Arsenic Exposure and Predicted 10-Year Atherosclerotic Cardiovascular Risk Using the Pooled Cohort Equations in U.S. Hypertensive Adults
Abstract
:1. Introduction
2. Methods
2.1. Study Population
2.2. Measures and Definitions
2.2.1. Urine Arsenic Measurement
2.2.2. Predicted 10-Year Risk of Hard ASCVD
2.2.3. Hypertension
2.2.4. Other Variables
2.3. Statistical Analysis
3. Results
3.1. Baseline Characteristics of the Study Population
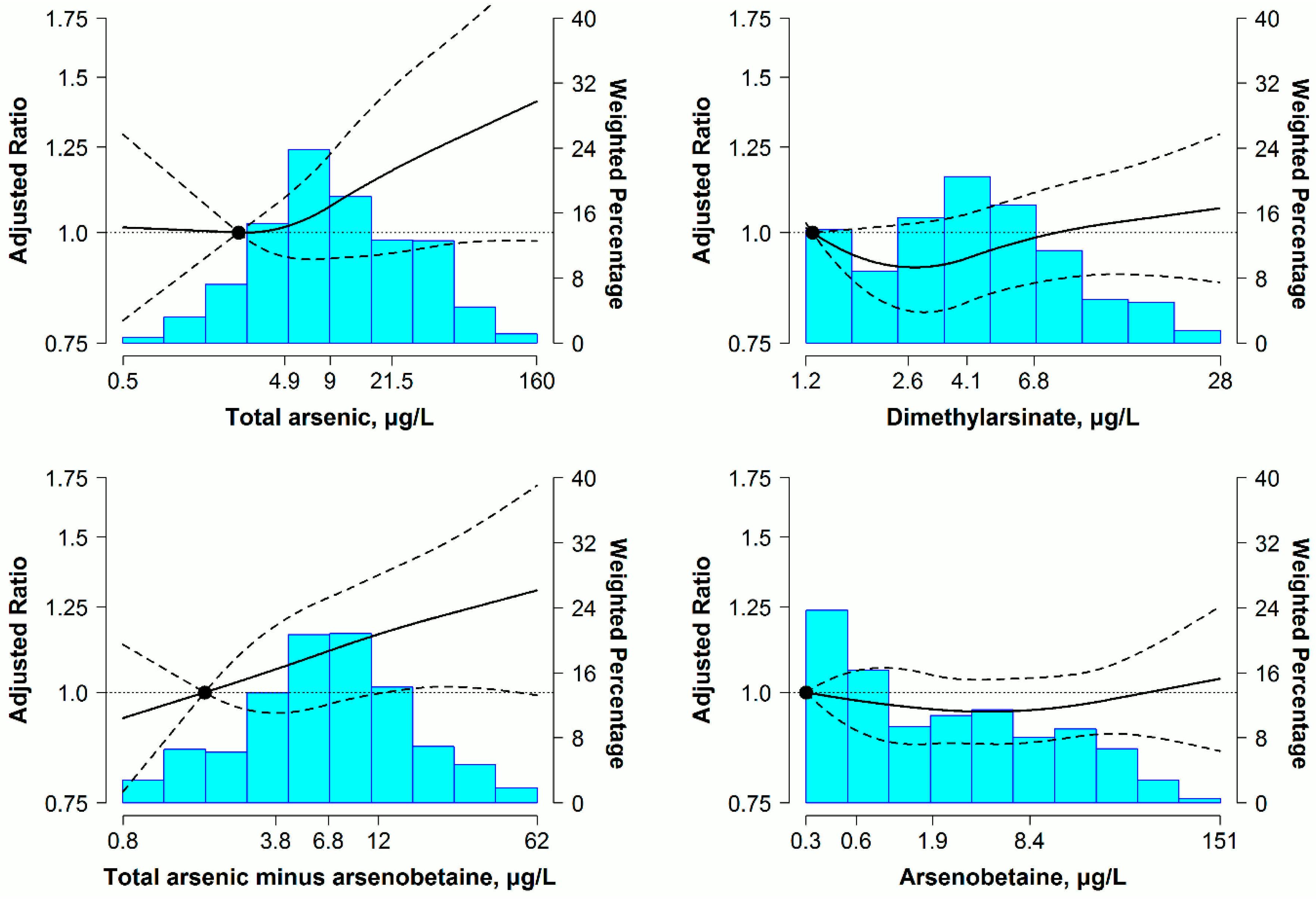
3.2. Relationship between Urine Arsenic and 10-Year Predicted ASCVD Risk
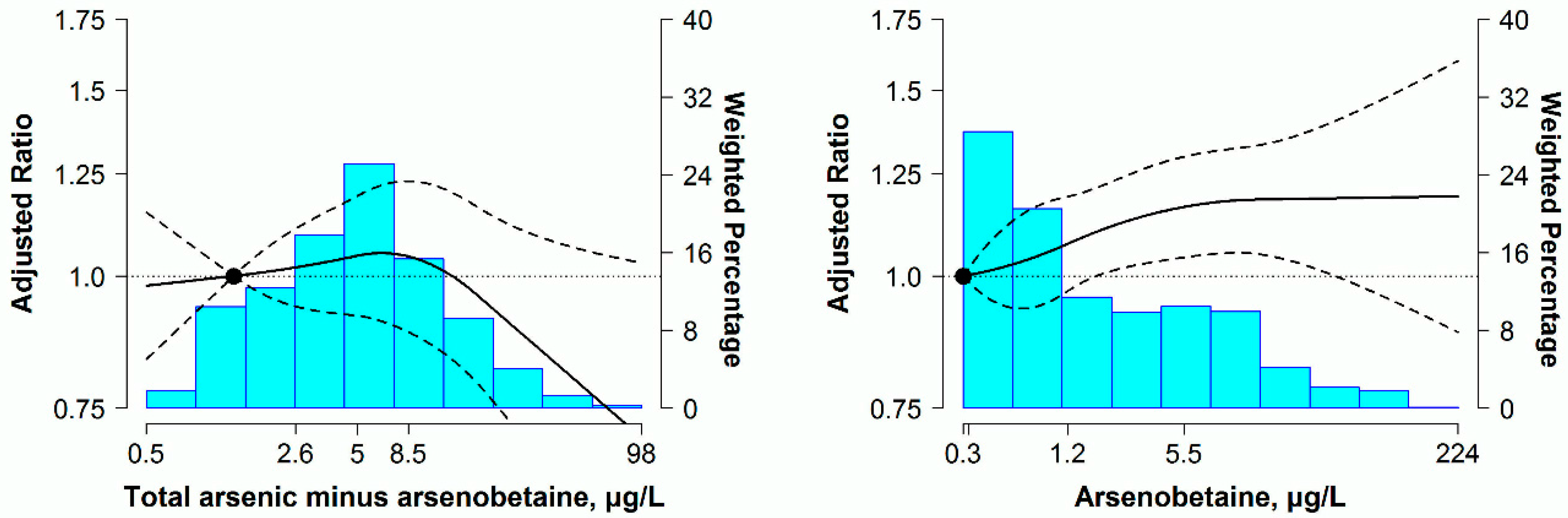
3.3. Sensitivity Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Guidelines for Drinking-Water Quality: Incorporating First and Second Addenda to Third Edition. Vol. 1, Recommendations. Available online: http://www.who.int/water_sanitation_health/dwq/fulltext.pdf (accessed on 29 March 2016).
- National Primary Drinking Water Regulations; Arsenic and Clarifications to Compliance and New Source Contaminants Monitoring; Final Rule. Available online: https://www.regulations.gov/document?D=EPA-HQ-OW-2001-0001-0228 (accessed on 20 October 2016).
- Wang, C.H.; Jeng, J.S.; Yip, P.K.; Chen, C.L.; Hsu, L.I.; Hsueh, Y.M.; Chiou, H.Y.; Wu, M.M.; Chen, C.J. Biological gradient between long-term arsenic exposure and carotid atherosclerosis. Circulation 2002, 105, 1804–1809. [Google Scholar] [CrossRef]
- Chen, C.J.; Chiou, H.Y.; Chiang, M.H.; Lin, L.J.; Tai, T.Y. Dose-response relationship between ischemic heart disease mortality and long-term arsenic exposure. Arterioscler. Thromb. Vasc. Biol. 1996, 16, 504–510. [Google Scholar] [CrossRef]
- Tseng, C.H.; Chong, C.K.; Tseng, C.P.; Hsueh, Y.M.; Chiou, H.Y.; Tseng, C.C.; Chen, C.J. Long-term arsenic exposure and ischemic heart disease in arseniasis-hyperendemic villages in Taiwan. Toxicol. Lett. 2003, 137, 15–21. [Google Scholar] [CrossRef]
- Chen, Y.; Graziano, J.H.; Parvez, F.; Liu, M.; Slavkovich, V.; Kalra, T.; Argos, M.; Islam, T.; Ahmed, A.; Rakibuz-Zaman, M.; et al. Arsenic exposure from drinking water and mortality from cardiovascular disease in Bangladesh: Prospective cohort study. BMJ 2011, 342, d2431. [Google Scholar] [CrossRef]
- Sidhu, M.S.; Desai, K.P.; Lynch, H.N.; Rhomberg, L.R.; Beck, B.D.; Venditti, F.J. Mechanisms of action for arsenic in cardiovascular toxicity and implications for risk assessment. Toxicology 2015, 331, 78–99. [Google Scholar] [CrossRef]
- Zhang, C.; Mao, G.; He, S.; Yang, Z.; Yang, W.; Zhang, X.; Qiu, W.; Ta, N.; Cao, L.; Yang, H.; et al. Relationship between long-term exposure to low-level arsenic in drinking water and the prevalence of abnormal blood pressure. J. Hazard. Mater. 2013, 262, 1154–1158. [Google Scholar] [CrossRef]
- Abhyankar, L.N.; Jones, M.R.; Guallar, E.; Navas-Acien, A. Arsenic exposure and hypertension: A systematic review. Environ. Health Perspect. 2012, 120, 494–500. [Google Scholar] [CrossRef]
- Jones, M.R.; Tellez-Plaza, M.; Sharrett, A.R.; Guallar, E.; Navas-Acien, A. Urine arsenic and hypertension in US adults: The 2003–2008 national health and nutrition examination survey. Epidemiology 2011, 22, 153–161. [Google Scholar] [CrossRef]
- Shiue, I.; Hristova, K. Higher urinary heavy metal, phthalate and arsenic concentrations accounted for 3%–19% of the population attributable risk for high blood pressure: US NHANES, 2009–2012. Hypertens. Res. 2014, 37, 1075–1081. [Google Scholar] [CrossRef]
- Navas-Acien, A.; Silbergeld, E.K.; Pastor-Barriuso, R.; Guallar, E. Arsenic exposure and prevalence of type 2 diabetes in US adults. JAMA 2008, 300, 814–822. [Google Scholar] [CrossRef]
- Mendez, M.A.; Gonzalez-Horta, C.; Sanchez-Ramirez, B.; Ballinas-Casarrubias, L.; Ceron, R.H.; Morales, D.V.; Terrazas, F.A.; Ishida, M.C.; Gutierrez-Torres, D.S.; Saunders, R.J.; et al. Chronic exposure to arsenic and markers of cardiometabolic risk: A cross-sectional study in Chihuahua, Mexico. Environ. Health Perspect. 2016, 124, 104–111. [Google Scholar] [CrossRef]
- Wu, F.; Jasmine, F.; Kibriya, M.G.; Liu, M.; Wojcik, O.; Parvez, F.; Rahaman, R.; Roy, S.; Paul-Brutus, R.; Segers, S.; et al. Association between arsenic exposure from drinking water and plasma levels of cardiovascular markers. Am. J. Epidemiol. 2012, 175, 1252–1261. [Google Scholar] [CrossRef]
- Chen, Y.; Wu, F.; Graziano, J.H.; Parvez, F.; Liu, M.; Paul, R.R.; Shaheen, I.; Sarwar, G.; Ahmed, A.; Islam, T.; et al. Arsenic exposure from drinking water, arsenic methylation capacity, and carotid intima-media thickness in Bangladesh. Am. J. Epidemiol. 2013, 178, 372–381. [Google Scholar] [CrossRef]
- Goff, D.C., Jr.; Lloyd-Jones, D.M.; Bennett, G.; Coady, S.; D’Agostino, R.B., Sr.; Gibbons, R.; Greenland, P.; Lackland, D.T.; Levy, D.; O’Donnell, C.J.; et al. 2013 ACC/AHA guideline on the assessment of cardiovascular risk: A report of the American college of cardiology/american heart association task force on practice guidelines. J. Am. Coll. Cardiol. 2014, 63, 2935–2959. [Google Scholar] [CrossRef]
- Karmali, K.N.; Goff, D.C., Jr.; Ning, H.; Lloyd-Jones, D.M. A systematic examination of the 2013 ACC/AHA pooled cohort risk assessment tool for atherosclerotic cardiovascular disease. J. Am. Coll. Cardiol. 2014, 64, 959–968. [Google Scholar] [CrossRef]
- Muntner, P.; Colantonio, L.D.; Cushman, M.; Goff, D.C., Jr.; Howard, G.; Howard, V.J.; Kissela, B.; Levitan, E.B.; Lloyd-Jones, D.M.; Safford, M.M. Validation of the atherosclerotic cardiovascular disease pooled cohort risk equations. JAMA 2014, 311, 1406–1415. [Google Scholar] [CrossRef]
- Kurzius-Spencer, M.; Burgess, J.L.; Harris, R.B.; Hartz, V.; Roberge, J.; Huang, S.; Hsu, C.H.; O’Rourke, M.K. Contribution of diet to aggregate arsenic exposures-an analysis across populations. J. Expo. Sci. Environ. Epidemiol. 2014, 24, 156–162. [Google Scholar] [CrossRef]
- Aylward, L.L.; Ramasamy, S.; Hays, S.M.; Schoeny, R.; Kirman, C.R. Evaluation of urinary speciated arsenic in NHANES: Issues in interpretation in the context of potential inorganic arsenic exposure. Regul. Toxicol. Pharmacol. 2014, 69, 49–54. [Google Scholar] [CrossRef]
- Tsuji, J.S.; Garry, M.R.; Perez, V.; Chang, E.T. Low-level arsenic exposure and developmental neurotoxicity in children: A systematic review and risk assessment. Toxicology 2015, 337, 91–107. [Google Scholar] [CrossRef]
- NHANES 2003–2004: Laboratory Component: Total Arsenic and Speciated Arsenic. Available online: https://wwwn.cdc.gov/Nchs/Nhanes/2003-2004/L06UAS_C.htm (accessed on 4 March 2016).
- NHANES 2005–2006: Laboratory Component: Total Arsenic and Speciated Arsenic. Available online: https://wwwn.cdc.gov/Nchs/Nhanes/2005-2006/UAS_D.htm (accessed on 4 March 2016).
- NHANES 2007–2008: Laboratory Component: Total Arsenic and Speciated Arsenic. Available online: https://wwwn.cdc.gov/Nchs/Nhanes/2007-2008/UAS_E.htm (accessed on 4 March 2016).
- NHANES 2009–2010: Laboratory Component: Total Arsenic and Speciated Arsenic. Available online: https://wwwn.cdc.gov/Nchs/Nhanes/2009-2010/UAS_F.htm (accessed on 5 March 2016).
- NHANES 2011–2012: Laboratory Component: Total Arsenic and Speciated Arsenic. Available online: https://wwwn.cdc.gov/Nchs/Nhanes/2011-2012/UAS_G.htm (accessed on 5 March 2016).
- Caldwell, K.L.; Jones, R.L.; Verdon, C.P.; Jarrett, J.M.; Caudill, S.P.; Osterloh, J.D. Levels of urinary total and speciated arsenic in the US population: National health and nutrition examination survey 2003–2004. J. Expo. Sci. Environ. Epidemiol. 2009, 19, 59–68. [Google Scholar] [CrossRef]
- NHANES 2007–2008 Data Documentation, Codebook, and Frequencies: Cholesterol—Total (TCHOL_E). Available online: https://wwwn.cdc.gov/Nchs/Nhanes/2007-2008/TCHOL_E.htm (accessed on 15 March 2016).
- NHANES 2007–2008 Data Documentation, Codebook, and Frequencies: Cholesterol—HDL (HDL_E). Available online: https://wwwn.cdc.gov/Nchs/Nhanes/2007-2008/HDL_E.htm (accessed on 15 March 2016).
- NHANES 2003–2004: Laboratory Component: Serum Cotinine. Available online: https://wwwn.cdc.gov/Nchs/Nhanes/2003-2004/L06COT_C.htm (accessed on 10 March 2016).
- Navas-Acien, A.; Francesconi, K.A.; Silbergeld, E.K.; Guallar, E. Seafood intake and urine concentrations of total arsenic, dimethylarsinate and arsenobetaine in the US population. Environ. Res. 2011, 111, 110–118. [Google Scholar] [CrossRef]
- NHANES 2007–2008 Data Documentation, Codebook, and Frequencies: Albumin & Creatinine—Urine (ALB_CR_E). Available online: https://wwwn.cdc.gov/Nchs/Nhanes/2007-2008/ALB_CR_E.htm (accessed on 15 March 2016).
- National Health and Nutrition Examination Survey: Analytic Guidelines, 1999–2010. Available online: http://www.cdc.gov/nchs/data/series/sr_02/sr02_161.pdf (accessed on 23 March 2016).
- Wu, M.M.; Chiou, H.Y.; Hsueh, Y.M.; Hong, C.T.; Su, C.L.; Chang, S.F.; Huang, W.L.; Wang, H.T.; Wang, Y.H.; Hsieh, Y.C.; et al. Effect of plasma homocysteine level and urinary monomethylarsonic acid on the risk of arsenic-associated carotid atherosclerosis. Toxicol. Appl. Pharmacol. 2006, 216, 168–175. [Google Scholar] [CrossRef]
- Wang, Y.H.; Wu, M.M.; Hong, C.T.; Lien, L.M.; Hsieh, Y.C.; Tseng, H.P.; Chang, S.F.; Su, C.L.; Chiou, H.Y.; Chen, C.J. Effects of arsenic exposure and genetic polymorphisms of p53, glutathione S-transferase M1, T1, and P1 on the risk of carotid atherosclerosis in Taiwan. Atherosclerosis 2007, 192, 305–312. [Google Scholar] [CrossRef]
- Sohel, N.; Persson, L.A.; Rahman, M.; Streatfield, P.K.; Yunus, M.; Ekstrom, E.C.; Vahter, M. Arsenic in drinking water and adult mortality: A population-based cohort study in rural Bangladesh. Epidemiology 2009, 20, 824–830. [Google Scholar] [CrossRef]
- Zierold, K.M.; Knobeloch, L.; Anderson, H. Prevalence of chronic diseases in adults exposed to arsenic-contaminated drinking water. Am. J. Public Health 2004, 94, 1936–1937. [Google Scholar] [CrossRef]
- Gong, G.; O’Bryant, S.E. Low-level arsenic exposure, AS3MT gene polymorphism and cardiovascular diseases in rural Texas counties. Environ. Res. 2012, 113, 52–57. [Google Scholar] [CrossRef]
- Moon, K.A.; Guallar, E.; Umans, J.G.; Devereux, R.B.; Best, L.G.; Francesconi, K.A.; Goessler, W.; Pollak, J.; Silbergeld, E.K.; Howard, B.V.; et al. Association between exposure to low to moderate arsenic levels and incident cardiovascular disease. A prospective cohort study. Ann. Intern. Med. 2013, 159, 649–659. [Google Scholar]
- Wu, F.; Molinaro, P.; Chen, Y. Arsenic exposure and subclinical endpoints of cardiovascular diseases. Curr. Environ. Health Rep. 2014, 1, 148–162. [Google Scholar] [CrossRef]
- Shi, Y.; Wei, Y.; Qu, S.; Wang, Y.; Li, Y.; Li, R. Arsenic induces apoptosis of human umbilical vein endothelial cells through mitochondrial pathways. Cardiovasc. Toxicol. 2010, 10, 153–160. [Google Scholar] [CrossRef]
- Bunderson, M.; Coffin, J.D.; Beall, H.D. Arsenic induces peroxynitrite generation and cyclooxygenase-2 protein expression in aortic endothelial cells: Possible role in atherosclerosis. Toxicol. Appl. Pharmacol. 2002, 184, 11–18. [Google Scholar] [CrossRef]
- Chou, Y.; Tsai, C.H.; Ueng, K.C.; Tian, T.Y.; Chen, S.C.; Yeh, H.I. Endothelial gap junctions are down-regulated by arsenic trioxide. Eur. J. Pharmacol. 2007, 569, 29–36. [Google Scholar] [CrossRef]
- Bunderson, M.; Brooks, D.M.; Walker, D.L.; Rosenfeld, M.E.; Coffin, J.D.; Beall, H.D. Arsenic exposure exacerbates atherosclerotic plaque formation and increases nitrotyrosine and leukotriene biosynthesis. Toxicol. Appl. Pharmacol. 2004, 201, 32–39. [Google Scholar] [CrossRef]
- Ellinsworth, D.C. Arsenic, reactive oxygen, and endothelial dysfunction. J. Pharmacol. Exp. Ther. 2015, 353, 458–464. [Google Scholar] [CrossRef]
- Thomas, S.R.; Chen, K.; Keaney, J.F., Jr. Oxidative stress and endothelial nitric oxide bioactivity. Antioxid. Redox Signal. 2003, 5, 181–194. [Google Scholar] [CrossRef]
- Simeonova, P.P.; Hulderman, T.; Harki, D.; Luster, M.I. Arsenic exposure accelerates atherogenesis in apolipoprotein e(−/−) mice. Environ. Health Perspect. 2003, 111, 1744–1748. [Google Scholar] [CrossRef]
- Srivastava, S.; Vladykovskaya, E.N.; Haberzettl, P.; Sithu, S.D.; D’Souza, S.E.; States, J.C. Arsenic exacerbates atherosclerotic lesion formation and inflammation in ApoE−/−mice. Toxicol. Appl. Pharmacol. 2009, 241, 90–100. [Google Scholar] [CrossRef]
- Hossain, E.; Ota, A.; Karnan, S.; Damdindorj, L.; Takahashi, M.; Konishi, Y.; Konishi, H.; Hosokawa, Y. Arsenic augments the uptake of oxidized LDL by upregulating the expression of lectin-like oxidized LDL receptor in mouse aortic endothelial cells. Toxicol. Appl. Pharmacol. 2013, 273, 651–658. [Google Scholar] [CrossRef]
- Hernanz, R.; Martinez-Revelles, S.; Palacios, R.; Martin, A.; Cachofeiro, V.; Aguado, A.; Garcia-Redondo, L.; Barrus, M.T.; de Batista, P.R.; Briones, A.M.; et al. Toll-like receptor 4 contributes to vascular remodelling and endothelial dysfunction in angiotensin II-induced hypertension. Br. J. Pharmacol. 2015, 172, 3159–3176. [Google Scholar] [CrossRef]
- Matrougui, K.; Abd Elmageed, Z.; Kassan, M.; Choi, S.; Nair, D.; Gonzalez-Villalobos, R.A.; Chentoufi, A.A.; Kadowitz, P.; Belmadani, S.; Partyka, M. Natural regulatory T cells control coronary arteriolar endothelial dysfunction in hypertensive mice. Am. J. Pathol. 2011, 178, 434–441. [Google Scholar] [CrossRef]
- Kassan, M.; Galan, M.; Partyka, M.; Saifudeen, Z.; Henrion, D.; Trebak, M.; Matrougui, K. Endoplasmic reticulum stress is involved in cardiac damage and vascular endothelial dysfunction in hypertensive mice. Arterioscler. Thromb. Vasc. Biol. 2012, 32, 1652–1661. [Google Scholar] [CrossRef]
- Li, D.J.; Evans, R.G.; Yang, Z.W.; Song, S.W.; Wang, P.; Ma, X.J.; Liu, C.; Xi, T.; Su, D.F.; Shen, F.M. Dysfunction of the cholinergic anti-inflammatory pathway mediates organ damage in hypertension. Hypertension 2011, 57, 298–307. [Google Scholar] [CrossRef]
- Gonzalez, G.E.; Rhaleb, N.E.; D’Ambrosio, M.A.; Nakagawa, P.; Liu, Y.; Leung, P.; Dai, X.; Yang, X.P.; Peterson, E.L.; Carretero, O.A. Deletion of interleukin-6 prevents cardiac inflammation, fibrosis and dysfunction without affecting blood pressure in angiotensin II-high salt-induced hypertension. J. Hypertens. 2015, 33, 144–152. [Google Scholar] [CrossRef]
- Li, Y.; Wu, Y.; Zhang, C.; Li, P.; Cui, W.; Hao, J.; Ma, X.; Yin, Z.; Du, J. γδT cell-derived interleukin-17A via an interleukin-1β-dependent mechanism mediates cardiac injury and fibrosis in hypertension. Hypertension 2014, 64, 305–314. [Google Scholar] [CrossRef]
- Kauser, K.; Rubanyi, G.M. Gender difference in endothelial dysfunction in the aorta of spontaneously hypertensive rats. Hypertension 1995, 25, 517–523. [Google Scholar] [CrossRef]
- Brandes, R.P.; Mugge, A. Gender differences in the generation of superoxide anions in the rat aorta. Life Sci. 1997, 60, 391–396. [Google Scholar] [CrossRef]
- Dantas, A.P.; Franco Mdo, C.; Silva-Antonialli, M.M.; Tostes, R.C.; Fortes, Z.B.; Nigro, D.; Carvalho, M.H. Gender differences in superoxide generation in microvessels of hypertensive rats: Role of NAD(P)H-oxidase. Cardiovasc. Res. 2004, 61, 22–29. [Google Scholar] [CrossRef]
- Camilletti, A.; Moretti, N.; Giacchetti, G.; Faloia, E.; Martarelli, D.; Mantero, F.; Mazzanti, L. Decreased nitric oxide levels and increased calcium content in platelets of hypertensive patients. Am. J. Hypertens. 2001, 14, 382–386. [Google Scholar] [CrossRef]
- Karagas, M.R.; Le, C.X.; Morris, S.; Blum, J.; Lu, X.; Spate, V.; Carey, M.; Stannard, V.; Klaue, B.; Tosteson, T.D. Markers of low level arsenic exposure for evaluating human cancer risks in a US population. Int. J. Occup. Med. Environ. Health 2001, 14, 171–175. [Google Scholar]
- Steinmaus, C.M.; Yuan, Y.; Smith, A.H. The temporal stability of arsenic concentrations in well water in western Nevada. Environ. Res. 2005, 99, 164–168. [Google Scholar] [CrossRef]

| Characteristics | Overall | Quartile 1 | Quartile 2 | Quartile 3 | Quartile 4 | p for Trend a |
|---|---|---|---|---|---|---|
| (≤4.92 μg/L) | (4.93–9.20 μg/L) | (9.21–21.56 μg/L) | (≥21.57 μg/L) | |||
| Age (y) | 57.0 (0.5) | 56.3 (0.9) | 56.1 (1.0) | 56.6 (0.9) | 59.1 (0.9) | 0.006 |
| Race (%) | ||||||
| Non-Hispanic white | 75.9 (2.1) | 81.3 (3.6) | 79.8 (3.2) | 72.3 (3.5) | 70.1 (4.4) | 0.03 |
| Non-Hispanic black | 11.0 (1.2) | 8.7 (2.1) | 10.7 (2.2) | 11.9 (2.4) | 12.9 (2.1) | 0.16 |
| Other | 13.1 (1.5) | 10.0 (2.9) | 9.5 (2.2) | 15.8 (2.6) | 17.0 (3.3) | 0.05 |
| High school education (%) | 80.6 (1.7) | 79.8 (3.9) | 82.0 (4.0) | 77.8 (3.8) | 82.7 (3.6) | 0.24 |
| BMI (kg/m2) | 30.4 (0.3) | 30.4 (0.7) | 30.1 (0.4) | 30.8 (0.6) | 30.1 (0.6) | 0.86 |
| Current smoking (%) | 19.0 (1.7) | 21.5 (4.1) | 24.4 (3.8) | 13.8 (3.2) | 16.3 (3.7) | 0.47 |
| Serum cotinine (ng/mL) | 69.4 (7.9) | 90.8 (18.4) | 73.9 (14.4) | 56.8 (16.1) | 56.0 (16.1) | 0.09 |
| Current antihypertension medication (%) | 81.2 (2.4) | 84.8 (4.5) | 70.2 (5.2) | 82.3 (3.9) | 87.6 (3.3) | 0.13 |
| Systolic blood pressure (mmHg) | 130.7 (1.0) | 130.9 (1.3) | 130.8 (1.5) | 129.1 (1.8) | 132.1 (2.3) | 0.82 |
| Diabetes (%) | 23.2 (2.1) | 20.3 (4.1) | 24.6 (4.3) | 23.3 (4.2) | 24.7 (5.1) | 0.90 |
| Total cholesterol (mg/dL) | 197.3 (2.0) | 201.3 (4.9) | 203.8 (4.4) | 192.5 (4.0) | 191.7 (4.0) | 0.12 |
| HDL cholesterol (mg/dL) | 46.8 (0.7) | 46.0 (1.4) | 47.2 (1.6) | 47.1 (1.2) | 47.0 (1.5) | 0.84 |
| Predicted 10-year ASCVD risk (%) b | 10.8 (5.7–19.5) | 10.8 (6.3–19.0) | 10.0 (5.3–19.4) | 9.8 (5.3–18.0) | 11.7 (6.6–23.2) | 0.71 |
| Urine creatinine (mg/dL) | 131.7 (3.4) | 82.7 (5.6) | 132.8 (7.6) | 149.5 (6.3) | 162.0 (6.3) | <0.001 |
| Urine arsenic (μg/L) b | ||||||
| Total arsenic | 9.2 (4.9–21.6) | 3.1 (2.1–4.2) | 7.0 (5.9–8.2) | 13.2 (10.7–16.5) | 39.0 (29.0–58.7) | |
| Dimethylarsinate | 4.1 (2.6–6.9) | 1.8 (1.2–2.6) | 3.5 (3.0–4.5) | 5.4 (4.0–7.4) | 9.0 (5.7–15.1) | <0.001 |
| Total arsenic minus arsenobetaine | 6.7 (3.8–11.5) | 2.3 (1.4–3.4) | 5.6 (4.6–6.7) | 9.2 (7.0–11.4) | 17.4 (11.5–27.7) | <0.001 |
| Arsenobetaine | 1.9 (0.6–8.4) | 0.7 (0.3–0.8) | 0.8 (0.3–2.1) | 3.7 (1.9–6.5) | 20.9 (12.8–32.9) | <0.001 |
| Characteristics | Overall | Quartile 1 | Quartile 2 | Quartile 3 | Quartile 4 | p for Trend a |
|---|---|---|---|---|---|---|
| (≤3.30 μg/L) | (3.31–6.71 μg/L) | (6.72–14.92 μg/L) | (≥14.93 μg/L) | |||
| Age (y) | 58.6 (0.5) | 58.1 (1.1) | 58.9 (0.7) | 60.0 (0.7) | 57.4 (1.1) | 0.53 |
| Race (%) | ||||||
| Non-Hispanic white | 75.5 (2.2) | 89.1 (2.3) | 73.8 (4.0) | 71.1 (3.1) | 68.0 (3.9) | 0.004 |
| Non-Hispanic black | 13.3 (1.6) | 5.4 (1.6) | 12.8 (2.4) | 16.0 (2.3) | 19.0 (3.0) | 0.003 |
| Other | 11.2 (1.4) | 5.5 (1.4) | 13.4 (3.0) | 12.9 (2.2) | 13.0 (2.4) | 0.17 |
| High school education (%) | 82.8 (1.6) | 86.5 (2.7) | 81.8 (3.2) | 83.5 (2.4) | 79.4 (3.6) | 0.35 |
| BMI (kg/m2) | 31.9 (0.4) | 30.6 (0.7) | 33.4 (1.0) | 32.4 (0.9) | 31.4 (0.7) | 0.42 |
| Current smoking (%) | 13.1 (1.5) | 14.0 (3.6) | 14.6 (3.3) | 8.3 (2.1) | 15.4 (3.6) | 0.78 |
| Serum cotinine (ng/mL) | 33.7 (3.6) | 45.6 (12.1) | 34.8 (7.4) | 21.2 (5.1) | 33.2 (7.4) | 0.91 |
| Current antihypertension medication (%) | 89.4 (1.4) | 88.4 (3.0) | 89.7 (3.2) | 91.8 (2.5) | 87.7 (2.5) | 0.78 |
| Systolic blood pressure (mmHg) | 130.9 (0.8) | 130.4 (1.4) | 129.6 (1.5) | 131.4 (1.8) | 132.1 (1.4) | 0.33 |
| Diabetes (%) | 22.3 (1.6) | 24.3 (3.7) | 25.9 (3.7) | 20.1 (3.6) | 18.8 (3.3) | 0.10 |
| Total cholesterol (mg/dL) | 206.7 (1.9) | 205.4 (4.1) | 210.5 (3.4) | 203.4 (3.5) | 207.6 (3.3) | 0.83 |
| HDL cholesterol (mg/dL) | 58.1 (0.8) | 55.4 (1.2) | 57.2 (1.8) | 59.8 (1.6) | 60.1 (1.8) | 0.06 |
| Predicted 10-year ASCVD risk (%) b | 6.3 (2.3–13.5) | 5.7 (2.0–13.5) | 6.6 (2.6–15.3) | 6.7 (2.9–12.4) | 5.9 (2.1–13.7) | 0.60 |
| Urine creatinine (mg/dL) | 90.7 (3.6) | 41.8 (2.2) | 90.1 (5.3) | 105.6 (4.4) | 125.9 (9.7) | <0.001 |
| Urine arsenic (μg/L) b | ||||||
| Total arsenic | 6.7 (3.3–14.9) | 2.1 (1.4–2.5) | 5.0 (4.3–5.8) | 9.2 (7.9–11.7) | 26.3 (19.7–48.0) | |
| Dimethylarsinate | 3.2 (1.9–5.4) | 1.2 (1.2–1.8) | 3.0 (2.2–3.6) | 4.4 (3.0–5.7) | 7.5 (4.4–12.2) | <0.001 |
| Total arsenic minus arsenobetaine | 4.8 (2.4–8.3) | 1.4 (1.0–2.0) | 4.1 (3.2–4.8) | 6.6 (5.1–7.9) | 15.2 (9.2–21.3) | <0.001 |
| Arsenobetaine | 1.2 (0.3–5.7) | 0.3 (0.3–0.8) | 0.8 (0.3–1.4) | 2.5 (0.9–4.8) | 12.4 (7.9–26.8) | <0.001 |
| Quartile 1 | Quartile 2 | Quartile 3 | Quartile 4 | p for Trend | |
|---|---|---|---|---|---|
| Total arsenic (μg/L) | ≤4.92 | 4.93–9.20 | 9.21–21.56 | ≥21.57 | |
| Model 1 | 1 (reference) | 1.02 (0.80–1.30) | 0.88 (0.73–1.07) | 0.98 (0.76–1.25) | 0.92 |
| Model 2 | 1 (reference) | 0.97 (0.85–1.11) | 0.97 (0.86–1.09) | 1.05 (0.92–1.19) | 0.10 |
| Model 3 | 1 (reference) | 1.00 (0.88–1.14) | 1.06 (0.92–1.22) | 1.24 (1.02–1.53) | 0.007 |
| Dimethylarsinate (μg/L) | ≤2.61 | 2.62–4.10 | 4.11–6.86 | ≥6.87 | |
| Model 1 | 1 (reference) | 0.99 (0.82–1.19) | 0.94 (0.80–1.12) | 0.95 (0.77–1.18) | 0.64 |
| Model 2 | 1 (reference) | 0.97 (0.89–1.06) | 0.98 (0.89–1.09) | 1.07 (0.98–1.17) | 0.03 |
| Model 3 | 1 (reference) | 0.98 (0.90–1.08) | 1.01 (0.90–1.12) | 1.13 (1.02–1.25) | 0.003 |
| Total arsenic minus arsenobetaine (μg/L) | ≤3.83 | 3.84–6.66 | 6.67–11.51 | ≥11.52 | |
| Model 1 | 1 (reference) | 1.03 (0.84–1.27) | 1.03 (0.83–1.29) | 1.00 (0.78–1.27) | 0.82 |
| Model 2 | 1 (reference) | 1.09 (0.98–1.20) | 1.06 (0.95–1.20) | 1.14 (1.01–1.29) | 0.04 |
| Model 3 | 1 (reference) | 1.10 (0.99–1.22) | 1.10 (0.97–1.23) | 1.22 (1.05–1.40) | 0.008 |
| Arsenobetaine (μg/L) | ≤0.60 | 0.61–1.92 | 1.93–8.39 | ≥8.40 | |
| Model 1 | 1 (reference) | 0.94 (0.77–1.14) | 0.86 (0.73–1.03) | 0.89 (0.74–1.08) | 0.49 |
| Model 2 | 1 (reference) | 0.96 (0.88–1.06) | 0.94 (0.85–1.03) | 0.98 (0.89–1.07) | 0.93 |
| Model 3 | 1 (reference) | 0.96 (0.88–1.06) | 0.94 (0.86–1.03) | 0.98 (0.90–1.07) | 0.79 |
| Quartile 1 | Quartile 2 | Quartile 3 | Quartile 4 | p for Trend | |
|---|---|---|---|---|---|
| Total arsenic (μg/L) | ≤3.30 | 3.31–6.71 | 6.72–14.92 | ≥14.93 | |
| Model 1 | 1 (reference) | 0.97 (0.81–1.16) | 0.85 (0.70–1.04) | 0.92 (0.73–1.16) | 0.67 |
| Model 2 | 1 (reference) | 1.01 (0.90–1.14) | 1.10 (0.98–1.24) | 1.12 (0.97–1.29) | 0.11 |
| Model 3 | 1 (reference) | 1.00 (0.88–1.13) | 1.07 (0.92–1.24) | 1.05 (0.85–1.29) | 0.80 |
| Dimethylarsinate (μg/L) | ≤1.89 | 1.90–3.15 | 3.16–5.36 | ≥5.37 | |
| Model 1 | 1 (reference) | 1.00 (0.80–1.23) | 1.07 (0.84–1.38) | 0.94 (0.71–1.23) | 0.45 |
| Model 2 | 1 (reference) | 1.13 (1.00–1.27) | 1.14 (0.98–1.32) | 1.14 (0.97–1.33) | 0.39 |
| Model 3 | 1 (reference) | 1.11 (0.99–1.26) | 1.11 (0.97–1.28) | 1.10 (0.92–1.30) | 0.80 |
| Total arsenic minus arsenobetaine (μg/L) | ≤2.40 | 2.41–4.82 | 4.83–8.33 | ≥8.34 | |
| Model 1 | 1 (reference) | 0.96 (0.80–1.16) | 0.93 (0.72–1.21) | 0.89 (0.70–1.14) | 0.34 |
| Model 2 | 1 (reference) | 1.03 (0.92–1.16) | 1.15 (0.98–1.34) | 1.08 (0.94–1.25) | 0.38 |
| Model 3 | 1 (reference) | 1.01 (0.89–1.14) | 1.11 (0.94–1.31) | 1.00 (0.85–1.19) | 0.64 |
| Arsenobetaine (μg/L) | ≤0.30 | 0.31–1.16 | 1.17–5.70 | ≥5.71 | |
| Model 1 | 1 (reference) | 1.03 (0.86–1.24) | 1.07 (0.90–1.27) | 1.00 (0.84–1.19) | 0.75 |
| Model 2 | 1 (reference) | 1.02 (0.92–1.12) | 1.10 (0.99–1.21) | 1.16 (1.03–1.30) | 0.03 |
| Model 3 | 1 (reference) | 1.02 (0.92–1.13) | 1.11 (1.00–1.23) | 1.20 (1.05–1.37) | 0.02 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nong, Q.; Zhang, Y.; Guallar, E.; Zhong, Q. Arsenic Exposure and Predicted 10-Year Atherosclerotic Cardiovascular Risk Using the Pooled Cohort Equations in U.S. Hypertensive Adults. Int. J. Environ. Res. Public Health 2016, 13, 1093. https://doi.org/10.3390/ijerph13111093
Nong Q, Zhang Y, Guallar E, Zhong Q. Arsenic Exposure and Predicted 10-Year Atherosclerotic Cardiovascular Risk Using the Pooled Cohort Equations in U.S. Hypertensive Adults. International Journal of Environmental Research and Public Health. 2016; 13(11):1093. https://doi.org/10.3390/ijerph13111093
Chicago/Turabian StyleNong, Qingjiao, Yiyi Zhang, Eliseo Guallar, and Qiuan Zhong. 2016. "Arsenic Exposure and Predicted 10-Year Atherosclerotic Cardiovascular Risk Using the Pooled Cohort Equations in U.S. Hypertensive Adults" International Journal of Environmental Research and Public Health 13, no. 11: 1093. https://doi.org/10.3390/ijerph13111093






