxides, and CaCO3) were incorporated, the R2 for the regression model was 0.99 for the EC50 values.
When the soil toxicity thresholds were based on extractable Ni with three EDTA extractions, CDFe was the most important single factor in predicting Ni toxicity in the unleached and leached soils. For unleached soils, CDFe as the single factor could explain 39% of the variance in the EC10 values, 47% of the variance in the EC20 values, and 62% of the variance in the EC50 values. Incorporating OC into the regression models for unleached soils gave a significant, but slightly smaller, improvement in predictability for the EC50 and EC20 thresholds. When three factors (CDFe, OC, and OXFe) were introduced into the regression models, the predictability of the regression models improved, with R2 = 0.82 for EC10 and R2 = 0.86 for EC20. For leached soils, CDFe as the single factor could explain 52% of the variance in the EC10 values, 74% of the variance in the EC20 values and 75% of the variance in the EC50 values. Factors such as OXFe, CDMn, and OXAl, improved the predictability of the regression models. When four factors (CDFe, OXFe, CDMn, and OXAl) were incorporated, the regression models had an R2 value of = 0.98 for the EC50 thresholds.
When the different regression equations were compared (Table 5 and Table 6), it was found that pH occasionally affected the toxicity thresholds expressed as extractable Ni based on one EDTA extraction, but that this effect disappeared when three EDTA extractions were used; this suggests that the soil constituents are more important than soil pH. However, whether one or three extractions were used, the EDTA extractable Ni as an indicator still needs to consider the effects of soil properties probably because most of EDTA extractable Ni is potentially available, not instantly available, to plants. The adjusted coefficients of determination for the EC50 predictive equations were higher than for the EC10 and EC20 predictive equations, which was probably due to the higher statistical uncertainty of the EC10 and EC20 values. Furthermore, the adjusted coefficients of determination for the unleached soil EC50 predictive equations were lower than those for the leached soils, which could be due to the high salinity and low pH induced by the addition of water soluble Ni salts to the soil.
3.5. Applications in Risk Assessment and Ecological Criteria Setting
Current soil quality criteria for trace metals are based on total metal concentrations, regardless of their availability and/or extractability in soils with high trace metal background values or their trace metal sources. Therefore, they do not assess the potential toxicity of trace metals and could lead to either underestimations or overestimations of trace metal pollution.
In this study, the relationships between total Ni added to soils and EDTA extractable Ni as affected by soil properties were set up. Furthermore, the regression models, based on EDTA extractions as a function of soil properties, accurately predicted Ni toxicity. These results provide a basis for establishing soil environmental quality criteria based on EDTA extractable Ni. Further study on the establishment of soil environmental quality criteria based on EDTA extractable Ni will improve the accuracy of Ni ecological risk assessments.
4. Conclusions
In this study, EDTA extractable Ni was significantly correlated with the total Ni added to soils, and soil pH was the most important factor affecting the relationships between EDTA extractable Ni and the actual Ni added to the soils. Three sequential EDTA extractions improved the prediction accuracy and stability of toxicity models based on EDTA extractions. The regression models for toxicity thresholds based on EDTA extractable Ni against soil properties showed that soil citrate dithionate extractable Fe was more important than soil pH when attempting to predict Ni toxicity in soils. These results showed that EDTA extractable Ni was better at assessing potential Ni toxicity in soils than the total Ni in a soil. Determining the extractability of soil trace metal elements is a key process in ecological risk assessment. Therefore, the creation of an extraction procedure that can determine the toxicity of soil trace metal elements will improve the accuracy of ecological risk assessments.
Acknowledgments
The authors would like to thank the “Research on Migration/Transformation and Safety Threshold of Heavy Metals in Farmland Systems” project (2016YFD0800406) and the Special Fund for Environmental Protection Scientific Research in the Public Interest (Project No. 201509032) for their financial support. The authors would also like to thank all the staff who managed the field experiments.
Author Contributions
Yibing Ma conceived and designed the experiments, and edited the manuscript; Guangyun Zhu, Bao Jiang, and Guohang Yang performed the experiments; Guangyun Zhu and Bao Jiang analyzed the data; Jumei Li contributed the reagents/materials/analysis tools; Guangyun Zhu wrote the manuscript.
Conflicts of Interest
The authors declare no conflict of interest. The founding sponsors had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; and in the decision to publish the results.
References
- Janssen, C.R.; Schamphelaere, K.D.; Heijerick, D.; Muyssen, B.; Lock, K.; Bossuyt, B.; Vangheluwe, M.; Sprang, P.V. Uncertainties in the Environmental Risk Assessment of Metals. Hum. Ecol. Risk Assess. 2000, 6, 1003–1018. [Google Scholar] [CrossRef]
- Lu, L.; Liu, G.; Wang, J.; Wu, Y. Bioavailability and mobility of heavy metals in soil in vicinity of a coal mine from Huaibei, China. Hum. Ecol. Risk Assess. 2017, 23, 1164–1177. [Google Scholar] [CrossRef]
- Wang, X.Q.; Wei, D.P.; Ma, Y.B.; Mclaughlin, M.J. Soil ecological criteria for nickel as a function of soil properties. Environ. Sci. Pollut. Res. 2018, 25, 2137–2146. [Google Scholar] [CrossRef] [PubMed]
- Smolders, E.; Oorts, K.; Sprang, P.V.; Schoeters, I.; Janssen, C.R.; Mcgrath, S.P.; Mclaughlin, M.J. Toxicity of trace metals in soil as affected by soil type and aging after contamination: Using calibrated bioavailability models to set ecological soil standards. Environ. Toxicol. Chem. 2009, 28, 1663–1670. [Google Scholar] [CrossRef] [PubMed]
- Weng, L.P.; Wolthoorn, A.; Lexmond, T.M.; Temminghoff, E.J.; van Riemsdijk, W.H. Understanding the effects of soil characteristics on phytotoxicity and bioavailability of nickel using speciation models. Environ. Sci. Technol. 2004, 38, 156–162. [Google Scholar] [CrossRef] [PubMed]
- Ren, X.; Zeng, G.; Tang, L.; Wang, J.; Wan, J.; Liu, Y.; Yu, J.; Yi, H.; Ye, S.; Deng, R. Sorption, transport and biodegradation—An insight into bioavailability of persistent organic pollutants in soil. Sci. Total. Environ. 2017, 610–611, 1154–1163. [Google Scholar] [CrossRef] [PubMed]
- Rao, C.R.M.; Sahuquillo, A.; Sanchez, J.F.L. A Review of the Different Methods Applied in Environmental Geochemistry For Single and Sequential Extraction of Trace Elements in Soils and Related Materials. Water Air Soil Pollut. 2008, 189, 291–333. [Google Scholar] [CrossRef]
- Ma, Y.B.; Uren, N.C. The fate and transformations of zinc added to soils. Soil Res. 1997, 35, 727–738. [Google Scholar] [CrossRef]
- Ma, Y.B.; Uren, N.C. Application of a new fractionation scheme for heavy metals in soils. Commun. Soil Sci. Plan Anal. 1995, 26, 3291–3303. [Google Scholar] [CrossRef]
- Krishnamurti, G.S.R. Chapter 20 Chemical methods for assessing contaminant bioavailability in soils. Dev. Soil Sci. 2008, 40, 495–520. [Google Scholar] [CrossRef]
- Quevauviller, P.; Rauret, G.; Rubio, R.; López-Sánchez, J.F.; Ure, A.; Bacon, J.; Muntau, H. Certified reference materials for the quality control of EDTA- and acetic acid-extractable contents of trace elements in sewage sludge amended soils (CRMs 483 and 484). Fresenius J. Anal. Chem. 1997, 357, 611–618. [Google Scholar] [CrossRef]
- Lilleengen, B.; Wibetoe, G. Graphite furnace atomic absorption spectrometry used for determination of total, EDTA and acetic acid extractable chromium and cobalt in soils. Anal. Bioanal. Chem. 2002, 372, 187–195. [Google Scholar] [CrossRef] [PubMed]
- Elliott, H.A.; Dempsey, B.A.; Maille, P.J. Content and fractionation of heavy metals in water treatment sludges. J. Environ. Qual. 1990, 19, 330–334. [Google Scholar] [CrossRef]
- Quevauviller, P.; Rauret, G.; López-Sánchez, J.F.; Rubio, R.; Ure, A.; Muntau, H. Certification of trace metal extractable contents in a sediment reference material (CRM 601) following a three-step sequential extraction procedure. Sci. Total Environ. 1997, 205, 223–234. [Google Scholar] [CrossRef]
- Alvarez, J.M.; Lopez-Valdivia, L.M.; Novillo, J.; Obrador, A.; Rico, M.I. Comparison of EDTA and sequential extraction tests for phytoavailability prediction of manganese and zinc in agricultural alkaline soils. Geoderma 2006, 132, 450–463. [Google Scholar] [CrossRef]
- Ma, Y.B.; Uren, N.C. Transformations of heavy metals added to soil—Application of a new sequential extraction procedure. Geoderma 1998, 84, 157–168. [Google Scholar] [CrossRef]
- Nannoni, F.; Protano, G. Chemical and biological methods to evaluate the availability of heavy metals in soils of the Siena urban area (Italy). Sci. Total Environ. 2016, 568, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Laing, G.D. Analysis and Fractionation of Trace Elements in Soils. In Trace Elements in Soils; Hooda, P.S., Ed.; Wiley-Blackwell: Chichester, UK, 2010; pp. 53–80. ISBN 9781405160377. [Google Scholar] [CrossRef]
- International Organization for Standardization (ISO). Soil Quality—Extraction of Trace Elements Using Dilute Nitric Acid; International Organization for Standardization (ISO): Geneva, Switzerland, 2016. [Google Scholar]
- Rauret, G. Extraction procedures for the determination of heavy metals in contaminated soil and sediment. Talanta 1998, 46, 449–455. [Google Scholar] [CrossRef]
- Quevauviller, P. Operationally defined extraction procedures for soil and sediment analysis. II. Certified reference materials. TrAC-Trend Anal. Chem. 1998, 17, 632–642. [Google Scholar] [CrossRef]
- Quevauviller, P. Operationally-defined extraction procedures for soil and sediment analysis. Part 3: New CRMs for trace-element extractable contents. TrAC-Trend Anal. Chem. 2002, 21, 774–785. [Google Scholar] [CrossRef]
- Echevarria, G.; Morel, J.L.; Fardeau, J.C.; Leclerccessac, E. Assessment of Phytoavailability of Nickel in Soils. J. Environ. Qual. 1998, 27, 1064–1070. [Google Scholar] [CrossRef]
- Nolan, A.L.; Ma, Y.B.; Lombi, E.; Mclaughlin, M.J. Speciation and isotopic exchangeability of nickel in soil solution. J. Environ. Qual. 2009, 38, 485–492. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.K.; Sinha, S. Chemical fractionation and heavy metal accumulation in the plant of Sesamum indicum (L.) var. T55 grown on soil amended with tannery sludge: Selection of single extractants. Chemosphere 2006, 64, 161–173. [Google Scholar] [CrossRef] [PubMed]
- Manouchehri, N.; Besancon, S.; Bermond, A. Major and trace metal extraction from soil by EDTA: Equilibrium and kinetic studies. Anal. Chim. Acta 2006, 559, 105–112. [Google Scholar] [CrossRef]
- Oorts, K.; Ghesquiere, U.E. Leaching and aging decrease nickel toxicity to soil microbial processes in soils freshly spiked with nickel chloride. Environ. Toxicol. Chem. 2007, 26, 1130–1138. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Zhang, H.T.; Ma, Y.B.; McLaughlin, M.J. Influences of soil properties and leaching on nickel toxicity to barley root elongation. Ecotoxcol. Environ. Saf. 2011, 74, 459–466. [Google Scholar] [CrossRef] [PubMed]
- Bernhard, A.Z.; Michael, J.M.; Michelle, K.S. The effect of acid digestion technique on the performance of nebulization systems used in inductively coupled plasma spectrometry. Commun. Soil Sci. Plant Anal. 1996, 27, 1331–1354. [Google Scholar] [CrossRef]
- International Organization for Standardization (ISO). Soil-Quality—Determination of Effects of Pollutants on Soil Flora—Part 1: Method for the Measuredment of Inhibition of Root Growth; International Organization for Standardization (ISO): Geneva, Switzerland, 1993. [Google Scholar]
- Haanstra, L.; Doelman, P.; Voshaar, J.H.O. The use of sigmoidal dose response curve in soil ecotoxicological research. Plant Soil. 1985, 84, 293–297. [Google Scholar] [CrossRef]
- Rayment, G.E.; Higginson, F.R. Australian Laboratory Handbook of Soil and Water Chemical Methods; Inkata Press Pty Ltd.: Melbourne, Australia, 1992; p. 330. ISBN 0909605688. [Google Scholar]
- Igor, M. Determination of carbon and nitrogen in samples of various soils by the dry combustion. Commun. Soil Sci. Plan Anal. 1997, 28, 1499–1511. [Google Scholar] [CrossRef]
- Sherrod, L.A.; Dunn, G.; Peterson, G.A.; Kolberg, R.L. Inorganic carbon analysis by modified pressure-calcimeter method. Soil Sci. Soc. Am. J. 2002, 66, 299–305. [Google Scholar] [CrossRef]
- Schwertmann, U. The differentiation of iron oxides in soils by a photochemical extraction with acid ammonium oxalate. J. Plant Nutr. Soil Sci. 1964, 105, 194–201. [Google Scholar] [CrossRef]
- McKeague, J.A.; Day, J.H. Differenzierung- and oxalate-extractable Fe and Al as aids in differentiating various classes of soils. Can. J. Soil Sci. 1966, 46, 13–22. [Google Scholar] [CrossRef]
- Mehra, O.P.; Jackson, M.L. Iron oxide removal from soils and clays by a dithionite-citrate system buffered with sodium bicarbonate. Clay Clay Miner. 1960, 7, 317–327. [Google Scholar] [CrossRef]
- Jackson, M.L.; Lim, C.H.; Zelazny, L.W.; Klute, A. Oxides, hydroxides, and aluminosilicates. In Methods of Soil Analysis. Part 1. Physical and Mineralogical Methods; Klute, A., Ed.; American Society of Agronomy, Inc.: Madison, WI, USA, 1986; pp. 101–150. [Google Scholar]
- Schabenberger, O.; Tharp, B.E.; Kells, J.J.; Penner, D. Statistical Tests for Hormesis and Effective Dosages in Herbicide Dose Response. Agron. J. 1999, 91, 713–721. [Google Scholar] [CrossRef]
- Guo, X.Y.; Ma, Y.B. Advances in the effects, mechanisms and modeling of hormesis in terrestrial ecosystems. Acta Ecol. Sin. 2009, 29, 4408–4419. (In Chinese) [Google Scholar]
- Jiang, B.; Ma, Y.B.; Su, D.C.; Wang, X.Q.; Li, B.; Wang, X.D.; Guo, X.Y. Stronger phytotoxicity of nickel than copper in acidic soils but not in alkaline soils. J. Residuals Sci. Technol. 2017, 29, 835–842. [Google Scholar] [CrossRef]
- Violante, A.; Cozzolino, V.; Perelomov, L.; Caporale, A.G.; Pigna, M. Mobility and bioavailability of heavy metals and metalloids in soil environments. J. Plant Nutr. Soil. Sci. 2010, 10, 266–290. [Google Scholar] [CrossRef]
- Cui, H.; Fan, Y.; Fang, G.; Zhang, H.; Su, B.; Zhou, J. Leachability, availability and bioaccessibility of Cu and Cd in a contaminated soil treated with apatite, lime and charcoal: A five-year field experiment. Ecotoxcol. Environ. Saf. 2016, 134, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Azouzi, R.; Charef, A.; Hamzaoui, A.H. Assessment of effect of pH, temperature and organic matter on zinc mobility in a hydromorphic soil. Environ. Earth Sci. 2015, 74, 1–14. [Google Scholar] [CrossRef]
- Choppala, G.; Bolan, N.; Lamb, D.; Kunhikrishnan, A. Comparative Sorption and Mobility of Cr(III) and Cr(VI) Species in a Range of Soils: Implications to Bioavailability. Water Air Soil Pollut. 2013, 224, 1–12. [Google Scholar] [CrossRef]
- Li, B.; Zhang, H.; Ma, Y.B.; McLaughlin, M.J. Relationships between soil properties and toxicity of copper and nickel to bok choy and tomato in Chinese soils. Environ. Toxicol. Chem. 2013, 32, 2372–2378. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).