Maize Purple Plant Pigment Protects Against Fluoride-Induced Oxidative Damage of Liver and Kidney in Rats
Abstract
:1. Introduction
2. Experimental Section
2.1. Chemicals and Reagents
2.2. Animals and Treatment
2.3. Determination of Fluoride
2.4. Organo-Somatic Index
2.5. Ultrastructure of Liver and Kidney
2.6. Determination of Serum and Tissue Oxidative Stress Markers
2.7. Determination of Bax and Bcl-2 Protein Expressions in Liver and Kidney of Rats by Western Blot Analysis
2.8. Statistical Analysis
3. Results
3.1. Distribution of Fluoride in Blood, Urine, Liver and Kidney
| Groups | N | Serum (μg/mL) | Urine (μg/mL) | Liver (μg/kg) | Kidney (μg/kg) |
|---|---|---|---|---|---|
| Group I | 10 | 0.098 ± 0.007 | 3.501 ± 0.404 | 198.20 ± 37.15 | 284.53 ± 44.96 |
| Group II | 10 | 0.391 ± 0.014 * | 26.559 ± 2.887 * | 581.03 ± 64.63 * | 914.32 ± 125.26 * |
| Group III | 10 | 0.370 ± 0.031 * | 26.014 ± 1.986 * | 611.08 ± 81.03 * | 923.42 ± 107.86 * |
| Group IV | 10 | 0.378 ± 0.027 * | 23.603 ± 2.018 * | 509.67 ± 75.47 * | 882.83 ± 83.55 * |
3.2. Organo-Somatic Index (OSI) of Liver and Kidney
| Groups | N | OSI of Liver | OSI of Two Kidneys |
|---|---|---|---|
| Group I | 20 | 2.58 ± 0.05 | 0.72 ± 0.01 |
| Group II | 20 | 2.61 ± 0.04 | 0.68 ± 0.01 * |
| Group III | 20 | 2.65 ± 0.05 | 0.70 ± 0.01 |
| Group IV | 20 | 2.61 ± 0.03 | 0.72 ± 0.01 # |
3.3. Ultrastructure Observation of Liver and Kidney

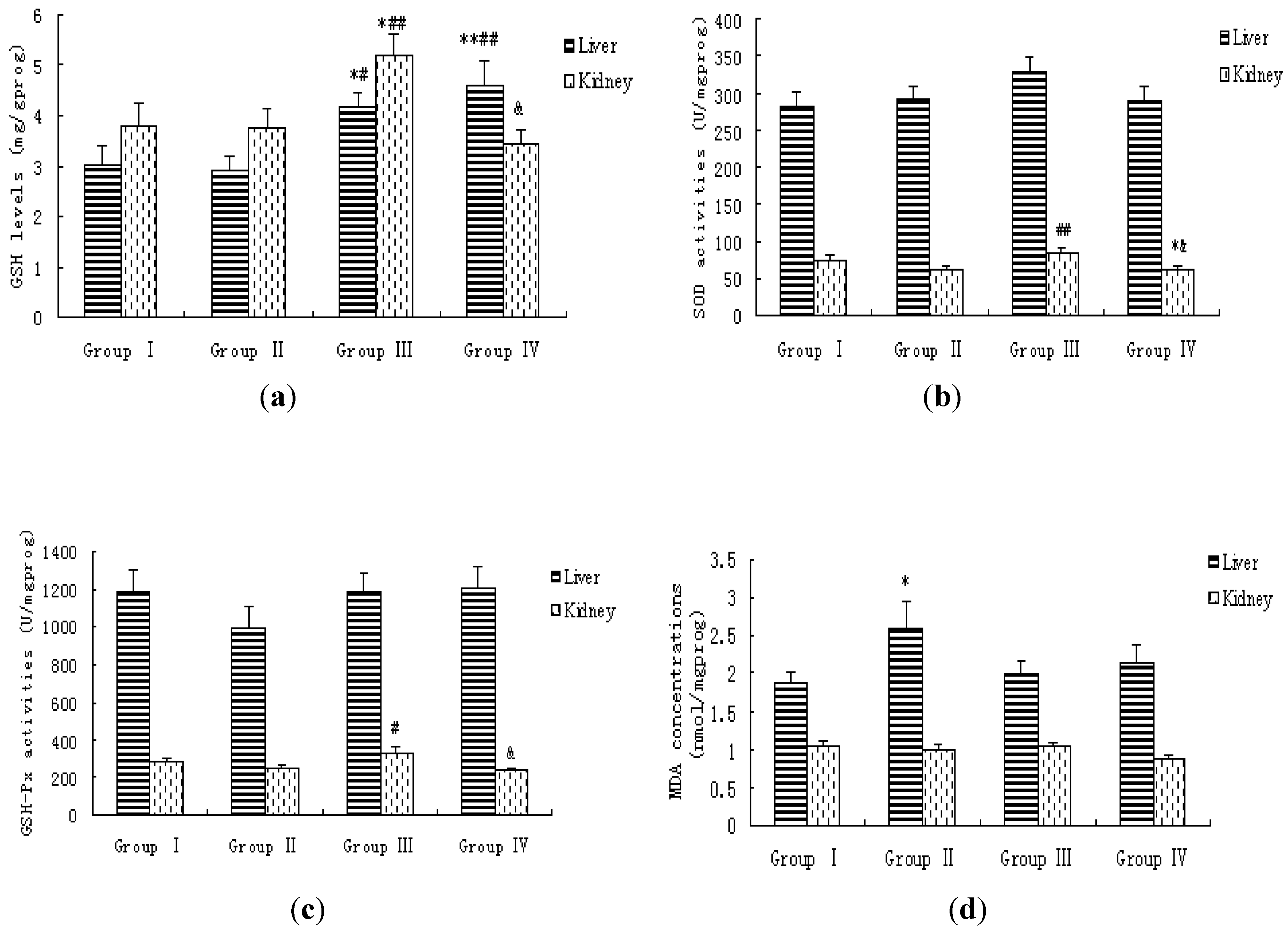
3.4. Lipid Peroxidation Levels and Antioxidative Status in Blood, Liver and Kidney of Rats
| Groups | N | T-AOC | T-SOD | GSH | GSH-Px | MDA |
|---|---|---|---|---|---|---|
| (U/mL) | (U/mL) | (mg/L) | (U/mL) | (nmol/mL) | ||
| Group I | 20 | 6.64 ± 0.31 | 133.72 ± 3.67 | 12.99 ± 1.08 | 1311.27 ± 54.39 | 3.34 ± 0.24 |
| Group II | 20 | 6.54 ± 0.30 | 133.01 ± 4.03 | 13.34 ± 0.98 | 1295.68 ± 53.85 | 4.27 ± 0.37 * |
| Group III | 20 | 6.55 ± 0.51 | 136.20 ± 4.92 | 11.44 ± 0.96 | 1462.25 ± 76.25 # | 3.19 ± 0.18 ## |
| Group IV | 20 | 7.02 ± 0.35 | 139.92 ± 4.62 | 14.17 ± 1.59 | 1230.43 ± 47.24 & | 3.53 ± 0.34 |
3.5. Bax and Bcl-2 Expressions in Liver and Kidney of Rats

4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lu, J.; Chen, H.; Xu, Q.; Zheng, J.; Liu, H.; Li, J.; Chen, K. Comparative proteomics of kidney samples from puffer fish Takifugu rubripes exposed to excessive fluoride: An insight into molecular response to fluorosis. Toxicol. Mech. Methods 2010, 20, 345–354. [Google Scholar] [CrossRef]
- Shashi, A.; Bhardwaj, M. Study on blood biochemical diagnostic indices for hepatic function biomarkers in endemic skeletal fluorosis. Biol. Trace Elem. Res. 2011, 143, 803–814. [Google Scholar] [CrossRef]
- Varol, E.; Icli, A.; Aksoy, F.; Bas, H.A.; Sutcu, R.; Ersoy, I.H.; Varol, S.; Ozaydin, M. Evaluation of total oxidative status and total antioxidant capacity in patients with endemic fluorosis. Toxicol. Ind. Health 2013, 29, 175–180. [Google Scholar]
- Barbier, O.; Arreola-Mendoza, L.; Del-Razo, L.M. Molecular mechanisms of fluoride toxicity. Chem. Biol. Interact. 2010, 188, 319–333. [Google Scholar] [CrossRef]
- Grucka-Mamczar, E.; Birkner, E.; Zalejska-Fiolka, J.; Machoy, Z.; Kasperczyk, S.; Błaszczyk, I. Influence of extended exposure to sodium fluoride and caffeine on the activity of carbohydrate metabolism enzymes in rat blood serum and liver. Fluoride 2007, 40, 62–66. [Google Scholar]
- He, L.F.; Chen, J.G. DNA damage, apoptosis and cell cycle changes induced by fluoride in rat oral mucosal cells and hepatocytes. World J. Gastroenterol. 2006, 12, 1144–1148. [Google Scholar]
- Kono, K.; Yoshida, Y.; Yamagata, H.; Watanabe, M.; Shibuya, Y.; Doi, K. Urinary fluoride monitoring of industrial hydrofluoric acid exposure. Environ. Res. 1987, 42, 415–420. [Google Scholar] [CrossRef]
- Kobayashi, C.A.; Leite, A.L.; Silva, T.L.; Santos, L.D.; Nogueira, F.C.; Oliveira, R.C.; Palma, M.S.; Domont, G.B.; Buzalaf, M.A. Proteomic analysis of kidney in rats chronically exposed to fluoride. Chem. Biol. Interact. 2009, 180, 305–311. [Google Scholar] [CrossRef]
- Xiong, X.; Liu, J.; He, W.; Xia, T.; He, P.; Chen, X.; Yang, K.; Wang, A. Dose-effect relationship between drinking water fluoride levels and damage to liver and kidney functions in children. Environ. Res. 2007, 103, 112–116. [Google Scholar] [CrossRef]
- Hassan, H.A.; Abdel-Aziz, A.F. Evaluation of free radical-scavenging and anti-oxidant properties of black berry against fluoride toxicity in rats. Food Chem. Toxicol. 2010, 48, 1999–2004. [Google Scholar]
- Nabavi, S.M.; Nabavi, S.F.; Habtemariam, S.; Moghaddam, A.H.; Latifi, A.M. Ameliorative effects of quercetin on sodium fluoride-induced oxidative stress in rat’s kidney. Ren. Fail. 2012, 34, 901–906. [Google Scholar] [CrossRef]
- Stawiarska-Pieta, B.; Paszczela, A.; Grucka-Mamczar, E.; Szaflarska-Stojko, E.; Birkner, E. The effect of antioxidative vitamins A and E and coenzyme Q on the morphological picture of the lungs and pancreata of rats intoxicated with sodium fluoride. Food Chem. Toxicol. 2009, 47, 2544–2550. [Google Scholar] [CrossRef]
- Zafra-Stone, S.; Yasmin, T.; Bagchi, M.; Chatterjee, A.; Vinson, J.A.; Bagchi, D. Berry anthocyanins as novel antioxidants in human health and disease prevention. Mol. Nutr. Food Res. 2007, 51, 675–683. [Google Scholar] [CrossRef]
- Devi, P.S.; Kumar, M.S.; Das, S.M. DNA Damage Protecting Activity and Free Radical Scavenging Activity of Anthocyanins from Red Sorghum (Sorghum bicolor) Bran. Biotechnol. Res. Int. 2012, 2012. [Google Scholar] [CrossRef]
- Pedreschi, R.; Cisneros-Zevallos, L. Antimutagenic and antioxidant properties of phenolic fractions from Andean purple corn (Zea mays L.). J. Agric. Food Chem. 2006, 54, 4557–4567. [Google Scholar] [CrossRef]
- Ramos-Escudero, F.; Muñoz, A.M.; Alvarado-Ortíz, C.; Alvarado, Á.; Yáñez, J.A. Purple corn (Zea mays L.) phenolic compounds profile and its assessment as an agent against oxidative stress in isolated mouse organs. J. Med. Food 2012, 15, 206–215. [Google Scholar] [CrossRef]
- Zhou, B.; Li, X.H.; Wang, X.H.; Guo, L.Y.; Zhang, Z.; Xu, C. Identification of main compositions in maize purple plant pigment. Nat. Prod. Res. Dev. 2008, 20, 842–845. (in Chinese). [Google Scholar]
- Zhang, Z.Y.; Han, B.; Qian, C. Rapid detection method for trace fluoride with microplate reader. Chin. J. Public Health 2011, 27, 255–256. (in Chinese). [Google Scholar]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Inkielewicz-Stepniak, I.; Czarnowski, W. Oxidative stress parameters in rats exposed to fluoride and caffeine. Food Chem. Toxicol. 2010, 48, 1607–1611. [Google Scholar] [CrossRef]
- Patel, P.D.; Chinoy, N.J. Influence of fluoride on biological free radical reactions in ovary of mice and its reversal. Fluoride 1998, 31, 27–30. [Google Scholar]
- Birkner, E.; Grucka-Mamczar, E.; Kasperczyk, S.; Zalejska-Fiolka, J.; Kasperczyk, A.; Chlubek, D.; Filipowska, B.; Birkner, B. The influence of sodium fluoride on the concentration of malondialdehyde and 7-ketocholesterol and the Activity of superoxide dismutase in blood Plasma of rabbits with experimental Hypercholesterolemia. Fluoride 2008, 41, 199–205. [Google Scholar]
- Shivarajashankara, Y.M.; Shivashankara, A.R.; Bhat, P.G.; Rao, S.H. Lipid peroxidation and antioxidant systems in the blood of young rats subjected to chronic fluoride toxicity. Indian J. Exp. Biol. 2003, 41, 857–860. [Google Scholar]
- Dubey, N.; Khan, A.M.; Raina, R. Sub-acute deltamethrin and fluoride toxicity induced hepatic oxidative stress and biochemical alterations in rats. Bull. Environ. Contam. Toxicol. 2013, 91, 334–338. [Google Scholar] [CrossRef]
- Altintas, L.; Essiz, D.; Eraslan, G.; Ince, S.; Arslanbas, E. Prophylactic effect of N-acetylcysteine against sodium fluoride-induced blood oxidative stress in mice. Food Chem. Toxicol. 2010, 48, 2838–2841. [Google Scholar] [CrossRef]
- Blaszczyk, I.; Grucka-Mamczar, E.; Kasperczyk, S.; Birkner, E. Influence of fluoride on rat kidney antioxidant system: Effects of methionine and vitamin E. Biol. Trace Elem. Res. 2008, 121, 51–59. [Google Scholar] [CrossRef]
- Chinoy, N.J.; Memon, M.R. Beneficial effects of some vitamins and calcium on fluoride and aluminium toxicity on gastrocnemius muscle and liver of male mice. Fluoride 2001, 34, 21–33. [Google Scholar]
- Hassan, H.A.; Yousef, M.I. Mitigating effects of antioxidant properties of black berry juice on sodium fluoride induced hepatotoxicity and oxidative stress in rats. Food Chem. Toxicol. 2009, 47, 2332–2337. [Google Scholar] [CrossRef]
- Rice-Evans, C.A. The relative antioxidant activities of plant-derived polyphenolic flavonoids. Free Radic. Res. 1995, 22, 3785–3793. [Google Scholar]
- Fan, Z.L.; Wang, Z.Y.; Zuo, L.L.; Tian, S.Q. Protective effect of anthocyanins from lingonberry on radiation-induced damages. Int. J. Environ. Res. Public Health 2012, 9, 4732–4743. [Google Scholar] [CrossRef]
- Tsuda, T. Mechanism for the peroxynitrite scavenging activity by anthocyanins. FEBS Lett. 2000, 484, 207–210. [Google Scholar] [CrossRef]
- Noda, Y. Antioxidant activity of nasunin, an anthocyanin in egg plant peels. Toxicology 2000, 148, 119–123. [Google Scholar] [CrossRef]
- Dabrowaska, E.; Letko, R.; Balunowska, M. Effect of sodium fluoride on the morphological picture of the rat liver exposed to NaF in drinking water. Adv. Med. Sci. 2006, 51, 91–95. [Google Scholar]
- Bouaziz, H.; Croute, F.; Boudawara, T.; Soleilhavoup, J.P.; Zeghal, N. Oxidative stress induced by fluoride in adult mice and their suckling pups. Exp. Toxicol. Pathol. 2007, 58, 339–349. [Google Scholar] [CrossRef]
- Jiao, Z.; Liu, J.; Wang, S. Antioxidant activities of total pigment extract from blackberries. Food Technol. Biotechnol. 2005, 43, 97–102. [Google Scholar]
- Szczepanski, M.; Kamianowska, M.; Kamianowski, G. Effects of fluorides on apoptosis and activation of human umbilical vein endothelial cells. Oral Dis. 2012, 18, 280–284. [Google Scholar] [CrossRef]
- Ghosh, J.; Das, J.; Manna, P.; Sil, P.C. Cytoprotective effect of arjunolic acid in response to sodium fluoride mediated oxidative stress and cell death via necrotic pathway. Toxicol. In Vitro 2008, 22, 1918–1926. [Google Scholar] [CrossRef]
- Lee, J.H.; Jung, J.Y.; Jeong, Y.J.; Park, J.H.; Yang, K.H.; Choi, N.K.; Kim, S.H.; Kim, W.J. Involvement of both mitochondrial- and death receptor-dependent apoptotic pathways regulated by Bcl-2 family in sodium fluoride-induced apoptosis of the human gingival fibroblasts. Toxicology 2008, 243, 340–347. [Google Scholar] [CrossRef]
- Cao, J.; Chen, J.; Wang, J.; Jia, R.; Xue, W.; Luo, Y.; Gan, X. Effects of fluoride on liver apoptosis and Bcl-2, Bax protein expression in freshwater teleost, Cyprinus carpio. Chemosphere 2013, 91, 1203–1212. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhang, Z.; Zhou, B.; Wang, H.; Wang, F.; Song, Y.; Liu, S.; Xi, S. Maize Purple Plant Pigment Protects Against Fluoride-Induced Oxidative Damage of Liver and Kidney in Rats. Int. J. Environ. Res. Public Health 2014, 11, 1020-1033. https://doi.org/10.3390/ijerph110101020
Zhang Z, Zhou B, Wang H, Wang F, Song Y, Liu S, Xi S. Maize Purple Plant Pigment Protects Against Fluoride-Induced Oxidative Damage of Liver and Kidney in Rats. International Journal of Environmental Research and Public Health. 2014; 11(1):1020-1033. https://doi.org/10.3390/ijerph110101020
Chicago/Turabian StyleZhang, Zhuo, Bo Zhou, Hiaohong Wang, Fei Wang, Yingli Song, Shengnan Liu, and Shuhua Xi. 2014. "Maize Purple Plant Pigment Protects Against Fluoride-Induced Oxidative Damage of Liver and Kidney in Rats" International Journal of Environmental Research and Public Health 11, no. 1: 1020-1033. https://doi.org/10.3390/ijerph110101020




