Introduction
Crude oil has been described as a complex mixture of over 6000 potentially different hydrocarbons and metals [
1]. Crude and refined petroleum, and oil field chemicals and emissions are highly complex chemical mixtures. Crude petroleum contains hundreds of compounds and the chemical composition varies between geologic formations [
2].
Knowledge of human responses to acute exposures to petroleum components comes from studies with several solvents containing benzene and petroleum [
3]. Recognized human biochemical and physiological responses associated with acute exposures to natural crudes are mainly transient and short lived unless the concentrations of the components are unusually high [
4]. Pollution implies that a particular level of contamination has deleterious effects, which may take the form of hazards to human health, interference with human activities, reduction of human amenities or harmful effect on living resources. Substantial amounts of those potentially toxic substances have been introduced into the environment. There is a concern that workers and other individuals exposed to crude oil might have an increased incidence of organ damage. After absorption via pulmonary or gastrointestinal routes, crude oil is transported in plasma initially bound to albumin and other larger proteins to the liver.
In Nigeria, crude oil is predominantly found in the riverine areas. Over the years the local population has used crude oil for various ailments such as gastrointestinal disorders, burns, foot rot and leg ulcers, poisoning and witchcraft. The present study, investigates the renal effect of bonny-light crude oil.
Materials and Methods
Matured albino rats weighing between 124-130g were obtained from the Animal Facility Center of the Department of Pharmacology and Toxicology, National Institute for Pharmaceutical Research and Development, Abuja, Nigeria. The animals were acclimated to housing conditions for at least one week prior to commencement of the experiment. Animals were housed singly at ambient temperature of 23 ± 3° C and a 12 h light, 12 h dark cycle. Water and food were provided ad libitum for the animals.
Animals were divided into 4 groups of 5 male rats each. The three test groups received by gavage for seven days freshly prepared Bonny light crude oil at doses of 200 mg/kg, 400 mg/kg and 800 mg/kg while the 4th group received a solution of Tween 80 in water only as control. These doses were based on that used by the local population in folkloric medicine. Food and fluid intake were measured daily. The final body weights of the rats were recorded.
Nigerian bonny-light crude oil was obtained from Nigerian National Petroleum Corporation (N.N.P.C.) Research Laboratory Port Harcourt, Nigeria. The bonny light crude oil was dissolved in Tween 80.
At the end of the seven days exposure period, the animals were weighed and sacrificed under chloroform anesthesia, and serum obtained. Sodium and potassium in serum were determined with Corning 410C clinical flame photometer (Corning Instruments, England) at 589 and 768nm respectively. Titrimetric method of Schales and Schales (1941) [
5] was used for the determination of chloride. The potassium and sodium were estimated using the flame emission spectrophotometer (corning brand). Urea was analysed spectrophotometrically at 525nm [
6]. Creatinine was determined using the method of Taussky 1956 [
7] at 520 nm. Serum potassium, sodium, bicarbonate and chloride were measured by the use of commercial kits (Biosystem, Spain).
The animals were then sacrificed under chloroform anaesthesia and the kidneys were harvested, weighed and fixed in 10% buffered formalin for 48 hrs. The kidneys were processed using an automatic tissue processor, embedded in paraffin wax, and sections (5µm thick) cut using a rotary microtome. The sections were stained by haematoxylin and eosin (H&E) method, and examined and photographed by using a light microscope. Two Histopathologists examined the sections, independently.
Statistical Analysis
The values were reported as means ° SEMs. Data were analyzed using the Student’s t-test, while Duncan’s multiple-range test was used to test for differences between treatment groups using Sigma-stat 2.0 software. P < 0.05 was considered statistically significant.
Results
Table 1 shows the effect of Bonny light crude oil on body weight, fluid intake, absolute and relative weights of kidney after 7 days of sub-chronic exposure to male rats. There was no significant difference in the mean fluid intake between the three dose levels. The animals demonstrated a progressive increase in body weight in the control and lower doses during the exposure period and there was a significant reduction in body weight in the high dose group (800mg/kg) compared to the control. There were no significant differences (
p ≥ 0.05) in the absolute and relative kidney weights of the animals in the three experimental groups.
The results show a significant increase (
p < 0.05) in serum creatinine in the 800mg/kg crude oil group; whereas there was significant increase in potassium in the 200 and 400mg/kg crude oil treated groups. There was also a significant decrease (
p < 0.05) in serum urea, sodium, bicarbonate and chloride in the bonny-light treated group when compared with the control (
Table 2).
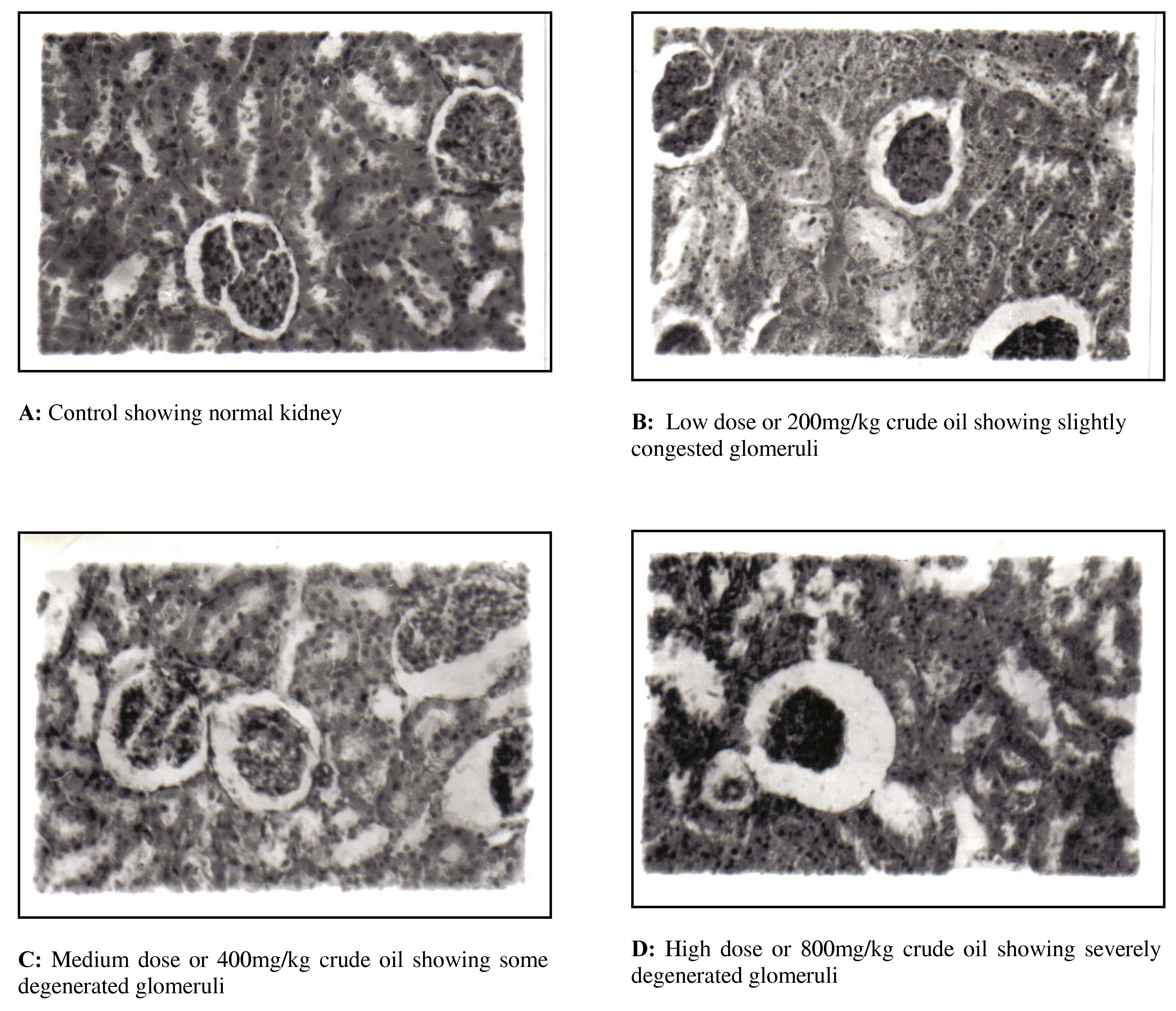
The histological changes of kidney in the different treatment groups are shown in
Figure 1. The control group showed normal histological structure. Low dose crude oil produced slightly congested glomeruli and hyperemia, a congestion caused intraglomerular space between the tuft and capsule. The glomeruli appeared infiltrated and congested creating wider spaces between the tuft and the capsule in the medium dose crude oil group. Also in this group some of the glomeruli appeared degenerated. The high dose group showed empty and widened capsules with severely degenerated glomeruli and noticeable necrosis.
Discussion
Exposure of humans and animal to crude oil, which is increasing in terms of the environmental levels, and application to body, may be toxic. Crude oil is used in folkloric medicine in the Niger-delta area of Nigeria for the treatment of various ailments including stomach up-set, wound, and burns [
8]. The route of administration is mostly oral and external application for burns and wounds. In several organs, mainly heart and liver, cell damage is followed by increased levels of a number of cytoplasmic enzymes in the blood, a phenomenon that provides the basis for clinical diagnosis of heart and liver diseases e.g. liver enzymes are usually raised in acute hepatotoxicity but tend to decrease with prolonged intoxication due to damage to the liver cells. The Nigerian Bonny crude oils are classified as light crude oils, with aromatic hydrocarbons accounting for up to 45% of the total hydrocarbons. As aromatic hydrocarbons are relatively soluble in water [
9], it is expected that the potential of this light crude oil to have adverse toxic effects is higher than for heavier, less water–soluble crude oils. It is known that lipophilic xenobiotics may have the characteristics of both electron uncouplers and energy inhibitors [
10]. Since a large proportion of the crude oil components is lipophilic in nature biological membrane may be the target sites where adverse effects occur.
The kidney can suffer considerable damage before losing sufficient function to modify the normal clinical indication of renal disease such as the serum creatinine concentration [
11]. Approximately 50% or more of renal capacity can be lost before serum creatinine become abnormal and disease is detectable clinically. A battery made up of a combination of different types of test can aid in the detection of damage by a nephrotoxin and also allows for the determination of various threshold damage. The detection of renal damage at a reversible stage is necessary before effective preventive measures can be taken to halt the progress of damage to the irreversible stage.
Table 1.
Effect of Bonny light crude oil on body weight, fluid intake, absolute and relative weights of kidney after 7 days of sub-chronic exposure to male rats.
Table 1.
Effect of Bonny light crude oil on body weight, fluid intake, absolute and relative weights of kidney after 7 days of sub-chronic exposure to male rats.
| Treatment Dose (mg/kg) | Final body weight (g) | Absolute kidney weight (g) | Relative kidney weight † (%) | Fluid intake ml/day/rat |
|---|
| Control | 180.3 ± 12.9 | 1.2 ± 0.7 | 0.67 ± 0.3 | 20.03 ± 2.39 |
| | (113±11.2) | | | |
| 200 | 166.1± 7.2* | 1.5 ± 0.5 | 0.90 ± 0.1 | 21.09 ± 2.34 |
| | (112.6 ± 12.9) | | | |
| 400 | 156.7± 11.8* | 1.3 ± 0.7 | 0.83 ± 0.2 | 22.89 ± 1.18 |
| | (114.0 ± 8.6) | | | |
| 800 | 136.8± 3.6* | 1.4 ± 0.4 | 1.02 ± 0.2 | 22.35 ± 4.16 |
| | (124.7 ± 21.8) | | | |
Table 2.
Effect of Bonny light crude oil on serum electrolytes, urea and creatinine after 7 days of sub-chronic exposure to male rats.
Table 2.
Effect of Bonny light crude oil on serum electrolytes, urea and creatinine after 7 days of sub-chronic exposure to male rats.
| Treatment Dose (mg/kg) | Urea (mmol/l) | Creatinine (mmol/l) | HCO3- (mmol/l) | Cl- (mmol/l) | Na+ (mmol/l) | K+ (mol/l) |
|---|
| Control | 7.6 ± 1.6 | 117.3 ± 2.5 | 26.3 ± 3.2 | 97.0 ± 2.4 | 145.3 ± 6.1 | 5.7 ± 2.4 |
| 200 | 5.7 ± 0.2 | 107.0 ± 1.7* | 30.6 ± 3.7# | 94.0 ± 2.0* | 144.1 ± 0.1 | 7.4 ± 0.1# |
| 400 | 4.9 ± 0.8* | 112.0 ± 5.1* | 23.0 ± 0.2 | 95.0 ± 2.0* | 143.6 ± 3.2 | 8.1 ± 0.7# |
| 800 | 2.4 ± 0.1* | 134.0 ± 4.0# | 21.0 ± 0.1* | 90.0 ± 2.1* | 140.6 ± 1.5* | 4.8 ± 0.4 |
Figure 1.
Photomicrographs of H&E stained sections of rat kidney (Magnification X250).
Figure 1.
Photomicrographs of H&E stained sections of rat kidney (Magnification X250).
The absolute and relative kidney weights, as well as body weight gain were significantly
(p < 0.05) decreased by exposure to the bonny–light crude oil. Researchers have reported that absolute kidney weight is a relatively sensitive indicator of nephrotoxicity for known nephrotoxicants, with nephrotoxicity defined as increased kidney weight (absolute or relative) [
12].
There were no significant (
p < 0.05) differences in the absolute and relative kidney weights of the animals in the three test groups. This observation seems not to conform with the above definition of toxicity probably due to the short duration of the exposure to the bonny-light crude oil. However the significant (
p < 0.05) increase in serum creatinine and potassium in the treated groups and also the significant (
p < 0.05) decrease in serum urea, sodium, bicarbonate and chloride in the bonny-light treated group when compared with the control is suggestive of renal pathology. This in part is in agreement with Kluwe definition of renal toxicity [
12].