Joint Exposure to Chemical and Nonchemical Neurodevelopmental Stressors in U.S. Women of Reproductive Age in NHANES
Abstract
:1. Introduction
2. Methods
2.1. The National Health and Nutrition Examination Surveys (NHANES) Data Set
2.2. Chemical NDT Stressor Exposure
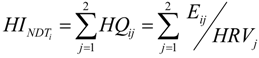
2.3. AL Biomarkers
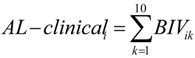
2.4. Data Analysis
3. Results
3.1. Pb and MeHg Concentrations and Joint NDT Exposure
3.2. Elevated Joint NDT Exposure and Chronic Stress
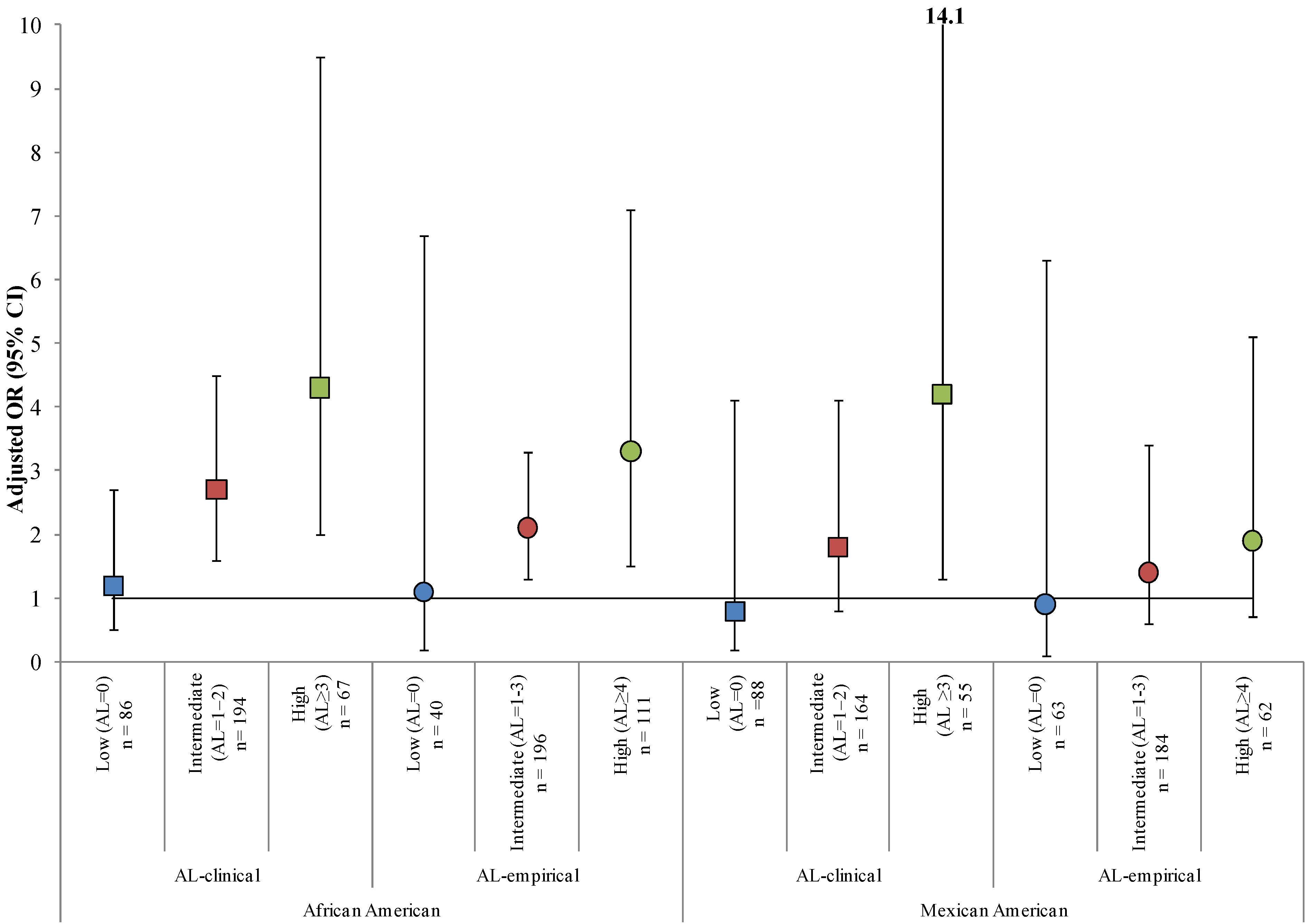
3.3. Effect Measure Modification by AL
3.4. Sensitivity Analyses
| All Women n (Weighted %) | Indicators of Elevated NDT Exposure a | Allostatic Load b | |||||
|---|---|---|---|---|---|---|---|
| HQPb > 1 n (Weighted %) | HQMeHg > 1 n (Weighted %) | HINDT > 1 n (Weighted %) | Low n (Weighted %) | Intermediate n (Weighted %) | High n (Weighted %) | ||
| Total Population | 1,250 | 159 (11) | 19 (2) | 324 (25) | 330 (28) | 632 (54) | 210 (20) |
| Race/Ethnicity | |||||||
| Caucasian | 551 (74) | 46 (9) | 10 (2) | 119 (22) | 156 (28) * | 274 (54) | 88 (19) |
| African American | 373 (15) | 52 (14) | 6 (2) | 104 (32) * | 86 (18) | 194 (55) | 67 (26) * |
| Mexican American | 326 (11) | 61 (22) * | 3 (1) | 101 (36) * | 88 (21) | 164 (56) | 55 (23) |
| Country of Birth | |||||||
| United States | 1,049 (90) | 92 (8) | 15 (2) * | 225 (23) | 278 (25) | 533 (54) | 175 (20) |
| Foreign | 201 (10) | 67 (33) * | 4 (2) | 99 (48) * | 52 (26) | 99 (52) | 35 (22) |
| Age (years) | |||||||
| 15−19 | 520 (16) | 43 (5) | 4 (1) | 79 (11) | 191 (43) | 254 (49) | 40 (8) |
| 20−28 | 252 (26) | 29 (9) | 3 (1) | 59 (20) * | 66 (30) * | 136 (58) * | 31 (12) * |
| 29−44 | 478 (58) | 87 (14) * | 12 (2) | 184 (33) * | 73 (19) * | 242 (54) | 139 (28) * |
| Highest Education c | |||||||
| Less than high school graduate | 327 (15) | 70 (22) * | 4 (1) | 114 (39) | 68 (18) * | 170 (53) | 69 (29) * |
| High school graduate | 302 (27) | 37 (11) | 3 (1) | 67 (21) | 74 (20) * | 152 (53) | 55 (27) * |
| Some college | 383 (35) | 30 (7) | 3 (1) | 75 (19) | 107 (28) * | 197 (56) | 54 (17) |
| College graduate or above | 194 (23) | 17 (8) | 9 (5) * | 57 (31) * | 67 (33) | 90 (54) | 26 (13) |
| Smoking Status (serum cotinine) | |||||||
| Nonsmoker (≤10 ng/mL) | 943 (72) | 100 (8) | 14 (2) | 214 (22) | 270 (27) | 485 (54) | 147 (19) |
| Smoker (>10 ng/mL) | 282 (28) | 56 (18) * | 5 (2) | 105 (37) * | 60 (21) * | 147 (55) | 62 (25) * |
| Iron Status Indicator d | |||||||
| Normal | 870 (74) | 105 (11) | 17 (2) | 235 (28) | 247 (27) * | 449 (55) | 122 (18) * |
| Abnormal | 379 (26) | 54 (11) | 2 (0) | 89 (21) | 83 (19) | 183 (52) | 87 (28) |
| OR (95% CI) | |||||
|---|---|---|---|---|---|
| Univariate | Multivariate | Multivariate by Allostatic Load b | |||
| Low | Intermediate | High | |||
| n = 1,181 | n = 316 | n = 609 | n = 203 | ||
| Race/Ethnicity | n = 1,250 | ||||
| Caucasian | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| African American | 1.7 (1.0, 2.6) | 2.2 (1.4, 3.3) | 1.2 (0.5, 2.7) | 2.7 (1.6, 4.5) | 4.3 (2.0, 9.5) |
| Mexican American | 2.0 (1.3, 3.0) | 1.4 (0.7, 2.6) | 0.8 (0.2, 4.1) | 1.9 (0.9, 4.0) | 4.2 (1.3, 14.1) |
| Country of Birth | n = 1,250 | ||||
| United States | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| Foreign | 3.1 (1.8, 5.4) | 3.3 (1.6, 6.8) | 10.3 (2.5, 43.2) | 2.2 (1.2, 4.3) | 1.8 (0.4, 8.1) |
| Age (years) | n = 1,250 | ||||
| 15−19 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| 20−28 | 2.1 (1.4, 3.2) | 1.8 (1.0, 3.2) | 2.5 (0.9, 7.4) | 1.6 (0.6, 4.2) | 19.1 (1.8, 201.6) |
| 29−44 | 4.1 (3.1, 5.5) | 3.5 (2.4, 5.1) | 6.3 (2.2, 17.7) | 3.9 (1.8, 8.4) | 33.3 (3.6, 305.5) |
| Highest Education c | n= 1,250 | ||||
| Less than high school graduate | 1.4 (0.8, 2.5) | 0.8 (0.5, 1.5) | 1.9 (0.5, 7.6) | 0.7 (0.3, 1.7) | 0.6 (0.1, 2.9) |
| High school graduate | 0.6 (0.3, 1.0) | 0.4 (0.2, 0.6) | 0.6 (0.2, 1.8) | 0.4 (0.2, 0.6) | 0.5 (0.1, 4.3) |
| Some college | 0.5 (0.3, 0.8) | 0.8 (0.5, 1.5) | 0.8 (0.4, 1.7) | 0.2 (0.1, 0.4) | 0.7 (0.2, 2.8) |
| College graduate or above | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| Smoking Status (serum cotinine) | n = 1,225 | ||||
| Nonsmoker (≤10 ng/mL) | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| Smoker (>10 ng/mL) | 2.0 (1.4, 3.0) | 3.0 (1.7, 5.0) | 2.0 (0.8, 4.6) | 3.8 (2.0, 7.4) | 3.4 (1.1, 10.6) |
| Iron Status Indicator d | n = 1,250 | ||||
| Normal | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| Abnormal | 0.7 (0.5, 1.0) | 0.6 (0.5, 0.8) | 1.1 (0.5, 2.4) | 0.5 (0.3, 0.7) | 0.7 (0.3, 1.4) |
4. Discussion
5. Conclusions
Supplementary Files
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lin, C.C.; Chen, Y.C.; Su, F.C.; Lin, C.M.; Liao, H.F.; Hwang, Y.H.; Hsieh, W.S.; Jeng, S.F.; Su, Y.N.; Chen, P.C. In utero exposure to environmental lead and manganese and neurodevelopment at 2 years of age. Environ. Res. 2013, 123, 52–57. [Google Scholar] [CrossRef]
- Jones, E.A.; Wright, J.M.; Rice, G.; Buckley, B.T.; Magsumbol, M.S.; Barr, D.B.; Williams, B.L. Metal exposures in an inner-city neonatal population. Environ. Int. 2010, 36, 649–654. [Google Scholar] [CrossRef]
- Lanphear, B.P.; Hornung, R.; Khoury, J.; Yolton, K.; Baghurst, P.; Bellinger, D.C.; Canfield, R.L.; Dietrich, K.N.; Bornschein, R.; Greene, T. Low-level environmental lead exposure and children’s intellectual function: An international pooled analysis. Environ. Health Persp. 2005, 113, 894–899. [Google Scholar] [CrossRef]
- Grandjean, P.; Weihe, P.; Nielsen, F.; Heinzow, B.; Debes, F.; Budtz-Jorgensen, E. Neurobehavioral deficits at age 7 years associated with prenatal exposure to toxicants from maternal seafood diet. Neurotoxicol. Teratol. 2012, 34, 466–472. [Google Scholar] [CrossRef]
- Thompson, M.R.; Boekelheide, K. Multiple environmental chemical exposures to lead, mercury and polychlorinated biphenyls among childbearing-aged women (NHANES 1999–2004): Body burden and risk factors. Environ. Res. 2013, 121, 23–30. [Google Scholar] [CrossRef]
- Yorifuji, T.; Debes, F.; Weihe, P.; Grandjean, P. Prenatal exposure to lead and cognitive deficit in 7- and 14-year-old children in the presence of concomitant exposure to similar molar concentration of methylmercury. Neurotoxicol. Teratol. 2011, 33, 205–211. [Google Scholar] [CrossRef]
- Bergman, K.; Sarkar, P.; Glover, V.; O’Connor, T.G. Maternal prenatal cortisol and infant cognitive development: Moderation by infant–mother attachment. Biol. Psychiatry 2010, 67, 1026–1032. [Google Scholar] [CrossRef]
- Jones, R.L.; Homa, D.M.; Meyer, P.A.; Brody, D.J.; Caldwell, K.L.; Pirkle, J.L.; Brown, M.J. Trends in blood lead levels and blood lead testing among US children aged 1 to 5 years, 1988–2004. Pediatrics 2009, 123, e376–e385. [Google Scholar] [CrossRef]
- Geronimus, A.T.; Hicken, M.; Keene, D.; Bound, J. “Weathering” and age patterns of allostatic load scores among blacks and whites in the United States. Am. J. Public Health 2006, 96, 826–833. [Google Scholar] [CrossRef]
- Peters, J.L.; Weisskopf, M.G.; Avron Spiro, J.S., III; Sparrow, D.; Nie, H.; Hu, H.; Wright, R.O.; Wright, R.J. Interaction of stress, lead burden, and age on cognition in older men: The VA Normative Aging Study. Environ. Health Persp. 2010, 118, 505–510. [Google Scholar]
- Cory-Slechta, D.A.; Virgolini, M.B.; Thiruchelvam, M.; Weston, D.D.; Bauter, M.R. Maternal stress modulates the effects of developmental lead exposure. Environ. Health Persp. 2004, 112, 717–730. [Google Scholar] [CrossRef]
- Rossi-George, A.; Virgolini, M.; Weston, D.; Thiruchelvam, M.; Cory-Slechta, D. Interactions of lifetime lead exposure and stress: Behavioral, neurochemical and HPA axis effects. Neurotoxicology 2011, 32, 83–99. [Google Scholar] [CrossRef]
- Rabinowitz, M.B.; Wetherill, G.; Kopple, J.D. Kinetic analysis of lead metabolism in healthy humans. J. Clin. Invest. 1976, 58, 260–270. [Google Scholar] [CrossRef]
- Smith, J.; Farris, F. Methyl mercury pharmacokinetics in man: A reevaluation. Toxicol. Appl. Pharmacol. 1996, 137, 245–252. [Google Scholar] [CrossRef]
- Hellhammer, D.; Stone, A.; Hellhammer, J.; Broderick, J. Measuring Stress. In Encyclopedia of Behavioural Neuroscience; Koob, G., Moal, M.L., Thompson, R., Eds.; Academic Press: Oxford, UK, 2010; pp. 186–191. [Google Scholar]
- McEwen, B.S. Protective and damaging effects of stress mediators. N. Engl. J. Med. 1998, 338, 171–179. [Google Scholar] [CrossRef]
- Sterling, P.; Eyer, J. Allostasis: A New Paradigm to Explain Arousal Pathology. In Handbook of Life Stress, Cognition and Health; Fisher, S., Reason, J., Eds.; Wiley: New York, NY, USA, 1988; pp. 629–649. [Google Scholar]
- Centers for Disease Control and Prevention (CDC). National Health and Nutrition Examination Survey: Public Data General Release File Documentation; CDC National Center for Health Statistics: Atlanta, GA, USA, 2006.
- Centers for Disease Control and Prevention (CDC). Laboratory Procedure Manual: Lead, Cadmium, and Mercury in Whole Blood; CDC National Center for Environmental Health: Atlanta, GA, USA, 2004.
- U.S. Environmental Protection Agency. Supplementary Guidance for Conducting Health Risk Assessment of Chemical Mixtures; EPA: Washington, DC, USA, 2000.
- Mahaffey, K.R.; Clickner, R.P.; Bodurow, C.C. Blood organic mercury and dietary mercury intake: National Health and Nutrition Examination Survey, 1999 and 2000. Environ. Health Persp. 2004, 112, 562–570. [Google Scholar] [CrossRef]
- U.S. Environmental Protection Agency (EPA). Methylmercury (MeHg) (CASRN 22967-92-6). In Integrated Risk Information System (IRIS); EPA: Washington, DC, USA, 2001. [Google Scholar]
- Jedrychowski, W.; Perera, F.; Jankowski, J.; Mrozek-Budzyn, D.; Mroz, E.; Flak, E.; Edwards, S.; Skarupa, A.; Lisowska-Miszczyk, I. Gender specific differences in neurodevelopmental effects of prenatal exposure to very low-lead levels: The prospective cohort study in three-year olds. Early Hum. Dev. 2009, 85, 503–510. [Google Scholar] [CrossRef]
- Amaral, J.H.; Rezende, V.B.; Quintana, S.M.; Gerlach, R.F.; Barbosa, F.; Tanus-Santos, J.E. The relationship between blood and serum lead levels in peripartum women and their respective umbilical cords. Basic Clin. Pharmacol. Toxicol. 2010, 107, 971–975. [Google Scholar] [CrossRef]
- Carlisle, J.C.; Dowling, K.C.; Siegel, D.M.; Alexeeff, G.V. A blood lead benchmark for assessing risks from childhood lead exposure. J. Environ. Sci. Health. A 2009, 44, 1200–1208. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention (CDC). What Do Parents Need to Know to Protect. Their Children? CDC: Atlanta, GA, USA, 2012.
- Juster, R.P.; McEwen, B.S.; Lupien, S.J. Allostatic load biomarkers of chronic stress and impact on health and cognition. Neurosci. Biobehav. Rev. 2010, 35, 2–16. [Google Scholar] [CrossRef]
- Chyu, L.; Upchurch, D.M. Racial and ethnic patterns of allostatic load among adult women in the United States: Findings from the National Health and Nutrition Examination Survey 1999–2004. J. Womens Health 2011, 20, 575–583. [Google Scholar] [CrossRef]
- Crimmins, E.M.; Johnston, M.; Hayward, M.; Seeman, T. Age differences in allostatic load: An index of physiological dysregulation. Exp. Gerontol. 2003, 38, 731–734. [Google Scholar] [CrossRef]
- Hawkley, L.C.; Lavelle, L.A.; Berntson, G.G.; Cacioppo, J.T. Mediators of the relationship between socioeconomic status and allostatic load in the Chicago Health, Aging, and Social Relations Study (CHASRS). Psychophysiology 2011, 48, 1134–1145. [Google Scholar] [CrossRef]
- Axelrad, D.A.; Cohen, J. Calculating summary statistics for population chemical biomonitoring in women of childbearing age with adjustment for age-specific natality. Environ. Res. 2011, 111, 149–155. [Google Scholar] [CrossRef]
- Fortin, M.C.; Cory-Slechta, D.A.; Ohman-Strickland, P.; Nwankwo, C.; Yanger, T.S.; Todd, A.C.; Moynihan, J.; Walton, J.; Brooks, A.; Fiedler, N. Increased lead biomarker levels are associated with changes in hormonal response to stress in occupationally exposed male participants. Environ. Health Persp. 2012, 120, 278–283. [Google Scholar]
- Geer, L.A.; Persad, M.D.; Palmer, C.D.; Steuerwald, A.J.; Dalloul, M.; Abulafia, O.; Parsons, P.J. Assessment of prenatal mercury exposure in a predominately Caribbean immigrant community in Brooklyn, NY. J. Environ. Monit. 2012, 14, 1035–1043. [Google Scholar] [CrossRef]
- Agency for Toxic Substances and Disease Registry (ATSDR). Interaction Profile for: Chlorpyrifos, Lead, Mercury, and Methylmercury; ATSDR: Atlanta, GA, USA, 2006.
- Aylward, L.L.; Kirman, C.R.; Schoeny, R.; Portier, C.J.; Hays, S.M. Evaluation of biomonitoring data from the CDC national exposure report in a risk assessment context: Perspectives across chemicals. Environ. Health Persp. 2013, 121, 287–294. [Google Scholar]
- U.S. Environmental Protection Agency (EPA). Integrated Risk Information System (IRIS); EPA: Washington, DC, USA, 2013.
- DeFur, P.L.; Evans, G.W.; Cohen Hubal, E.A.; Kyle, A.D.; Morello-Frosch, R.A.; Williams, D.R. Vulnerability as a function of individual and group resources in cumulative risk assessment. Environ. Health Persp. 2007, 115, 817–824. [Google Scholar] [CrossRef]
- Nelson, J.W.; Scammell, M.K.; Hatch, E.E.; Webster, T.F. Social disparities in exposures to bisphenol A and polyfluoroalkyl chemicals: A cross-sectional study within NHANES 2003–2006. Environ. Health 2012, 11, 1–15. [Google Scholar] [CrossRef]
- Crimmins, E.M.; Kim, J.K.; Alley, D.E.; Karlamangla, A.; Seeman, T. Hispanic paradox in biological risk profiles. Am. J. Public Health 2007, 97, 1305–1310. [Google Scholar] [CrossRef]
- Martin, J.; Hamilton, B.; Sutton, P.; Ventura, S.; Menacker, F.; Munson, M. Births: Final Data for 2003; National Center for Health Statistics: Hyattsville, MD, USA, 2005. [Google Scholar]
- Hertz-Picciotto, I.; Schramm, M.; Watt-Morse, M.; Chantala, K.; Anderson, J.; Osterloh, J. Patterns and determinants of blood lead during pregnancy. Am. J. Epidemiol. 2000, 152, 829–837. [Google Scholar] [CrossRef]
- Woodruff, T.J.; Zota, A.R.; Schwartz, J.M. Environmental chemicals in pregnant women in the United States: NHANES 2003–2004. Environ. Health Persp. 2011, 119, 878–885. [Google Scholar] [CrossRef]
- Kudielka, B.M.; Wust, S. Human models in acute and chronic stress: assessing determinants of individual hypothalamus-pituitary-adrenal axis activity and reactivity. Stress 2010, 13, 1–14. [Google Scholar] [CrossRef]
- Morello-Frosch, R.; Shenassa, E.D. The environmental “riskscape” and social inequality: Implications for explaining maternal and child health disparities. Environ. Health Persp. 2006, 114, 1150–1153. [Google Scholar] [CrossRef]
- Morrison, S.; Shenassa, E.D.; Mendola, P.; Wu, T.; Schoendorf, K. Allostatic load may not be associated with chronic stress in pregnant women, NHANES 1999–2006. Ann. Epidemiol. 2013, 23, 294–297. [Google Scholar] [CrossRef]
- Wallace, M.E.; Harville, E.W. Allostatic load and birth outcomes among white and black women in New Orleans. Matern Child Health J. 2013, 17, 1025–1029. [Google Scholar] [CrossRef]
- Clougherty, J.E.; Levy, J.I.; Kubzansky, L.D.; Ryan, P.B.; Suglia, S.F.; Canner, M.J.; Wright, R.J. Synergistic effects of traffic-related air pollution and exposure to violence on urban asthma etiology. Environ. Health Persp. 2007, 115, 1140–1146. [Google Scholar] [CrossRef]
- Chen, E.; Schreier, H.M.; Strunk, R.C.; Brauer, M. Chronic traffic-related air pollution and stress interact to predict biologic and clinical outcomes in asthma. Environ. Health Persp. 2008, 116, 970–975. [Google Scholar] [CrossRef]
- Shankardass, K.; McConnell, R.; Jerrett, M.; Milam, J.; Richardson, J.; Berhane, K. Parental stress increases the effect of traffic-related air pollution on vhildhood ssthma incidence. Proc. Natl. Acad. Sci. USA. 2009, 106, 12406–12411. [Google Scholar] [CrossRef]
- Hicken, M.T.; Gee, G.C.; Connell, C.; Snow, R.C.; Morenoff, J.; Hu, H. Black-white blood pressure disparities: Depressive symptoms and differential vulnerability to blood lead. Environ. Health Persp. 2013, 121, 205–209. [Google Scholar]
- Read, S.; Grundy, E. Allostatic Load—A Challenge to Measure Multisystem Physiological Dysregulation. Pathways Node at NCRM; National Centre for Research Methods: Oxford, UK, 2012. [Google Scholar]
- Seplaki, C.L.; Goldman, N.; Glei, D.; Weinstein, M. A comparative analysis of measurement approaches for physiological dysregulation in an older population. Exp. Gerontol. 2005, 40, 438–449. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Evans, A.M.; Rice, G.E.; Teuschler, L.K.; Wright, J.M. Joint Exposure to Chemical and Nonchemical Neurodevelopmental Stressors in U.S. Women of Reproductive Age in NHANES. Int. J. Environ. Res. Public Health 2014, 11, 4384-4401. https://doi.org/10.3390/ijerph110404384
Evans AM, Rice GE, Teuschler LK, Wright JM. Joint Exposure to Chemical and Nonchemical Neurodevelopmental Stressors in U.S. Women of Reproductive Age in NHANES. International Journal of Environmental Research and Public Health. 2014; 11(4):4384-4401. https://doi.org/10.3390/ijerph110404384
Chicago/Turabian StyleEvans, Amanda M., Glenn E. Rice, Linda K. Teuschler, and J. Michael Wright. 2014. "Joint Exposure to Chemical and Nonchemical Neurodevelopmental Stressors in U.S. Women of Reproductive Age in NHANES" International Journal of Environmental Research and Public Health 11, no. 4: 4384-4401. https://doi.org/10.3390/ijerph110404384




