Assessment of Infantile Mineral Imbalances in Autism Spectrum Disorders (ASDs)
Abstract
:1. Introduction
2. Mineral Disorders in Autism
2.1. Infantile Zinc Deficiency

2.2. Infantile Magnesium/Calcium Deficiency
| Mineral | Number of Cases with Deficiency | Rate (%) of Deficiency |
|---|---|---|
| Zn | 584 | 29.7 |
| Mg | 347 | 17.6 |
| Ca | 114 | 5.8 |
| Co | 40 | 2.0 |
| Fe | 17 | 0.9 |
| Cr | 12 | 0.6 |
| Mn | 4 | 0.2 |
| Cu | 4 | 0.2 |
2.3. Toxic Metal Burdens
| Toxic Metal | Number of Cases with High Burden | Rate (%) of High Burden | Maximum (ppm) | Ratio to Reference |
|---|---|---|---|---|
| Al | 339 | 17.2 | 79.4 | 21.1 |
| Cd | 168 | 8.5 | 5.5 | 782.0 |
| Pb | 94 | 4.8 | 24.9 | 57.4 |
| Hg | 56 | 2.8 | 36.3 | 9.3 |
| As | 52 | 2.6 | 1.7 | 33.5 |
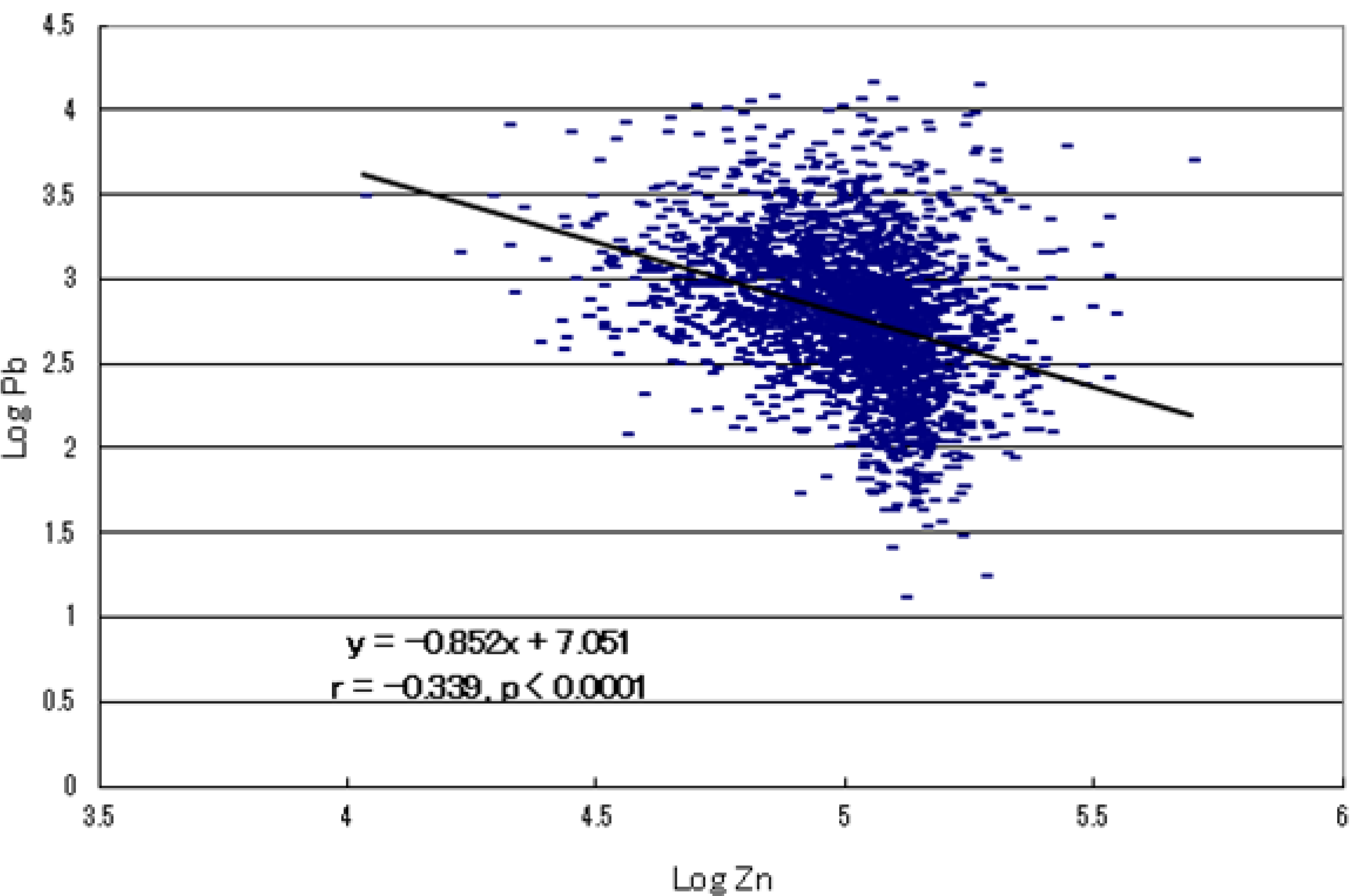
2.4. Metallomics Profiles in Autistic Infants
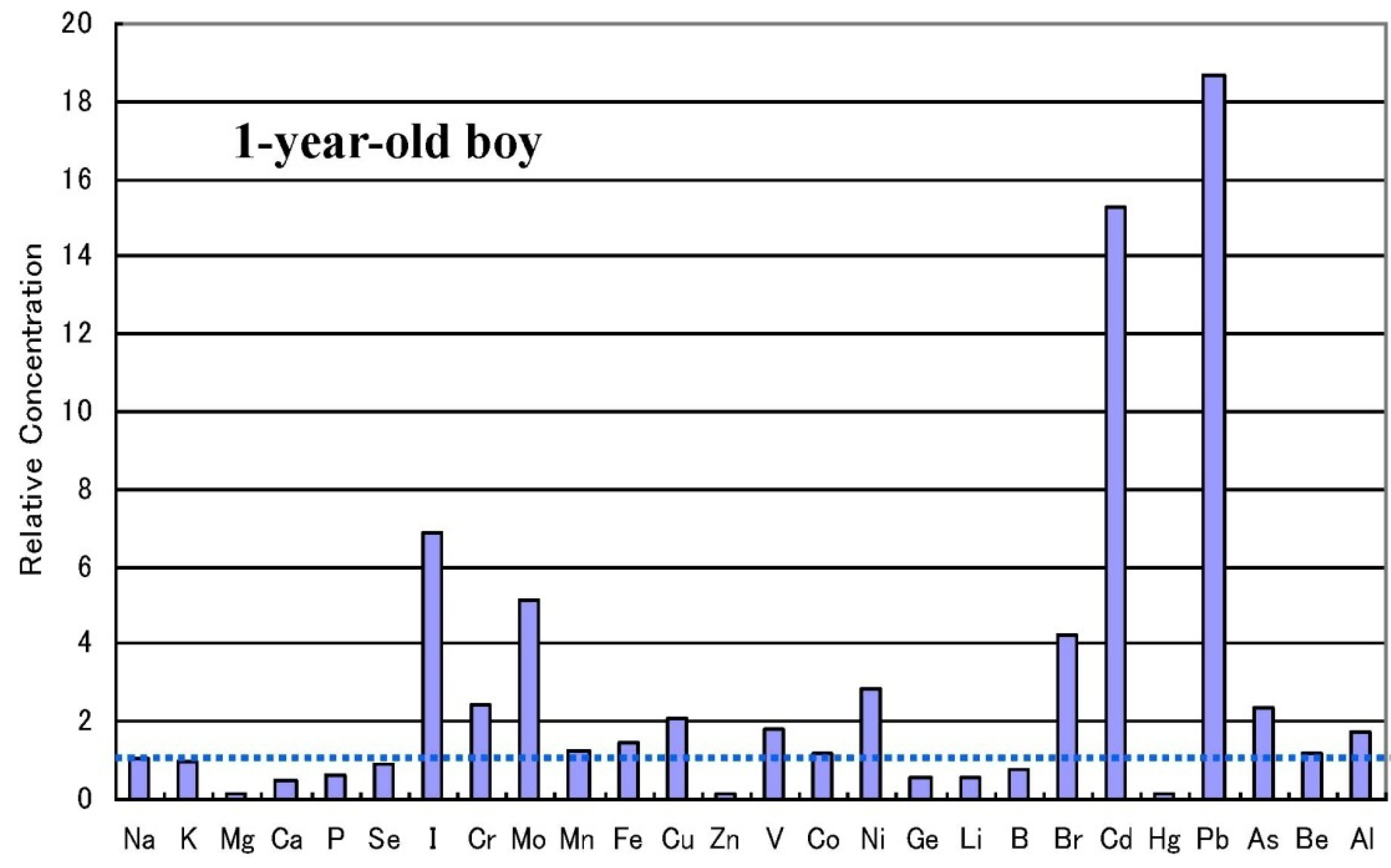
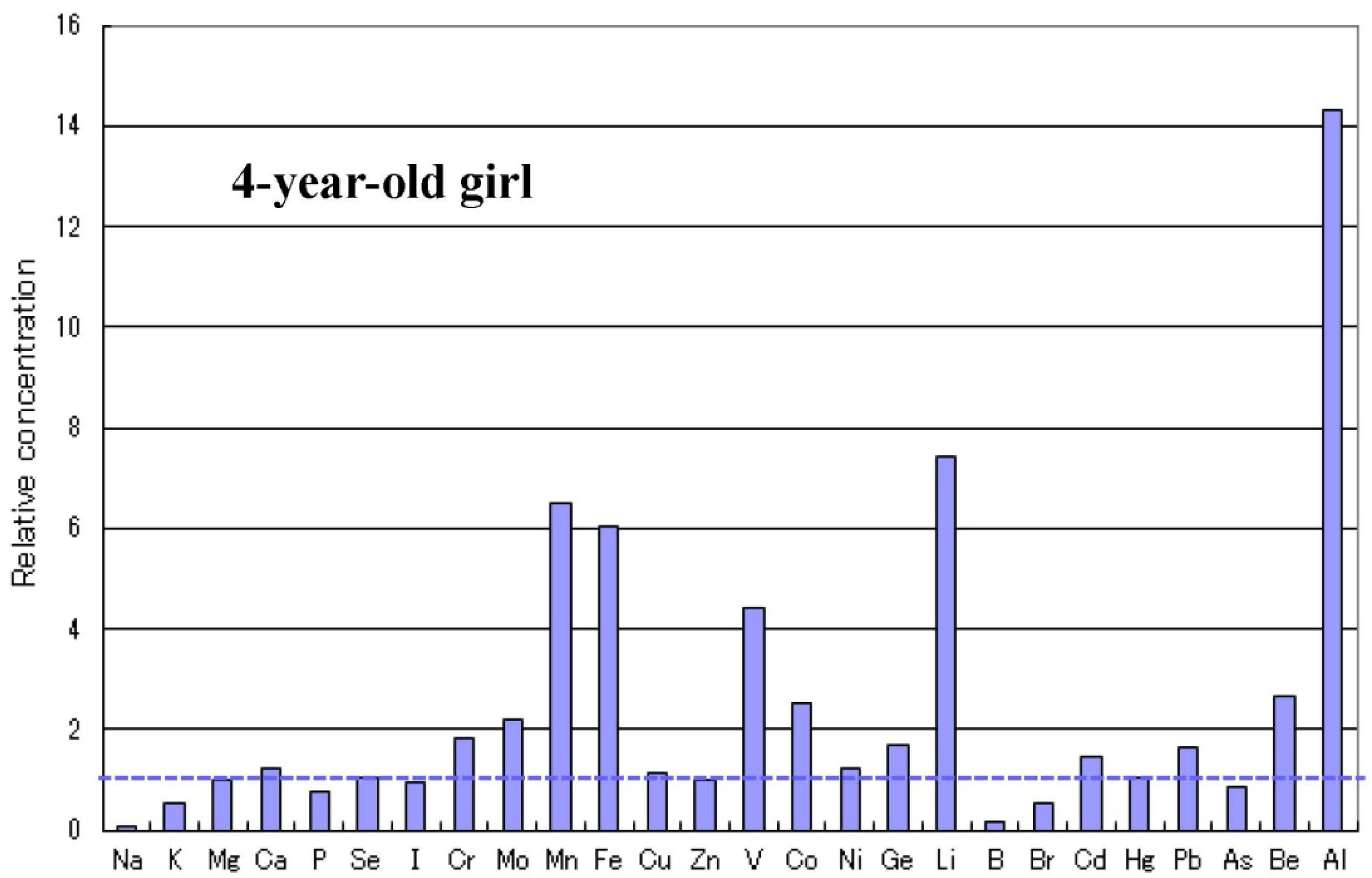
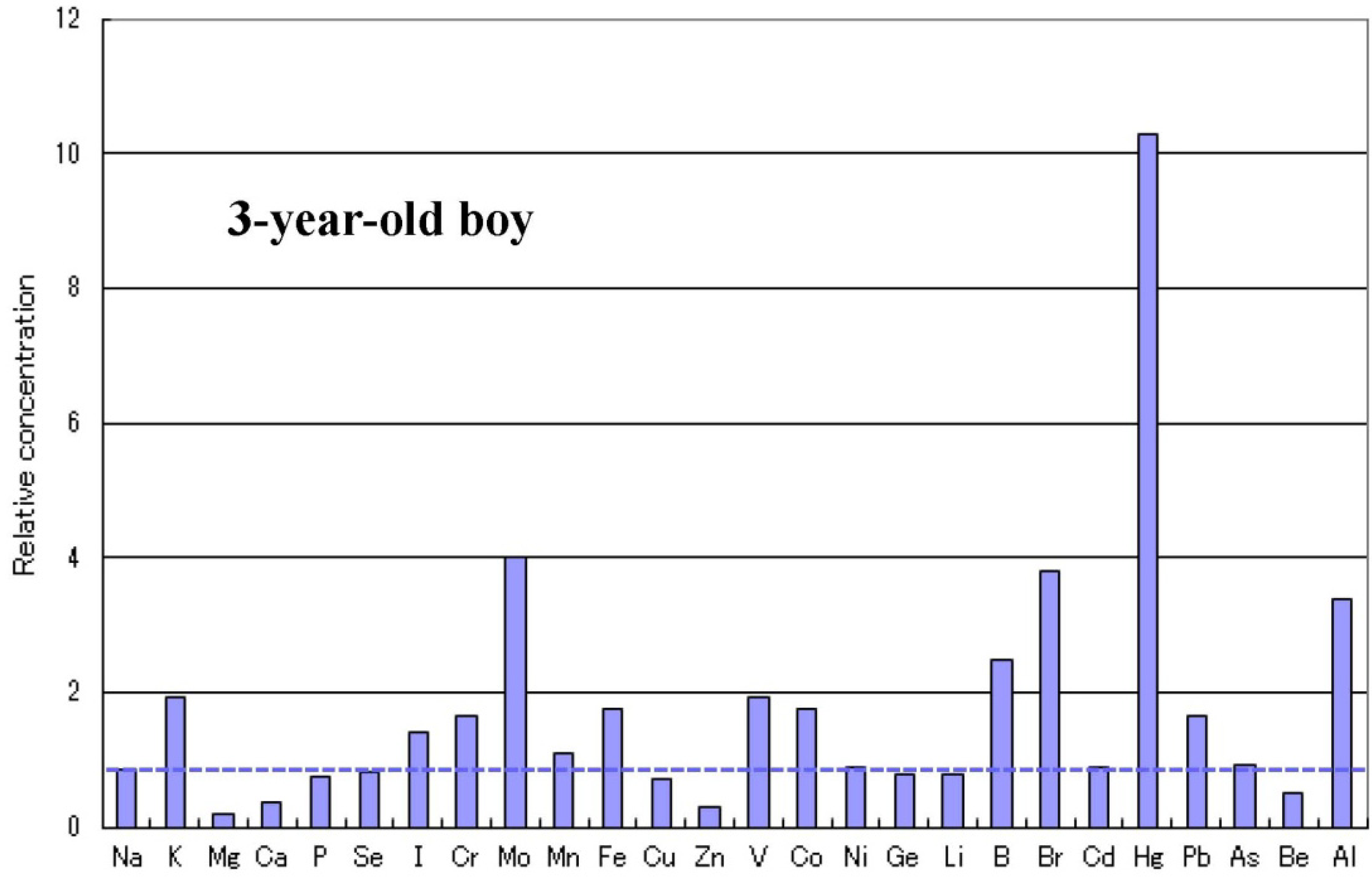
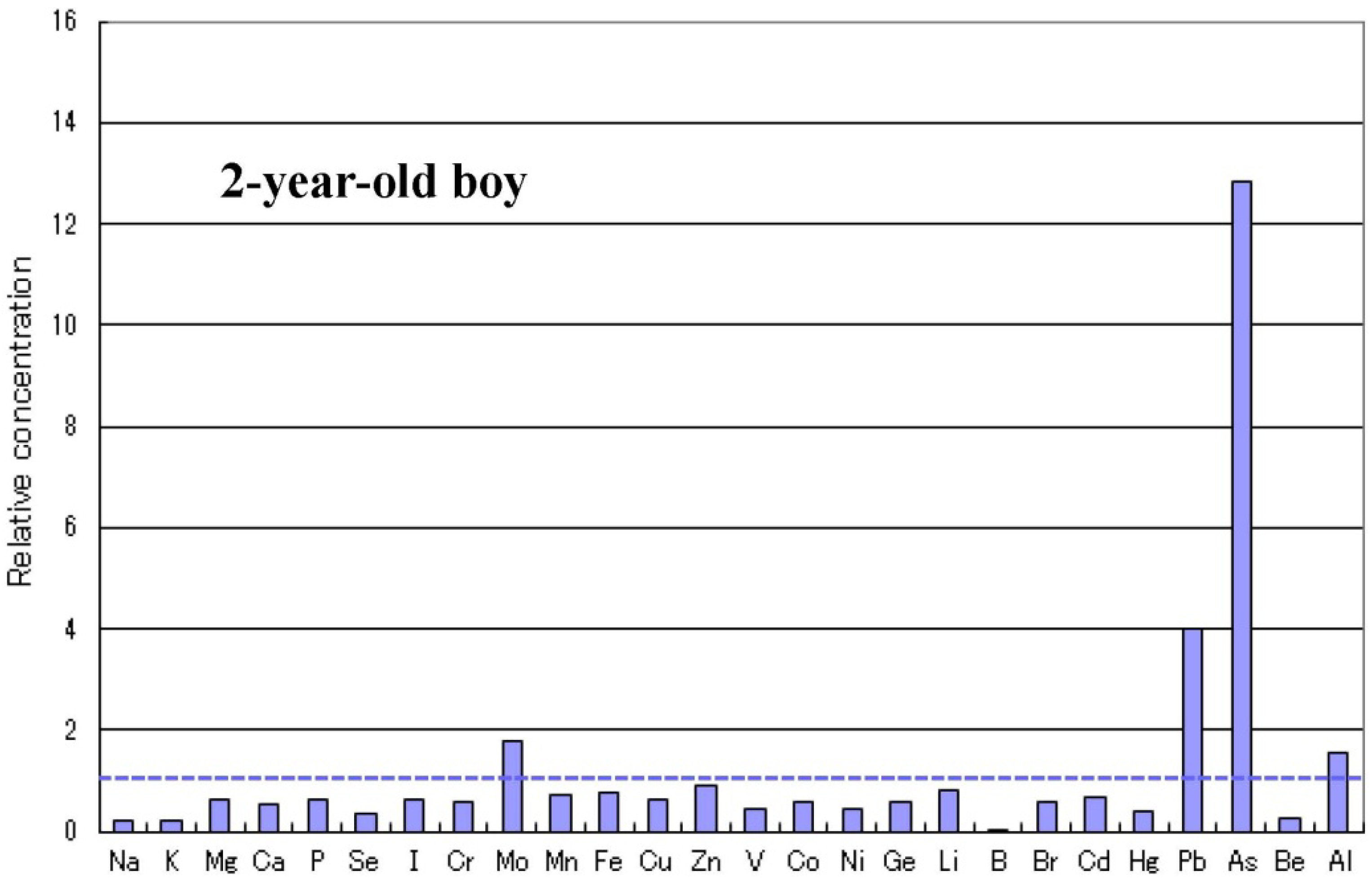
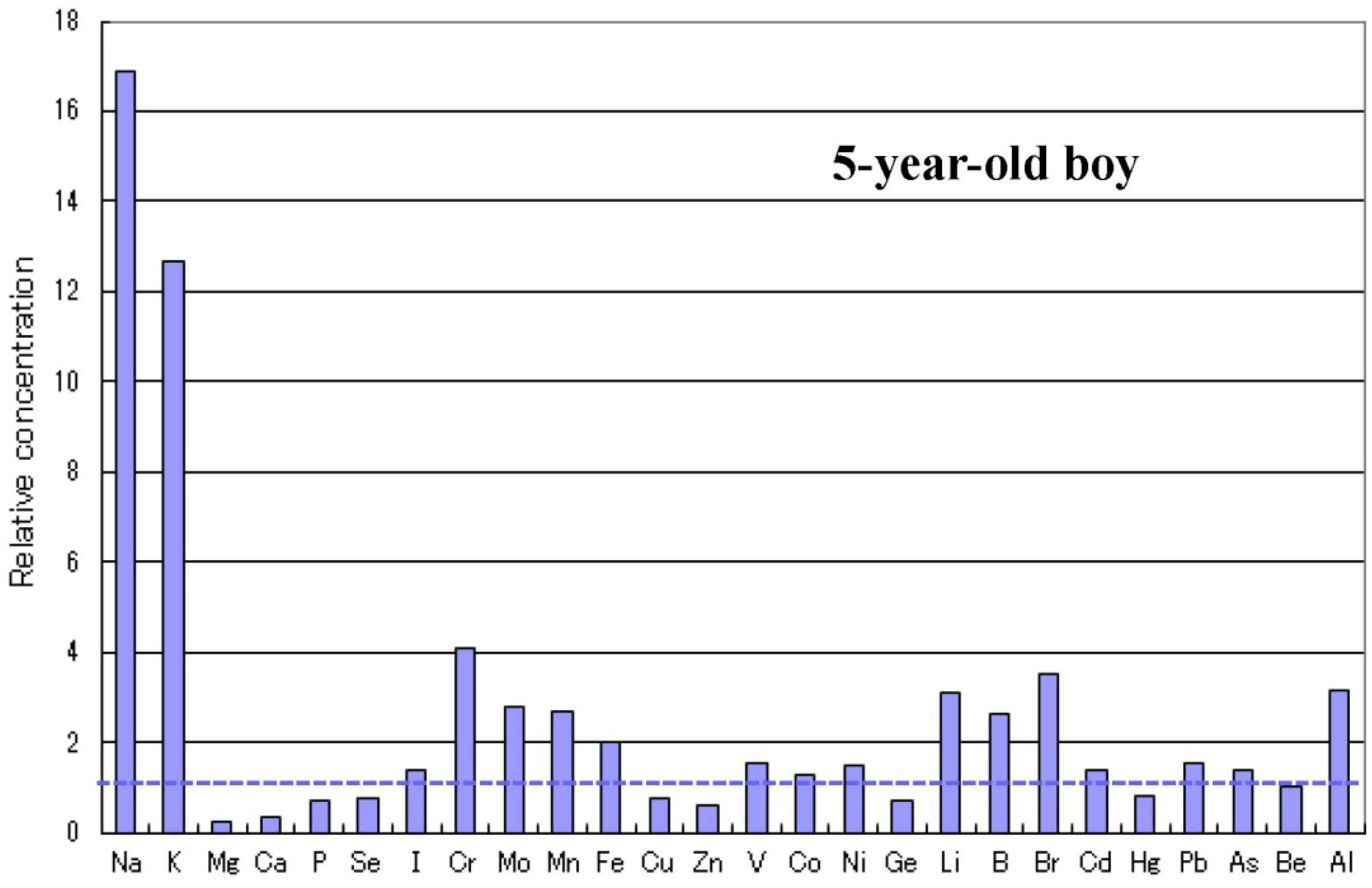
2.5. Infantile Time Window in Neurodevelopment and for Therapy
2.6. Autism-Related Genes and Epigenetic Alteration by Mineral Disorders
3. Discussion
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Weintraub, K. Autism counts. Nature 2011, 479, 22–24. [Google Scholar]
- Pinto, D.; Pagnamenta, A.T.; Klei, L.; Anney, R.; Merico, D.; Regan, R.; Conroy, J.; Magalhaes, T.R.; Correia, C.; Abrahams, B.S.; et al. Functional impact of global rare copy number variation in autism spectrum disorders. Nature 2010, 466, 368–372. [Google Scholar]
- Chakrabarti, S.; Fombonne, E. Pervasive developmental disorders in preschool children: Confirmation of high prevalence. Amer. J. Psychiat. 2005, 162, 1133–1141. [Google Scholar] [CrossRef]
- Bailey, A.; le Couteur, A.; Gottesman, I.; Bolton, P.; Simonoff, E.; Yuzda, E.; Rutter, M. Autism as a strongly genetic disorder: Evidence from a British twin study. Psychol. Med. 1995, 25, 63–77. [Google Scholar] [CrossRef]
- Marshall, C.R.; Noor, A.; Vincent, J.B.; Lionel, A.C.; Feuk, L.; Skaug, J.; Shago, M.; Moessner, R.; Pinto, D.; Ren, Y.; et al. Structural variation of chromosomes in autism spectrum disorder. Amer. J. Hum. Genet. 2008, 82, 477–488. [Google Scholar] [CrossRef]
- Kim, S.J.; Silva, R.M.; Flores, C.G.; Jacob, S.; Guter, S.; Valcante, G.; Zaytoun, A.M.; Cook, E.H.; Badner, J.A. A quantitative association study of SLC25A12 and restricted repetitive behavior traits in autism spectrum disorders. Mol. Autism 2011, 2. [Google Scholar] [CrossRef]
- Murdoch, J.D.; State, M.W. Recent developments in the genetics of autism spectrum disorders. Curr. Opin. Genet. Dev. 2013, 23, 310–315. [Google Scholar] [CrossRef]
- Huguet, G.; Ey, E.; Bourgeron, T. The genetic landscapes of autism spectrum disorders. Ann. Rev. Genomics Hum. Genet. 2013, 14, 191–213. [Google Scholar] [CrossRef]
- Hughes, V. Autism: Complex disorder. Nature 2012, 491, S2–S3. [Google Scholar] [CrossRef]
- Dufault, R.; Schnoll, R.; Lukiw, W.J.; Leblanc, B.; Cornett, C.; Patrick, L.; Wallinga, D.; Gilbert, S.G.; Crider, R. Mercury exposure, nutritional deficiencies and metabolic disruption may affect learning in children. Behav. Brain Funct. 2009, 5, 44–58. [Google Scholar] [CrossRef]
- Palmer, R.F.; Blanchard, S.; Wood, R. Proximity to point sources of environmental mercury release as a predictor of autism prevalence. Health Place 2009, 15, 18–24. [Google Scholar] [CrossRef]
- Majewska, M.D.; Urbanowicz, E.; Rok-Bujko, P.; Namyslowska, I.; Mierzejewsky, P. Age-dependent lower or higher levels of hair mercury in autistic children than in healthy controls. Acta Neurobiol. Exp. 2010, 70, 196–208. [Google Scholar]
- O’Rahilly, S. Human genetics illuminates the paths to metabolic disease. Nature 2009, 462, 307–314. [Google Scholar] [CrossRef]
- James, S.J.; Shpyleva, S.; Melnyk, S.; Pavliv, O.; Pogribny, I.P. Complex epigenetic regulation of engrailed-2 (EN-2) homeobox gene in the autism cerebellum. Transl. Psychiat. 2013, 3, e232. [Google Scholar] [CrossRef]
- Jin, Y.H.; Clark, A.B.; Slebos, R.J.; Al-Refai, H.; Taylor, J.A.; Kunkel, T.A.; Resnick, M.A.; Gordenin, D.A. Cadmium is a mutagen that acts by inhibiting mismatch repair. Nat. Genet. 2003, 34, 239–241. [Google Scholar] [CrossRef]
- Takiguchi, M.; Achanzar, W.E.; Qu, W.; Li, G.; Waalkes, M.P. Effects of cadmium on DNA-(cytosine-5) methyltransferase activity and DNA methylation status during cadmium-induced cellular transformation. Exp. Cell Res. 2003, 286, 355–365. [Google Scholar] [CrossRef]
- Arita, A.; Costa, M. Epigenetics in metal carcinogenesis: Nickel, arsenic, chromium and cadmium. Metallomics 2009, 1, 222–228. [Google Scholar] [CrossRef]
- Perera, F.; Herbstman, J. Prenatal environmental exposure, epigenetics, and disease. Reprod. Toxicol. 2011, 31, 363–373. [Google Scholar] [CrossRef]
- Jakovcevski, M.; Akbarian, S. Epigenetic mechanisms in neurological disease. Nat. Med. 2012, 18, 1194–1204. [Google Scholar] [CrossRef]
- Ciesielski, T.; Weuve, J.; Bellinger, D.C.; Schwartz, J.; Lanphear, B.; Wright, R.O. Cadmium exposure and neurodevelopmental outcomes in U.S. children. Environ. Health Perspect. 2012, 120, 758–763. [Google Scholar] [CrossRef]
- Rodushkin, I.; Axelsson, M.D. Application of double focusing sector field ICP-MS for multi elemental characterization of human hair and nails. Part I. Analytical methodology. Sci. Total Environ. 2000, 250, 83–100. [Google Scholar] [CrossRef]
- Goulle, J.P.; Mahieu, L.; Castermant, J.; Neveu, N.; Bonneau, L.; Lainé, G.; Bouige, D.; Lacroix, C. Metal and metalloid multi-elementary ICP-MS validation in whole blood, plasma, urine and hair: Reference values. Forensic Sci. Int. 2005, 153, 39–44. [Google Scholar] [CrossRef]
- Wang, C.T.; Chang, W.T.; Zeng, W.F.; Lin, C.H. Concentrations of calcium, copper, iron, magnesium, potassium, sodium and zinc in adult female hair with different body mass indexes in Taiwan. Clin. Chem. Lab. Med. 2005, 43, 389–393. [Google Scholar]
- Munakata, M.; Onuma, A.; Kobayashi, Y.; Haginoya, K.; Yokoyama, H.; Fujiwara, I.; Yasuda, H.; Tsutsui, T.; Iinuma, K. A preliminary analysis of trace elements in the scalp hair of patients with severe motor disabilities receiving enteral nutrition. Brain Dev. 2006, 28, 521–525. [Google Scholar] [CrossRef]
- Yasuda, H.; Yonashiro, T.; Yoshida, K.; Ishii, T.; Tsutsui, T. Mineral imbalance in children with autistic disorders. Biomed. Res. Trace Elem. 2005, 16, 285–291. [Google Scholar]
- Yasuda, H.; Yonashiro, T.; Yoshida, K.; Ishii, T.; Tsutsui, T. Relationship between body mass index and minerals in male Japanese adults. Biomed. Res. Trace Elem. 2006, 17, 316–321. [Google Scholar]
- Yasuda, H.; Yoshida, K.; Segawa, M.; Tokuda, R.; Tsutsui, T.; Yasuda, Y.; Magara, S. Metallomics study using hair mineral analysis and multiple logistic regression analysis: Relationship between cancer and minerals. Environ. Health Prev. Med. 2009, 14, 261–266. [Google Scholar] [CrossRef]
- Ochi, A.; Ishimura, E.; Tsujimoto, Y.; Kakiya, R.; Tabata, T.; Mori, K.; Tahara, H.; Shoji, T.; Yasuda, H.; Nishizawa, Y.; et al. Elemental concentrations in scalp hair, nutritional status and health-related quality of life in haemodialysis patients. Ther. Apher. Dial. 2012, 16, 127–133. [Google Scholar] [CrossRef]
- Yasuda, H.; Yonashiro, T.; Yoshida, K.; Ishii, T.; Tsutsui, T. High toxic metal levels in scalp hair of infants and children. Biomed. Res. Trace Elem. 2005, 16, 39–45. [Google Scholar]
- Yasuda, H.; Yoshida, K.; Segawa, M.; Tokuda, R.; Yasuda, Y.; Tsutsui, T. High accumulation of aluminium in hairs of infants and children. Biomed. Res. Trace Elem. 2008, 19, 57–62. [Google Scholar]
- Yasuda, H.; Yoshida, K.; Yasuda, Y.; Tsutsui, T. Two age-related accumulation profiles of toxic metals. Curr. Aging Sci. 2012, 5, 105–111. [Google Scholar] [CrossRef]
- Yasuda, H.; Yoshida, K.; Yasuda, Y.; Tsutsui, T. Infantile zinc deficiency: Association with autism spectrum disorders. Sci. Rep. 2011, 1. [Google Scholar] [CrossRef]
- Yasuda, H.; Yasuda, Y.; Tsutsui, T. Estimation of autistic children by metallomics analysis. Sci. Rep. 2013, 3, ep01199. [Google Scholar] [CrossRef]
- Landa, R.J. Diagnosis of autism spectrum disorders in the first 3 years of life. Nat. Clin. Pract. Neurol. 2008, 4, 138–147. [Google Scholar] [CrossRef]
- Huang, Y.Z.; Pan, E.; Xiong, Z.Q.; McNamara, J.O. Zinc-mediated transactivation of TrkB potentiates the hippocampal mossy fiber-CA3 pyramidal synapse. Neuron 2008, 57, 546–558. [Google Scholar] [CrossRef]
- O’Roak, B.J.; Deriziotis, P.; Lee, C.; Vives, L.; Schwartz, J.J.; Girirajan, S.; Karakoc, E.; Mackenzie, A.P.; Ng, S.B.; Baker, C.; et al. Exome sequencing in sporadic autism spectrum disorders identifies severe de novo mutations. Nat. Genet. 2011, 43, 585–589. [Google Scholar] [CrossRef]
- Sanders, S.J.; Murtha, M.T.; Gupta, A.R.; Murdoch, J.D.; Raubeson, M.J.; Willsey, A.J.; Ercan-Sencicek, A.G.; DiLullo, N.M.; Parikshak, N.N.; Stein, J.L.; et al. De novo mutations revealed by whole exome sequencing are strongly associated with autism. Nature 2012, 485, 237–241. [Google Scholar] [CrossRef]
- Grabrucker, A.M. Environmental factors in autism. Front. Psychist. 2013, 3, 1–13. [Google Scholar]
- Dufault, R.; Lukiw, W.J.; Crider, R.; Schnoll, R.; Wallinga, D.; Deth, R. A macroepigenetic approach to identify factors responsible for the autism epidemic in the United States. Clin. Epigenetics 2012, 4, 6. [Google Scholar] [CrossRef]
- Grabrucker, A.M. A role for synaptic zinc in ProSAP/Shank PSD scaffold malformation in autism spectrum disorders. Dev. Neurobiol. 2013. [Google Scholar] [CrossRef]
- Fukada, T.; Yamasaki, S.; Nishida, K.; Murakami, M.; Hirano, T. Zinc homeostasis and signalling in health and diseases: Zinc signalling. J. Biol. Inorg. Chem. 2011, 16, 1123–1134. [Google Scholar] [CrossRef]
- Andreini, C.; Banci, L.; Bertini, I.; Rosato, A. Counting the zinc-proteins encoded in the human genome. J. Proteome Res. 2006, 5, 196–201. [Google Scholar] [CrossRef]
- Takeda, A. Movement of zinc and its functional significance in the brain. Brain Res. Rev. 2000, 34, 137–148. [Google Scholar] [CrossRef]
- Takeda, A.; Nakamura, M.; Fujii, H.; Tamano, H. Synaptic Zn(2+) homeostasis and its significance. Metallomics 2013, 5, 417–423. [Google Scholar] [CrossRef]
- Prasad, A.S. Impact of the discovery of human zinc deficiency on health. J. Am. Coll. Nutr. 2009, 28, 257–265. [Google Scholar] [CrossRef]
- Arnold, L.E.; di Silvestro, R.A. Zinc in attention-deficit/hyperactivity disorder. J. Child Adolesc. Psychopharmacol. 2005, 15, 619–627. [Google Scholar] [CrossRef]
- di Girolamo, A.M.; Raminez-Zea, M. Role of zinc in maternal and child mental health. Amer. J. Clin. Nutr. 2009, 89, S940–S945. [Google Scholar] [CrossRef]
- Scheplyagina, L.A. Impact of the mother’s zinc deficiency on the woman’s and new-borns health status. J. Trace Elem. Med. Biol. 2005, 19, 29–35. [Google Scholar] [CrossRef]
- Plum, L.M.; Rink, L.; Haase, H. The essential toxin: Impact of zinc on human health. Int. J. Environ. Res. Public Health 2010, 7, 1342–1365. [Google Scholar] [CrossRef]
- Yorbik, O.; Akay, C.; Sayal, A.; Cansever, A.; Sohmen, T.; Cavdar, A.O. Zinc status in autistic children. J. Trace Elem. Exp. Med. 2004, 17, 101–107. [Google Scholar] [CrossRef]
- Fido, A.; Al-Saad, S. Toxic trace elements in the hair of children with autism. Autism 2005, 9, 290–298. [Google Scholar] [CrossRef]
- Adams, J.B.; Holloway, C.E.; George, F.; Quig, D. Analyses of toxic metals and essential minerals in the hair of Arizona children with autism and associated conditions, and their mothers. Biol. Trace Elem. Res. 2006, 110, 193–209. [Google Scholar] [CrossRef]
- Faber, S.; Zinn, G.M.; Kern, J.C.; Kingston, H.M. The plasma zinc/serum copper ratio as a biomarker in children with autism spectrum disorders. Biomarkers 2009, 14, 171–180. [Google Scholar] [CrossRef]
- Priya, M.D.L.; Geetha, A. Level of trace elements (copper, zinc, magnesium and selenium) and toxic elements (lead and mercury) in the hair and nail of children with autism. Biol. Trace Elem. Res. 2011, 142, 148–158. [Google Scholar] [CrossRef]
- Gebremedhin, S.; Enquselassie, F.; Umeta, M. Prevalence of prenatal zinc deficiency and its association with socio-demographic, dietary and health care related factors in Rural Sidama, Southern Ethiopia: A cross-sectional study. BMC Public Health 2011, 11, 898–907. [Google Scholar] [CrossRef]
- Kurita, H.; Ohsako, S.; Hashimoto, S.; Yoshinaga, J.; Tohyama, C. Prenatal zinc deficiency-dependent epigenetic alterations of mouse metallothionein-2 gene. J. Nutr. Biochem. 2013, 24, 256–266. [Google Scholar] [CrossRef]
- Arnold, L.E.; Bozzolo, H.; Hollway, J.; Cook, A.; di Silvestro, R.A.; Bozzolo, D.R.; Crowl, L.; Ramadan, Y.; Williams, C. Serum zinc correlates with parent- and teacher- rated inattention in children with attention-deficit/hyperactivity disorder. J. Child Adoles. Psychopharmacol. 2005, 15, 628–636. [Google Scholar] [CrossRef]
- Yorbik, O.; Ozdag, M.F.; Olgun, A.; Senol, M.G.; Bek, S.; Akman, S. Potential effects of zinc on information processing in boys with attention deficit hyper-activity disorder. Prog. Neuropsychopharmacol. Biol. Psychiatry 2008, 32, 662–667. [Google Scholar] [CrossRef]
- di Girolamo, A.M.; Ramirez-Zea, M.; Wang, M.; Flores-Ayala, R.; Martorell, R.; Neufeld, L.M.; Ramakrishnan, U.; Sellen, D.; Black, M.M.; Stein, A.D. Randomized trial of the effect of zinc supplementation on the mental health of school-age children in Guatemala. Am. J. Clin. Nutr. 2010, 92, 1241–1250. [Google Scholar] [CrossRef]
- Akhondzadeh, S.; Mohammadi, M.R.; Khademi, M. Zinc sulfate as an adjunct to methylphenidate for the treatment of attention deficit hyperactivity disorder in children: A double blind and randomised trial [ISRCTN64132371]. BMC Psychiatry 2004, 4, 9–14. [Google Scholar] [CrossRef]
- Kozielec, T.; Starobrat-Hermelin, B. Assessment of magnesium levels in children with attention deficit hyperactivity disorder (ADHD). Magnes. Res. 1997, 10, 143–148. [Google Scholar]
- Starobrat-Hermelin, B.; Kozielec, T. The effects of magnesium physiological supplementation on hyperactivity in children with attention deficit hyperactivity disorder (ADHD). Positive response to magnesium oral loading test. Magnes. Res. 1997, 10, 149–156. [Google Scholar]
- Mousain-Bose, M.; Roche, M.; Rapin, J.; Bali, J.P. Magnesium VitB6 intake reduces central nervous system hyperexcitability in children. J. Amer. Coll. Nutr. 2004, 23, S545–S548. [Google Scholar] [CrossRef]
- Mousain-Bose, M.; Roche, M.; Polge, A.; Pradal-Prat, D.; Rapin, J.; Bali, J.P. Improvement of neurobehavioral disorders in children supplemented with magnesium-vitanin B6 II. Pervasive developmental disorder-autism. Magnes. Res. 2006, 19, 53–62. [Google Scholar]
- Ochi, A.; Ishimura, E.; Tsujimoto, Y.; Kakiya, R.; Tabata, T.; Mori, K.; Fukumoto, S.; Tahara, H.; Shoji, T.; Yasuda, H.; et al. Hair magnesium, but not serum magnesium, is associated with left ventricular wall thickness in hemodialysis patients. Circ. J. 2013. [Google Scholar] [CrossRef]
- Dufner-Beattie, J.; Kuo, Y.M.; Gitschier, J.; Andrews, G.K. The adaptive response to dietary zinc in mice involves the differential cellular localization and zinc regulation of the zinc transporters ZIP4 and ZIP5. J. Biol. Chem. 2004, 279, 49082–49090. [Google Scholar] [CrossRef]
- Lichten, L.A.; Cousins, R.J. Mammalian zinc transporters: Nutritional and physiologic regulation. Ann. Rev. Nutr. 2009, 29, 153–176. [Google Scholar] [CrossRef]
- Goyer, R.A. Toxic and essential metal interactions. Ann. Rev. Nutr. 1997, 17, 37–50. [Google Scholar] [CrossRef]
- Mahaffey, K.R.; Gartside, P.S.; Glueck, C.J. Blood lead levels and dietary calcium intake in 1 to 11 year-old children: The second national health and nutrition examination survey, 1976 to 1980. Pediatrics 1986, 78, 257–262. [Google Scholar]
- Shannon, M.; Graef, J.W. Lead intoxication in children with pervasive developmental disorders. J. Toxicol. Clin. Toxicol. 1996, 34, 177–181. [Google Scholar] [CrossRef]
- Eubig, P.A.; Agular, A.; Schantz, S.L. Lead and PCBs as risk factors for attention deficit/hyperactivity disorder. Environ. Health Perspect. 2010, 118, 1654–1667. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention (CDC). CDC National Surveillance Data (1997–2009), National Centers for Environmental Health 2012. Available online: http://www.cdc.gov/nceh/lead/data/national.htm. (accessed on 6 November 2013).
- Jones, R.L.; Homa, D.M.; Meyer, P.A.; Brody, D.J.; Caldwell, K.L.; Pirkle, J.L.; Brown, M.J. Trends in blood lead levels and blood testing among US children aged 1 to 5 years, 1988–2004. Pediatrics 2009, 123, 376–385. [Google Scholar] [CrossRef]
- Advisory Committee on Childhood Lead Poisoning Prevention, of the Centers for Disease Control and Prevention. Low Level Lead Exposure Harms Children: A Renewed Call for Primary Prevention: Report to the CDCP; ACCLPP: Atlanta, GA, USA, 2012; pp. 1–54.
- Needleman, H.L.; Schell, A.; Bellinger, D.; Leviton, A.; Allred, E.N. The long-term effects of exposure to low doses of lead in childhood. An 11-year follow up report. N. Engl. J. Med. 1990, 322, 83–88. [Google Scholar] [CrossRef]
- Binns, H.J.; Campbell, C.; Brown, M.J. Interpreting and managing blood lead levels of less than 10 micro g/dL in children and reducing childhood exposure to lead: Recommendations of the centers for disease control and prevention advisory committee on childhood lead poisoning prevention. Pediatrics 2007, 120, e1285–e1298. [Google Scholar] [CrossRef]
- Bellinger, D.C. Lead neurotoxicity and socioeconomic status: Conceptual and analytical issues. Neurotoxicology 2008, 29, 828–832. [Google Scholar] [CrossRef]
- Gump, B.B.; Stewart, P.; Reihman, J.; Lonky, E.; Darvill, T.; Parsons, P.J.; Granger, D.A. Low-level prenatal and postnatal blood lead (Pb) exposure and adrenocortical responses to acute stress in children. Environ. Health Perspect. 2008, 116, 249–255. [Google Scholar]
- Nigg, J.T.; Knottnerus, G.M.; Martel, M.M.; Nikoas, M.; Cavanagh, V.; Karmaus, W.; Rappley, M.D. Low blood lead levels associated with clinically diagnosed attention-deficit/hyperactivity disorder and mediated by weak cognitive control. Biol. Psychiatry 2008, 63, 325–331. [Google Scholar] [CrossRef]
- Dixon, S.L.; Gaitens, J.M.; Jacobs, D.E.; Strauss, W.; Nagaraja, L.; Pivetz, T.; Wilson, J.W.; Ashley, P.J. Exposure of U.S. children to residential dust lead, 1999–2004: II. The contribution of lead-contaminated dust to children’s blood lead levels. Environ. Health Perspect. 2009, 117, 468–474. [Google Scholar]
- American Academy of Pediatrics. Lead exposure in children: Prevention, detection, and management: Statement of policy reaffirmation. Psychiatry 2009, 123, 1421–1422. [Google Scholar]
- Deocampo, D.M.; Reed, P.J.; Kalenuik, A.P. Road dust lead (Pb) in two neighbourhoods of urban Atlanta, (GA, USA). Int. J. Environ. Res. Public Health 2012, 9, 2020–2030. [Google Scholar] [CrossRef]
- Hall, G.; Tinklenberg, J. Determination of Ti, Zn, and Pb in lead-based house paints by EDXRF. J. Anal. At. Spectrom. 2003, 18, 775–778. [Google Scholar] [CrossRef]
- Lewis, P.A. Inorganic Colored Pigments. In Paint and Coating Testing Manual, 14th ed.; Koleske, J.V., Ed.; ASTM: West Conshohocken, PA, USA, 1995; pp. 1–950. [Google Scholar]
- Symanski, E.; Hertz-Picciotto, I. Blood lead levels in relation to menopause, smoking, and pregnancy history. Amer. J. Epidemiol. 1995, 141, 1047–1058. [Google Scholar]
- Razagui, I.B.; Ghribi, I. Maternal and neonatal scalp hair concentrations of zinc, cadmium, and lead: Relationship to some lifestyle factors. Biol. Trace Elem. Res. 2005, 106, 1–28. [Google Scholar] [CrossRef]
- Gulson, B.L.; Jameson, C.W.; Mahaffey, K.R.; Mizon, K.J.; Korsch, M.J.; Vimpani, G. Pregnancy increases mobilization of lead from maternal skeleton. J. Lab. Clin. Med. 1997, 130, 51–62. [Google Scholar] [CrossRef]
- Sanders, A.P.; Flood, K.; Chiang, S.; Herring, A.H.; Wolf, L.; Fry, R.C. Towards prenatal biomonitoring in North Carolina: Assessing arsenic, cadmium, mercury and lead levels in pregnant women. PLoS One 2012, 7, e31354. [Google Scholar] [CrossRef]
- Parajuli, R.P.; Fujiwara, T.; Umezaki, M.; Watanabe, C. Association of cord blood levels of lead, arsenic, and zinc with neurodevelopmental indicators in newborns: A birth cohort study in Chitwan Valley, Nepal. Environ. Res. 2012, 10, 509–519. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yasuda, H.; Tsutsui, T. Assessment of Infantile Mineral Imbalances in Autism Spectrum Disorders (ASDs). Int. J. Environ. Res. Public Health 2013, 10, 6027-6043. https://doi.org/10.3390/ijerph10116027
Yasuda H, Tsutsui T. Assessment of Infantile Mineral Imbalances in Autism Spectrum Disorders (ASDs). International Journal of Environmental Research and Public Health. 2013; 10(11):6027-6043. https://doi.org/10.3390/ijerph10116027
Chicago/Turabian StyleYasuda, Hiroshi, and Toyoharu Tsutsui. 2013. "Assessment of Infantile Mineral Imbalances in Autism Spectrum Disorders (ASDs)" International Journal of Environmental Research and Public Health 10, no. 11: 6027-6043. https://doi.org/10.3390/ijerph10116027
APA StyleYasuda, H., & Tsutsui, T. (2013). Assessment of Infantile Mineral Imbalances in Autism Spectrum Disorders (ASDs). International Journal of Environmental Research and Public Health, 10(11), 6027-6043. https://doi.org/10.3390/ijerph10116027




