An Evaluation of Microbial and Chemical Contamination Sources Related to the Deterioration of Tap Water Quality in the Household Water Supply System
Abstract
:1. Introduction
2. Materials and Methods
2.1. Feed Water
| Items | Cold water | Hot water |
|---|---|---|
| DOC (mg/L) | 1.5 ± 0.1 | 1.4 ± 0.1 |
| Turbidity (NTU) | <0.4 | <0.4 |
| pH | 7.1 ± 0.05 | 7.4 ± 0.05 |
| Conductivity (µS/cm) | 180 ± 5 | 180 ± 5 |
| Residual chlorine (mg/L) | <0.3 | <0.1 |
| UV-254 | 0.009 ± 0.001 | 0.010 ± 0.001 |
| DO (mg/L) | 10 ± 2.5 | 10 ± 2.5 |
2.2. Pipe System
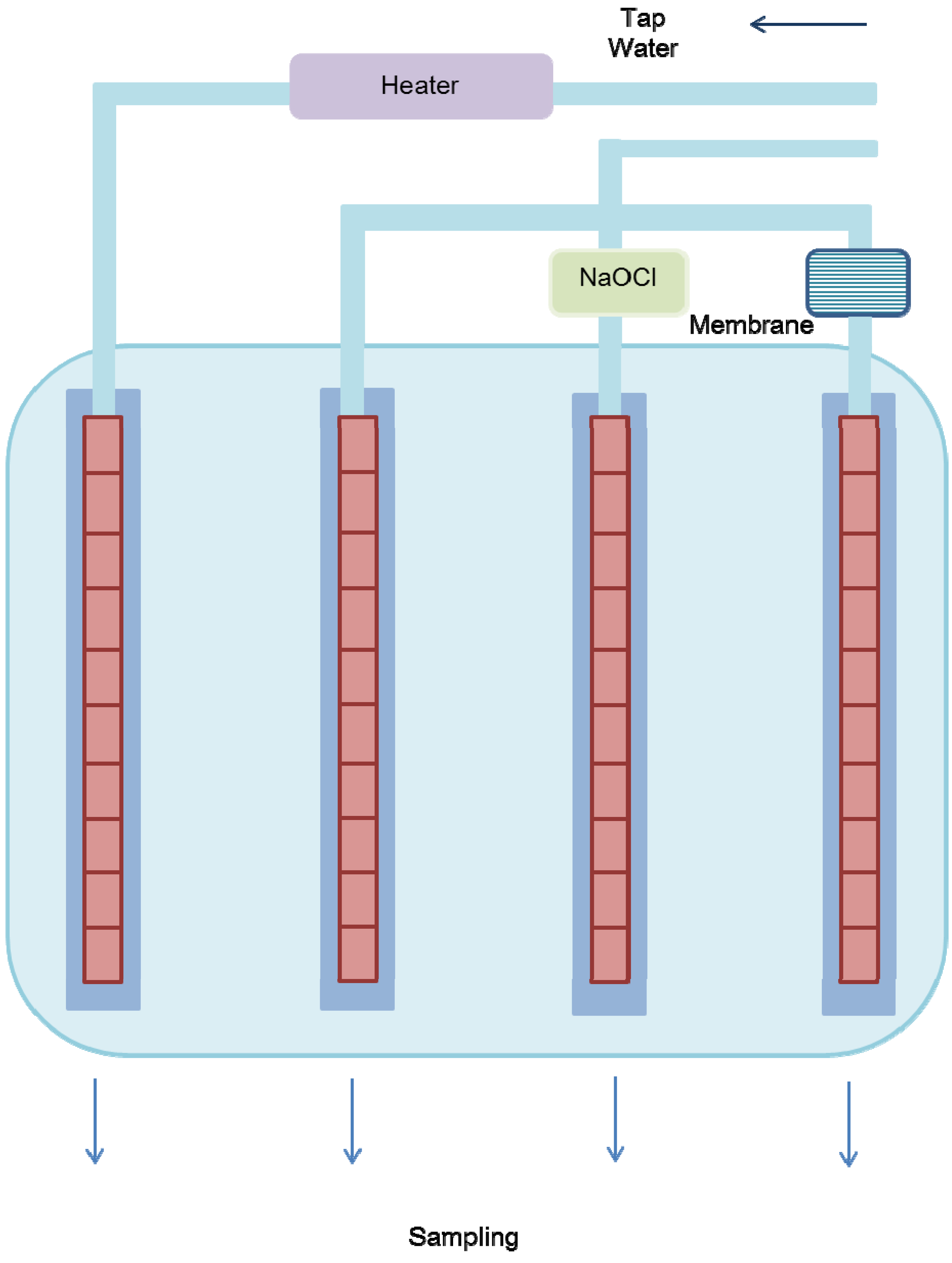
2.3. Chemical Analysis
2.4. Bacterial Analysis
3. Results and Discussion
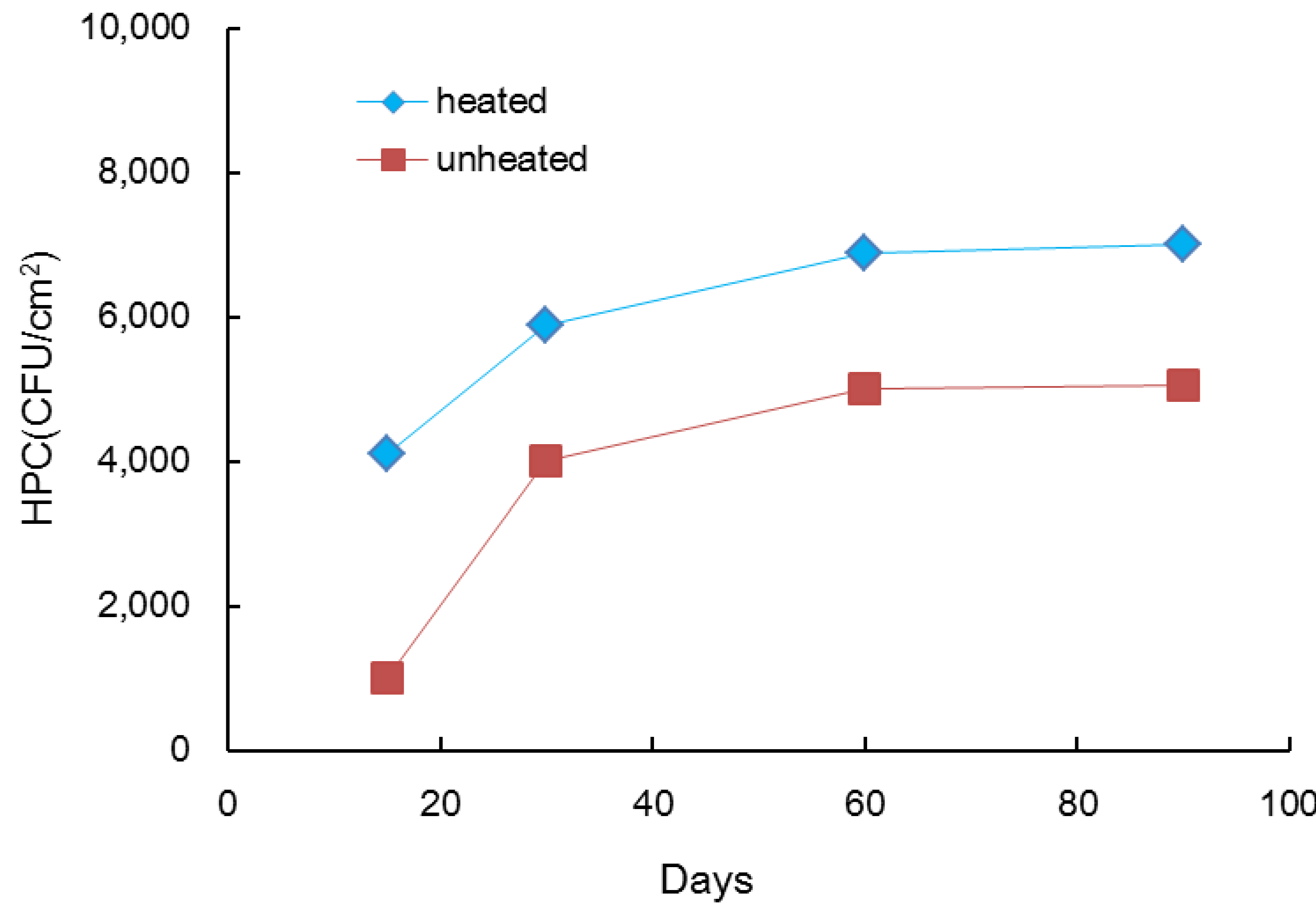
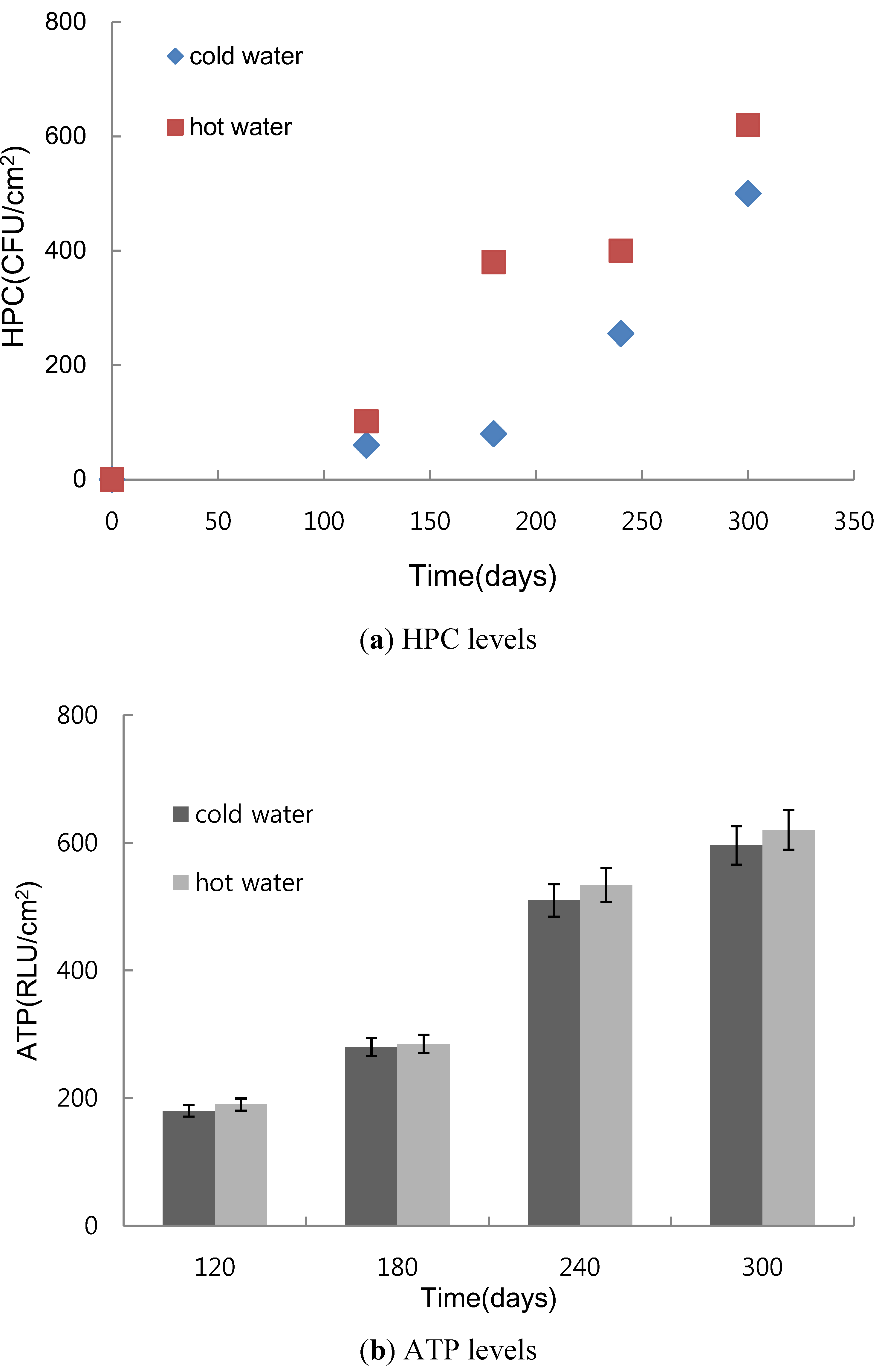
3.1. Biofilm Formation in Hot Water Plumbing
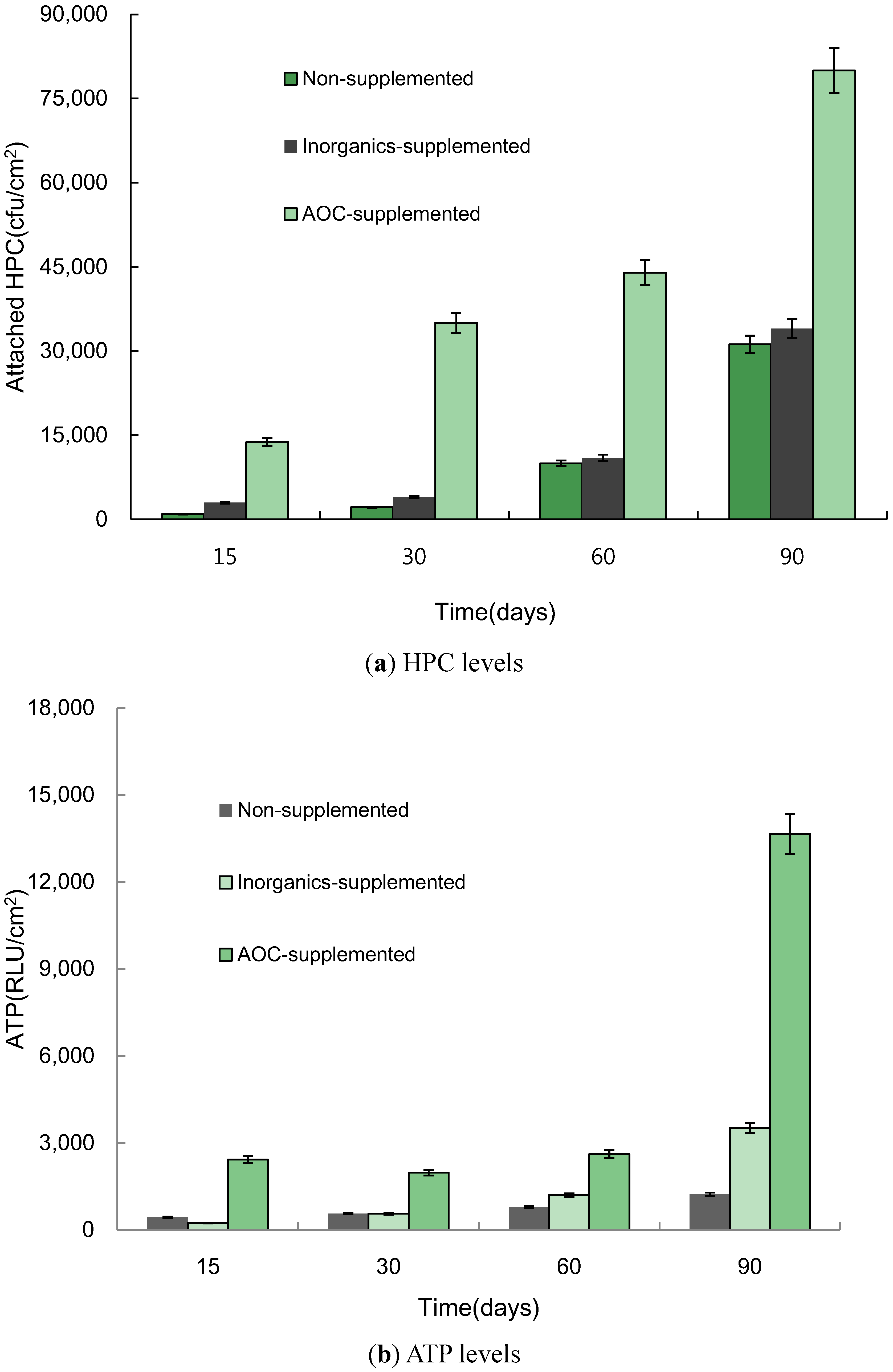
3.2. Effect of the Addition of Nutrient Sources on Biofilm Formation
3.3. Multiplication of Microorganisms on Shower Heads
| Home | CFU/cm2 | School | CFU/cm2 |
|---|---|---|---|
| H1 | 48 | S1 | 88 |
| H2 | 120 | S2 | 185 |
| H3 | 389 | S3 | 322 |
| H4 | 422 | S4 | 281 |
| H5 | 145 | S5 | 455 |
| H6 | 100 | S6 | 392 |
| H7 | 144 | S7 | 430 |
| H8 | 101 | S8 | 395 |
| H9 | 398 | S9 | 196 |
| H10 | 370 | S10 | 267 |
| Sample | Microorganisms | Sample | Microorganisms |
|---|---|---|---|
| B1 | Acinetobacter guillouiae | B6 | Acidovorax temperans |
| Sphingobium lactosutens | |||
| Acidovorax temperans | |||
| Sphingomonas paucimobilis | |||
| B2 | Stenotrophomonas maltophilia | B7 | Chryseobacterium hominis |
| Sphingobium lactosutens | |||
| Acinetobacter guillouiae | Methylobacterium aminovorans | ||
| Stenotrophomonas maltophilia | |||
| Acidovorax temperans | |||
| B3 | Stenotrophomonas maltophilia | B8 | Stenotrophomonas maltophilia |
| Gordonia sputi | Sphingomonas paucimobilis | ||
| Microbacterium lacticum | |||
| Microbacterium lacus | Acinetobacter guillouiae | ||
| B4 | Acidovorax delafieldii | B9 | Staphylococcus warneri |
| Pseudoxanthomonas mexicana | |||
| Sphingomonas koreensis | Agromyces soli | ||
| Rhodococcus corynebacterioides | |||
| Chryseobacterium defluvii | Sphingopyxis chilensis | ||
| Stenotrophomonas maltophilia | |||
| B5 | Sphingobacterium siyangense | B10 | Acidovorax temperans |
| Sphingomonas paucimobilis | |||
| Sphingomonas koreensis | |||
| Sphingomonas ginsenosidimutansMicrobacteriumoxydans |
3.4. Migration of VOC from Plastic Pipes
| Items | PB | PVC | PP | PE | cPVC |
|---|---|---|---|---|---|
| Vinylacetate | 1.2 | 0.0 | 0.7 | 0.8 | 0.9 |
| N,N-Dimethylaniline | 0.4 | 0.5 | 0.5 | 0.3 | 0.3 |
| cis-1,2-Dichloroethylene | 0.4 | 0.0 | 0.1 | 0.4 | 0.3 |
| Epichlorohydrin | 0.4 | 0.3 | 0.7 | 0.3 | 0.4 |
| Styrene | 0.3 | 0.3 | 0.4 | 0.4 | 0.2 |

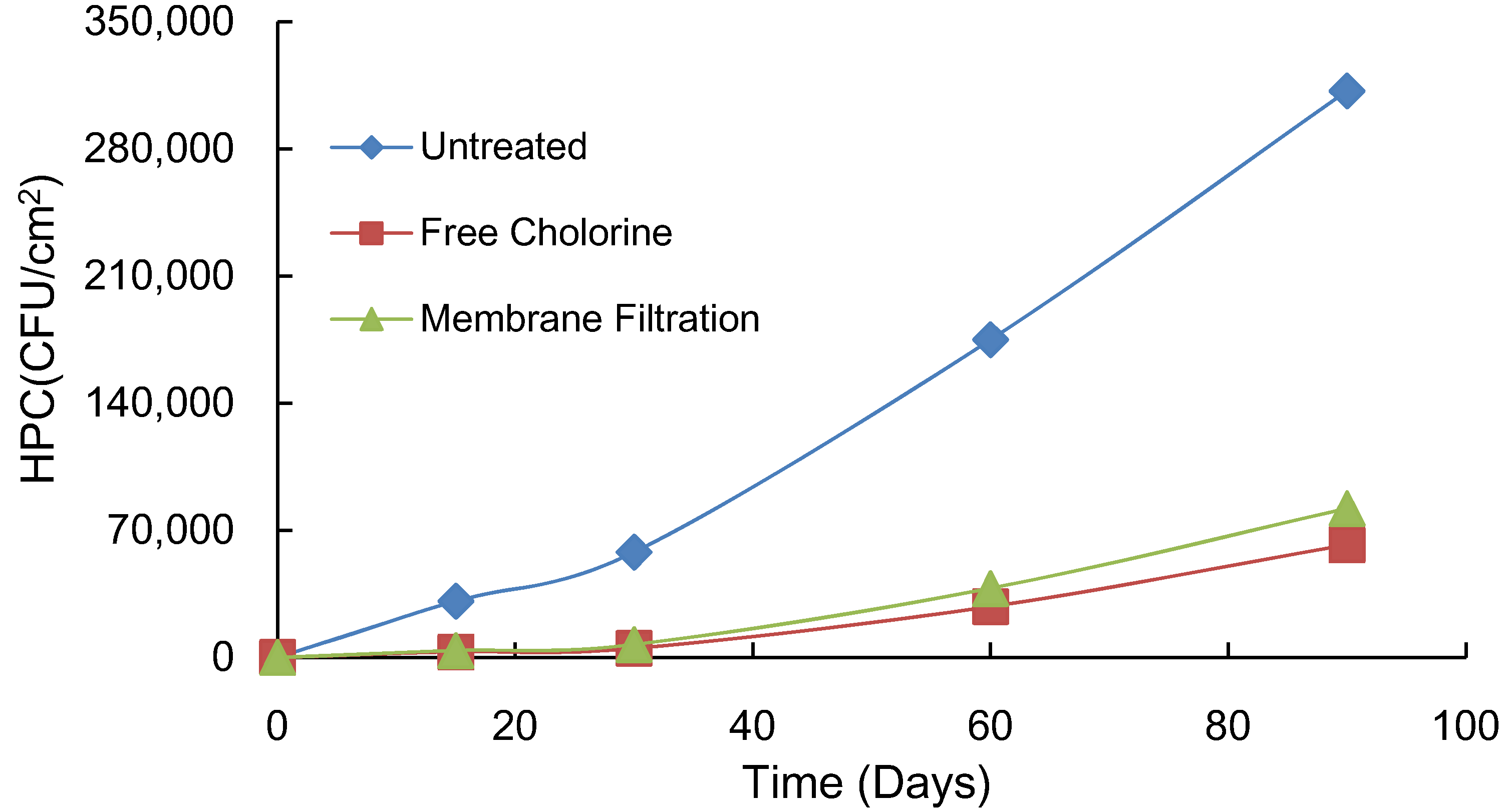
3.5. Effects of Chlorine and Membrane Filtration on Biofilms
4. Conclusions
- (1)
- The results of the pipe coupon test indicated that biofilm accumulation in pipes is stimulated by adding AOC and inorganics supplement. However, biofilm formation was significantly greater for AOC than for inorganics supplement in stainless steel pipe. Bacterial growth in biofilms was stimulated by the heating of the tap water. Based on these results, the removal efficiency of nutrient residuals through the treatment processes in water treatment plants may affect biofilm formation in the distribution system. Both chlorination and microfiltration reduced biofilm formation, as measured by HPCs on pipe walls.
- (2)
- After inspection of the internal surfaces in PVC systems, significant pipe loss was observed on pipe surfaces after 1 years of operation, and VOC substances, such as vinylacetate, NN-DMA, cis-1,2-dichloroethylene, epichlorohydrin, and styrene were identified in five kinds of plastic pipes (PVC, PB, PP, PE, and cPVC).
- (3)
- The predominant microorganisms on household shower heads taken from “N” city included Stenotrophomonas maltophilia, Sphingomonas paucimobilis, Acidovorax temperans, and Microbacterium lacticum. However, Legionella was not found. Some species detected are recognized as opportunistic and potential human pathogens in causing nosocomial infection, such as Stenotrophomonas maltophilia and Sphingomonas paucimobilis. The existence of these microorganisms in the water supply system could cause considerable risks to immnocompromised people. In these cases, shower heads may act as possible pollutant sources for the microbial contamination of water. Further research is required to characterize the persistence of Stenotrophomonas maltophilia and Sphingomonas paucimobilis in the presence of water treatment, including chlorine disinfection, of the water distribution system.
Acknowledgements
Conflicts of Interest
References
- Yoon, T.H.; Lee, Y.J. Bacterial regrowth in water distribution systems and its relationship to the water quality: Case study of two distribution systems in Korea. J. Microbiol. Biotechnol. 2004, 14, 262–267. [Google Scholar]
- Schwartz, T.; Hoffmann, S.; Obst, U. Formation and bacterial composition of young, natural biofilms obtained from public bank filtered drinking water sysem. Wat. Res. 1988, 32, 2787–2797. [Google Scholar] [CrossRef]
- Burke, V.; Robinson, J.; Gracey, M.; Peterson, D.; Partridge, K. Isolation of Aeromonas hydrophila from a metropolitan water supply: Seasonal correlation with clinical isolates. Appl. Environ. Microbiol. 1984, 48, 361–366. [Google Scholar]
- Engel, H.W.B.; Berwald, L.G.; Hvelaar, A.H. The occurrence of Mycobacterium kansasii in tap water. Tuberculosis 1980, 61, 21–26. [Google Scholar]
- Mackay, W.G.; Gribbon, L.T.; Barer, M.R.; Reid, D.C. Biofilms in drinking water systems—A possible reservoir for Helicobacterpylory. Water Sci. Technol. 1998, 38, 181–185. [Google Scholar]
- Wadowsky, R.M.; Yee, R.B.; Mezmar, L.; Wing, E.J.; Dowling, N.J. Hot water systems as sources of Legionella pneumpholia in hospital and non-hospital plumbing fixtures. Appl. Environ. Microbiol. 1982, 43, 1104–1110. [Google Scholar]
- Siller, W.G.; Dewar, W.A.; Whitehead, C.C. Cystic dilatation of the seminiferous tubules in the fowl: A sequel of sodium intoxication. J. Pathol. 1972, 107, 191–197. [Google Scholar] [CrossRef]
- Block, J.C.; I. Sibille, I; Gatel, D; Reasoner, D.J.; Lykins, B.; Clark, R.M. Biodiversity in Drinking Water Distribution System. In Microbiological Quality of Water; Sutcliffe, D.W., Ed.; Freshwater Biological Association Ambleside: London, UK, 1995. [Google Scholar]
- Huh, J.R.; Choi, K.H.; Park, S.S. Study on the tap water drinking promotion with analysis of current status in Seoul citizen. (in Korean). J. Korean Soc. Environ. Educ. 2011, 24, 78–93. [Google Scholar]
- Park, H.K.; Ryu, S.C.; Jeon, S.Y. Study on the water quality for efficiency management in the water tanks. (in Korean). J. Environ. Sci. Int. 2009, 18, 1339–1347. [Google Scholar]
- Lee, H.D.; Bae, C.H.; Park, J.H. Indirect diagnosis evaluation of service pipes according to the periods of water supply. (in Korean). J. Wat. Treat. 2002, 10, 25–37. [Google Scholar]
- Nam, S.C.; Lee, Y.J. Investigation of nutrients controlling bacterial regrowth in a PEX and stainless steel plumbing water system. Asian J. Chem. 2010, 22, 2427–2441. [Google Scholar]
- Kilb, B.; Lange, B.; Schaule, G.; Flemming, H.; Wingender, J. Contamination of drinking water by coliforms from biofilms grown on rubber-coated valves. Int. J. Hyg. Environ. Health. 2003, 206, 563–573. [Google Scholar] [CrossRef]
- Sadiki, A.L.; Williams, D.T. A study on organitoin levels in Canadian drinking waer distributed through PVC pipes. Chemosphere 1999, 38, 1541–1548. [Google Scholar] [CrossRef]
- American Public Health Association; American Water Works Association; Water Environment Federation. Standard Methods for the Examination of Water and Wastewater, 20th ed.APHA, AWWA and WEF: Washington, DC, USA, 2001.
- Ministry of Environmental in Korea, Standard Method for the Examination of Drinking Water in Korea; Dong Wha Tech Publishing: Paju, Korea, 2009.
- Sambrook, J.; Fritsch, E.F.; Maniatis, T. Molecular Cloning: A Laboratory Manual, 2nd ed.; Cold Spring Harbor Laboratory: Cold Spring Harbor, NY, USA, 1989. [Google Scholar]
- Chun, J.; Lee, J.H.; Jung, Y.; Kim, M.; Kim, S.; Kim, B.K.; Lim, Y.W. EzTaxon: A web-based tool for the identification of prokaryotes based on 16S ribosomal RNA gene sequences. Int. J. Syst. Evol. Microbiol. 2007, 57, 2259–2261. [Google Scholar] [CrossRef]
- Skjevrak, I.; Lund, V.; Ormerod, K.; Herikstad, H. Volatile organic compounds in natural biofilm in polyethylene pipes supplied with lake water and treated water from the distribution network. Wat. Res. 2005, 39, 4133–4144. [Google Scholar] [CrossRef]
- Lee, Y.J. Factors controlling bacterial growth in drinking water distribution pipes. Asian J. Chem. 2013, 25, 1629–1634. [Google Scholar]
- Lee, Y.J. Effects of temperature on corrosion and bacterial growth in water distribution pipes. Asian J. Chem. 2008, 20, 6535–6550. [Google Scholar]
- Borella, P.; Montagne, M.T.; Spica, V.R.; Stami, S.; Stancanelli, G.; Triassi, M.; Neglia, R.; Marchesi, I.; Fantuzzi, G.; Tato, D.; et al. Legionella infection risk from domestic hot water. Emerg. Infect. Dis. 2004, 10, 457–464. [Google Scholar] [CrossRef]
- Borella, P.; Montagna, M.T.; Stampi, S.; Stancanelli, G.; Romano-Spica, V.; Triassi, M.; Marchesi, I.; Bargellini, A.; Tatò, D.; Napoli, C.; et al. Legionella contamination in hot water of Italian hotels. Appl. Environ Microbiol. 2005, 71, 5805–5813. [Google Scholar] [CrossRef]
- Lee, Y.J. Investigation of chemical and bacterial quality in Korean public bath facilities. Asian J. Chem. 2009, 21, 2321–2330. [Google Scholar]
- Appenzeller, B.M.R.; Batté, M.; Mathieu, L.; Block, J.C.; Lahoussine, V.; Cavard, J.; Gatel, D. Effect of adding phosphate to drinking water on bacterial growth in slightly and highly corroded pipes. Wat. Res. 2001, 35, 1100–1105. [Google Scholar] [CrossRef]
- Hsu, B.M.; Chen, C.H.; Wan, M.T.; Cheng, H.W. Legionella prevalence in hot spring recreation areas of Taiwan. Wat. Res. 2006, 40, 3267–3272. [Google Scholar] [CrossRef]
- Kim, S.H.; Kim, M.J.; Kee, H.Y.; Kim, T.S.; Seo, J.J.; Kim, E.S.; Park, J.T.; Chung, J.K. Surveillance of Legionella contamination on water supply systems of public utilizing facilities in Gwangju, Korea. (in Korean). J. Bacteriol. Virol. 2010, 40, 19–28. [Google Scholar] [CrossRef]
- Funke, G.; Haase, G.; Schnitzler, N.; Schrage, N.; Reinert, R.R. Endophthalmitis due to Microbacterium species: Case report and review of microbacterium infections. Clin. Infect. Dis. 1997, 24, 713–716. [Google Scholar] [CrossRef]
- Kitamura, Y.; Sawabe, E; Ohkusu, K; Tojo, N.; Tohda, S. First report of sepsis caused by Rhodococcus corynebacterioides in a patient with myelodysplastic syndrome. J. Clin. Microbiol. 2012, 50, 1089–1091. [Google Scholar] [CrossRef]
- Kamath, U.; Singer, C.; Isenberg, H.D. Clinical significance of Staphylococcus warneri bacteremia. J. Clin. Microbiol. 1992, 30, 261–264. [Google Scholar]
- Brooke, J.S. Stenotrophomonas maltophilia: An emerging global opportunistic pathogen. Clin. Microbiol. Rev. 2012, 25, 2–41. [Google Scholar] [CrossRef]
- Ryu, J.H.; Beuchat, L.R. Biofilm formation by Escherichia coli O157:H7 on stainless steel: Effect of exopolysaccharide and curli production on its resistance to chlorine. Appl. Environ. Microbiol. 2005, 71, 247–254. [Google Scholar] [CrossRef]
- Tan, C.K.; Liaw, S.J.; Yu, C.J.; Teng, L.J.; Hsueh, P.R. Extensively drug-resistant Stenotrophomonas maltophilia in a tertiary care hospital in Taiwan: Microbiologic characteristics, clinical features, and outcomes. Diagn. Microbiol. Infect. Dis. 2008, 60, 205–210. [Google Scholar] [CrossRef]
- Smalley, D.L.; Hansen, V.R.; Baselski, V.S. Susceptibility of Pseudomonas paucimobilis to 24 antimicrobial agents. Antimicrob. Agents Chemother. 1983, 23, 161–162. [Google Scholar] [CrossRef]
- Lin, J.N.; Lai, C.H.; Chen, Y.H.; Lin, H.L.; Huang, C.K.; Chen, W.F.; Wanga, J.L.; Chung, H.C.; Lianga, S.H.; Lin, H.H. Sphingomonas paucimobilis bacteremia in humans:16 case reports and a literature review. J. Microbiol. Immunol. Infect. 2010, 43, 35–42. [Google Scholar] [CrossRef]
- Wang, Y.; Fan, Y.; Gu, J.D. Microbial degradation of the endocrine-disrupting chemicals phthalic acid and dimethyl phthalate ester under aerobic conditions. Bull. Environ. Contam. Toxicol. 2003, 71, 810–818. [Google Scholar] [CrossRef]
- Heijstra, B.D.; Pichler, F.B.; Liang, Q.; Blaza, R.G.; Turner, S.J. Extracellular DNA and Type IV pili mediate surface attachment by Acidovorax temperans. Antonie Van Leeuwenhoek 2009, 95, 343–349. [Google Scholar] [CrossRef]
- Bulut, S.; Waites, W.M.; Mitchell, J.R. Effects of combined shear and thermal forces on destruction of Microbacterium lacticum. Appl. Environ. Microbiol. 1999, 65, 4464–4469. [Google Scholar]
- Mininstry of Health and Welfare in Korea, A study on the Analytical Methods and National Surveys of Trace Hazardous Compounds in Drinking Water; (in Korean). Korea Institute of Science and Technology: Seoul, Korea, 1993.
- Kolman, A.; Chovanec, M.; Osterman-Golkar, S. Genotoxic effects of ethylene oxide, propylene oxide and epichlorohydrin in humans: Update review (1990–2001). Mutat. Res. 2002, 512, 173–194. [Google Scholar] [CrossRef]
- Jo, C.H.; Kim, S.K.; Jung, S.W.; Kim, T.G. A Study on Taste and Odor Induced by PVC Hose for Tap Water. In Proceedings of the Korea Society of Water and Wastewater and Korea Society on Water Environment, Daejon, Chungnam, Korea, 4 November 2004; pp. 13–19.
- Heim, T. Impact of Polymeric Plumbing Materials on Drinking Water Quality and Aesthetics. Master Thesis, Virginia Polytechnic Institute and State University, Blacksburg, VA, USA, April 2006. [Google Scholar]
- Al-Malack, M.H. Migration of lead from unplasticized polyvinyl chloride pipes. J. Hazard. Mater. 2001, 82, 263–274. [Google Scholar] [CrossRef]
- Lee, Y. Impacts of biodegradable organic matter and pipe materials on bacterial multiplication in water supply systems. Asian J. Chem. 2010, 22, 1555–1568. [Google Scholar]
- Kim, D.H.; Lee, D.J.; Kim, K.P.; Bae, C.H.; Joo, H.E. Computing the dosage and analysing the effect of optimal rechlorination for adequate residual chlorine in water distribution system. (in Korean). J. Korean Soc. Environ. Eng. 2010, 32, 916–927. [Google Scholar]
- Miettinen, I.T.; Vartiainen, T.; Nissinen, T.; Tuhkanen, T.; Martikainen, P.J. Microbial growth in drinking waters treated with ozone, ozone/hydrogen peroxide or chlorine. Ozone Sci. Eng. 1998, 20, 303–315. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lee, Y. An Evaluation of Microbial and Chemical Contamination Sources Related to the Deterioration of Tap Water Quality in the Household Water Supply System. Int. J. Environ. Res. Public Health 2013, 10, 4143-4160. https://doi.org/10.3390/ijerph10094143
Lee Y. An Evaluation of Microbial and Chemical Contamination Sources Related to the Deterioration of Tap Water Quality in the Household Water Supply System. International Journal of Environmental Research and Public Health. 2013; 10(9):4143-4160. https://doi.org/10.3390/ijerph10094143
Chicago/Turabian StyleLee, Yoonjin. 2013. "An Evaluation of Microbial and Chemical Contamination Sources Related to the Deterioration of Tap Water Quality in the Household Water Supply System" International Journal of Environmental Research and Public Health 10, no. 9: 4143-4160. https://doi.org/10.3390/ijerph10094143




