1. Introduction
Itai-itai disease is the most severe form of chronic cadmium (Cd) toxicosis, originally discovered in the Jinzu River basin (Toyama Prefecture, Japan); it is characterized by osteomalacia, severe osteoporosis and renal tubular dysfunction [
1]. Studies of itai-itai disease patients and residents of Cd-polluted areas in Japan showed increased low-molecular weight proteins such as α
1-microglobulin (α
1-MG), β
2-microglobulins (β
2-MG) and retinol binding protein (RBP), enzymuria such as N-acetyl-β-
d-glucosamidinase (NAG) and glucosuria due to renal tubular dysfunction [
2,
3]. Increased amino acids in the urine are also found among Japanese residents of the Cd-polluted areas, and the total amino acids and proline in urine have been used as biomarkers of renal tubular dysfunction [
1,
2,
3].
In 2003, environmental Cd contamination has been discovered in the Mae Sot District of Tak Province, located in the northwestern region of Thailand [
4,
5]. The source of the contamination is believed to be a zinc-rich area located north of the irrigation creeks where a zinc mine had been operated for more than 20 years [
4]. The paddy fields in 12 villages of the Mae Sot District were found to contain markedly elevated Cd, and the urinary Cd levels showed that the Cd body burden was high in these residents [
5]. Teeyakasem
et al. reported that increased Cd levels in the urine were associated with increased levels of renal tubular dysfunction biomarkers such as urinary β
2-MG, α
1-MG and NAG [
6]. Swaddiwudhipong
et al. also reported an increased serum creatinine (Cr) and decreased glomerular filtration rate (GFR) associated with increased urinary Cd in the inhabitants with urinary Cd ≥ 5 μg/g Cr [
5], which the WHO has suggested to be indicative of Cd contamination [
7]. Dose-response relationships of urinary β
2-MG and NAG with urinary Cd were found, suggesting that renal tubular dysfunction was caused by Cd exposure in this area [
8]. Moreover, Nambunmee
et al. reported increased biomarkers of bone resorption associated with decreased calcium reabsorption, indicating the influence of Cd exposure on bone effects in the Thai population [
9]. However, the most characteristic health effect in the present Thai Cd-polluted area is a high prevalence of urinary tract stones related to increased urinary Cd [
10,
11], a phenomenon that has not been reported in Cd-exposed residents in other countries.
Recently, metabolomics analysis has been performed to identify the alteration of metabolites in the affected people exposed to environmental chemicals and toxicants, and indicate specific biomarkers for early detection [
12]. Urinary metabolomics study is the simultaneous analysis of different classes of metabolite in urine, such as organic acids, amino acid, sugar and sugar alcohol, to be able to compare metabolic profile of exposed cases and controls [
13,
14]. There are some methods to detect metabolites in urine using different techniques such as gas chromatography-mass spectrometry (GC-MS), liquid chromatography-mass spectrometry (LC-MS), and nuclear magnetic resonance-spectrometry (NMR). Particularly, metabolomics analysis using GC-MS is good at quantification of sugar, organic acids and amino acids in urine [
13], which are often observed among residents in Cd polluted area [
1,
2,
3].
Therefore, we firstly performed a metabolomics analysis based on GC-MS to identify specific biomarker candidates for Cd toxicosis cases in Thailand. Then, as the next step, we investigated the relationships between the biomarker found by metabolomics and urinary Cd among exposed residents in the Mae Sot area to confirm the association between the level of the detected biomarker and Cd exposure in a Thai population.
2. Experimental Section
2.1. Study Subjects
A total of 168 residents (83 men and 85 women; mean age 61.6 years [range: 47–86 years]) living in Cd-polluted Mae Sot in the Tak province were selected from 700 subjects who participated in a health impact survey conducted in 2007 [
9,
15]. The subjects in the Cd-polluted area consisted of two groups, according to the levels of β
2-MG and Cd in urine collected in 2007: (1) the Cd toxicosis group (47 men and 37 women) with marked renal tubular dysfunction (β
2-MG ≥ 1,000 μg/g Cr) and high Cd exposure (urinary Cd ≥ 5 μg/g Cr) and (2) the normal function group (36 men and 48 women) with no clear renal dysfunction (β
2-MG < 300 μg/g Cr) and low Cd exposure (urinary Cd < 5 μg/g Cr). In 2012, we collected morning urine specimens from these 169 residents, measured the Cd, β
2-MG and NAG levels in urine, and selected ten typical Cd toxicosis cases (four men and six women) for metabolomics analysis based on the following criteria: Cd ≥ 5 μg/g Cr, β
2-MG ≥ 1,000 μg/g Cr and NAG ≥ 8 U/g Cr in the urine collected in 2012.
In 2012, a total of 100 residents (50 men and 50 women) living in a non-polluted area in the same district were recruited as controls. The mean age of control subjects was 61.0 years (range: 43–87 years). The control subjects underwent the same urinary analysis as the exposed subjects; from this group, age- and gender-matched 10 controls were selected for metabolomics analysis based on the following criteria: no history of renal disease or diabetes mellitus, Cd < 2 μg/g Cr, β
2-MG < 300 μg/g Cr and NAG < 8 U/g Cr in the urine in 2012. The mean values with standard deviation (SD) of age, body size, urinary Cd, renal markers and anemia markers and the prevalence of hypertension, diabetes mellitus and nephrolithiasis for typical Cd toxicosis cases and metabolomics controls are shown in
Table 1. No significant differences in age, BMI or prevalence of common diseases were found between the cases and controls, but the levels of Cd, β
2-MG, NAG and amino acids in the urine, serum Cr and anemia markers for the Cd toxicosis cases were significantly higher than those for the controls.
As the second step after identifying the specific biomarker candidate to Cd toxicosis by metabolomics analysis, the biomarker was measured in urine samples taken in 2012 from all 169 residents in the Cd-polluted area and from the 100 controls in a non-polluted area. The association between the urinary Cd and new biomarker levels was investigated to confirm that the new biomarker is a good indicator of the health effects induced by Cd exposure.
The study protocol was approved by the Research Ethical Committee of the Faculty of Medicine, Chiang Mai University (Approval No. 004/2012). Because the subjects were selected before the survey, we used the data from a previous survey conducted in 2007 by a research team including medical staffs of Mae Sot General Hospital, researchers of Chiang Mai University and Kanazawa Medical University. Before the survey was performed, a medical doctor and medical staffs of Mae Sot General Hospital informed the participants about the study objectives and asked them for their consent to be enrolled in this study.
Table 1.
Clinical characteristics of cadmium toxicosis cases and controls in a non-polluted area.
Table 1.
Clinical characteristics of cadmium toxicosis cases and controls in a non-polluted area.
| | Control (N = 10) | Cd Toxicosis Cases (N = 10) | |
|---|
| | Mean | SD | Min–Max | Mean | SD | Min–Max | P-Value |
|---|
| Age | years | 67 | 8.6 | 48–77 | 71 | 10.6 | 49–82 | NS |
| Gender ratio | Men/Women | 4/6 | | | 4/6 | | | |
| Height | cm | 151.6 | 4.9 | 143–159 | 149.5 | 8.7 | 137–168 | NS |
| Weight | kg | 56.3 | 6.7 | 45–65 | 47.4 | 11.9 | 29–65 | NS |
| BMI | kg/m2 | 23.8 | 3.1 | 18.0–28.9 | 21.3 | 5.4 | 12.1–31.8 | NS |
| Urinary Cd # | μg/g Cr | 0.87 | 1.6 | 0.43–2.1 | 11.5 | 1.2 | 9.2–18.7 | *** |
| Urinary β2-MG # | μg/g Cr | 132 | 1.7 | 62–278 | 33,266 | 2.3 | 10,366–138,413 | *** |
| Urinary NAG # | U/g Cr | 4.1 | 1.4 | 2.6–6.5 | 14.5 | 1.5 | 9.5–29.4 | *** |
| Urinary amino acids # | μg/g Cr | 96 | 1.3 | 70.9–171.9 | 124 | 1.2 | 99.6–167.4 | * |
| Urinary proline # | μg/g Cr | 4.1 | 1.4 | 2.3–6.6 | 5.8 | 1.6 | 2.8–11.5 | NS |
| Serum Cr | mg/dl | 0.93 | 0.18 | 0.6–1.2 | 1.62 | 0.7 | 1.1–3.4 | * |
| RBC | ×10,000 | 508 | 44 | 434–599 | 406 | 56 | 333–514 | *** |
| Hb | g/dl | 13.5 | 1.7 | 11.3–16.3 | 11.5 | 1.1 | 10.2–13.6 | ** |
| Ht | % | 42.2 | 4.5 | 36.4–50.3 | 35.1 | 3.2 | 31.1–41.0 | ** |
| Hypertension | N(%) | 5 | (50) | | 6 | (60) | | NS |
| Diabetes meritus | N(%) | 0 | (0) | | 0 | ( 0) | | NS |
| Nephrolithiasis | N(%) | 1 | (10) | | 3 | (30) | | NS |
2.2. Urine Collection and the Measurement of Renal and Exposure Markers
Approximately 25–30 mL of urine was collected from each subject in the early morning before breakfast. The urine pH was measured immediately using a pH indicator strip (Merck, Darmstadt, Germany) and adjusted to ≥pH 6 to avoid the degradation of β2-MG in acidic urine. The urine specimens were transported in a cool container and frozen at −20 °C in the hospital laboratory within 3 hours (average 2.5 h) after collection. To measure renal and exposure markers, the samples were transported to Kanazawa Medical University, Japan in a container with dry ice and kept frozen until analysis.
Urinary Cd was analyzed using a graphite furnace atomic-absorption spectrometer (Shimadzu AA 6300, Kyoto, Japan). The analytical techniques were validated, and the quality assurance of the analysis used urine standard reference material No. 2670 (The National Institute of Standards, Washington, DC, USA) as a quality control. The concentration of urinary β
2-MG was measured via enzyme immunoassay using a latex agglutination immunoassay (Eiken Chemical, Tokyo, Japan); urinary NAG was measured via a colorimetric assay using the NAG test kit (Shionogi Pharmaceuticals, Osaka, Japan). Urinary calcium (Ca), magnesium (Mg) and phosphorus (P) were determined via colorimetric assays (OCPC for Ca, xylidyl blue method for Mg and phosphomolybdic acid method for P; Clinimate test kit, Sekisui Medical Ltd., Tokyo, Japan) using an automated analyzer (BioMajesty JCA-BM1650, JEOL Ltd., Akishima, Japan). The urinary total amino acids and proline of the typical cases and their age- and gender-matched controls were measured using the colorimetric assay developed by Fukushima
et al. [
16]. All urinary markers were corrected by the urinary Cr concentrations, as determined by the kinetic measurement based on the Jeffe reaction.
2.4. Measurements of Detected Biomarker and Minerals in Urine for All Samples on the Second Step of the Study
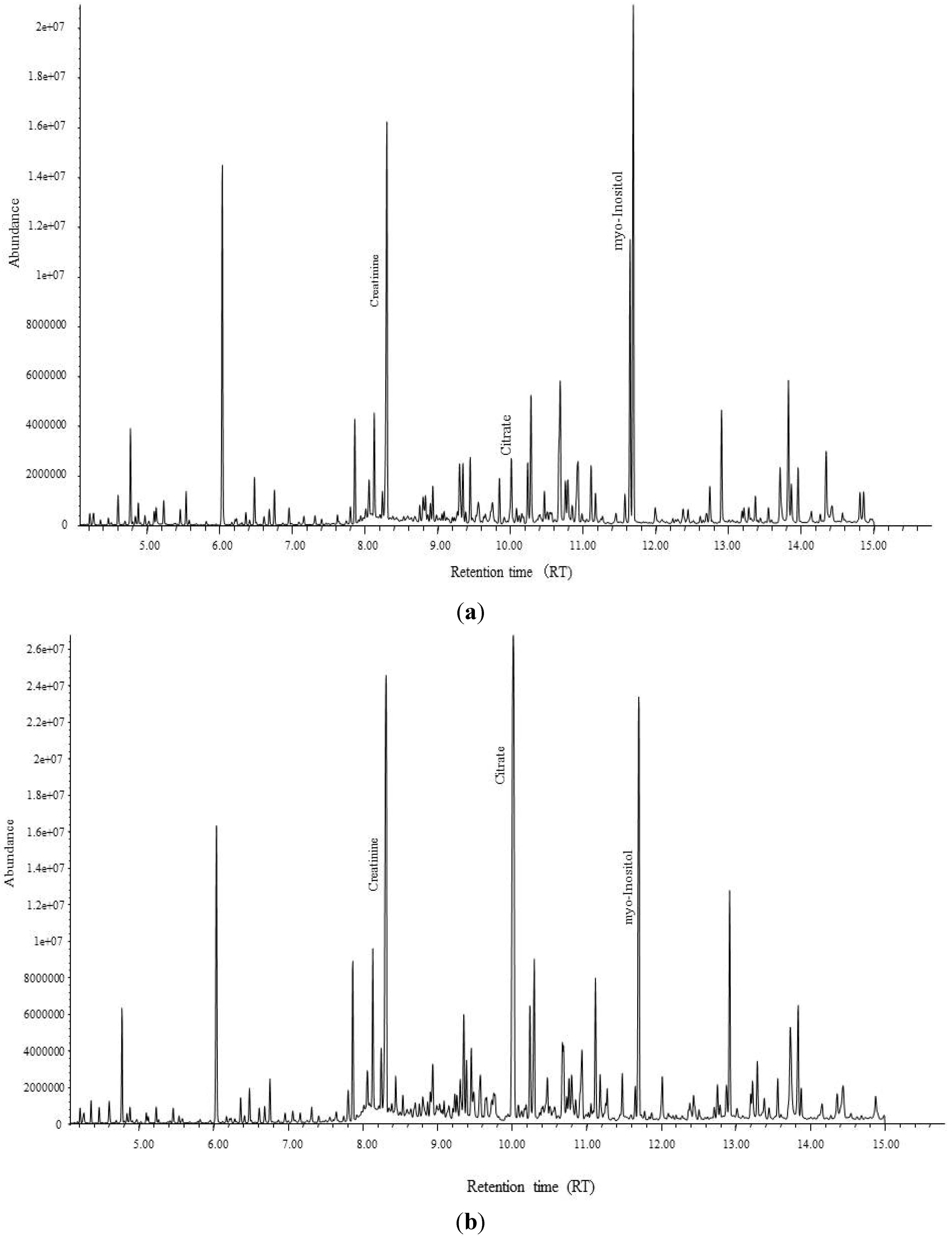
Because urinary citrate was detected as a specific biomarker candidate for the health effects of Cd pollution in the Thai population, urinary citrate was measured using an enzymatic method (F-kit Citrate, Roche Diagnostics GmbH, Wibringen, Germany) in all subjects living in Cd-polluted and non-polluted areas. The ratios of Ca to citrate and Mg to citrate were calculated and used to determine the risk factors related to urinary stone formation.
2.5. Data Analysis
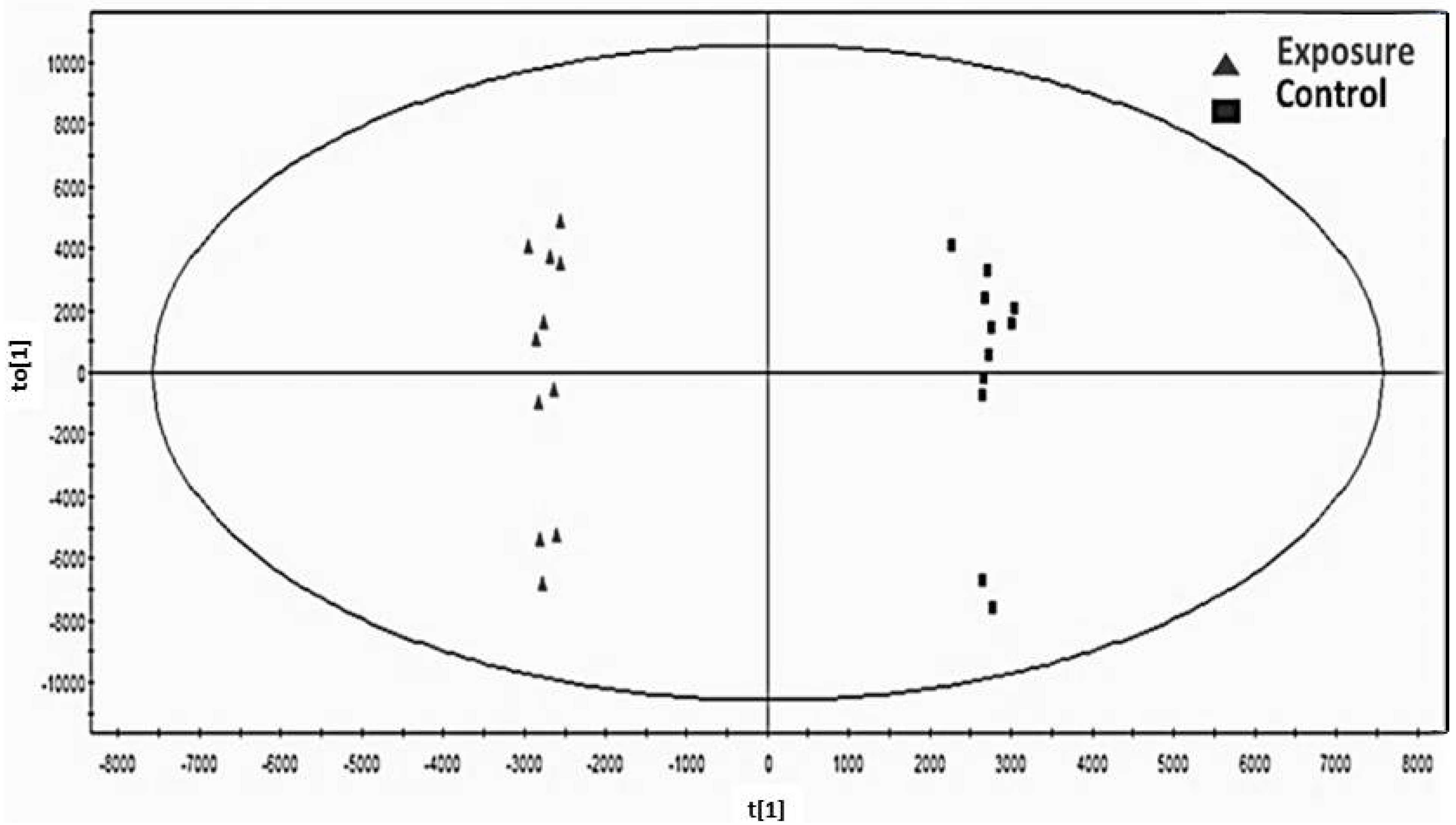
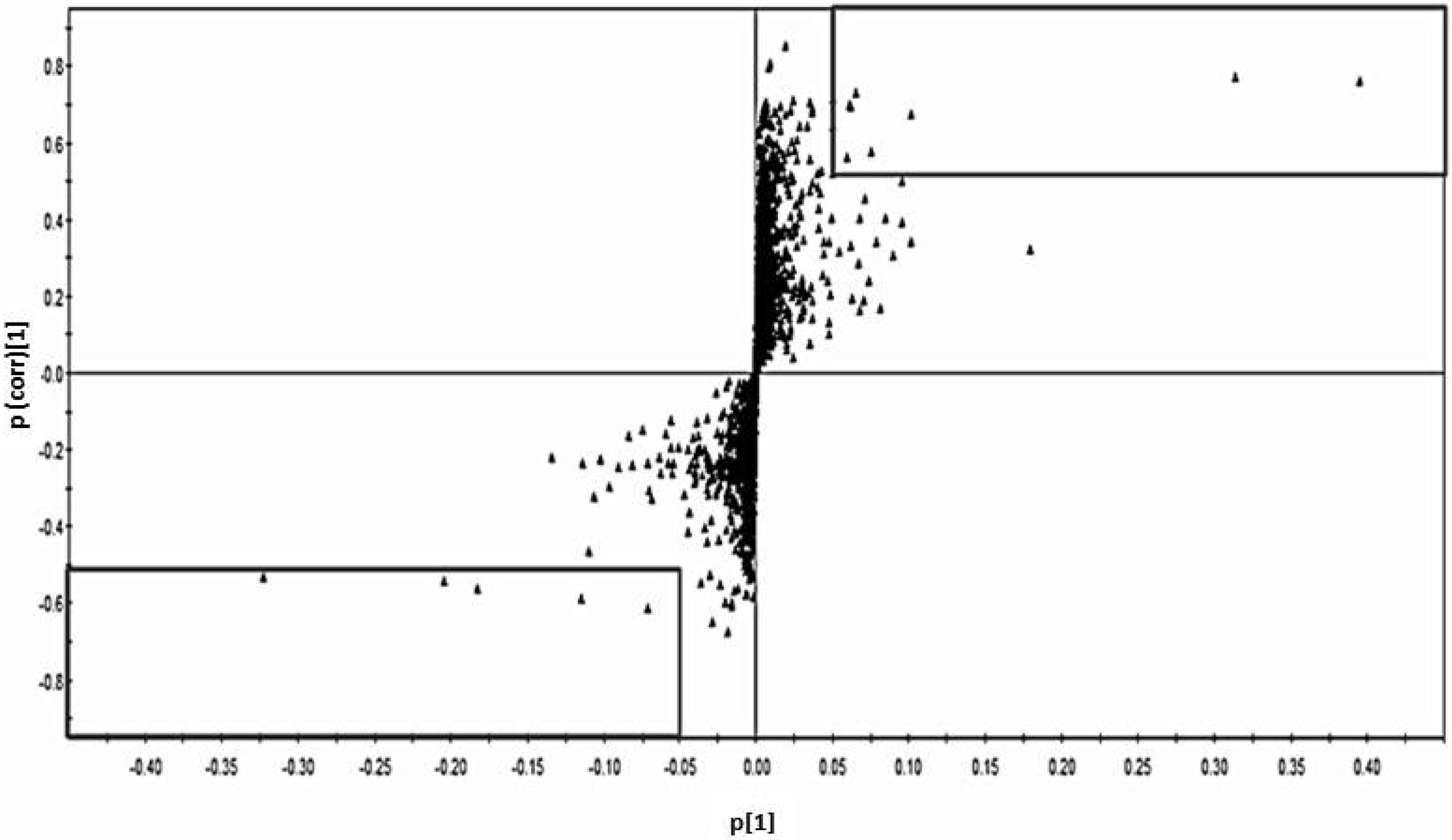
The general characteristic markers of the subjects and the quantification measurements of the metabolites determined with GC-MS were compared between the Cd-exposed and control groups using the paired t-test. The difference in the new biomarker and related factors among three Cd exposure categories were compared using one-way ANOVA. The relationships among urinary Cd, renal markers and the candidate metabolite were analyzed using Spearman’s correlation analysis and a linear regression model after adjusting for covariates. These statistical analyses above were performed using Statistical Package SPSS, Version 15 (IBM Inc., New York, NY, USA). To identify a metabolite candidate that could differentiate Cd toxicosis cases from controls, orthogonal partial least squares discriminate analysis (OPLS-DA) was performed using SIMCA-P+ software, Version 12 (Umetrics, Umea, Sweden). Score-plots (S-plots) for the separation of case-control data and variable importance (VIP) for the confirmation of the importance or power of the selected candidates were used to select metabolite candidates.
 ) and control group (■) with no variation between two groups.
) and control group (■) with no variation between two groups.
 ) and control group (■) with no variation between two groups.
) and control group (■) with no variation between two groups.