Antimicrobial Effect of 7-O-Butylnaringenin, a Novel Flavonoid, and Various Natural Flavonoids against Helicobacter pylori Strains
Abstract
:1. Introduction
2. Experimental Section
2.1. Bacterial Strains
2.2. Flavonoids

2.3. Screening by Paper Disc Diffusion Method
2.4. Assay of Antimicrobial Effects on H. pylori
2.5. Flavonoid Inhibition of Urease in H. pylori
2.6. Preparation of H. pylori for Scanning Electron Microscopy (SEM)
2.7. Statistical Analysis
3. Results and Discussion
3.1. Screening by Paper Disc Diffusion Method
| Flavonoid | Conc. (mM) | H. pylori 26695 | H. pylori 51 | H. pylori SS1 |
|---|---|---|---|---|
| Kaempferol | 2.5 | - | - | - |
| 5 | ++ | - | + | |
| 10 | ++ | + | + | |
| 20 | ++ | + | ++ | |
| Quercetin | 2.5 | - | - | - |
| 5 | - | - | - | |
| 10 | - | - | + | |
| 20 | + | - | + | |
| Apigenin | 2.5 | - | - | - |
| 5 | - | - | - | |
| 10 | - | - | - | |
| 20 | - | - | - | |
| Naringenin | 2.5 | - | - | - |
| 5 | +++ | +++ | +++ | |
| 10 | +++ | +++ | +++ | |
| 20 | +++ | +++ | +++ | |
| Luteolin | 2.5 | - | - | - |
| 5 | + | + | - | |
| 10 | ++ | ++ | + | |
| 20 | ++ | ++ | + | |
| Hesperetin | 2.5 | - | - | - |
| 5 | +++ | +++ | +++ | |
| 10 | +++ | +++ | +++ | |
| 20 | +++ | +++ | +++ | |
| Hesperidin | 2.5 | - | - | - |
| 5 | - | - | - | |
| 10 | - | - | - | |
| 20 | - | - | - | |
| Genkwanin | 2.5 | - | - | - |
| 5 | - | - | - | |
| 10 | - | - | - | |
| 20 | - | - | - | |
| 7-O-Butylnaringenin | 2.5 | - | - | - |
| 5 | - | ++ | + | |
| 10 | + | + | + | |
| 20 | ++ | ++ | ++ |
3.2. Evaluation of Antimicrobial Effects on H. pylori Strains
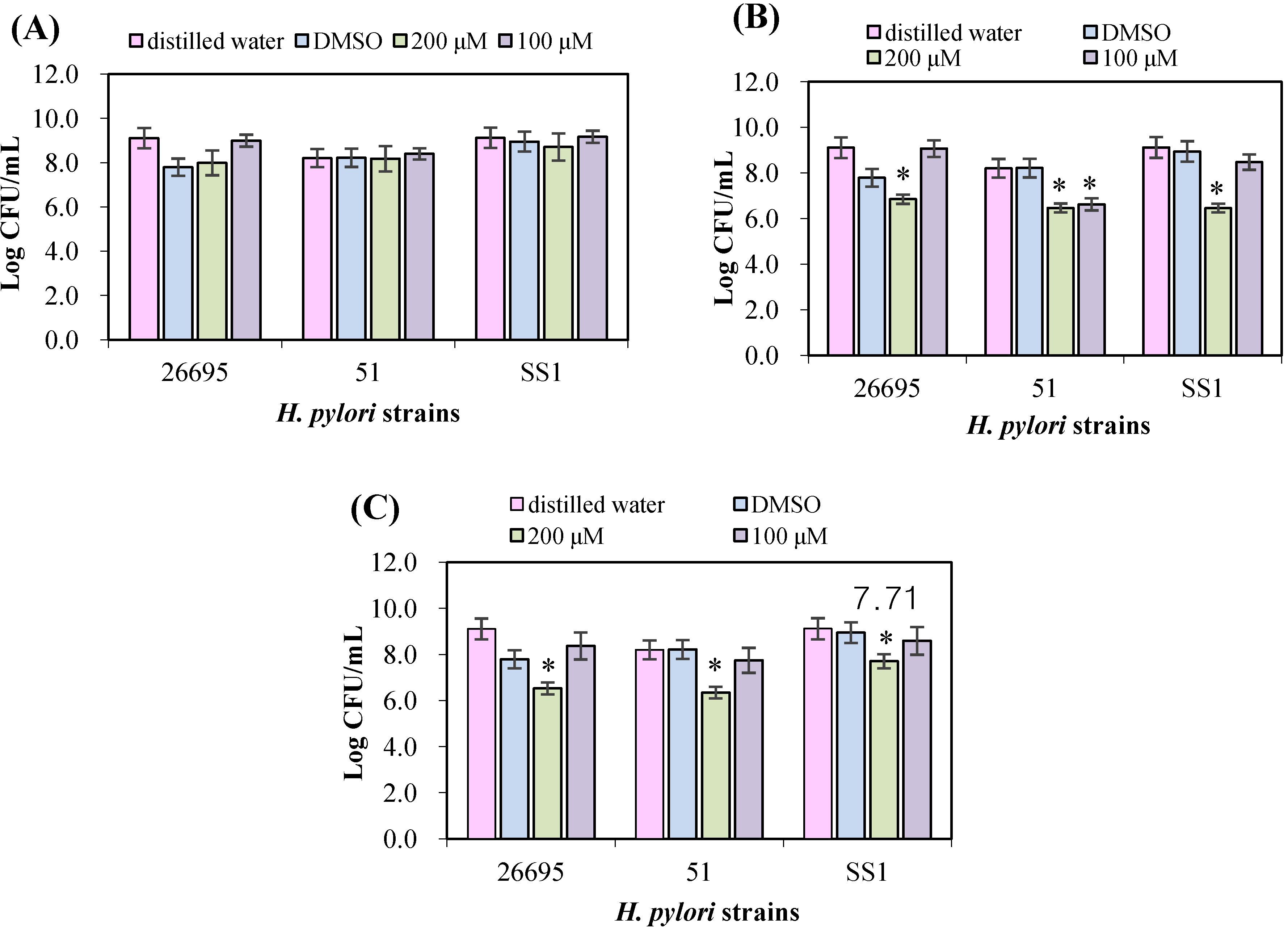
3.3. Urease Inhibition by Flavonoids in H. pylori
| Flavonoids | Naringenin | Hesperetin | 7-O-Butylnaringenin | |
|---|---|---|---|---|
| Strains | ||||
| H. pylori 26695 | 1.33 ± 0.13 *,ax | 0.93 ± 0.15 ax | 27.49 ± 0.32 bx | |
| H. pylori 51 | 16.55 ± 1.23 ax | 7.31 ± 0.23 ax | 61.93 ± 1.45 by | |
| H. pylori SS1 | 13.64 ± 0.98 ax | 6.32 ± 0.11 ax | 70.75 ± 3.56 by | |
3.4. Morphological Study of H. pylori Using SEM

4. Conclusions
Acknowlegments
Conflicts of Interest
References
- Marshall, B.J. Campylobacter pyloridis and gastritis. J. Infect. Dis. 1986, 153, 650–657. [Google Scholar] [CrossRef]
- Fukai, T.; Marumo, A.; Kaitou, K.; Kanda, T.; Terada, S.; Nomura, T. Anti-Helicobacter pylori flavonoids from licorice extract. Life Sci. 2002, 71, 1449–1463. [Google Scholar]
- Jiang, X.; Doyle, M.P. Optimizing enrichment culture conditions for detecting Helicobacter pylori in foods. J. Food Prot. 2002, 65, 1949–1954. [Google Scholar]
- Smith, J.L. The role of gastric acid in preventing foodborne disease and how bacteria overcome acid conditions. J. Food Prot. 2003, 66, 1292–1303. [Google Scholar]
- Stamatis, G.; Kyriazopoulos, P.; Golegou, S.; Basayiannis, A.; Skaltsas, S.; Skaltsa, H. In vitro anti-Helicobacter pylori activity of Greek herbal medicines. J. Ethnopharmacol. 2003, 88, 175–179. [Google Scholar]
- Shin, J.E.; Kim, J.M.; Bae, E.A.; Hyun, Y.J.; Kim, D.H. In vitro inhibitory effects of flavonoids on growth, infection and vacuolation of Helicobacter pylori. Planta Med. 2005, 71, 197–201. [Google Scholar]
- Stingel, K.; Reuse, H.D. Staying alive overdose: How does Helicobacter pylori control urease activity? Int. J. Med. Microbiol. 2005, 295, 307–315. [Google Scholar]
- Hazell, S.L.; Lee, A. Campylobacter pyloridis, urease, hydrogen ion back diffusion and gastric ulcers. Lancet 1986, 5, 15–17. [Google Scholar] [CrossRef]
- Hazell, S.L.; Borody, T.J.; Lee, A. Campylobacter pyloridis gastritis. I: Detection of urease as a marker of bacterial colonization and gastritis. Am. J. Gastroenterol. 1987, 82, 292–296. [Google Scholar]
- Muhammad, A.; Farooq, A.; Muhammad, R.J.; Muhammad, A.I.; Umer, R. Green synthesis of silver nanoparticles through reduction with Solanum xanthocarpum L. Berry extract: Characterization, antimicrobial and urease inhibitory activities against Helicobacter pylori. Int. J. Mol. Sci. 2012, 13, 9923–9941. [Google Scholar]
- Jiang, X.; Doyle, M.P. Effect of environmental and substrate factors on survival and growth of Helicobacter pylori. J. Food Prot. 1998, 61, 929–933. [Google Scholar]
- Nicoline, F.T.; Lucy, M.N.; Roland, N.N. DNA sequence analysis of South African Helicobacter pylori vacuolating cytotoxin gene (vacA). Int. J. Mol. Sci. 2011, 12, 7459–7468. [Google Scholar]
- Masuda, H.; Naohide, K.; Woo, G.J.; Shin, I.S. Inhibitory effects of gochoonangi (Wasabia japonica) against Helicobacter pylori and its urease activity. Food Sci. Biotechnol. 2004, 13, 191–196. [Google Scholar]
- Silvia, M.; Claudia, D.A.; Andrea, C.; Silvia, F.; Francesca, B.; Annalisa, S.; Natale, F.; Claudio, R. Antimicrobial activity against Helicobacter pylori strains and antioxidant properties of blackberry leaves (Rubus ulmifolius) and isolated compounds. Int. J. Antimicrob. Agric. 2009, 34, 50–59. [Google Scholar]
- Muhammad, A.; Farooq, A.; Fauqia, N.; Tahir, M.; Nazamid, S. Anti-Helicobacter pylori and urease inhibition activities of some traditional medicinal plants. Molecules 2013, 18, 2135–2149. [Google Scholar]
- Stojanovic, G.; Radulovic, N.; Hashimoto, T.; Palic, R. In vitro antimicrobial activity of extracts of four Achillea species: The composition of Achillea clavennae L. (Asteraceae) extract. J. Ethnopharmacol. 2005, 101, 185–190. [Google Scholar]
- Maria, H.; Concepción, S.-M.; Sonia, P.T. Flavonoid-flavonoid interaction and its effect on their antioxidant activity. Food Chem. 2010, 121, 691–696. [Google Scholar]
- Bang, M.H.; Jung, Y.J.; Kim, W.K. Effect of several flavonoids on cancer cell motility in human fibrosarcoma HT 1080 cells. Food Sci. Biotechnol. 2004, 13, 739–743. [Google Scholar]
- Kim, K.T.; Moon, S.H.; Yeo, E.J.; Han, Y.S.; Nah, S.Y.; Paik, H.-D. Inhibitory effect of 7-O-butyl naringenin on growth of Helicobacter pylori ATCC 26695. Food Sci. Biotechnol. 2006, 15, 466–468. [Google Scholar]
- Ghaleb, A.; Bassam, A.-S.; Kamel, A. Antibacterial activities of some plant extracts alone and in combination with different antimicrobials against multidrug-resistant Pseudomonas aeruginosa strains. Asian Pac. J. Trop. Med. 2010, 3, 266–269. [Google Scholar]
- Son, D.J.; Lee, S.E.; Park, B.S. Inhibitory effects of naturally occurring flavonoids on a human intestinal bacterium, Clostridium perfringens. Food Sci. Biotechnol. 2003, 12, 180–182. [Google Scholar]
- Tim, C.T.P.; Lamb, A.J. Antimicrobial activity of flavonoids. Int. J. Antimicrob. Agric. 2005, 26, 343–356. [Google Scholar]
- Tim, C.T.P.; Hamilton, V.E.S.; Lamb, A.J. Assessment of the antibacterial activity of selected flavonoids and consideration of discrepancies between previous reports. Microbial. Res. 2003, 158, 281–289. [Google Scholar]
- Christy, E.M.L.; Clarke, A.M.; Munzhelele, T.; Ezekiel, G.; Noxolo, F.M.; Roland, N.N. Selected South African honeys and their extracts possess in vitro anti-Helicobacter pylori activity. Arch. Med. Res. 2010, 41, 324–331. [Google Scholar]
- Yerra, K.R.; Lien, H.-M.; Lin, Y.H.; Hsu, Y.-M.; Yeh, C.-T.; Chen, C.C.; Lai, C.H.; Tzeng, Y.M. Antibacterial activities of Anisomeles indica constituents and their inhibition effect on Helicobacter pylori-induced inflammation in human gastric epithelial cells. Food Chem. 2012, 132, 780–787. [Google Scholar]
- Kim, K.T.; Paik, H.-D. Propolis as a Multi-Functional Natural Material for Health. In Natural Products as Future Medicinal Agents; Hawthorne, S., Ed.; Transworld Research Network: Kerala, India, 2008; pp. 33–48. [Google Scholar]
- Malekzadeh, F.; Ehsanifar, H.; Shahamat, M.; Levin, M.; Colwell, R.R. Antibacterial activity of black myrobalan (Terminalia chebulaetz) against Helicobacter pylori. Int. J. Antimicrob. Agric. 2001, 18, 85–88. [Google Scholar]
- Mobley, H.L.T.; Cortesia, M.J.; Roseuthal, L.E.; Jones, B.D. Characterization of urease from Campylobacter pylori. J. Clin. Microbiol. 1988, 26, 831–836. [Google Scholar]
- Tsugawa, H.; Suzuki, H.; Nakagawa, I.; Nishizawa, T.; Saito, Y.; Suematsu, M.; Hibi, T. Alpha-ketoglutarate oxidoreductase, an essential salvage enzyme of energy metabolism, in coccoid form of Helicobacter pylori. Biochem. Biophys. Res. Commun. 2008, 376, 46–51. [Google Scholar]
- Paulo, L.; Oleastro, M.; Gallardo, E.; Queiroz, J.A.; Domi, F. Anti-Helicobacter pylori and urease inhibitory activities of resveratrol and red wine. Food Res. Int. 2011, 44, 964–969. [Google Scholar]
- Becker-Ritta, A.B.; Martinelli, A.H.S.; Mitidierib, S.; Federa, V.; Wassermanna, G.E.; Santia, L.; Vainsteina, M.H.; Oliveirad, J.T.A.; Fiuzae, L.M.; Pasqualia, G.; et al. Antifungal activity of plant and bacterial ureases. Toxicon 2007, 50, 971–983. [Google Scholar]
- Shin, S.Y.; Bajpai, V.K.; Kim, H.R.; Kang, S.C. Antibacterial activity of eicosapentaenoic acid (EPA) against foodborne and food spoilage microorganisms. LWT-Food Sci. Technol. 2007, 40, 1515–1519. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Moon, S.H.; Lee, J.H.; Kim, K.-T.; Park, Y.-S.; Nah, S.-Y.; Ahn, D.U.; Paik, H.-D. Antimicrobial Effect of 7-O-Butylnaringenin, a Novel Flavonoid, and Various Natural Flavonoids against Helicobacter pylori Strains. Int. J. Environ. Res. Public Health 2013, 10, 5459-5469. https://doi.org/10.3390/ijerph10115459
Moon SH, Lee JH, Kim K-T, Park Y-S, Nah S-Y, Ahn DU, Paik H-D. Antimicrobial Effect of 7-O-Butylnaringenin, a Novel Flavonoid, and Various Natural Flavonoids against Helicobacter pylori Strains. International Journal of Environmental Research and Public Health. 2013; 10(11):5459-5469. https://doi.org/10.3390/ijerph10115459
Chicago/Turabian StyleMoon, Sun Hee, Jae Hoon Lee, Kee-Tae Kim, Yong-Sun Park, Seung-Yeol Nah, Dong Uk Ahn, and Hyun-Dong Paik. 2013. "Antimicrobial Effect of 7-O-Butylnaringenin, a Novel Flavonoid, and Various Natural Flavonoids against Helicobacter pylori Strains" International Journal of Environmental Research and Public Health 10, no. 11: 5459-5469. https://doi.org/10.3390/ijerph10115459
APA StyleMoon, S. H., Lee, J. H., Kim, K.-T., Park, Y.-S., Nah, S.-Y., Ahn, D. U., & Paik, H.-D. (2013). Antimicrobial Effect of 7-O-Butylnaringenin, a Novel Flavonoid, and Various Natural Flavonoids against Helicobacter pylori Strains. International Journal of Environmental Research and Public Health, 10(11), 5459-5469. https://doi.org/10.3390/ijerph10115459






