Absorption and Metabolism of Xanthophylls
Abstract
:1. Introduction
2. Bioaccessibility of Carotenoids
3. Intestinal Absorption of Xanthophylls
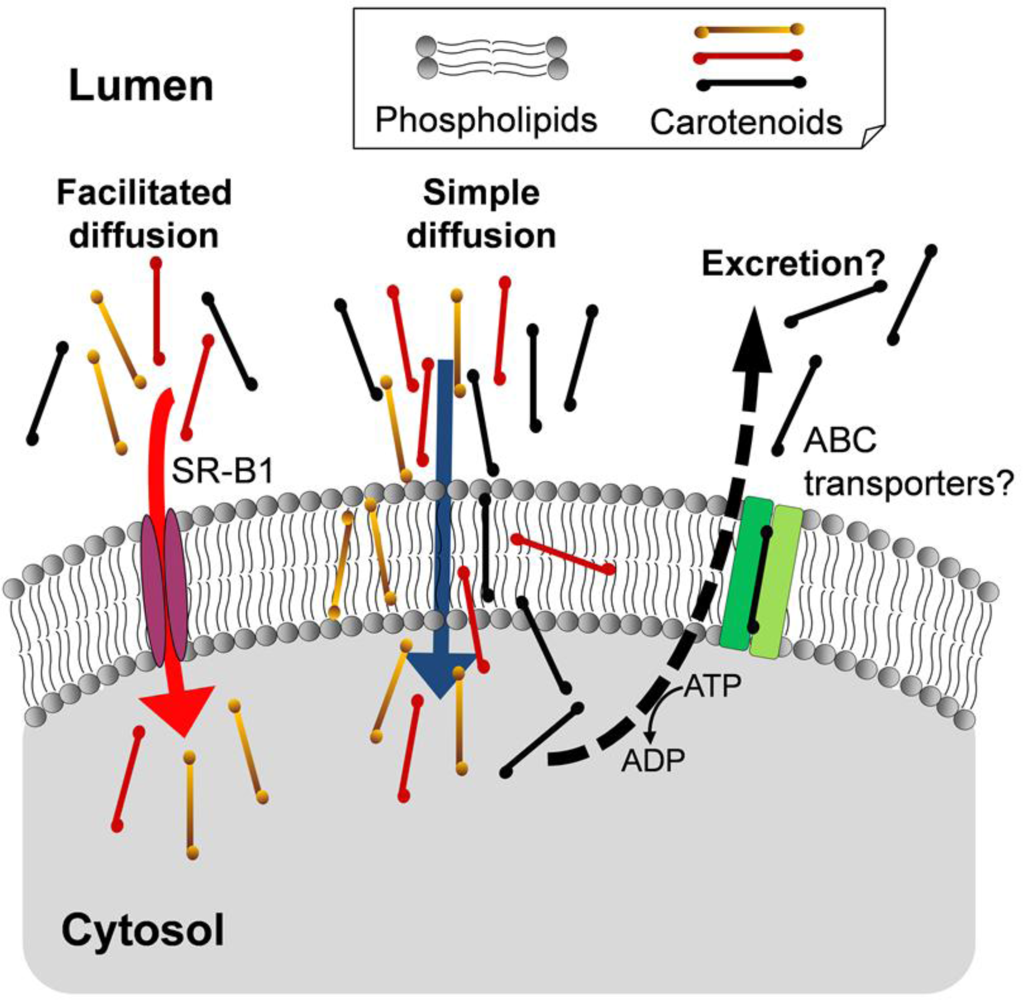
4. Mechanisms of the Intestinal Absorption
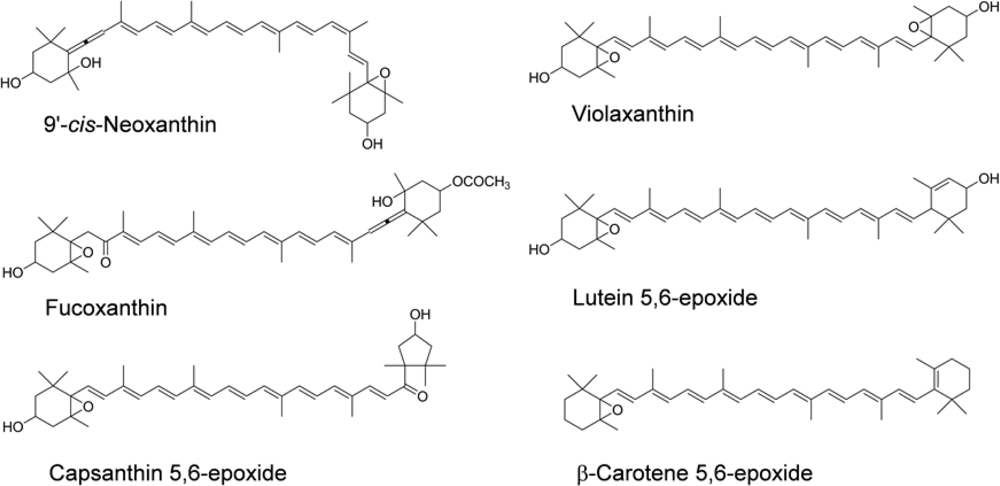
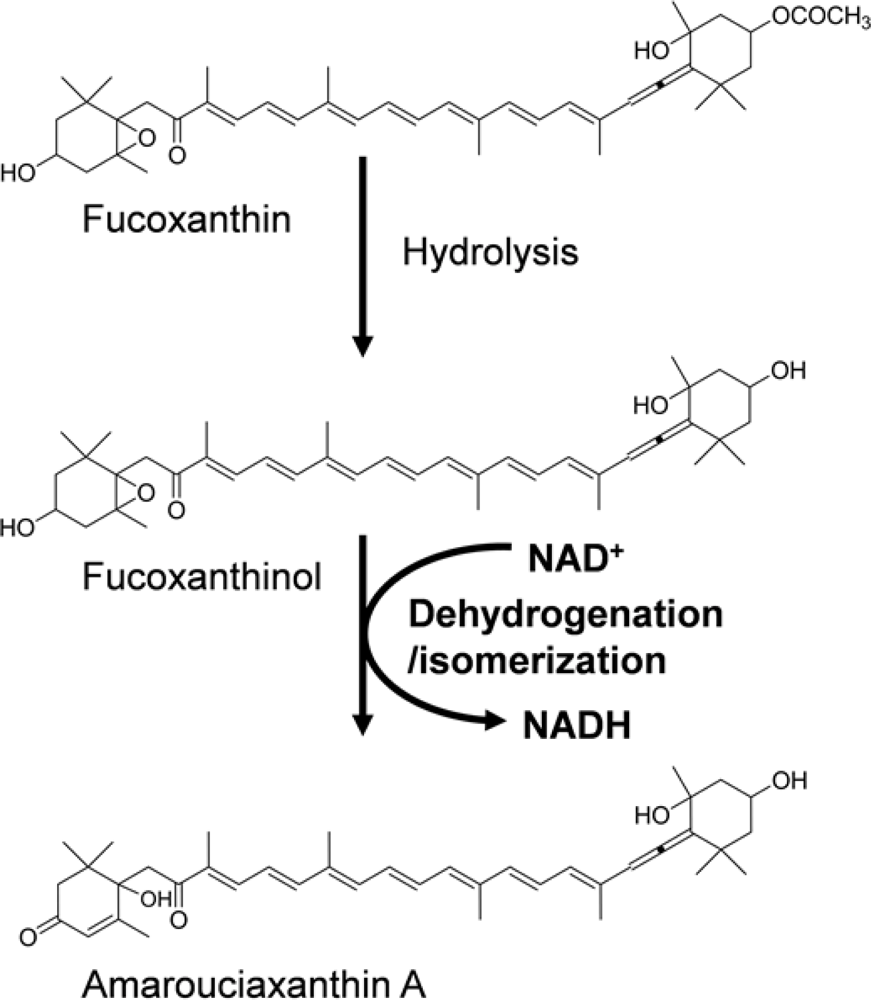
5. Metabolism of Xanthophylls in Mammals
6. Conclusions
- Samples Availability: Available from the authors.
References
- Miller, NJ; Sampson, J; Candeias, LP; Bramley, PM; Rice-Evans, CA. Antioxidant activities of carotenes and xanthophylls. FEBS Lett 1996, 384, 240–242. [Google Scholar]
- Di Mascio, P; Kaiser, S; Sies, H. Lycopene as the most efficient biological carotenoid singlet oxygen quencher. Arch Biochem Biophys 1989, 274, 532–538. [Google Scholar]
- Junghans, A; Sies, H; Stahl, W. Macular pigments lutein and zeaxanthin as blue light filters studied in liposomes. Arch Biochem Biophys 2001, 391, 160–164. [Google Scholar]
- Katsuura, S; Imamura, T; Bando, N; Yamanishi, R. Beta-carotene and beta-cryptoxanthin but not lutein evoke redox and immune changes in RAW264 murine macrophages. Mol Nutr Food Res 2009, 53, 1396–1405. [Google Scholar]
- Yamaguchi, M; Weitzmann, MN. The bone anabolic carotenoid beta-cryptoxanthin enhances transforming growth factor-beta1-induced SMAD activation in MC3T3 preosteoblasts. Int J Mol Med 2009, 24, 671–675. [Google Scholar]
- Uchiyama, S; Yamaguchi, M. Oral administration of beta-cryptoxanthin prevents bone loss in ovariectomized rats. Int J Mol Med 2006, 17, 15–20. [Google Scholar]
- Aizawa, K; Inakuma, T. Dietary capsanthin, the main carotenoid in paprika (Capsicum annuum), alters plasma high-density lipoprotein-cholesterol levels and hepatic gene expression in rats. Br J Nutr 2009, 102, 1760–1766. [Google Scholar]
- Hussein, G; Sankawa, U; Goto, H; Matsumoto, K; Watanabe, H. Astaxanthin, a carotenoid with potential in human health and nutrition. J Nat Prod 2006, 69, 443–449. [Google Scholar]
- Yuan, JP; Peng, J; Yin, K; Wang, JH. Potential health-promoting effects of astaxanthin: A high-value carotenoid mostly from microalgae. Mol Nutr Food Res 2011, 55, 150–165. [Google Scholar]
- Okuzumi, J; Nishino, H; Murakoshi, M; Iwashima, A; Tanaka, Y; Yamane, T; Fujita, Y; Takahashi, T. Inhibitory effects of fucoxanthin, a natural carotenoid, on n-myc expression and cell cycle progression in human malignant tumor cells. Cancer Lett 1990, 55, 75–81. [Google Scholar]
- Hosokawa, M; Wanezaki, S; Miyauchi, K; Kurihara, H; Kohno, H; Kawabata, J; Odashima, S; Takahashi, K. Apoptosis-inducing effect of fucoxanthin on human leukemia cell line HL-60. Food Sci Technol Res 1999, 5, 243–246. [Google Scholar]
- Kotake-Nara, E; Terasaki, M; Nagao, A. Characterization of apoptosis induced by fucoxanthin in human promyelocytic leukemia cells. Biosci Biotechnol Biochem 2005, 69, 224–227. [Google Scholar]
- Kotake-Nara, E; Kushiro, M; Zhang, H; Sugawara, T; Miyashita, K; Nagao, A. Carotenoids affect proliferation of human prostate cancer cells. J Nutr 2001, 131, 3303–3306. [Google Scholar]
- Kotake-Nara, E; Sugawara, T; Nagao, A. Antiproliferative effect of neoxanthin and fucoxanthin on cultured cells. Fish Sci 2005, 71, 459–461. [Google Scholar]
- Okuzumi, J; Takahashi, T; Yamane, T; Kitao, Y; Inagake, M; Ohya, K; Nishino, H; Tanaka, Y. Inhibitory effects of fucoxanthin, a natural carotenoid, on N-ethyl-N′-nitro-N-nitrosoguanidine-induced mouse duodenal carcinogenesis. Cancer Lett 1993, 68, 159–168. [Google Scholar]
- Sakai, S; Sugawara, T; Matsubara, K; Hirata, T. Inhibitory effect of carotenoids on the degranulation of mast cells via suppression of antigen-induced aggregation of high affinity IgE receptors. J Biol Chem 2009, 284, 28172–28179. [Google Scholar]
- Maeda, H; Hosokawa, M; Sashima, T; Miyashita, K. Dietary combination of fucoxanthin and fish oil attenuates the weight gain of white adipose tissue and decreases blood glucose in obese/diabetic KK-Ay mice. J Agric Food Chem 2007, 55, 7701–7706. [Google Scholar]
- Khachik, F; Beecher, GR; Goli, MB; Lusby, WR. Separation, identification, and quantification of carotenoids in fruits, vegetables and human plasma by high performance liquid chromatography. Pure Appl Chem 1991, 63, 71–90. [Google Scholar]
- Khachik, F; Spangler, CJ; Smith, JC, Jr; Canfield, LM; Steck, A; Pfander, H. Identification, quantification, and relative concentrations of carotenoids and their metabolites in human milk and serum. Anal Chem 1997, 69, 1873–1881. [Google Scholar]
- Slifka, KA; Bowen, PE; Stacewicz-Sapuntzakis, M; Crissey, SD. A survey of serum and dietary carotenoids in captive wild animals. J Nutr 1999, 129, 380–390. [Google Scholar]
- Richelle, M; Enslen, M; Hager, C; Groux, M; Tavazzi, I; Godin, JP; Berger, A; Métairon, S; Quaile, S; Piguet-Welsch, C; et al. Both free and esterified plant sterols reduce cholesterol absorption and the bioavailability of beta-carotene and alpha-tocopherol in normocholesterolemic humans. Am J Clin Nutr 2004, 80, 171–177. [Google Scholar]
- Maiani, G; Castón, MJ; Catasta, G; Toti, E; Cambrodón, IG; Bysted, A; Granado-Lorencio, F; Olmedilla-Alonso, B; Knuthsen, P; Valoti, M; et al. Carotenoids: actual knowledge on food sources, intakes, stability and bioavailability and their protective role in humans. Mol Nutr Food Res 2009, 53(Suppl. 2), S194–S218. [Google Scholar]
- Holst, B; Williamson, G. Nutrients and phytochemicals: from bioavailability to bioefficacy beyond antioxidants. Curr Opin Biotechnol 2008, 19, 73–82. [Google Scholar]
- Zaripheh, S; Erdman, JW, Jr. Factors that influence the bioavailablity of xanthophylls. J Nutr 2002, 132, 531S–534S. [Google Scholar]
- Yonekura, L; Nagao, A. Intestinal absorption of dietary carotenoids. Mol Nutr Food Res 2007, 51, 107–115. [Google Scholar]
- Rock, CL; Lovalvo, JL; Emenhiser, C; Ruffin, MT; Flatt, SW; Schwartz, SJ. Bioavailability of beta-carotene is lower in raw than in processed carrots and spinach in women. J Nutr 1998, 128, 913–916. [Google Scholar]
- Small, DM; Penkett, SA; Chapman, D. Studies on simple and mixed bile salt micelles by nuclear magnetic resonance spectroscopy. Biochim Biophys Acta 1969, 176, 178–189. [Google Scholar]
- Yeum, KJ; Russell, RM. Carotenoid bioavailability and bioconversion. Annu Rev Nutr 2002, 22, 483–504. [Google Scholar]
- Hollander, D; Ruble, PE, Jr. Beta-carotene intestinal absorption: Bile, fatty acid, pH, and flow rate effects on transport. Am J Physiol Endocrinol Metab 1978, 235, E686–E691. [Google Scholar]
- Scita, G; Aponte, GW; Wolf, G. Uptake and cleavage of β-carotene by cultures of rat small intestinal cells and human lung fibroblasts. J Nutr Biochem 1992, 3, 118–123. [Google Scholar]
- Sugawara, T; Kushiro, M; Zhang, H; Nara, E; Ono, H; Nagao, A. Lysophosphatidylcholine enhances carotenoid uptake from mixed micelles by Caco-2 human intestinal cells. J Nutr 2001, 131, 2921–2927. [Google Scholar]
- Sugawara, T; Baskaran, V; Tsuzuki, W; Nagao, A. Brown algae fucoxanthin is hydrolyzed to fucoxanthinol during absorption by Caco-2 human intestinal cells and mice. J Nutr 2002, 132, 946–951. [Google Scholar]
- Asai, A; Sugawara, T; Ono, H; Nagao, A. Biotransformation of fucoxanthinol into amarouciaxanthin A in mice and HepG2 cells. Drug Metab Dispos 2004, 32, 205–211. [Google Scholar]
- Asai, A; Terasaki, M; Nagao, A. An epoxide–furanoid rearrangement of spinach neoxanthin occurs in the gastrointestinal tract of mice and in vitro: Formation and cytostatic activity of neochrome stereoisomers. J Nutr 2004, 134, 2237–2243. [Google Scholar]
- Sangeetha, RK; Bhaskar, N; Divakar, S; Baskaran, V. Bioavailability and metabolism of fucoxanthin in rats: structural characterization of metabolites by LC-MS (APCI). Mol Cell Biochem 2010, 333, 299–310. [Google Scholar]
- Baskaran, V; Sugawara, T; Nagao, A. Phospholipids affect the intestinal absorption of carotenoids in mice. Lipids 2003, 38, 705–711. [Google Scholar]
- Matsuno, T; Ookubo, M. A new carotenoid, halocynthiaxanthin from the sea squirt, Halocynthia Roretzi. Tetrahedron Lett 1981, 22, 4659–4660. [Google Scholar]
- Matsuno, T; Ookubo, M; Komori, T. Carotenoids of tunicates, III. The structural elucidation of two new marine carotenoids, amarouciaxanthin A and B. J Nat Prod 1985, 48, 606–613. [Google Scholar]
- Strand, A; Herstad, O; Liaaen-Jensen, S. Fucoxanthin metabolites in egg yolks of laying hens. Comp Biochem Physiol A Mol Integr Physiol 1998, 119, 963–974. [Google Scholar]
- Matsuno, T; Ohkubo, M; Toriiminami, Y; Tsushima, M; Sakaguchi, S; Minami, T; Maoka, T. Carotenoids in food chain between freshwater fish and aquatic insects. Comp Biochem Physiol A Mol Integr Physiol 1999, 124, 341–345. [Google Scholar]
- Asai, A; Yonekura, L; Nagao, A. Low bioavailability of dietary epoxyxanthophylls in humans. Br J Nutr 2008, 100, 273–277. [Google Scholar]
- Yonekura, L; Nagao, A. Soluble fibers inhibit carotenoid micellization in vitro and uptake by Caco-2 cells. Biosci Biotechnol Biochem 2009, 73, 196–199. [Google Scholar]
- Pérez-Gálvez, A; Martin, HD; Sies, H; Stahl, W. Incorporation of carotenoids from paprika oleoresin into human chylomicrons. Br J Nutr 2003, 89, 787–793. [Google Scholar]
- Barua, AB; Olson, JA. Xanthophyll epoxides, unlike beta-carotene monoepoxides, are not detectibly absorbed by humans. J Nutr 2001, 131, 3212–3215. [Google Scholar]
- Barua, AB. Intestinal absorption of epoxy-beta-carotenes by humans. Biochem J 1999, 339(Pt. 2), 359–362. [Google Scholar]
- Reboul, E; Abou, L; Mikail, C; Ghiringhelli, O; André, M; Portugal, H; Jourdheuil-Rahmani, D; Amiot, MJ; Lairon, D; Borel, P. Lutein transport by Caco-2 TC-7 cells occurs partly by a facilitated process involving the scavenger receptor class B type I (SR-BI). Biochem J 2005, 387(Pt. 2), 455–461. [Google Scholar]
- Kiefer, C; Sumser, E; Wernet, MF; Von Lintig, J. A Class B scavenger receptor mediates the cellular uptake of carotenoids in Drosophila. Proc Natl Acad Sci USA 2002, 99, 10581–10586. [Google Scholar]
- Borel, P; Moussa, M; Reboul, E; Lyan, B; Defoort, C; Vincent-Baudry, S; Maillot, M; Gastaldi, M; Darmon, M; Portugal, H; et al. Human plasma levels of vitamin E and carotenoids are associated with genetic polymorphisms in genes involved in lipid metabolism. J Nutr 2007, 137, 2653–2659. [Google Scholar]
- Moussa, M; Landrier, JF; Reboul, E; Ghiringhelli, O; Coméra, C; Collet, X; Fröhlich, K; Böhm, V; Borel, P. Lycopene absorption in human intestinal cells and in mice involves scavenger receptor class B type I but not Niemann-Pick C1-Like 1. J Nutr 2008, 138, 1432–1436. [Google Scholar]
- O’Sullivan, L; Aisling, SA; O’Brien, NM. Investigation of beta-carotene and lutein transport in Caco-2 cells: carotenoid-carotenoid interactions and transport inhibition by ezetimibe. Int J Vitam Nutr Res 2009, 79, 337–347. [Google Scholar]
- During, A; Hussain, MM; Morel, DW; Harrison, EH. Carotenoid uptake and secretion by CaCo-2 cells: Beta-carotene isomer selectivity and carotenoid interactions. J Lipid Res 2002, 43, 1086–1095. [Google Scholar]
- During, A; Harrison, EH. Mechanisms of provitamin A (carotenoid) and vitamin A (retinol) transport into and out of intestinal Caco-2 cells. J Lipid Res 2007, 48, 2283–2294. [Google Scholar]
- During, A; Dawson, HD; Harrison, EH. Carotenoid transport is decreased and expression of the lipid transporters SR-BI, NPC1L1, and ABCA1 is downregulated in Caco-2 cells treated with ezetimibe. J Nutr 2005, 135, 2305–2312. [Google Scholar]
- van Bennekum, A; Werder, M; Thuahnai, ST; Han, CH; Duong, P; Williams, DL; Wettstein, P; Schulthess, G; Phillips, MC; Hauser, H. Class B scavenger receptor-mediated intestinal absorption of dietary beta-carotene and cholesterol. Biochemistry 2005, 44, 4517–4525. [Google Scholar]
- Lobo, GP; Hessel, S; Eichinger, A; Noy, N; Moise, AR; Wyss, A; Palczewski, K; von Lintig, J. ISX is a retinoic acid-sensitive gatekeeper that controls intestinal beta, beta-carotene absorption and vitamin A production. FASEB J 2010, 24, 1656–1666. [Google Scholar]
- Fransen, HP; de Jong, N; Wolfs, M; Verhagen, H; Verschuren, WM; Lütjohann, D; von Bergmann, K; Plat, J; Mensink, RP. Customary use of plant sterol and plant stanol enriched margarine is associated with changes in serum plant sterol and stanol concentrations in humans. J Nutr 2007, 137, 1301–1306. [Google Scholar]
- Yu, L; von Bergmann, K; Lutjohann, D; Hobbs, HH; Cohen, JC. Selective sterol accumulation in ABCG5/ABCG8-deficient mice. J Lipid Res 2004, 45, 301–307. [Google Scholar]
- Herron, KL; McGrane, MM; Waters, D; Lofgren, IE; Clark, RM; Ordovas, JM; Fernandez, ML. The ABCG5 polymorphism contributes to individual responses to dietary cholesterol and carotenoids in Eggs. J Nutr 2006, 136, 1161–1165. [Google Scholar]
- Shen, J; Cross, ST; Tang-Liu, DD; Welty, DF. Evaluation of an immortalized retinal endothelial cell line as an in vitro model for drug transport studies across the blood-retinal barrier. Pharm Res 2003, 20, 1357–1363. [Google Scholar]
- Gyémánt, N; Tanaka, M; Molnár, P; Deli, J; Mándoky, L; Molnár, J. Reversal of multidrug resistance of cancer cells in vitro: modification of drug resistance by selected carotenoids. Anticancer Res 2006, 26, 367–374. [Google Scholar]
- Molnár, J; Gyémánt, N; Mucsi, I; Molnár, A; Szabó, M; Körtvélyesi, T; Varga, A; Molnár, P; Tóth, G. Modulation of multidrug resistance and apoptosis of cancer cells by selected carotenoids. In Vivo 2004, 18, 237–244. [Google Scholar]
- Ugocsai, K; Varga, A; Molnár, P; Antus, S; Molnár, J. Effects of selected flavonoids and carotenoids on drug accumulation and apoptosis induction in multidrug-resistant colon cancer cells expressing MDR1/LRP. In Vivo 2005, 19, 433–438. [Google Scholar]
- Khachik, F; Bernstein, PS; Garland, DL. Identification of lutein and zeaxanthin oxidation products in human and monkey retinas. Invest Ophthalmol Vis Sci 1997, 38, 1802–1811. [Google Scholar]
- Khachik, F; de Moura, FF; Zhao, DY; Aebischer, CP; Bernstein, PS. Transformations of selected carotenoids in plasma, liver, and ocular tissues of humans and in nonprimate animal models. Invest Ophthalmol Vis Sci 2002, 43, 3383–3392. [Google Scholar]
- Bhosale, P; Bernstein, PS. Quantitative measurement of 3′-oxolutein from human retina by normal-phase high-performance liquid chromatography coupled to atmospheric pressure chemical ionization mass spectrometry. Anal Biochem 2005, 345, 296–301. [Google Scholar]
- Bhosale, P; Zhao, DY; Serban, B; Bernstein, PS. Identification of 3-methoxyzeaxanthin as a novel age-related carotenoid metabolite in the human macula. Invest Ophthalmol Vis Sci 2007, 48, 1435–1440. [Google Scholar]
- Yonekura, L; Kobayashi, M; Terasaki, M; Nagao, A. Keto-carotenoids are the major metabolites of dietary lutein and fucoxanthin in mouse tissues. J Nutr 2010, 140, 1824–1831. [Google Scholar]
- Etoh, H; Utsunomiya, Y; Komori, A; Murakami, Y; Oshima, S; Inakuma, T. Carotenoids in human blood plasma after ingesting paprika juice. Biosci Biotechnol Biochem 2000, 64, 1096–1098. [Google Scholar]
- Zeng, S; Furr, HC; Olson, JA. Metabolism of carotenoid analogs in humans. Am J Clin Nutr 1992, 56, 433–439. [Google Scholar]
- Khachik, F. An efficient conversion of (3R,3′R,6′R)-lutein to (3R,3′S,6′R)-lutein (3′-epilutein) and (3R,3′R)-zeaxanthin. J Nat Prod 2003, 66, 67–72. [Google Scholar]
- Khachik, F; Englert, G; Beecher, GR; Smith, JC, Jr. Isolation, structural elucidation, and partial synthesis of lutein dehydration products in extracts from human plasma. J Chromatogr B Biomed Appl 1995, 670, 219–233. [Google Scholar]
- Kiefer, C; Hessel, S; Lampert, JM; Vogt, K; Lederer, MO; Breithaupt, DE; von Lintig, J. Identification and characterization of a mammalian enzyme catalyzing the asymmetric oxidative cleavage of provitamin A. J Biol Chem 2001, 276, 14110–14116. [Google Scholar]
- Hu, KQ; Liu, C; Ernst, H; Krinsky, NI; Russell, RM; Wang, XD. The biochemical characterization of ferret carotene-9′,10′-monooxygenase catalyzing cleavage of carotenoids in vitro andin vivo. J Biol Chem 2006, 281, 19327–19338. [Google Scholar]
- Mein, JR; Dolnikowski, GG; Ernst, H; Russell, RM; Wang, XD. Enzymatic formation of apo-carotenoids from the xanthophyll carotenoids lutein, zeaxanthin and β-cryptoxanthin by ferret carotene-9′,10′-monooxygenase. Arch Biochem Biophys 2011, 506, 109–121. [Google Scholar]
- Våge, DI; Boman, IA. A nonsense mutation in the beta-carotene oxygenase 2 (BCO2) gene is tightly associated with accumulation of carotenoids in adipose tissue in sheep (Ovis aries). BMC Genet 2010, 11, 10. [Google Scholar]
- Amengual, J; Lobo, GP; Golczak, M; Li, HN; Klimova, T; Hoppel, CL; Wyss, A; Palczewski, K; von Lintig, J. A mitochondrial enzyme degrades carotenoids and protects against oxidative stress. FASEB J 2011, 25, 948–959. [Google Scholar]

© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kotake-Nara, E.; Nagao, A. Absorption and Metabolism of Xanthophylls. Mar. Drugs 2011, 9, 1024-1037. https://doi.org/10.3390/md9061024
Kotake-Nara E, Nagao A. Absorption and Metabolism of Xanthophylls. Marine Drugs. 2011; 9(6):1024-1037. https://doi.org/10.3390/md9061024
Chicago/Turabian StyleKotake-Nara, Eiichi, and Akihiko Nagao. 2011. "Absorption and Metabolism of Xanthophylls" Marine Drugs 9, no. 6: 1024-1037. https://doi.org/10.3390/md9061024



