Children’s Blood Lead Seasonality in Flint, Michigan (USA), and Soil-Sourced Lead Hazard Risks
Abstract
:1. Introduction
2. Background
2.1. Lead Contamination of Urban Soils
2.2. Association Between Soil Lead and Blood Lead
2.3. Atmospheric Lead and Soil Seasonality
2.4. Blood Lead Seasonality Studies
2.5. Lead in Drinking Water as a Source of Blood Lead Seasonality
2.6. Other Potential Sources of Blood Lead Seasonality
2.7. Blood Lead Levels in Michigan and the United States
3. Methods and Results
3.1. Quarterly Blood Lead Incidence Trend—Flint, Michigan
3.2. Soil Lead Data—Flint, Michigan
4. Discussion
Blood Lead Seasonality and Flint
5. Conclusions and Recommendations
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kovarik, W. Ethyl-leaded gasoline: How a classic occupational disease became an international public health disaster. Int. J. Occup. Environ. Health 2005, 11, 384–397. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, D.E. Lead-based paint as a major source of childhood lead poisoning: A review of the evidence. In Lead in Paint, Soil and Dust: Health Risks, Exposure Studies, Control Measures, Measurement Methods, and Quality Assurance; ASTM International: West Conshohocken, PA, USA, 1995. [Google Scholar]
- Troesken, W. Lead water pipes and infant mortality at the turn of the twentieth century. J. Hum. Resour. 2008, 43, 553–575. [Google Scholar] [CrossRef]
- Candelone, J.P.; Hong, S.; Pellone, C.; Boutron, C.F. Post-Industrial Revolution changes in large-scale atmospheric pollution of the northern hemisphere by heavy metals as documented in central Greenland snow and ice. J. Geophys. Res. Atmos. 1995, 100, 16605–16616. [Google Scholar] [CrossRef]
- Manton, W.I.; Angle, C.R.; Stanek, K.L.; Reese, Y.R.; Kuehnemann, T.J. Acquisition and retention of lead by young children. Environ. Res. 2000, 82, 60–80. [Google Scholar] [CrossRef] [PubMed]
- United States Environmental Protection Agency. Child-Specific Exposure Factors Handbook (External Review Draft) 2006. Available online: http://cfpub.epa.gov/ncea/risk/recordisplay.cfm?deid=56747 (accessed on 5 February 2016).
- Roberts, J.R.; Roberts, J.; Reigart, J.R.; Ebeling, M.; Hulsey, T.C. Time required for blood lead levels to decline in nonchelated children. J. Toxicol. Clin. Toxicol. 2001, 39, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Nevin, R. How lead exposure relates to temporal changes in IQ, violent crime, and unwed pregnancy. Environ. Res. 2000, 83, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Nigg, J.T.; Knottnerus, G.M.; Martel, M.M.; Nikolas, M.; Cavanagh, K.; Karmaus, W.; Rappley, M.D. Low blood lead levels associated with clinically diagnosed attention-deficit/hyperactivity disorder and mediated by weak cognitive control. Biol. Psychiatry 2008, 63, 325–331. [Google Scholar] [CrossRef] [PubMed]
- Lucchini, R.; Aschner, M.; Bellinger, D.C.; Caito, S. Neurotoxicology of Metals. In Handbook on the Toxicology of Metals; Academic Press: San Diego, CA, USA, 2014; pp. 299–311. [Google Scholar]
- Needleman, H. Lead poisoning. Annu. Rev. Med. 2004, 55, 209–222. [Google Scholar] [CrossRef] [PubMed]
- United States Department of Health National Toxicology Program (USDH-NTP). Health Effects of Low-Level Lead Evaluation NTP Monograph on Health Effects of Low-Level Lead (June 2012). Available online: http://ntp.niehs.nih.gov/pubhealth/hat/noms/lead/index.html (accessed on 5 February 2016).
- United States Environmental Protection Agency (USEPA). Learn about Lead. Available online: http://www.epa.gov/lead/learn-about-lead#effects (accessed on 5 February 2016).
- Agency for Toxic Substances and Disease Registry (ATDSR). Lead Toxicity: What are the Physiological Effects of Lead Exposure? Available online: http://www.atsdr.cdc.gov/csem/csem.asp?csem=7&po=10 (accessed on 5 February 2016).
- Mielke, H.W.; Laidlaw, M.A.; Gonzales, C.R. Estimation of leaded (Pb) gasoline’s continuing material and health impacts on 90 US urbanized areas. Environ. Int. 2011, 37, 248–257. [Google Scholar] [CrossRef] [PubMed]
- Mielke, H.W.; Reagan, P.L. Soil is an important pathway of human lead exposure. Environ. Health Perspect. 1998, 106, 217. [Google Scholar] [CrossRef] [PubMed]
- Filippelli, G.M.; Laidlaw, M.A.; Latimer, J.C.; Raftis, R. Urban lead poisoning and medical geology: An unfinished story. GSA Today 2005, 15, 4–11. [Google Scholar] [CrossRef]
- Hunt, A.; Johnson, D.L. Suspension and re-suspension of dry soil indoors following track-in on footwear. Environ. Geochem. Health 2012, 34, 355–363. [Google Scholar] [CrossRef] [PubMed]
- Hunt, A.; Johnson, D.L.; Griffith, D.A. Mass transfer of soil indoors by track-in on footwear. Sci. Total Environ. 2006, 370, 360–371. [Google Scholar] [CrossRef] [PubMed]
- Johnson, D.L. A first generation dynamic ingress, redistribution and transport model of soil track-in: DIRT. Environ. Geochem. Health 2008, 30, 589–596. [Google Scholar] [CrossRef] [PubMed]
- Brunekreef, B.; Noy, D.; Biersteker, K.; Boleij, J. Blood lead levels of Dutch city children and their relationship to lead in the environment. J. Air Pollut. Control. Assoc. 1983, 33, 872–876. [Google Scholar] [CrossRef] [PubMed]
- Laidlaw, M.A.; Mielke, H.W.; Filippelli, G.M.; Johnson, D.L.; Gonzales, C.R. Seasonality and children’s blood lead levels: Developing a predictive model using climatic variables and blood lead data from Indianapolis, Indiana, Syracuse, New York, and New Orleans, Louisiana (USA). Environ. Health Perspect. 2005, 1, 793–800. [Google Scholar] [CrossRef]
- Laidlaw, M.A.; Zahran, S.; Mielke, H.W.; Taylor, M.P.; Filippelli, G.M. Re-suspension of lead contaminated urban soil as a dominant source of atmospheric lead in Birmingham, Chicago, Detroit and Pittsburgh, USA. Atmos. Environ. 2012, 49, 302–310. [Google Scholar] [CrossRef]
- Zahran, S.; Laidlaw, M.A.; McElmurry, S.P.; Filippelli, G.M.; Taylor, M. Linking source and effect: Resuspended soil lead, air lead, and children’s blood lead levels in Detroit, Michigan. Environ. Sci. Technol. 2013, 47, 2839–2845. [Google Scholar] [CrossRef] [PubMed]
- Laidlaw, M.A.; Zahran, S.; Pingitore, N.; Clague, J.; Devlin, G.; Taylor, M.P. Identification of lead sources in residential environments: Sydney Australia. Environ. Pollut. 2014, 184, 238–246. [Google Scholar] [CrossRef] [PubMed]
- Zahran, S.; Mielke, H.W.; McElmurry, S.P.; Filippelli, G.M.; Laidlaw, M.A.; Taylor, M.P. Determining the relative importance of soil sample locations to predict risk of child lead exposure. Environ. Int. 2013, 60, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Mielke, H.W.; Dugas, D.; Mielke, P.W.; Smith, K.S.; Smith, S.L.; Gonzales, C.R. Associations between lead dust contaminated soil and childhood blood lead: A case study of urban New Orleans and rural Lafourche Parish, Louisiana, USA. Environ. Health Perspect. 1997, 105, 950–954. [Google Scholar] [CrossRef] [PubMed]
- Bickel, M.J. Spatial and Temporal Relationships between Blood Lead and Soil Lead Concentrations in Detroit, Michigan. Master’s Thesis, Wayne State University, Detroit, MI, USA, 2010. Available online: http://digitalcommons.wayne.edu/cgi/viewcontent.cgi?article=1046&context=oa_theses (accessed on 5 February 2016). [Google Scholar]
- Johnson, D.L.; Bretsch, J.K. Soil lead and children’s blood lead levels in Syracuse, NY, USA. Environ. Geochem. Health 2002, 24, 375–385. [Google Scholar] [CrossRef]
- Davis, J.J.; Gulson, B.L. Ceiling (attic) dust: A “museum” of contamination and potential hazard. Environ. Res. 2005, 99, 177–194. [Google Scholar] [CrossRef] [PubMed]
- Laidlaw, M.A.; Taylor, M.P. Potential for childhood lead poisoning in the inner cities of Australia due to exposure to lead in soil dust. Environ. Pollut. 2011, 159, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Greene, N.A.; Morris, V.R. Assessment of public health risks associated with atmospheric exposure to PM2.5 in Washington, DC, USA. Int. J. Environ. Res. Public Health 2006, 3, 86–97. [Google Scholar] [CrossRef] [PubMed]
- Melaku, S.; Morris, V.; Raghavan, D.; Hosten, C. Seasonal variation of heavy metals in ambient air and precipitation at a single site in Washington, DC. Environ. Pollut. 2008, 155, 88–98. [Google Scholar] [CrossRef] [PubMed]
- United States Environmental Protection Agency (USEPA). Seasonal Rhythms of BLL Levels: Boston, 1979–1983: Final Report; EPA 747-R-94-003; U.S. Environmental Protection Agency: Washington, DC, USA, 1995.
- United States Environmental Protection Agency (USEPA). Seasonal Trends in BLL Levels in Milwaukee: Statistical Methodology; EPA 747-R-95-010; USEPA: Washington, DC, USA, 1996.
- Billick, I.H.; Curran, A.S.; Shier, D.R. Analysis of pediatric blood lead levels in New York City for 1970–1976. Environ. Health Perspect. 1979, 31, 83. [Google Scholar] [CrossRef]
- Yiin, L.M.; Rhoads, G.G.; Lioy, P.J. Seasonal influences on childhood lead exposure. Environ. Health Perspect. 2000, 108, 177–182. [Google Scholar] [CrossRef] [PubMed]
- Paode, R.D.; Sofuoglu, S.C.; Sivadechathep, J.; Noll, K.E.; Holsen, T.M.; Keeler, G.J. Dry deposition fluxes and mass size distributions of Pb, Cu, and Zn measured in southern Lake Michigan during AEOLOS. Environ. Sci. Technol. 1998, 32, 1629–1635. [Google Scholar] [CrossRef]
- Laidlaw, M.A.; Filippelli, G.M. Resuspension of urban soils as a persistent source of lead poisoning in children: A review and new directions. Appl. Geochem. 2008, 23, 2021–2039. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention (CDC). Morbidity and Mortality Weekly Report (MMWR) Childhood Blood Lead Levels—United States, 2007–2012. Available online: http://www.cdc.gov/mmwr/preview/mmwrhtml/mm6254a5.htm?s_cid=mm6254a5_x (accessed on 5 February 2016).
- Hunter, J.M. The summer disease: An integrative model of the seasonality aspects of childhood lead poisoning. Soc. Sci. Med. 1977, 11, 691–703. [Google Scholar] [CrossRef]
- Hunter, J.M. The summer disease: Some field evidence on seasonality in childhood lead poisoning. Soc. Sci. Med. 1978, 12, 85–94. [Google Scholar] [CrossRef]
- Havlena, J.; Kanarek, M.S.; Coons, M. Factors associated with the seasonality of blood lead levels among preschool Wisconsin children. Wis. Med. J. 2009, 108, 151. [Google Scholar]
- Rothenberg, S.J.; Williams, F.A., Jr.; Delrahim, S.; Khan, F.; Kraft, M.; Lu, M.; Manalo, M.; Sanchez, M.; Wooten, D.J. Blood lead levels in children in south central Los Angeles. Arch. Environ. Health. 1996, 51, 383–388. [Google Scholar] [CrossRef] [PubMed]
- Johnson, D.L.; McDade, K.; Griffith, D. Seasonal variation in paediatric blood lead levels in Syracuse, NY, USA. Environ. Geochem. Health 1996, 18, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Haley, V.B.; Talbot, T.O. Seasonality and trend in blood lead levels of New York State children. BMC Pediatr. 2004, 4, 1. [Google Scholar] [CrossRef] [PubMed]
- Interagency Monitoring of Protected Visual Environments. Available online: http://vista.cira.colostate.edu/improve/ (accessed on 25 February 2015).
- Gulson, B.; Taylor, A. Relationship of Pb in house dust and ambient air. Modern Chem. Appl. 2014, 2. [Google Scholar] [CrossRef]
- Schock, M.R. Causes of temporal variability of lead in domestic plumbing systems. Environ. Monitor. Assess. 1990, 15, 59–82. [Google Scholar] [CrossRef] [PubMed]
- Schaut, G.C. The Action of a Chlorinated Water Supply upon Lead Pipe. Am. J. Pharmacol. 1942, 114, 241–249. [Google Scholar]
- Moore, M.R. Plumbosolvency of waters. Nature 1973, 243, 222–223. [Google Scholar] [CrossRef]
- Gregory, R.; Jackson, P.J. Central Water Treatment to Reduce Lead Solubility. In Proceedings of the VIVA Annual Conference, Dallas, TX, USA, 24–27 June 1984.
- Cartier, C.; Laroche, L.; Deshommes, E.; Nour, S.; Richard, G.; Edwards, M.; Prévost, M. Investigating dissolved lead at the tap using various sampling protocols. Am. Water Works Assoc. J. 2011, 103, 55. [Google Scholar]
- Ngueta, G.; Prevost, M.; Deshommes, E.; Abdous, B.; Gauvin, D.; Levallois, P. Exposure of young children to household water lead in the Montreal area (Canada): The potential influence of winter-to-summer changes in water lead levels on children’s blood lead concentration. Environ. Int. 2014, 73, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Farfel, M.R.; Chisolm, J.J., Jr. Health and environmental outcomes of traditional and modified practices for abatement of residential lead-based paint. Am. J. Public Health 1990, 80, 1240–1245. [Google Scholar] [CrossRef] [PubMed]
- Jain, R.B. Trends and variability in blood lead concentrations among US children and adolescents. Environ. Sci. Pollut. Res. 2016, 1. [Google Scholar] [CrossRef] [PubMed]
- Michigan Department of Health and Human Services. Healthy Homes and Lead Poisoning Prevention: General Information. Available online: http://www.michigan.gov/mdhhs/0,5885,7-339-73971_4911_4913---,00.html (accessed on 25 February 2016).
- Filippelli, G.M.; Risch, M.; Laidlaw, M.A.; Nichols, D.E.; Crewe, J. Geochemical legacies and the future health of cities: A tale of two neurotoxins in urban soils. Elem. Sci. Anthr. 2015, 3, 000059. [Google Scholar] [CrossRef]
- Michigan Department of Environmental Quality (MDEQ). Michigan Department of Health and Human Services (MDHHS) Blood Lead Level Test Results for Flint Zip Codes 48501-48507, Genesee County, and the State of Michigan Summary as of 22 January 2016. Available online: http://www.michigan.gov/documents/flintwater/Flint_Blood_Testing_Report_12Jan22_512221_7.pdf (accessed on 5 February 2016).
- Hanna-Attisha, M.; LaChance, J.; Sadler, R.C.; Schnepp, A.C. Elevated blood lead levels in children associated with the Flint drinking water crisis: A spatial analysis of risk and public health response. Am. J. Public Health 2016, 106, 283–290. [Google Scholar] [CrossRef]
- WebPlotDigitizer: Web Based Tool to Extract Data from Plots, Images, and Maps. Available online: http://arohatgi.info/WebPlotDigitizer/ (accessed on 5 February 2016).
- Cade, B.S.; Richards, J.R. Blossom Statistical Package. Available online: https://www.fort.usgs.gov/products/23736 (accessed on 5 February 2016).
- Edible Flint Website. Available online: http://www.edibleflint.org/ (accessed on 5 February 2016).
- University of Massachusetts, Amherst. Soil Lead: Testing, Interpretation, & Recommendations. Available online: http://soiltest.umass.edu/sites/soiltest.umass.edu/files/fact-sheets/pdf/SPTTL_5%20Soil%20Lead.pdf (accessed on 5 February 2016).
- McBride, M.B.; Mathur, R.R.; Baker, L.L. Chemical extractability of lead in field-contaminated soils: Implications for estimating total lead. Commun. Soil Sci. Plant Anal. 2011, 42, 1581–1593. [Google Scholar]
- Minca, K.K.; Basta, N.T. Comparison of plant nutrient and environmental soil tests to predict Pb in urban soils. Sci. Total Environ. 2013, 445, 57–63. [Google Scholar]
- Mielke, H.W.; Gonzales, C.; Powell, E.; Mielke, P.W. Evolving from Reactive to Proactive Medicine: Community Lead (Pb) and Clinical Disparities in Pre- and Post-Katrina New Orleans. Int. J. Environ. Res. Public Health 2014, 11, 7482–7491. [Google Scholar]
- Mielke, H.W.; Covington, T.P.; Mielke, P.W.; Wolman, F.J.; Powell, E.T.; Gonzales, C.R. Soil intervention as a strategy for lead exposure prevention: The New Orleans lead-safe childcare playground project. Environ. Pollut. 2011, 159, 2071–2077. [Google Scholar] [CrossRef] [PubMed]
- Mielke, H.W. Nature and extent of metal-contaminated soils in urban environments (keynote talk). Environ. Geochem. Health 2016, 1, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Chambers, L.G.; Chin, Y.-U.; Filippelli, G.M.; Gardner, C.B.; Herndon, E.M.; Long, D.T.; Lyons, W.B.; Macpherson, G.L.; McElmurry, S.P.; McLean, C.E.; et al. Developing the scientific framework for urban geochemistry. Appl. Geochem. 2016, 67, 1–20. [Google Scholar] [CrossRef]
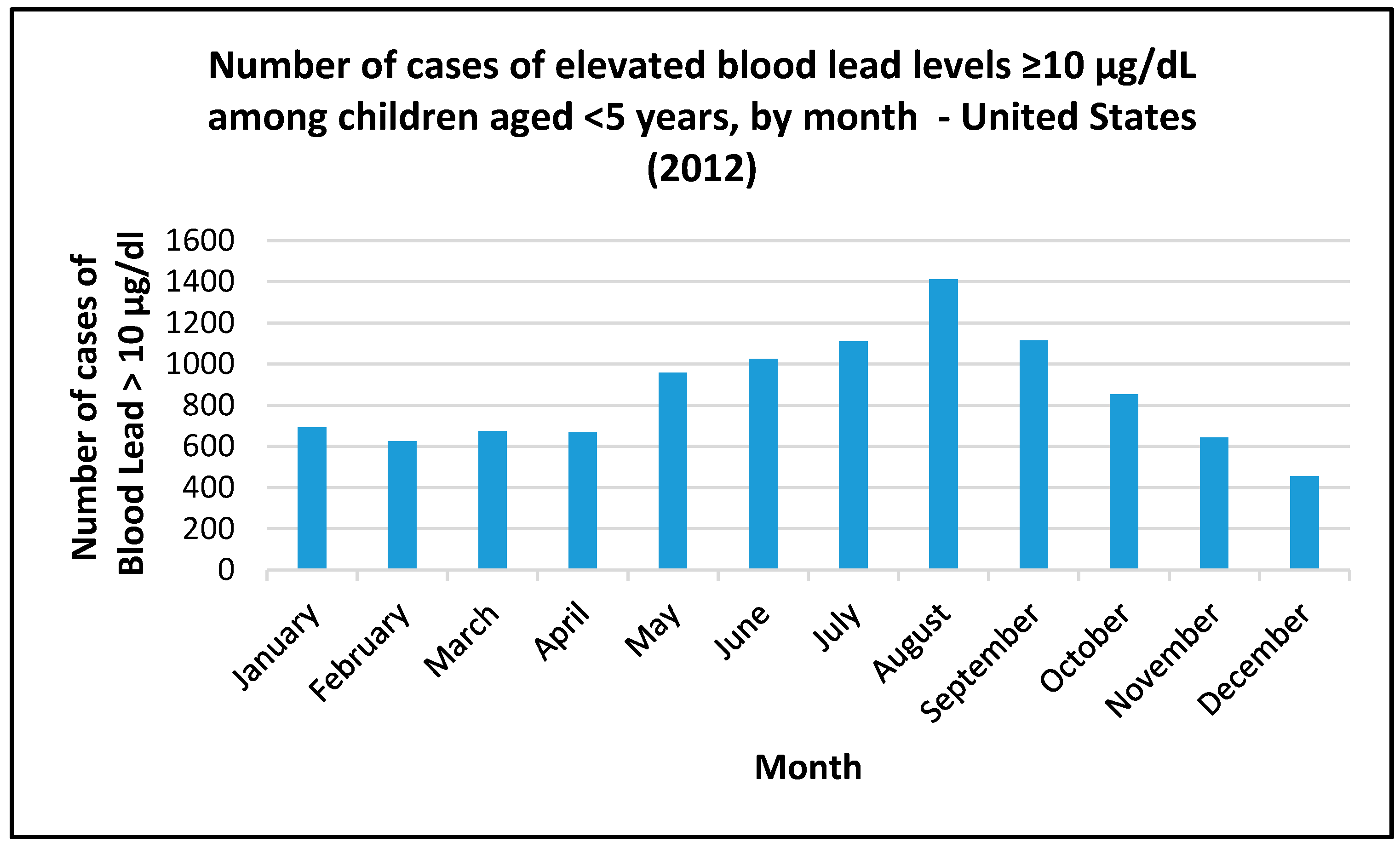

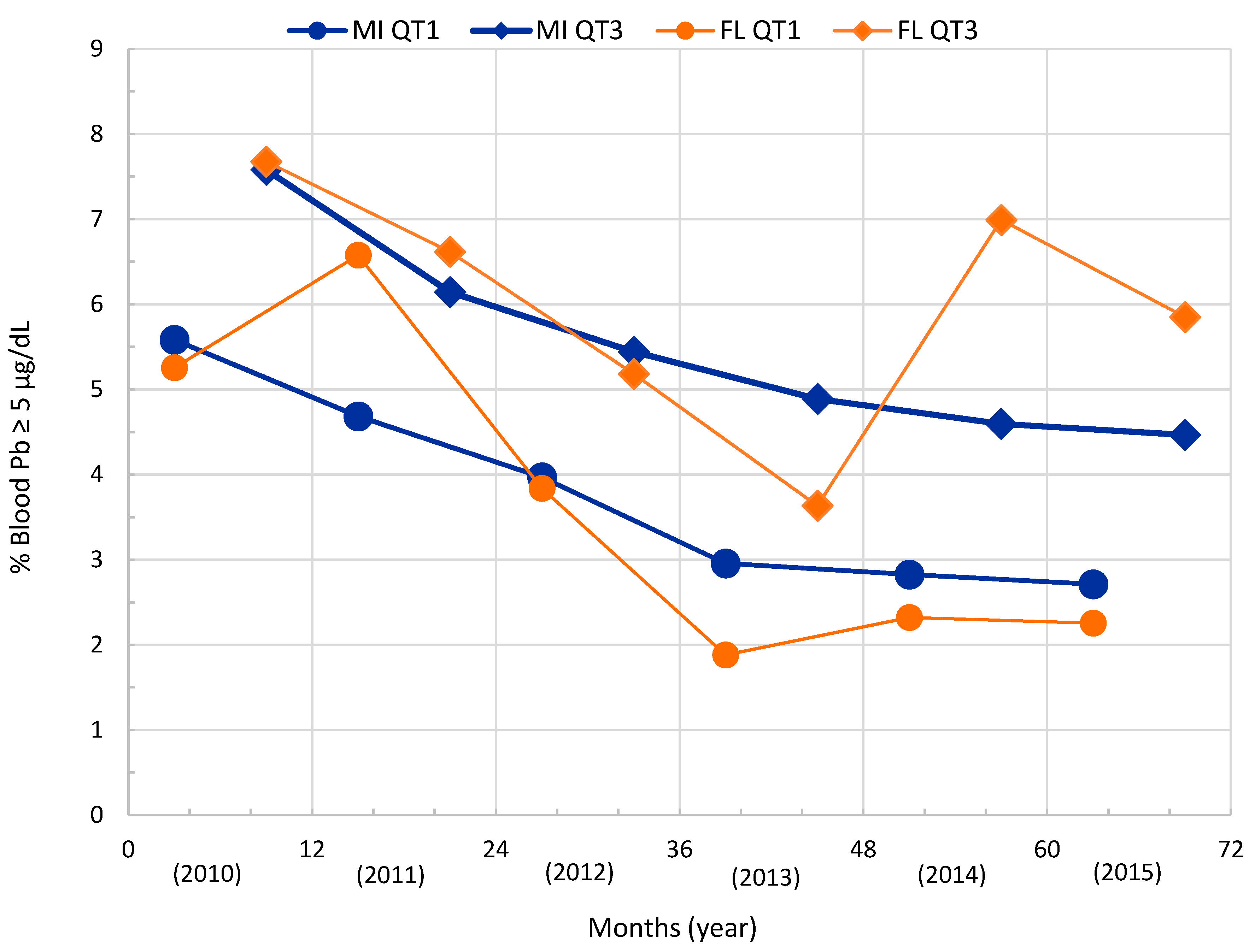
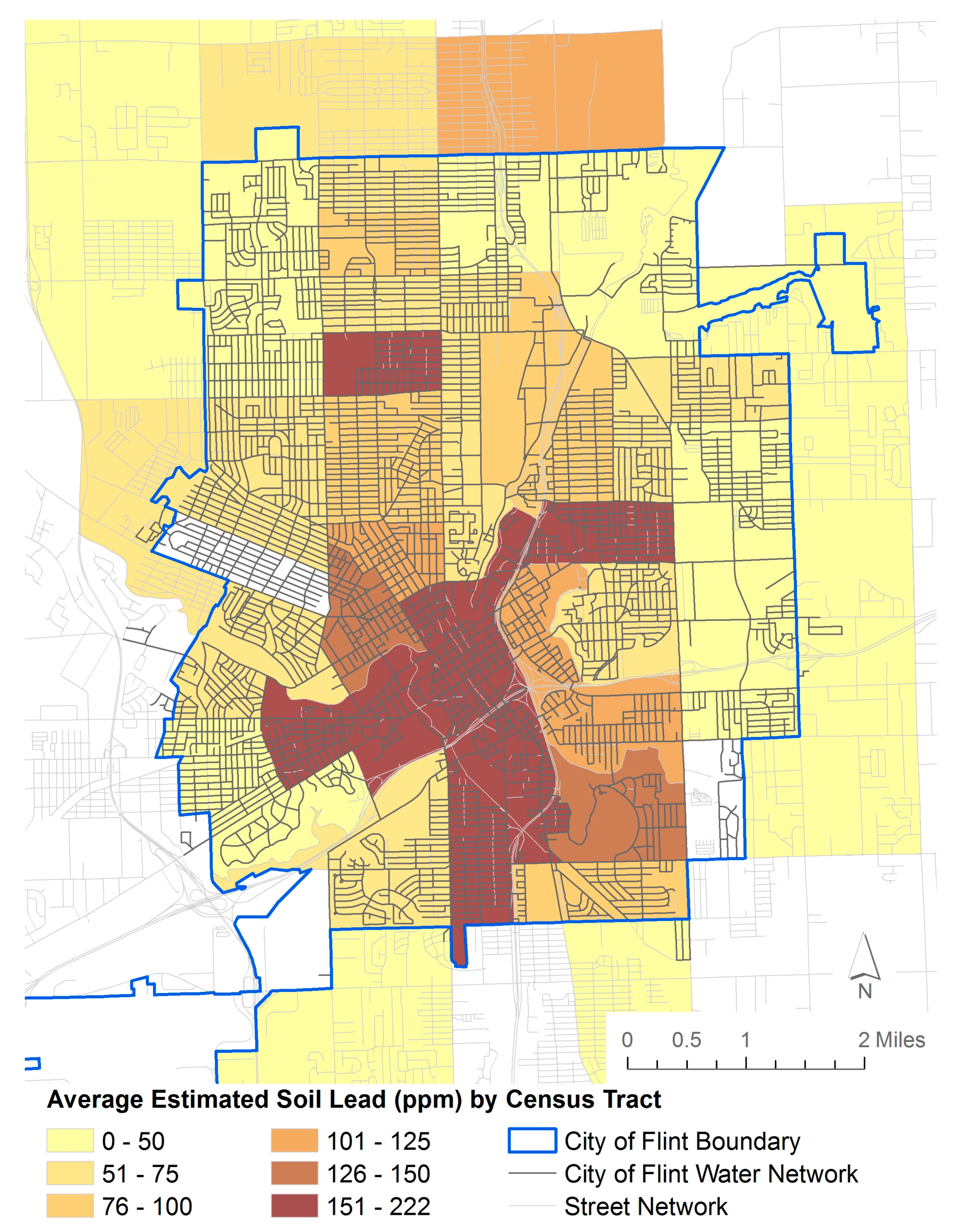
| Location | Genesee | Flint |
|---|---|---|
| Michigan | 0.877 * | 0.816 * |
| Genesee | - | 0.977 * |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Laidlaw, M.A.S.; Filippelli, G.M.; Sadler, R.C.; Gonzales, C.R.; Ball, A.S.; Mielke, H.W. Children’s Blood Lead Seasonality in Flint, Michigan (USA), and Soil-Sourced Lead Hazard Risks. Int. J. Environ. Res. Public Health 2016, 13, 358. https://doi.org/10.3390/ijerph13040358
Laidlaw MAS, Filippelli GM, Sadler RC, Gonzales CR, Ball AS, Mielke HW. Children’s Blood Lead Seasonality in Flint, Michigan (USA), and Soil-Sourced Lead Hazard Risks. International Journal of Environmental Research and Public Health. 2016; 13(4):358. https://doi.org/10.3390/ijerph13040358
Chicago/Turabian StyleLaidlaw, Mark A.S., Gabriel M. Filippelli, Richard C. Sadler, Christopher R. Gonzales, Andrew S. Ball, and Howard W. Mielke. 2016. "Children’s Blood Lead Seasonality in Flint, Michigan (USA), and Soil-Sourced Lead Hazard Risks" International Journal of Environmental Research and Public Health 13, no. 4: 358. https://doi.org/10.3390/ijerph13040358
APA StyleLaidlaw, M. A. S., Filippelli, G. M., Sadler, R. C., Gonzales, C. R., Ball, A. S., & Mielke, H. W. (2016). Children’s Blood Lead Seasonality in Flint, Michigan (USA), and Soil-Sourced Lead Hazard Risks. International Journal of Environmental Research and Public Health, 13(4), 358. https://doi.org/10.3390/ijerph13040358









