The Performance of Children Prenatally Exposed to HIV on the A-Not-B Task in Kilifi, Kenya: A Preliminary Study
Abstract
:1. Introduction
2. Methods
2.1. Study Site
2.2. Participants
2.3. General Procedures
2.4. Measures and Procedures
2.4.1. A-Not-B
2.4.2. KDI
| Variables | Reference population | HIV exposed | HIV infected | p |
|---|---|---|---|---|
| Sample size (Girls) | 319 (159) | 17 (6) | 31 (14) | |
| Age in months | 18.84 (8.43) | 17.72 (8.77) | 21.10 (8.86) | 0.300 |
| Maternal education a | 3.40 (3.50) | 4.35 (4.07) | 4.80 (3.55) | 0.069 |
| Psychomotor b | 0.08(0.84) | 0.11 (0.60) | −0.91(1.86) | 0.000 |
| Weight-for-Age | −1.24 (1.08) | −1.28 (.79) | −2.12 (1.36) | 0.000 |
2.4.3. Weight-for-Age
2.4.4. Maternal Education
2.5. Data Analysis Strategies
3. Results
3.1. HIV Exposure and Background Variables
3.2. A-Not-B Task
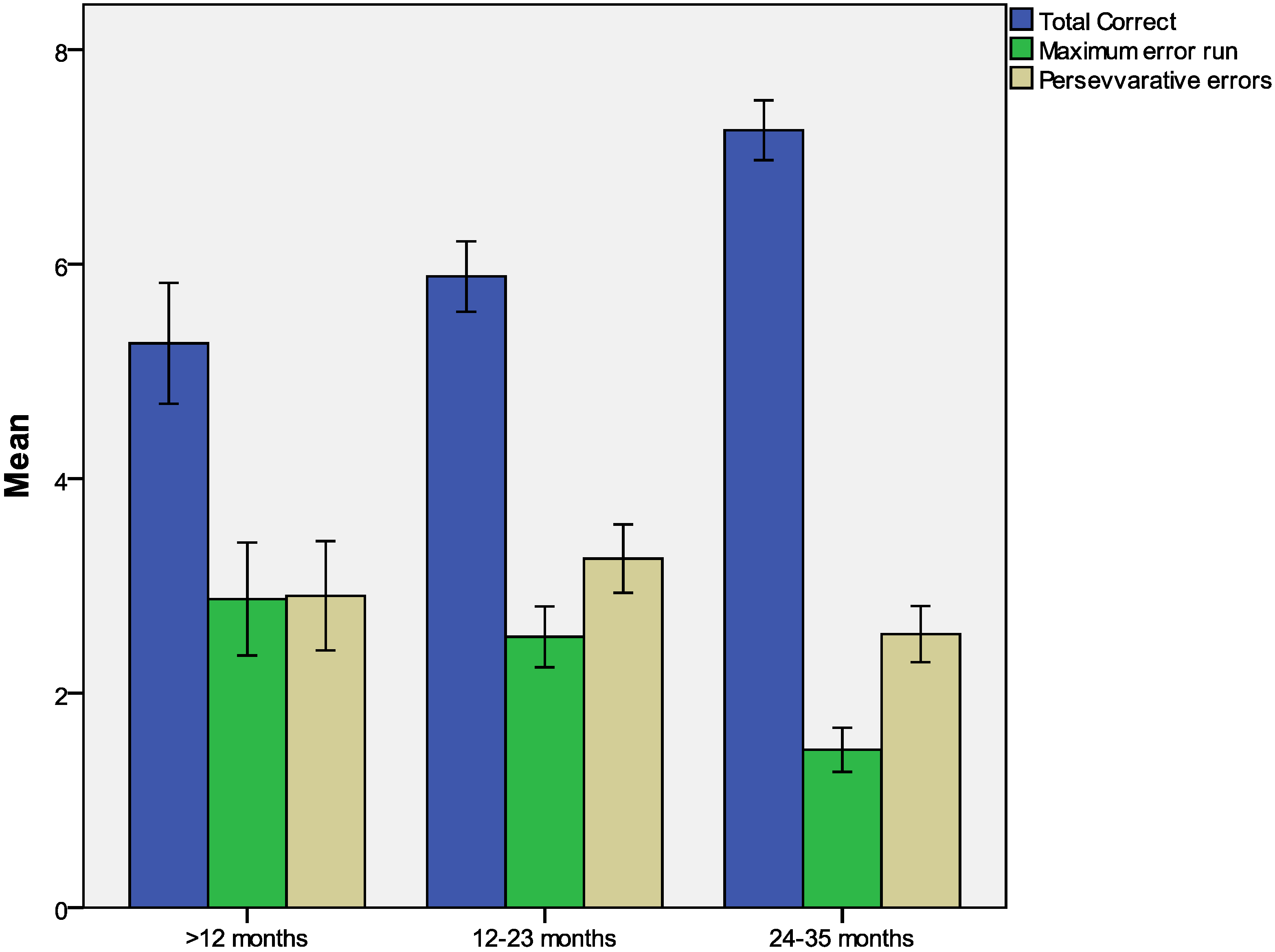
| Variables | Reference population | HIV exposed but uninfected | HIV infected | p c |
|---|---|---|---|---|
| Children not completing task N (%) | 54 (16.9) | 6 (35.3) | 12 (41.9%) | 0.004 |
| Total correct M (SD) a,b | −0.001 (1.74) | 0.720 (2.15) | −0.1936 (1.20) | 0.347 |
| Perseverative errors M (SD) a,b | −0.000 (1.70) | −0.200 (1.41) | −0.252 (1.48) | 0.777 |
| Maximum run of error M (SD) a,b | 0.001 (1.54) | −0.22 (1.69) | 0.335 (1.01) | 0.569 |
3.3. Predictors of Inability to Complete A-Not-B
| B | S.E. | Odds Ratio | Sig. | |
|---|---|---|---|---|
| Being Underweight | −0.654 | 0.298 | 0.520 | 0.028 |
| Aged 24–35 months a | 1.270 | 0.378 | 3.560 | 0.001 |
| HIV exposed uninfected b | −1.083 | 0.554 | 0.339 | 0.050 |
| HIV infected b | −1.228 | 0.427 | 0.293 | 0.004 |
4. Discussion
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Abubakar, A.; van Baar, A.L.; van de Vijver, F.J.R.; Holding, P.; Newton, C.R.J.C. Pediatric HIV and neurodevelopment in Africa: A systematic review. Trop. Med. Int. Health 2008, 13, 880–887. [Google Scholar] [CrossRef]
- Van Rie, A.; Mupuala, A.; Dow, A. Impact of the HIV/AIDS epidemic on the neurodevelopment of preschool-aged children in Kinshasa, Democratic Republic of the Congo. Pediatrics 2008, 122, 123–128. [Google Scholar] [CrossRef]
- Lezak, M. Neuropsychological Assessment, 3rd ed.; Oxford University Press: New York, NY, USA, 1995. [Google Scholar]
- Knight, R.; Stuss, D. Prefrontal Cortex: The Present and the Future. In Principles of Frontal Lobe Function; Stuss, D., Knight, R., Eds.; Oxford University Press: New York, NY, USA, 2002; pp. 573–597. [Google Scholar]
- Thompson, P.M.; Dutton, R.A.; Hayashi, K.M.; Toga, A.W.; Lopez, O.L.; Aizenstein, H.T.; Becker, J.T. Thinning of the cerebral cortex visualized in HIV/AIDS reflects cd4 and tlymphocyte decline. Proc. Natl. Acad. Sci. USA 2005, 102, 15647–15652. [Google Scholar]
- Melrose, R.J.; Tinaz, S.; Castello, J.M.B.; Courtney, M.G.; Stern, C.G. Compromise to fronto-stratial functioning in HIV: An FMRI investigation of the semantic event sequencing. Behav. Brain Res. 2008, 188, 337–347. [Google Scholar]
- Isquith, P.K.; Crawford, J.S.; Espy, K.A.; Gioia, G.A. Assessment of executive function in preschool-aged children. Ment. Retard. Dev. Disabil. Res. Rev. 2005, 11, 209–215. [Google Scholar] [CrossRef]
- Bisiacchi, P.S.; Suppiej, A.; Laverda, A. Neuropsychological evaluation of neurologically asymptomatic HIV-infected children. Brain Cogn. 2000, 43, 49–52. [Google Scholar]
- Koekkoek, S.; de Sonneville, L.M.J.; Wolfs, T.W.M.; Licht, R.; Geelen, S.P.M. Neurocognitive function profile in HIV-infected school age children. Eur. J. Paediatr. Neurol. 2007, 12, 290–297. [Google Scholar]
- Zelazo, P.D.; Müller, U. Executive Function in Typical and Atypical Developmen. In Blackwell Handbook of Childhood Cognitive Development; Goswami, U., Ed.; Blackwell Publishing: Herzberg, Germay, 2004. [Google Scholar]
- Reznick, J.S.; Morrow, J.D.; Goldman, B.D.; Synder, J. The onset of working memory in infants. Infancy 2004, 6, 145–154. [Google Scholar] [CrossRef]
- Jing, S.; Monay, H.; O’Callanghan, M. A comparison of executive functions in very preterm and term infants at 8 months corrected age. Early Hum. Dev. 2008, 85, 225–230. [Google Scholar]
- Schatz, J.; Roberts, C.W. Neurobehavioral impact of sickle cell disease in early childhood. J. Int. Neuropsychol. Soc. 2007, 13, 933–943. [Google Scholar]
- Lipina, S.J.; Martelli, M.I.; Vuelta, B.; Colombo, T.A. Perfomance on the A-not-B task of Argentinian infants from unsatisfied and satisified basic need homes. Interam. J. Psychol. 2005, 39, 49–60. [Google Scholar]
- Beirman, K.L.; Nix, R.L.; Greenberg, M.T.; Blair, C.; Domitrovitch, C.E. Executive functions and school readiness intervention: Impact, moderation and mediation in the head start REDI programme. Dev. Psychopathol. 2008, 20, 821–843. [Google Scholar]
- Filteau, S. The HIV‐exposed, uninfected african child. Trop. Med. Int. Health 2009, 14, 276–287. [Google Scholar] [CrossRef]
- Kandawasvika, G.Q.; Ogundipe, E.; Gumbo, F.Z.; Kurewa, E.N.; Mapingure, M.P.; Stray‐Pedersen, B. Neurodevelopmental impairment among infants born to mothers infected with human immunodeficiency virus and uninfected mothers from three peri‐urban primary care clinics in Harare, Zimbabwe. Dev. Med. Child Neurol. 2011, 53, 1046–1052. [Google Scholar] [CrossRef]
- English, M.; Ngama, M.; Musumba, C.; Wamola, B.; Bwika, J.; Mohammed, S.; Ahmed, M.; Mwarumba, S.; Ouma, B.; McHugh, K.; et al. Causes and outcome of young infant admissions to a Kenyan district hospital. Arch. Dis. Child. 2003, 88, 438–443. [Google Scholar] [CrossRef]
- Abubakar, A.; Holding, P.A.; van Baar, A.; Newton, C.; van de Vijver, F.J.R. Monitoring psychomotor development of children in resource poor settings: An evaluation of Kilifi Developmental Inventory. Ann. Trop. Pediatr. 2008, 28, 217–226. [Google Scholar] [CrossRef]
- Espy, K.A.; Kaufmann, P.M.; McDiarmid, M.D.; Glisky, M.L. Executive functioning in preschool children: Performance on A-not-B and other delayed response format tasks. Brain Cogn. 1999, 41, 178–199. [Google Scholar] [CrossRef]
- Diamond, A. The development and neural bases of memory functions as indexed by the AB and delayed response tasks in human infants and infant monkeys. Ann. N. Y. Acad. Sci. 1990, 608, 267–317. [Google Scholar] [CrossRef]
- Diamond, A. Looking closely at infants’ performance and experimental procedures in the A-not-B task. Behav. Brain Sci. 2001, 24, 38–41. [Google Scholar] [CrossRef]
- Diamond, A. The Early Development of Executive Functions. In Lifespan cognition: Mechanisms of Change; Bialystok, E., Craik, F., Eds.; Oxford University Press: New York, NY, USA, 2006; pp. 70–95. [Google Scholar]
- Who Anthro 2005 Software and Macros for Assessing Child Growth and Development; 2006 Version; World Health Organization: Geneva, Switzerland, 2005.
- Abubakar, A.; Holding, P.; Newton, C.R.; van Baar, A.; van de Vijver, F.J. The role of weight for age and disease stage in poor psychomotor outcome of HIV-infected children in Kilifi, Kenya. Dev. Med. Child Neurol. 2009, 51, 968–973. [Google Scholar] [CrossRef]
- Wolters, P.L.; Brouwers, P.; Moss, H.A.; Pizzo, P.A. Differential receptive and expressive language functioning of children with symptomatic HIV disease and relation to CT scan brain abnormalities. Pediatrics 1995, 95, 112–119. [Google Scholar]
- Brown, L.K.; Lourie, K.J.; Pao, M. Children and adolescents living with HIV and AIDS: A review. J. Child Psychol. Psychiatry 2000, 41, 81–96. [Google Scholar] [CrossRef]
- Feldman, R.; Eidelman, A.I.; Rotenberg, N. Parenting stress, infant emotion regulation, maternal sensitivity, and the cognitive development of triplets: A model for parent and child influences in a unique ecology. Child Dev. 2004, 75, 1774–1791. [Google Scholar] [CrossRef]
- Thompson, R.A.; Calkins, S.D. The double-edged sword: Emotional regulation for children at risk. Dev. Psychopathol. 1996, 8, 163–182. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Abubakar, A.; Holding, P.; Van Baar, A.; Newton, C.R.J.C.; Van de Vijver, F.J.R.; Espy, K.A. The Performance of Children Prenatally Exposed to HIV on the A-Not-B Task in Kilifi, Kenya: A Preliminary Study. Int. J. Environ. Res. Public Health 2013, 10, 4132-4142. https://doi.org/10.3390/ijerph10094132
Abubakar A, Holding P, Van Baar A, Newton CRJC, Van de Vijver FJR, Espy KA. The Performance of Children Prenatally Exposed to HIV on the A-Not-B Task in Kilifi, Kenya: A Preliminary Study. International Journal of Environmental Research and Public Health. 2013; 10(9):4132-4142. https://doi.org/10.3390/ijerph10094132
Chicago/Turabian StyleAbubakar, Amina, Penny Holding, Anneloes Van Baar, Charles. R. J. C. Newton, Fons. J. R. Van de Vijver, and Kimberly Andrews Espy. 2013. "The Performance of Children Prenatally Exposed to HIV on the A-Not-B Task in Kilifi, Kenya: A Preliminary Study" International Journal of Environmental Research and Public Health 10, no. 9: 4132-4142. https://doi.org/10.3390/ijerph10094132





