Ion Exchange Chromatography and Mass Spectrometric Methods for Analysis of Cadmium-Phytochelatin (II) Complexes
Abstract
:1. Introduction
2. Experimental Section
2.1. Chemicals
2.2. Ion Exchange Chromatography
2.3. Matrix-Assisted Laser Desorption/Ionization Time-of-Flight Mass Spectrometry
3. Results and Discussion
3.1. Optimization of IEC for Cd-PC Complex Determination
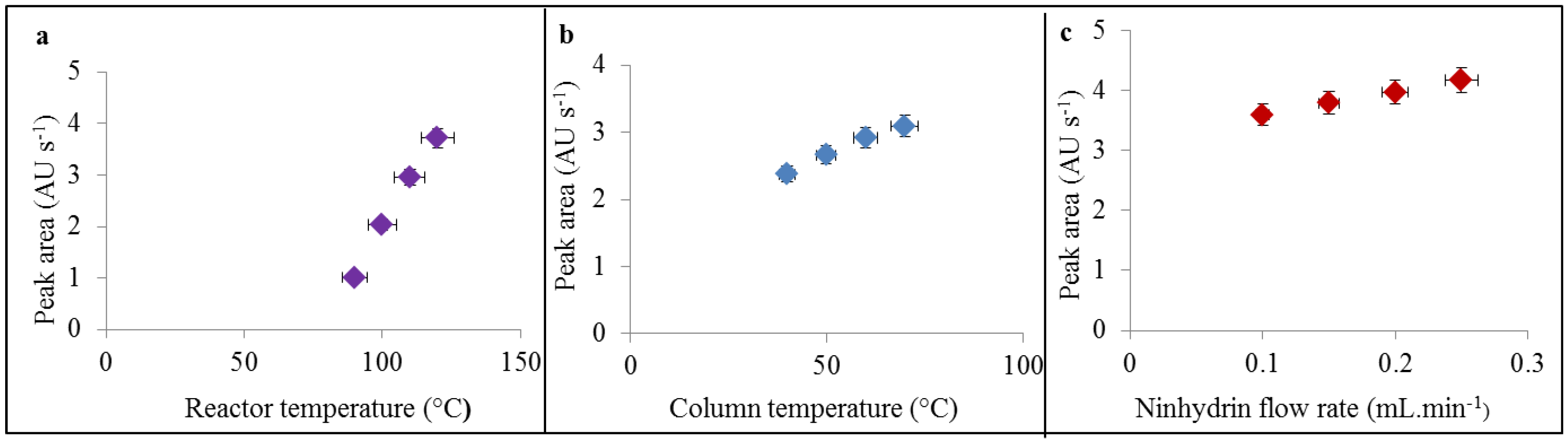
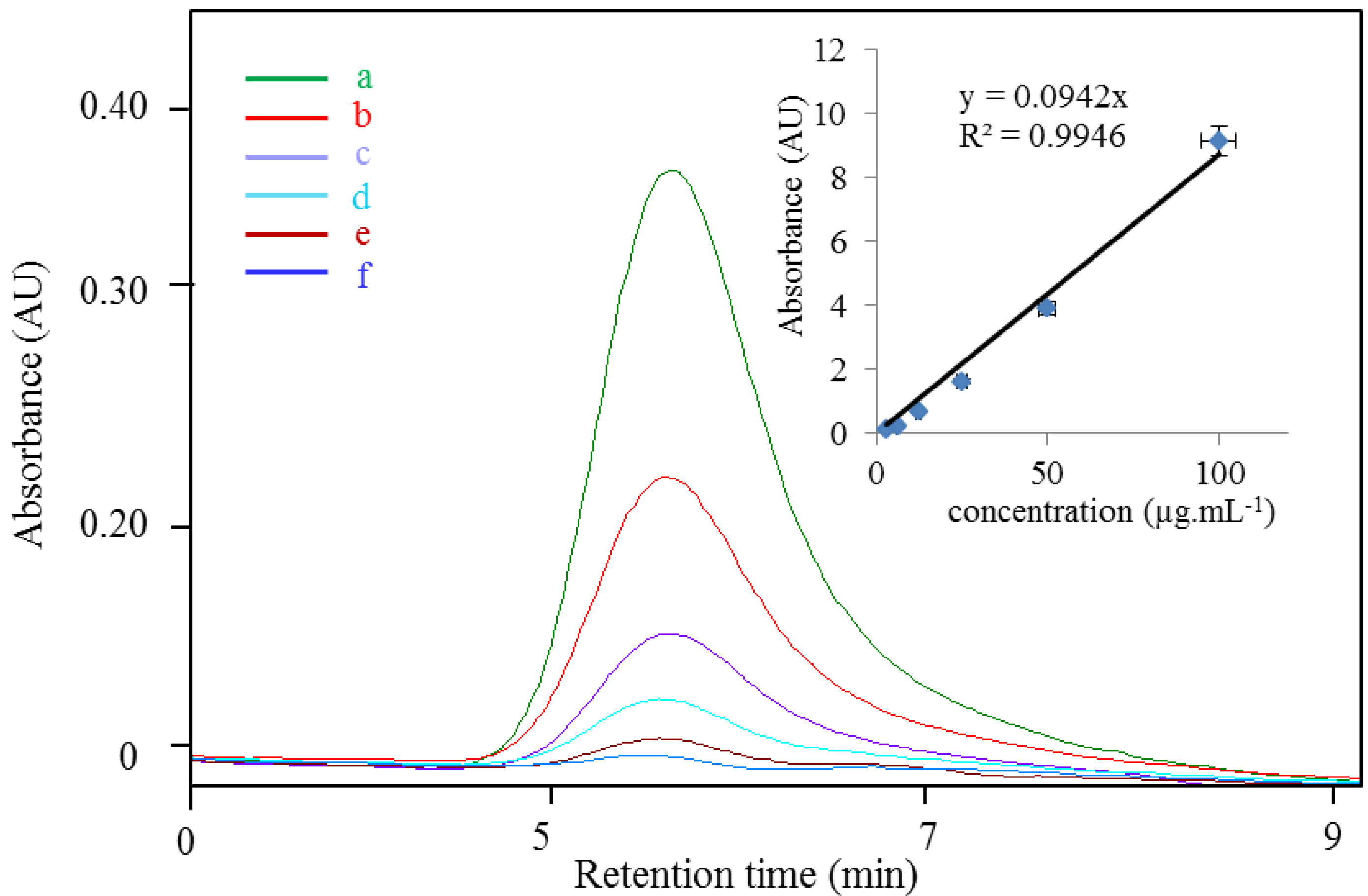
3.2. Study of Cd-PC2 Complexes by IEC and MALDI-TOF


4. Conclusions
Acknowledgements
References
- Rauser, W.E. Phytochelatins and related peptides—Structure, biosynthesis, and function. Plant Physiol. 1995, 109, 1141–1149. [Google Scholar]
- Cobbett, C.S. Phytochelatins and their roles in heavy metal detoxification. Plant Physiol. 2000, 123, 825–832. [Google Scholar] [CrossRef]
- Hall, J.L. Cellular mechanisms for heavy metal detoxification and tolerance. J. Exp. Bot. 2002, 53, 1–11. [Google Scholar] [CrossRef]
- Cruz, B.H.; Diaz-Cruz, J.M.; Sestakova, I.; Velek, J.; Arino, C.; Esteban, M. Differential pulse voltammetric study of the complexation of Cd(II) by the phytochelatin (gamma-Glu-Cys)(2)Gly assisted by multivariate curve resolution. J. Electroanal. Chem. 2002, 520, 111–118. [Google Scholar] [CrossRef]
- Gekeler, W.; Grill, E.; Winnacker, E.L.; Zenk, M.H. Algae sequester heavy-metals via synthesis of phytochelatin complexes. Arch. Microbiol. 1988, 150, 197–202. [Google Scholar] [CrossRef]
- Grill, E.; Loffler, S.; Winnacker, E.L.; Zenk, M.H. Phytochelatins, the heavy-metal-binding peptides of plants, are synthesized from glutathione by a specific gamma-glutamylcysteine dipeptidyl transpeptidase (phytochelatin synthase). Proc. Natl. Acad. Sci. USA 1989, 86, 6838–6842. [Google Scholar]
- Leopold, I.; Gunter, D.; Neumann, D. Application of high performance liquid chromatography—Inductively coupled plasma mass spectrometry to the investigation of phytochelatin complexes and their role in heavy metal detoxification in plants. Analusis 1998, 26, M28–M32. [Google Scholar]
- Krystofova, O.; Adam, V.; Babula, P.; Zehnalek, J.; Beklova, M.; Havel, L.; Kizek, R. Effects of various doses of selenite on stinging nettle (Urtica dioica L.). Int. J. Environ. Res. Public Health 2010, 7, 3804–3815. [Google Scholar] [CrossRef]
- Skladanka, J.; Adam, V.; Zitka, O.; Krystofova, O.; Beklova, M.; Kizek, R.; Havlicek, Z.; Slama, P.; Nawrath, A. Investigation into the effect of molds in grasses on their content of low molecular mass thiols. Int. J. Environ. Res. Public Health 2012, 9, 3789–3805. [Google Scholar] [CrossRef]
- Miller, G.; Begonia, G.; Begonia, M.F.T. Selected morphological characteristics, lead uptake and phytochelatin synthesis by coffeeweed (Sesbania exaltata Raf.) grown in elevated levels of lead-contaminated soil. Int. J. Environ. Res. Public Health 2011, 8, 2401–2417. [Google Scholar] [CrossRef]
- Potesil, D.; Petrlova, J.; Adam, V.; Vacek, J.; Klejdus, B.; Zehnalek, J.; Trnkova, L.; Havel, L.; Kizek, R. Simultaneous femtomole determination of cysteine, reduced and oxidized glutathione, and phytochelatin in maize (Zea mays L.) kernels using high-performance liquid chromatography with electrochemical detection. J. Chromatogr. A 2005, 1084, 134–144. [Google Scholar] [CrossRef]
- Petrlova, J.; Mikelova, R.; Stejskal, K.; Kleckerova, A.; Zitka, O.; Petrek, J.; Havel, L.; Zehnalek, J.; Adam, V.; Trnkova, L.; et al. Simultaneous determination of eight biologically active thiol compounds using gradient elution-Liquid Chromatography with Coul-Array detection. J. Sep. Sci. 2006, 29, 1166–1173. [Google Scholar] [CrossRef]
- Diopan, V.; Stejskal, K.; Galiova, M.; Adam, V.; Kaiser, J.; Horna, A.; Novotny, K.; Liska, M.; Havel, L.; Zehnalek, J.; et al. Determination of plant thiols by liquid chromatography coupled with coulometric and amperometric detection in lettuce treated by lead(II) ions. Electroanalysis 2010, 22, 1248–1259. [Google Scholar] [CrossRef]
- Zitka, O.; Krystofova, O.; Sobrova, P.; Adam, V.; Zehnalek, J.; Beklova, M.; Kizek, R. Phytochelatin synthase activity as a marker of metal pollution. J. Hazard. Mater. 2011, 192, 794–800. [Google Scholar] [CrossRef]
- Zitka, O.; Merlos, M.A.; Adam, V.; Ferrol, N.; Pohanka, M.; Hubalek, J.; Zehnalek, J.; Trnkova, L.; Kizek, R. Electrochemistry of copper(II) induced complexes in mycorrhizal maize plant tissues. J. Hazard. Mater. 2012, 203, 257–263. [Google Scholar] [CrossRef]
- Supalkova, V.; Huska, D.; Diopan, V.; Hanustiak, P.; Zitka, O.; Stejskal, K.; Baloun, J.; Pikula, J.; Havel, L.; Zehnalek, J.; et al. Electroanalysis of plant thiols. Sensors 2007, 7, 932–959. [Google Scholar] [CrossRef]
- Zitka, O.; Skutkova, H.; Krystofova, O.; Sobrova, P.; Adam, V.; Zehnalek, J.; Havel, L.; Beklova, M.; Hubalek, J.; Provaznik, I.; et al. Rapid and ultrasensitive method for determination of phytochelatin(2) using high performance liquid chromatography with electrochemical detection. Int. J. Electrochem. Sci. 2011, 6, 1367–1381. [Google Scholar]
- Jaeckel, P.; Krauss, G.; Menge, S.; Schierhorn, A.; Rucknagel, P.; Krauss, G.J. Cadmium induces a novel metallothionein and phytochelatin 2 in an aquatic fungus. Biochem. Biophys. Res. Commun. 2005, 333, 150–155. [Google Scholar] [CrossRef]
- Cavanillas, S.; Gusmao, R.; Arino, C.; Diaz-Cruz, J.M.; Esteban, M. Voltammetric analysis of phytochelatin complexation in ternary metal mixtures supported by multivariate analysis and ESI-MS. Electroanalysis 2012, 24, 309–315. [Google Scholar] [CrossRef]
- Chen, L.Q.; Guo, Y.F.; Yang, L.M.; Wang, Q.Q. SEC-ICP-MS and ESI-MS/MS for analyzing in vitro and in vivo Cd-phytochelatin complexes in a Cd-hyperaccumulator Brassica chinensis. J. Anal. At. Spectrom. 2007, 22, 1403–1408. [Google Scholar] [CrossRef]
- Najmanova, J.; Neumannova, E.; Leonhardt, T.; Zitka, O.; Kizek, R.; Macek, T.; Mackova, M.; Kotrba, P. Cadmium-induced production of phytochelatins and speciation of intracellular cadmium in organs of Linum usitatissimum seedlings. Ind. Crop. Prod. 2012, 36, 536–542. [Google Scholar] [CrossRef]
- Raab, A.; Feldmann, J.; Meharg, A.A. The nature of arsenic-phytochelatin complexes in Holcus lanatus and Pteris cretica. Plant Physiol. 2004, 134, 1113–1122. [Google Scholar] [CrossRef]
- Rigas, P.G. Review: Liquid chromatography-post-column derivatization for amino acid analysis: Strategies, instrumentation, and applications. Instrum. Sci. Technol. 2012, 40, 161–193. [Google Scholar] [CrossRef]
- Li-Chan, E.C.Y.; Sultanbawa, F.; Losso, J.N.; Oomah, B.D.; Mazza, G. Characterization of phytochelatin-like complexes from flax (Linum usitatissimum) seed. J. Food Biochem. 2002, 26, 271–293. [Google Scholar]
- Nejdl, L.; Sochor, J.; Zitka, O.; Cernei, N.; Ruttkay-Nedecky, B.; Kopel, P.; Babula, P.; Adam, V.; Hubalek, J.; Kizek, R. Spectrometric and chromatographic study of reactive oxidants hypochlorous and hypobromous acids and their interactions with taurine. Chromatographia 2013, in press. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Rodrigo, M.A.M.; Cernei, N.; Kominkova, M.; Zitka, O.; Beklova, M.; Zehnalek, J.; Kizek, R.; Adam, V. Ion Exchange Chromatography and Mass Spectrometric Methods for Analysis of Cadmium-Phytochelatin (II) Complexes. Int. J. Environ. Res. Public Health 2013, 10, 1304-1311. https://doi.org/10.3390/ijerph10041304
Rodrigo MAM, Cernei N, Kominkova M, Zitka O, Beklova M, Zehnalek J, Kizek R, Adam V. Ion Exchange Chromatography and Mass Spectrometric Methods for Analysis of Cadmium-Phytochelatin (II) Complexes. International Journal of Environmental Research and Public Health. 2013; 10(4):1304-1311. https://doi.org/10.3390/ijerph10041304
Chicago/Turabian StyleRodrigo, Miguel Angel Merlos, Natalia Cernei, Marketa Kominkova, Ondrej Zitka, Miroslava Beklova, Josef Zehnalek, Rene Kizek, and Vojtech Adam. 2013. "Ion Exchange Chromatography and Mass Spectrometric Methods for Analysis of Cadmium-Phytochelatin (II) Complexes" International Journal of Environmental Research and Public Health 10, no. 4: 1304-1311. https://doi.org/10.3390/ijerph10041304



