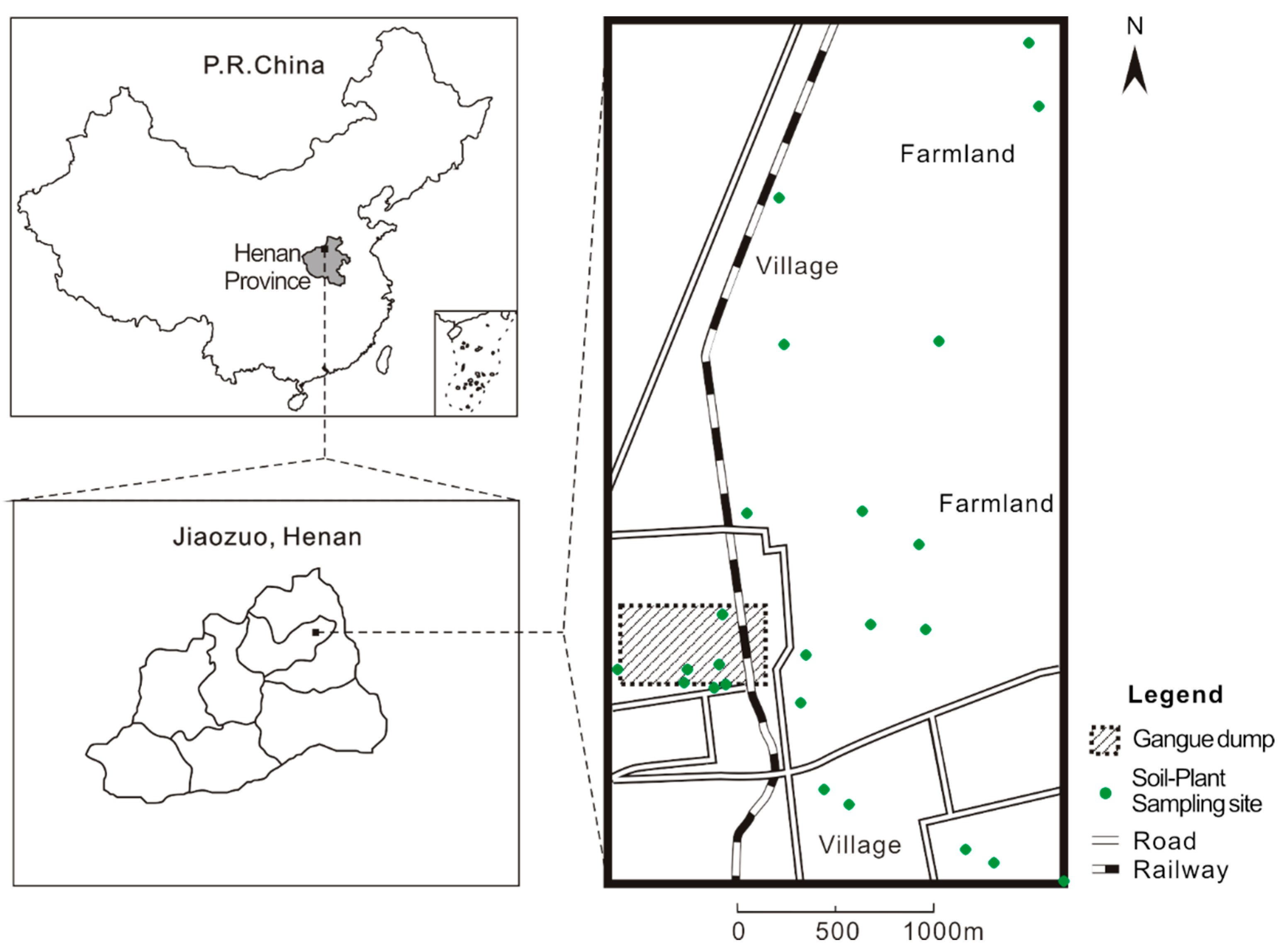
3.1. Soil HM Contamination
The concentration of the soil Cd, Pb and Zn exceeded the corresponding local soil HM background value (
Table 2). The Cd concentration varied from 0.17 to 1.38 mg·kg
−1. Five samples exceeded the level 1 China National Standard (CNS, GB15618-1995,
Table 1) and six samples exceeded level 2 CNS. Cd concentration of all samples were higher than Henan soil background. The Pb concentration was 19.25–91.13 mg·kg
−1. There are ten samples exceeded level 1 CNS and twelve exceeded Henan background [
30]. The highest Zn concentration was 172.38 ± 30.93 mg·kg
−1. Five sample exceeded level 1 CNS. The Cu and Cr concentration of all the samples were within level 1 CNS, while Cu concentration of several samples slightly exceeded the background value.
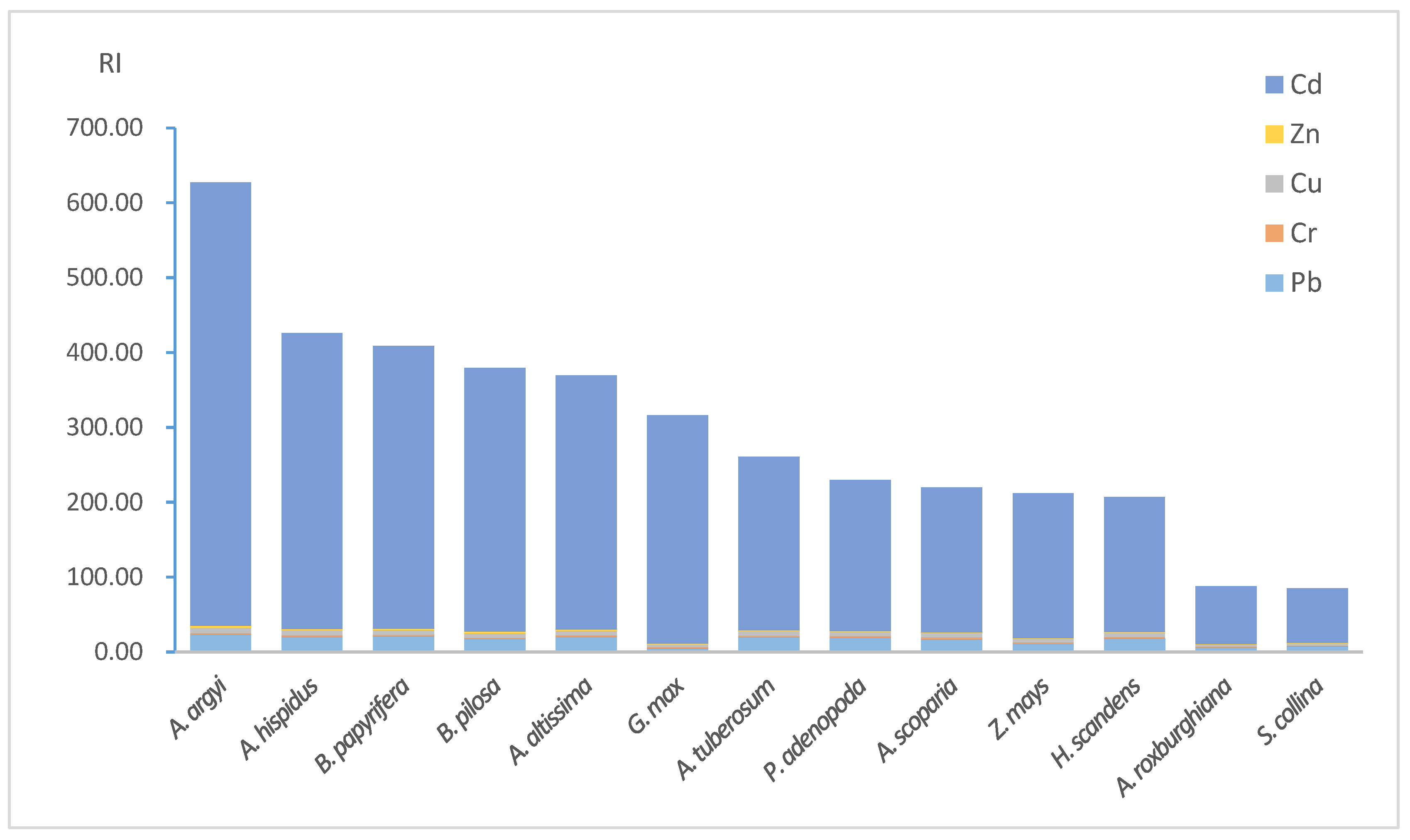
As shown in
Figure 2, the highest RI was up to 627.30. There were 1, 5, 5 and 2 samples for very high risk, considerable risk, moderate risk and low risk, respectively.
Figure 2 also shows that the contribution of Cd monomial risks varied from 85.48 percent to 96.48 percent with an average of 90.51 percent among all the samples. The results of potential ecological risk assessment indicated the gangue dump and the adjacent land was contaminated by Cd, Cu, Pb and Zn, and Cd contributed the most of the RI. Cd exposure could result in carcinogenicity, mutagenicity, and teratogenicity, endocrine disruption, biological calcium regulation interference, renal failure and chronic anemia [
31,
32,
33]. In addition, the reference dose (RfD) of oral soil digestion is provided by USEPA and Leung et al. [
34,
35], which is 0.001, 0.003, 0.04, 0.0035 and 0.3 mg·(d·kg
−1) for Cd, Cr, Cu, Pb and Zn. The RfD of Cd is much lower than those of the others. Also, some researchers stated that Cd is the HM of most concern because it is the “only metal that might pose human or animal health risks at plant tissue concentrations that are not generally phytotoxic” [
36]. Along with the potential ecological index assessment result, Cd should therefore be considered as the target element for the screening of native phytoextractors.
3.3. Screening for Native Phytoextractors
BCF is a more important measure than shoot metal concentration when considering the potential of a given candidate species for phytoextraction [
23]. As shown in
Table 5, the plant samples with BCF greater than 1 were mainly for Cd element. The highest one was in AG sample of
Artemisia roxburghiana Bess. (8.22), and the lowest one was in maize grain (0.27). Only a few samples had BCF higher than 1 for the other HM elements.
Accumulation and exclusion are two tolerance strategies evolved by plants to live in HM polluted medium [
42,
43,
44,
45]. HM accumulators are defined by the TF > 1, while root HM transport to shoot in excluders is restricted and TF < 1 [
43,
46,
47]. A TF value greater than 1 indicates the translocation of HM from root to AG parts [
48]. The translocation factors of the studied plant species were shown in
Table 6. The Cd TFs of
Artemisia roxburghiana Bess.,
Artemisia scoparia Waldst.
Et Kit.,
Bidens pilosa Linn., and
Artemisia argyi Lévl.
et Van. were higher than 1. They could be considered as Cd accumulators. The other ones behaved as Cd excluders, suggesting Cd immobilization in the roots.
The dominance of species is a combination of abundance and cover of plants, which indicates the biomass and adaptability in certain habitats. With the introduction of the habitat classification by the potential ecological risk assessment, the adaptability factor can reflect the plants’ vitality in the environment under different levels of HM stress. The adaptability factor could be a useful supplementary criterion other than BCF and TF for phytoextractor screening from a community ecological point of view, though it is a “black box” of the plants’ adaptability indication, and lacks quantitative tolerance study like the response of biomass, length, chlorophyll (CHL), superoxide dismutase (SOD), peroxidase (POD), malondialdehyde (MDA), and soluble protein (SP) within the plants under HM stress. Furthermore, DNA changes determined by random amplified polymorphic DNA (RAPD) could to be employed as a useful tool to identify the genotoxic effects caused by HMs within plant individuals [
49]. Therefore, the results obtained from the field survey should be validated with the methods mentioned above.
The previous study stated that adverse effects were observed in plant tissues stressed by high HM content, such as reduction of biomass, length and total protein contents [
50]. In this study, all plant species in
Table 2 did not show obvious symptom of phytotoxicity. They were divided into 4 categories (G1–G4) based on their dominance status and the pollution level of their habitats. (G1): the dominances of the plants were higher than 75% at CRS sites, which indicated high adaptability under intensive HM pollution stress. There were 5 species in this group:
Bidens pilosa Linn.,
Artemisia argyi Lévl.
et Van.,
Artemisia scoparia Waldst.
Et Kit.,
Humulus scandens (Lour.) Merr., and
Arthraxon hispidus (Trin.) Makino. (G2): the dominances were C (40–75%) at CRS sites.
Broussonetia papyrifera (Linn.) L'Hér.
ex Vent. belonged to this group. (G3) stood for D dominances status (>75%) at MRS sites, including
Salsola collina Pall.,
Artemisia roxburghiana Bess.,
Cynodon dactylon (Linn.) Pers.,
Setaria viridis (Linn.) Beauv., and
Eleusine indica (Linn.) Gaertn. These species had high adaptability under moderate HM stress. (G4) contained the rest of the plants, including the species of dominance status C at MRS sites and dominance status R.
Herbs are more promising for phytoextraction than shrubs and trees due to their high growth rate, high biomass, and more adaptability under stress [
51]. Therefore, the following herbs like
Bidens pilosa Linn.,
Artemisia argyi Lévl.
et Van.,
Artemisia scoparia Waldst.
Et Kit.,
Artemisia roxburghiana Bess., and
Arthraxon hispidus (Trin.) Makino were chosen to be discussed based on bioconcentration factors, translocation factors and adaptability factors analyzed above.
Bidens pilosa Linn. belonged to G1. The Cd content, BCF and TF was 1.16 ± 0.30 mg·kg
−1, 1.41, and 3.05, when the root soil concentration was 0.82 ± 0.57 mg·kg
−1. According to Yoon et al. [
10], plant species with both BCF and TF greater than 1 have the potential to be used for phytoextraction.
Bidens pilosa met this criterion in this study.
Bidens pilosa was considered as a hyperaccumulator under laboratory conditions in previous studies [
52]. The Cd concentration of AG was higher than 100 mg·kg
−1, the BF and TF values were all greater than 1.0, when soil Cd concentration was 8 and 16 mg·kg
−1 in the form of CdCl
2 solution. The roots were able to uptake HM from aqueous phase. However, soil HMs in the field were mainly insoluble and unavailable for uptake by plants due to strong binding of HM ions to soil particles and precipitation [
4]. This could explain the BCF difference between present study and the laboratory experiment. In addition, the biomass showed no significant reduction and the HM stress did not show any significant impact on CHL, SOD, POD, MDA, and SP, which showed high tolerance to Cd [
52]. The high adaptability at polluted sites, BCF, TF greater than 1, and the potential to be a hyperaccumulator suggested
Bidens pilosa Linn. could be used as a native phytoextractor for soil Cd phytoremediation.
In the previous study [
53], the AG content, BCF and TF of Cd in
Artemisia argyi Lévl.
et Van. were 35.5 ± 3.2 mg·kg
−1, 3.55 and 3.11 in the soil treated by 10 mg·kg
−1 for Cd (CdCl
2·2.5H
2O), 1000 mg·kg
−1 for Pb (Pb(CH
3COO)
2·3H
2O), 400 mg·kg
−1 for Cu (CuSO
4·5H
2O) and 1000 mg·kg
−1 for Zn (ZnSO
4·7H
2O). The biomass reduced significantly by about 50%. This suggested that
Artemisia argyi Lévl.
et Van. did not show hyperaccumulator characteristics and lacked strong tolerance under the high level of HM co-stress. In this study,
Artemisia argyi Lévl.
et Van. was classified as a G1 plant. Its Cd content, BCF and TF were 2.67 ± 0.69 mg·kg
−1, 1.93 and 1.78. The results showed it could be considered as a potential phytoextractor. The difference of accumulation performance could be explained by the lower root soil Cd content as well as the lower bioavailability caused by the difference between the natural soil and the soil-HM compound formulated in the laboratory.
Artemisia scoparia Waldst.
Et Kit. was reported having certain ability to extract HM from contaminated soil in previous research [
54]. Its AG content, BCF and TF of Cd were 3.33 ± 0.55 mg·kg
−1, 0.69 and 1.25 in the soil with Cd concentration of 4.76 ± 1.2 mg·kg
−1 at a mine-affect area, which did not meet the requirement for phytoextractor due to the low BCF. According to another investigation [
55],
Artemisia scoparia Waldst.
Et Kit. from different sites showed quite contradictory phytoextraction performances. The AG content, BCF and TF of the sample from site 3 were 4.02 ± 0.89 mg·kg
−1, 10.05 and 4.14, while the parameter from site 4 were 0.34 ± 0.12 mg·kg
−1, 1.31 and 0.79. The total soil Cd concentration were similar at two sites, which were 0.30 ± 0.04 for site 3 and 0.26 ± 0.05 mg·kg
−1 for site 4. The difference between DTPA extractable Cd (0.04 ± 0.004 for site 3 and 0.01 ± 0.001 mg·kg
−1 for site 4) suggested that the phytoextraction characteristics of
Artemisia scoparia Waldst.
Et Kit. relied on the DTPA extractable Cd in the soil and might lack the ability to mobilize carbonate-bound and residual state Cd.
Artemisia scoparia Waldst.
Et Kit. was divided into G1 group in present study. Its AG content, BCF and TF were 2.62 ± 0.16 mg·kg
−1, 5.82 and 5.70, which had the potential as a phytoextractor on the sites with high bioavailable Cd content.
Artemisia roxburghiana Bess. belonged to G3 in this case. The soil pollution level was moderate: 23.96 ± 4.81, 12.25 ± 3.29, 52.55 ± 15.37 and 0.18 ± 0.06 mg·kg
−1 for Pb, Cu, Zn and Cd. The BCF and TF of AG for Pb, Cu, Zn and Cd was 0.27 and 4.11, 1.57 and 2.85, 1.67 and 4.48, 8.22 and 6.73, respectively. The lack of dominance at CRS sites might limit its remediation application. Lei and Duan [
56] found that
Artemisia roxburghiana Bess.’s BCF for Pb, Zn and Cu were less than 0.005, and the HM content remained a low level at very highly polluted sites (over 7000, 35,000 and 400 mg·kg
−1 for Pb, Zn and Cu), showing weak phytoremediation capacity. However,
Artemisia roxburghiana Bess. was also found to have the ability to reduce a considerable amount of Pb, Zn and Cu concentration and increase the soil fertility in rhizosphere simultaneously. Although
Artemisia roxburghiana Bess. was unfit for phytoextraction at considerable risk sites, it showed the potential to treat the soil at moderate risk sites due to the high BCF and TF as well as the ability of soil fertility improvement.
Arthraxon hispidus (Trin.) Makino was classified as one of G1 species in this study. The Cd content, BCF and TF of AG was 0.54 ± 0.05 mg·kg
−1, 0.59 and 0.39. Our results were in consistency with the previous experiment conducted in Cd(NO
3)
2 treated soil with 3.00 mg·kg
−1 of total Cd. The BCF and TF of AG was 4.17 and 0.95, also showing the exclusion characteristics [
57]. The BCF difference between the two study could be explained by high bioavailable Cd content due to the implementation of Cd(NO
3)
2 during the sample formulation. The exclusion strategic of
Arthraxon hispidus (Trin.) Makino suggested it had little potential for phytoextraction, but it had the potential to be used for phytostabilization at considerable risk sites.