Terpenes from Marine-Derived Fungi
Abstract
:1. Introduction
2. Examples of Terpenoids
2.1. Monoterpenes
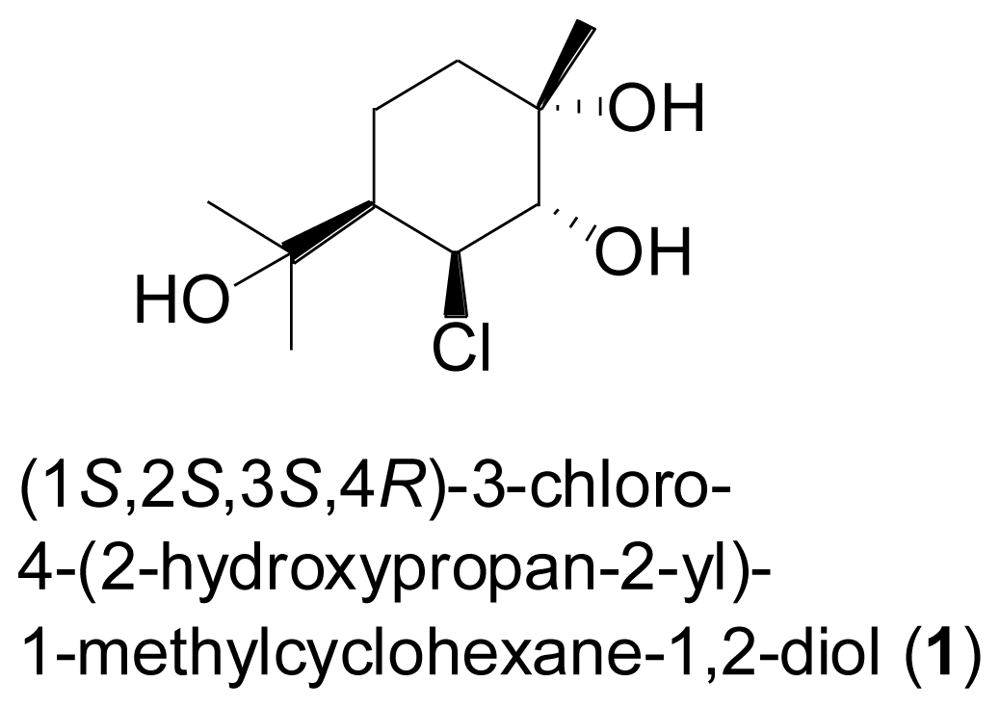
2.2. Sesquiterpenes
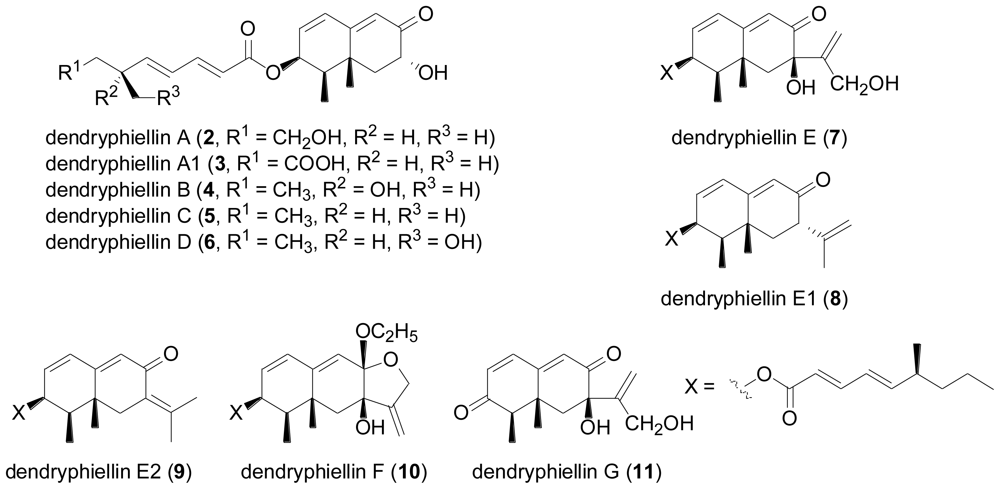

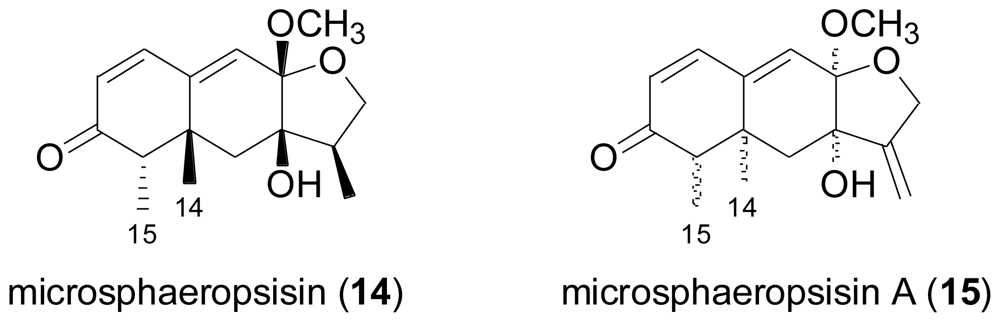
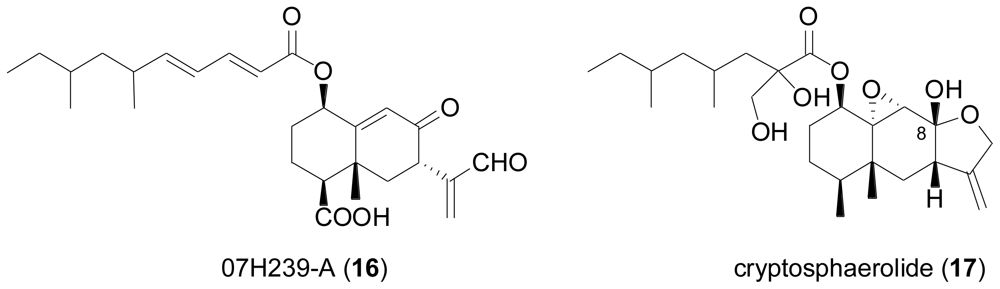
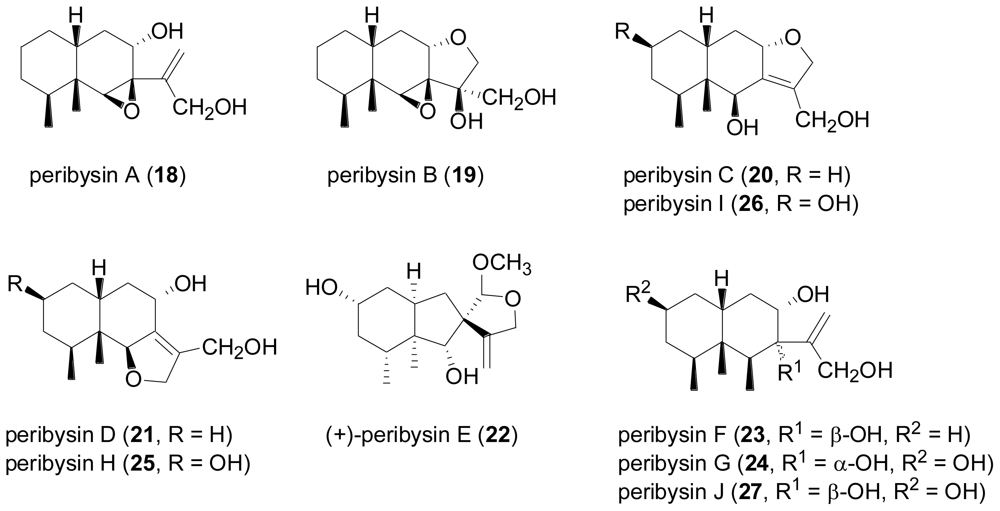
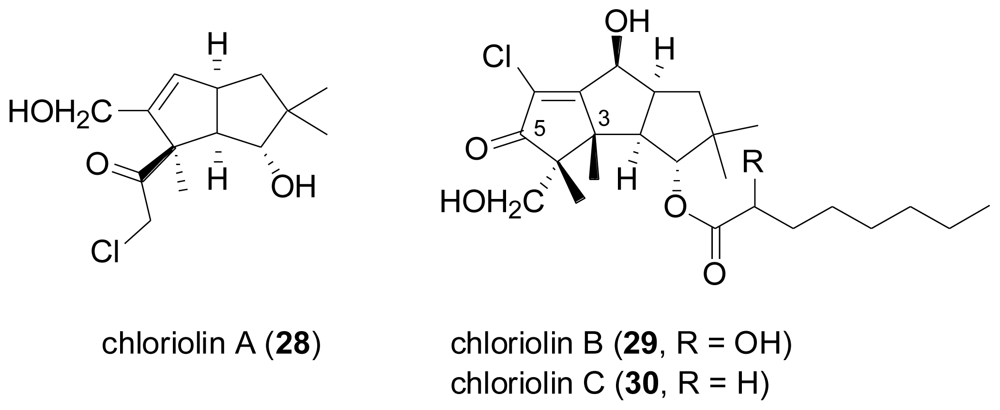
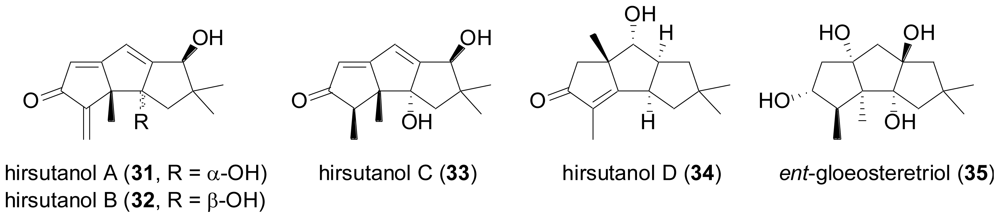
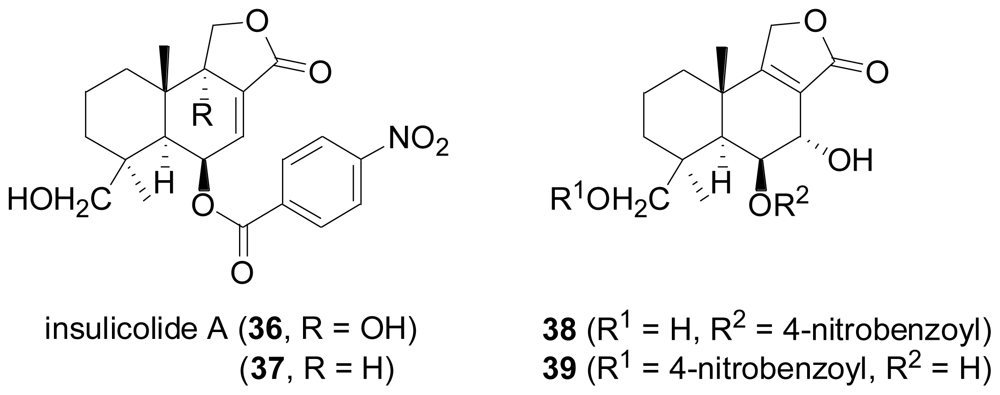
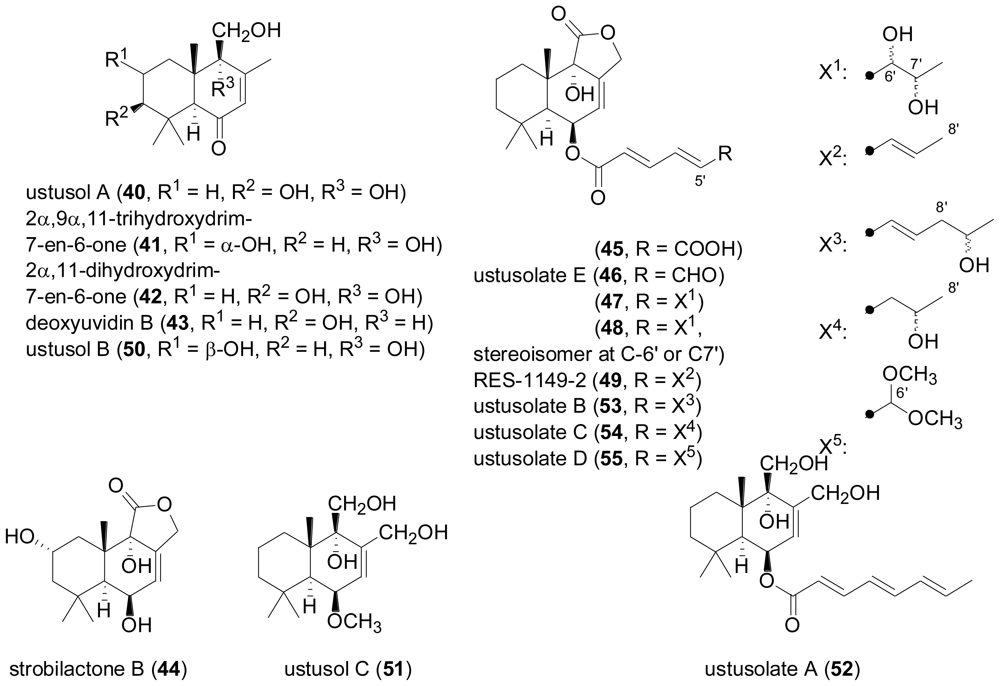
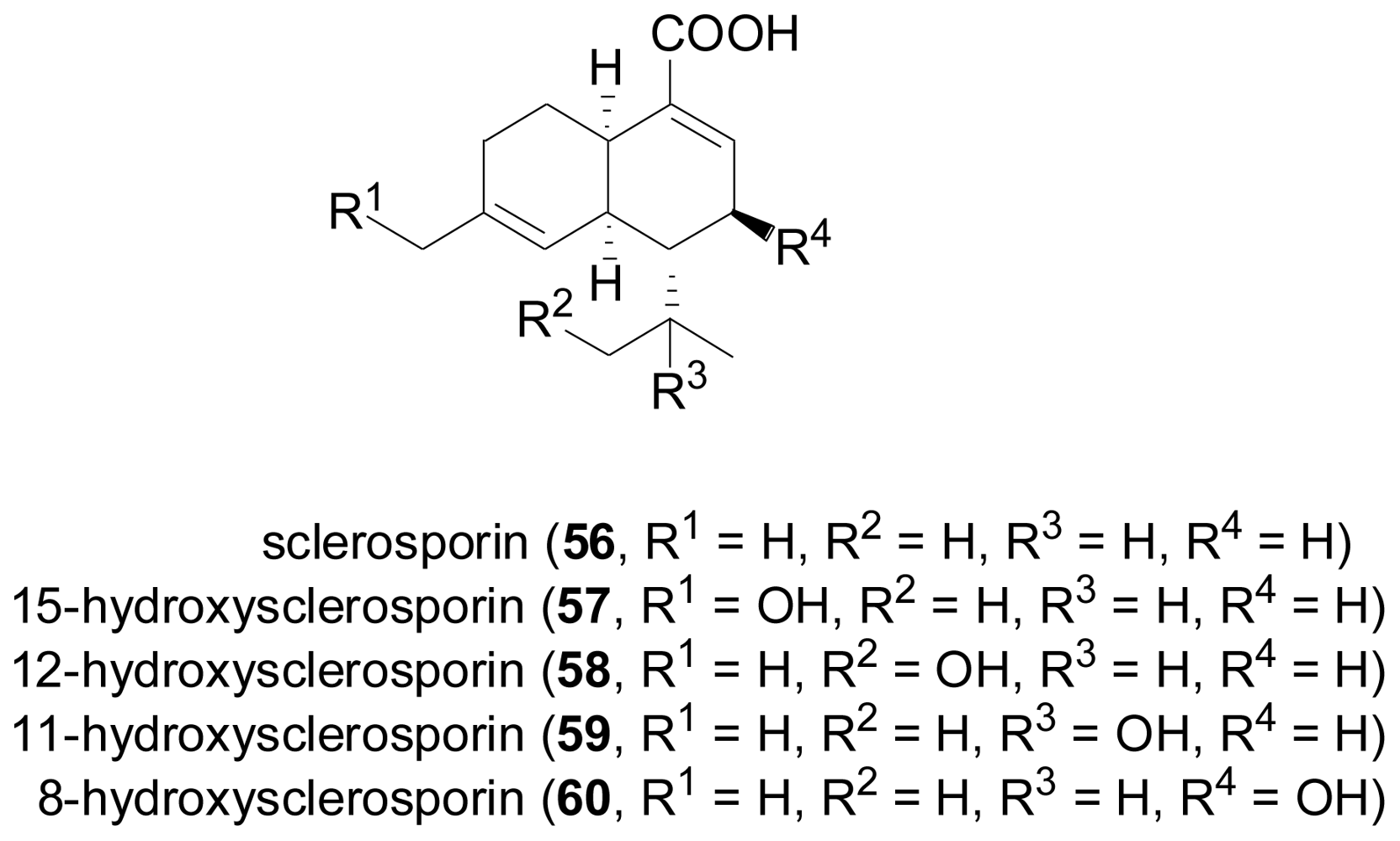
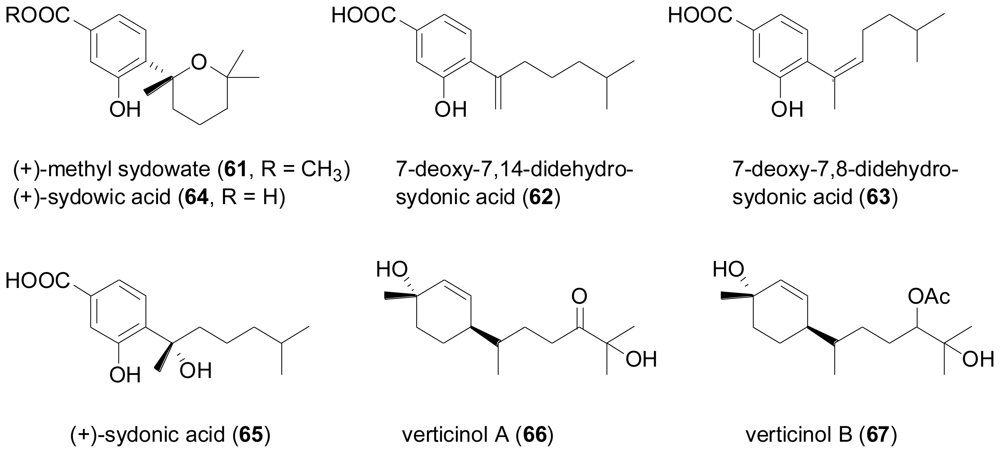
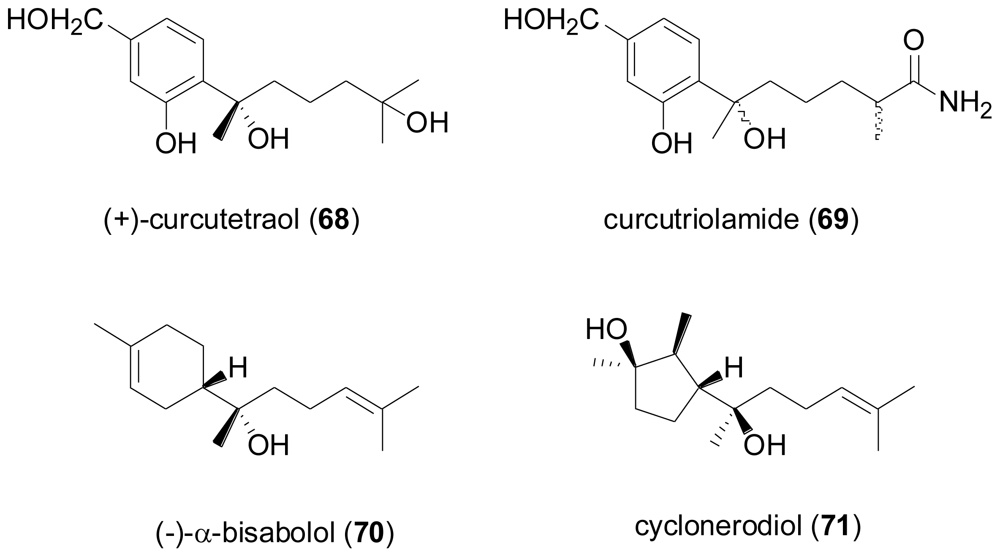
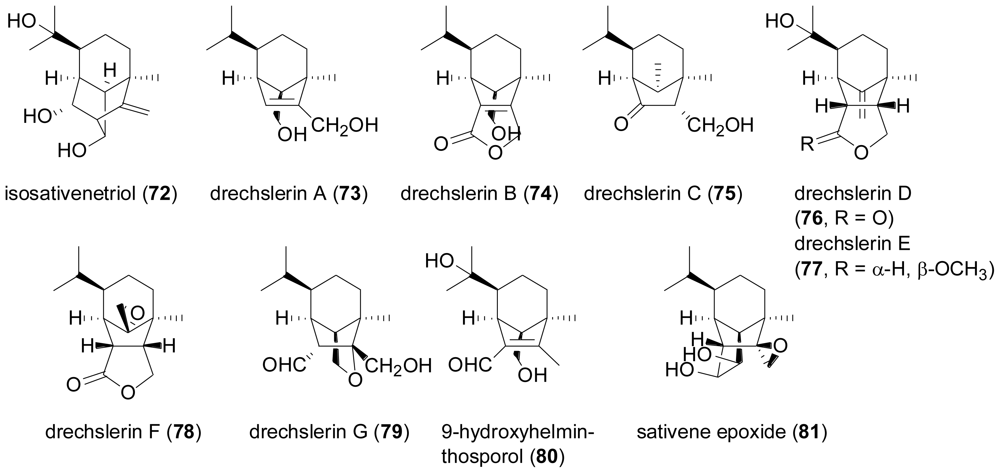
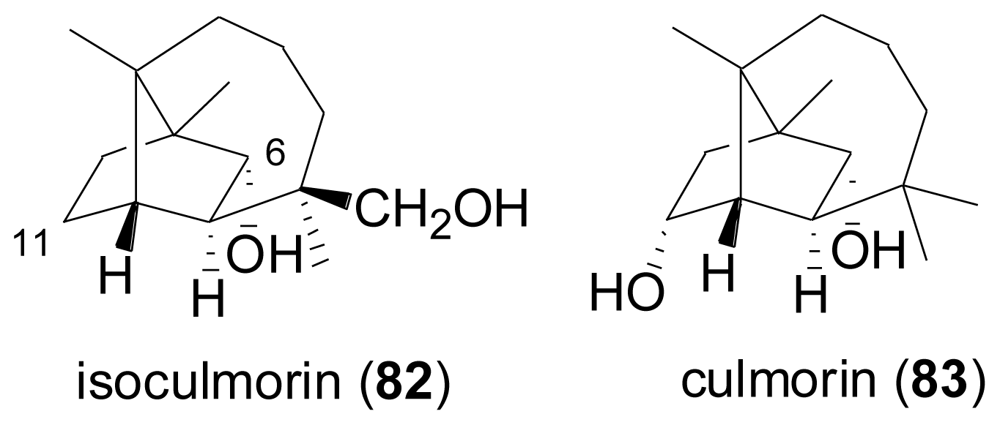
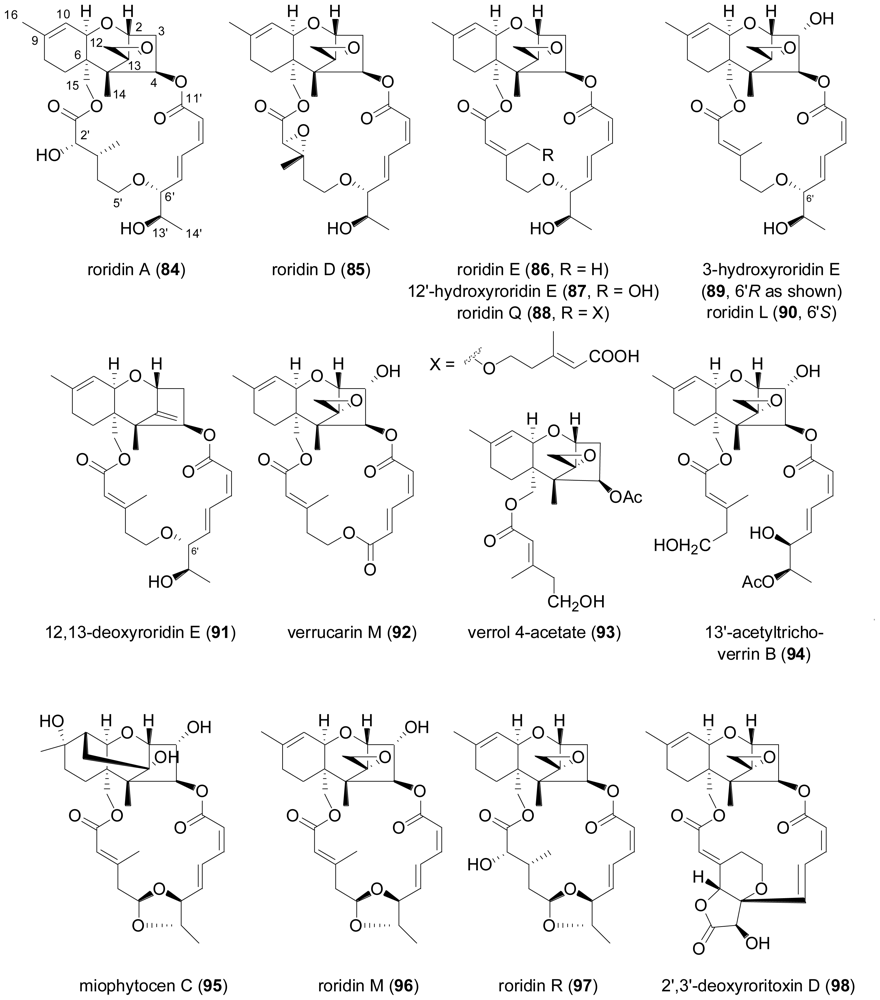
2.3. Diterpenes
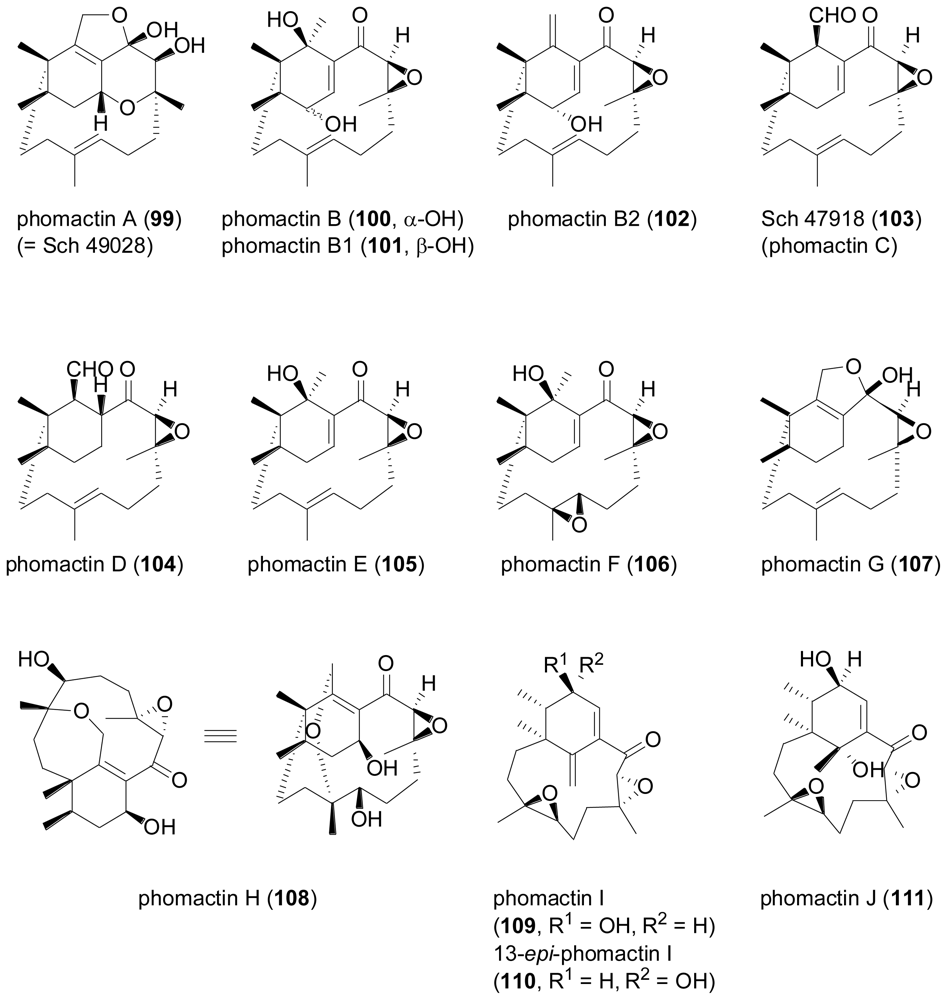
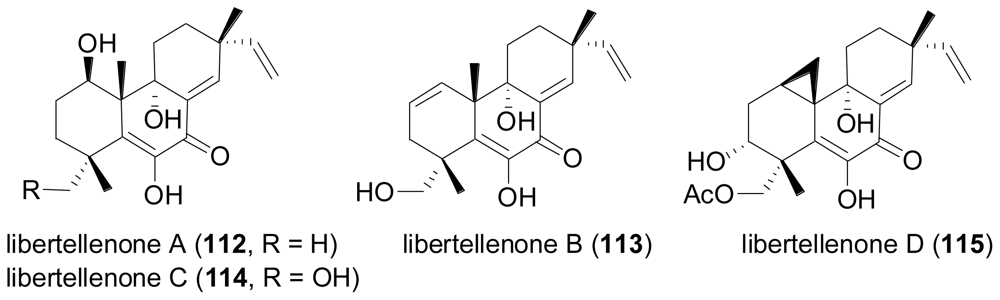
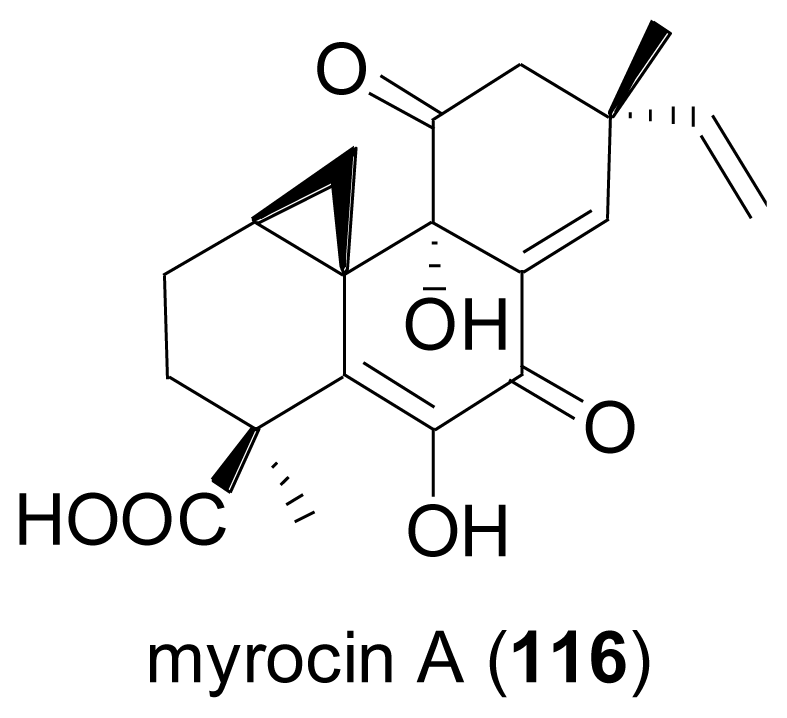
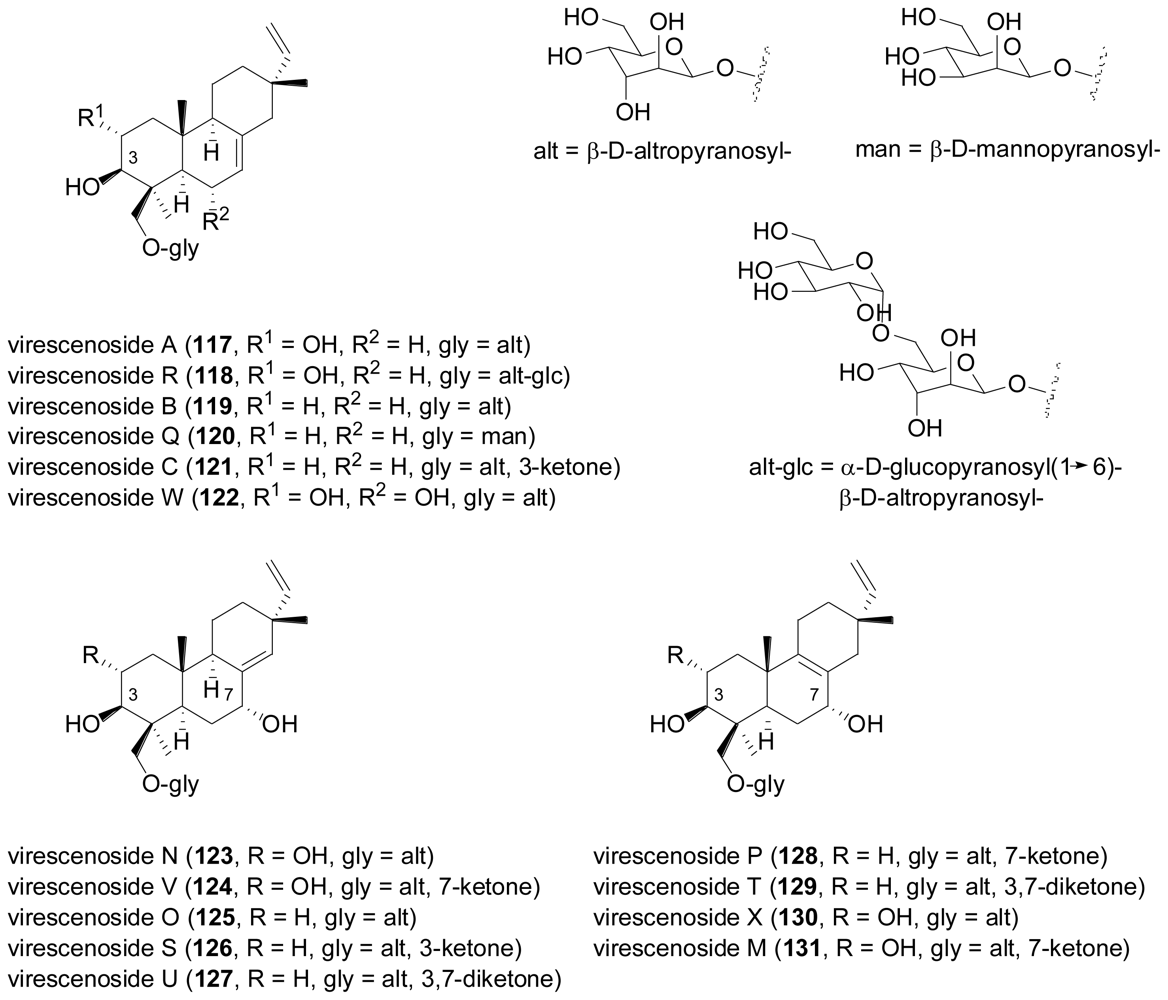
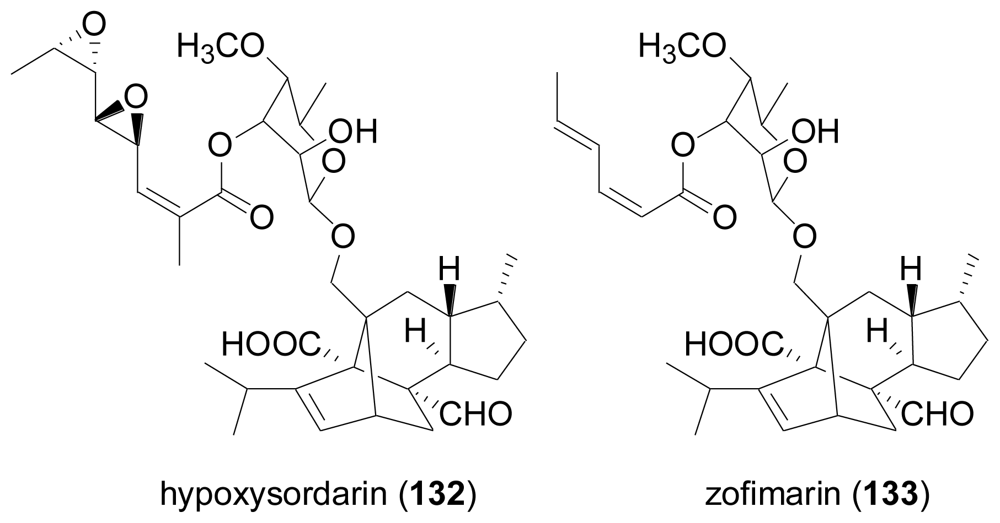
2.4. Sesterterpenes
2.5. Triterpenes
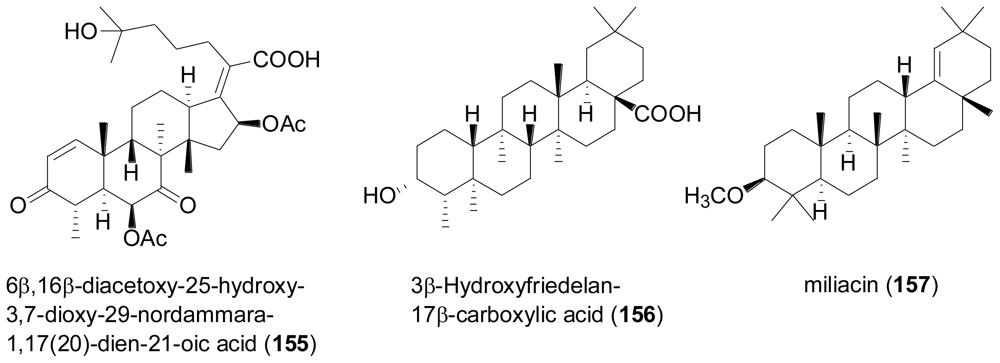
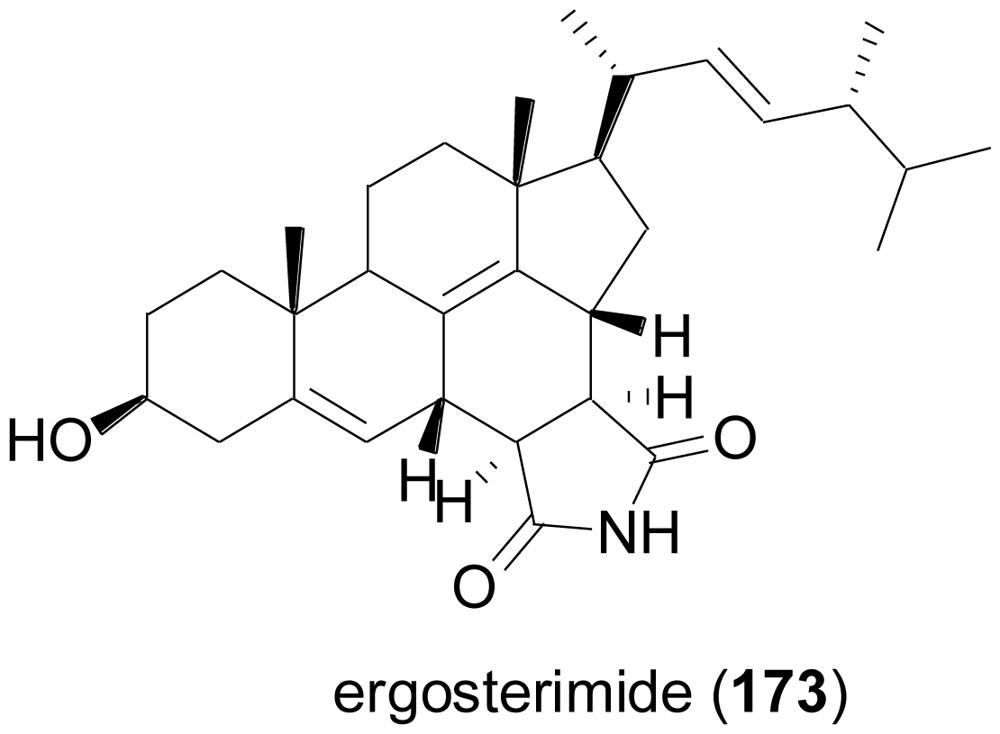
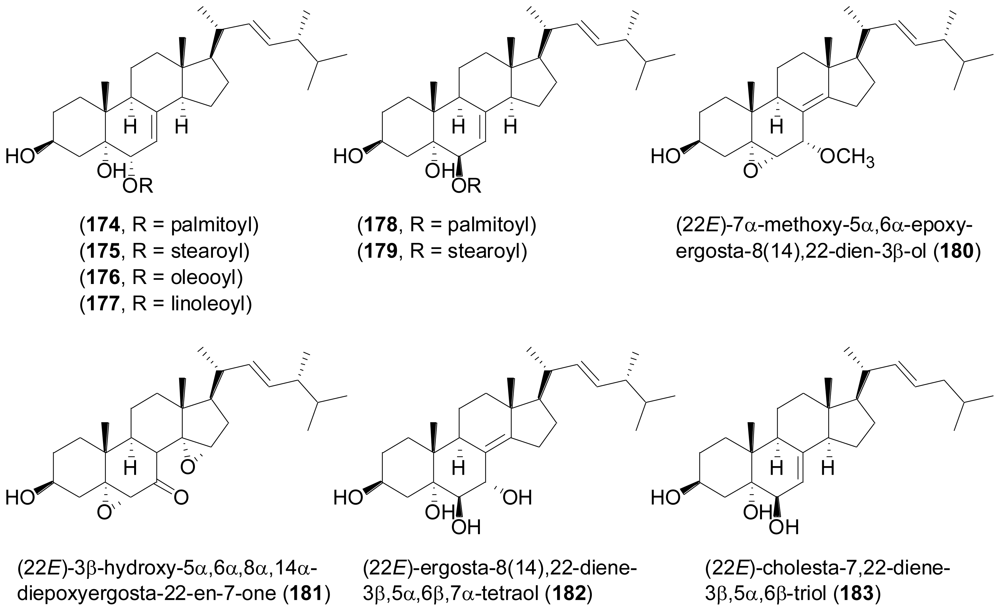
2.6. Steroids
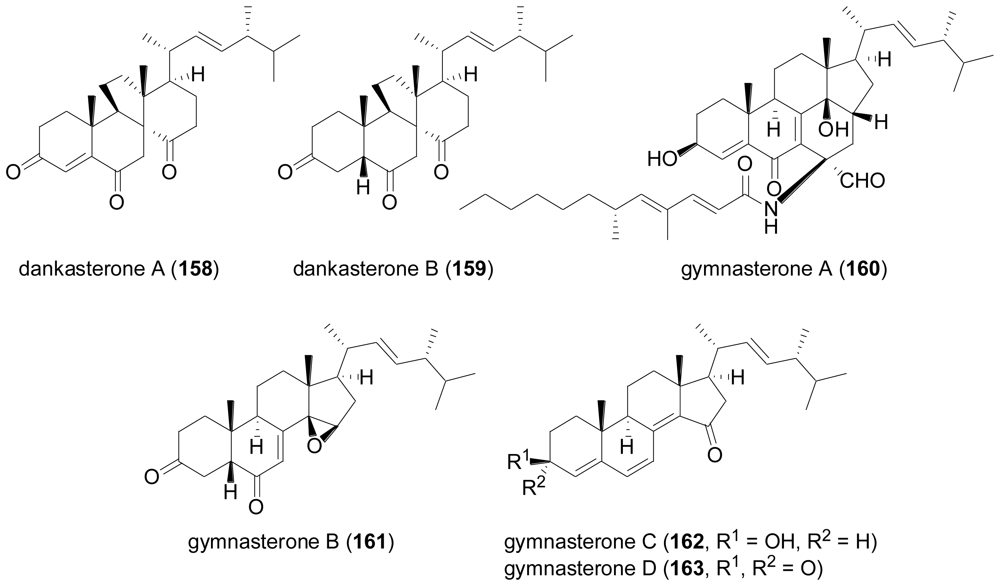
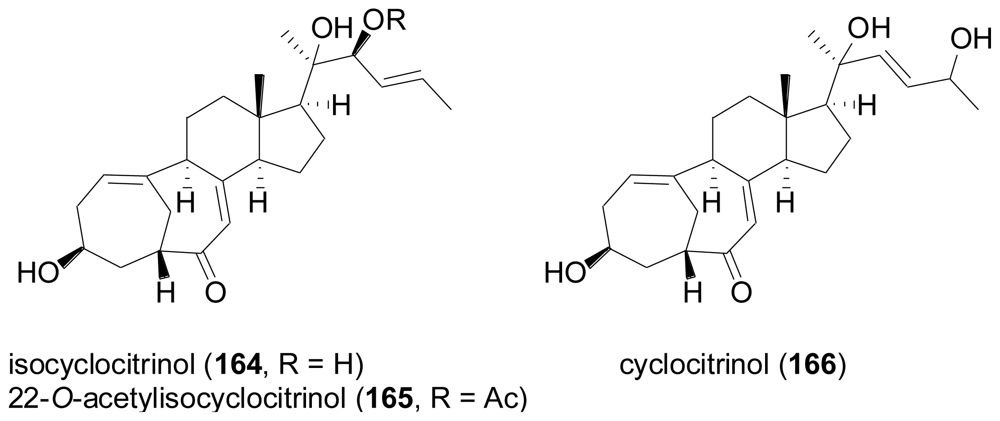
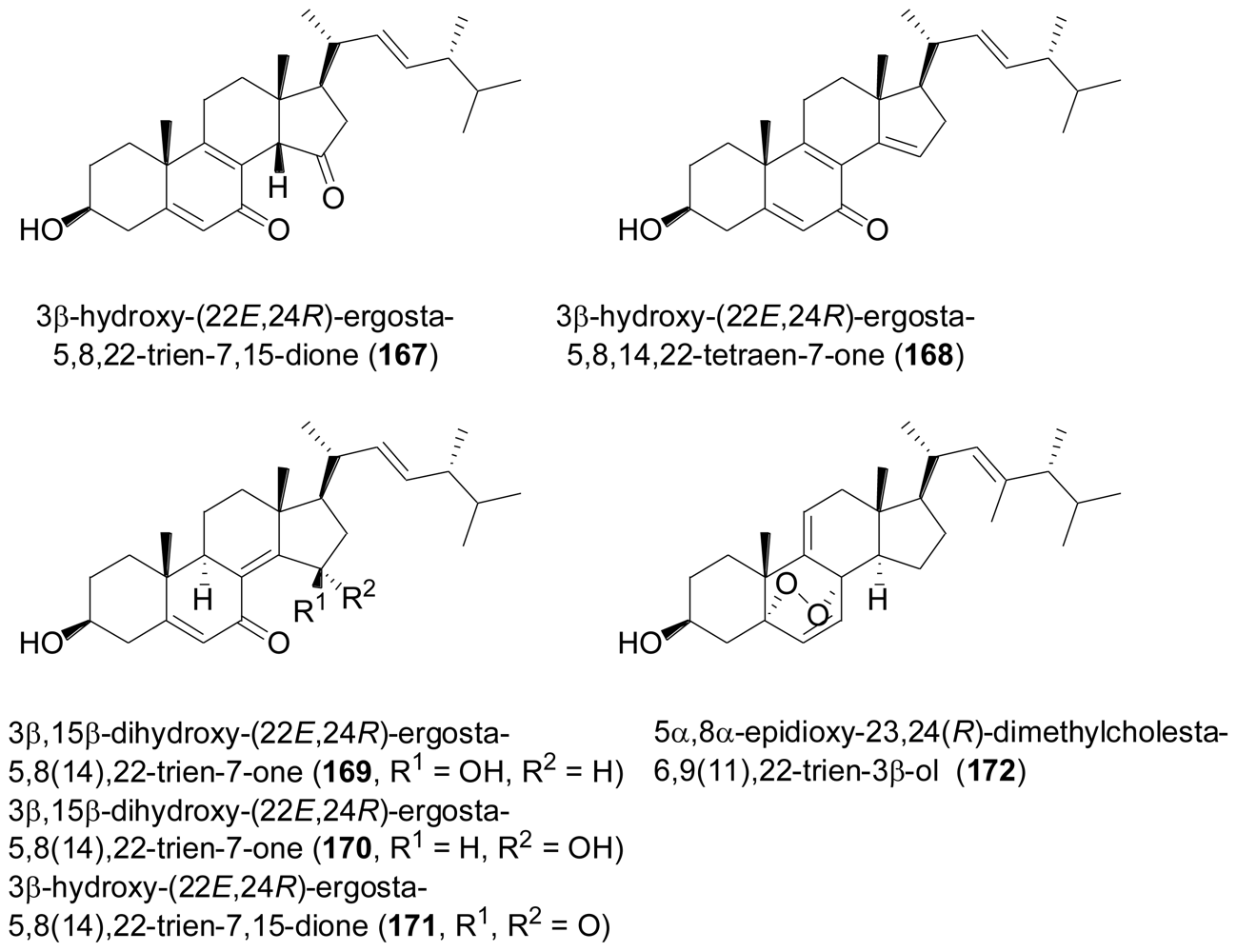
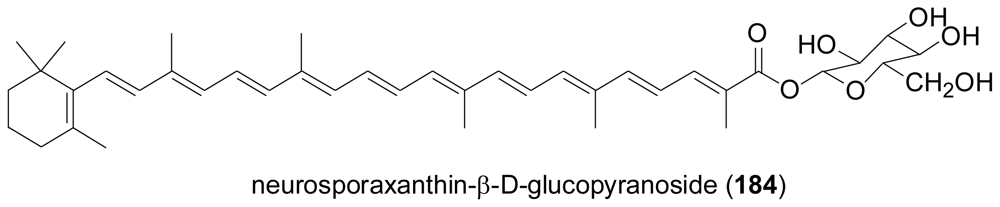
2.7. Tetraterpenes (Carotenoids)
3. Conclusions
- Samples Availability: Available from the authors.
References
- Bugni, TS; Ireland, CM. Marine-derived fungi: a chemically and biologically diverse group of microorganisms. Nat. Prod. Rep 2004, 21, 143–163. [Google Scholar]
- Ebel, R. Proksch, P, Müller, WEG, Eds.; Secondary metabolites from marine-derived fungi. In Frontiers in Marine Biotechnology; Horizon Bioscience: Norwich, UK, 2006; pp. 73–144. [Google Scholar]
- Saleem, M; Ali, MS; Hussain, S; Jabbar, A; Ashraf, M; Lee, YS. Marine natural products of fungal origin. Nat. Prod. Rep 2007, 24, 1142–1152. [Google Scholar]
- Huang, HR; Xia, XK; She, ZG; Lin, YC; Vrijmoed, LLP; Jones, EBG. A new chloro-monoterpene from the mangrove endophytic fungus Tryblidiopycnis sp. (4275). J. Asian Nat. Prod. Res 2006, 8, 609–612. [Google Scholar]
- Guerriero, A; Dambrosio, M; Cuomo, V; Vanzanella, F; Pietra, F. Dendryphiellin A, the first fungal trinor-eremophilane. Isolation from the marine deuteromycete Dendryphiella salina (Sutherland) Pugh et Nicot. Helv. Chim. Acta 1988, 71, 57–61. [Google Scholar]
- Guerriero, A; Dambrosio, M; Cuomo, V; Vanzanella, F; Pietra, F. Novel trinor-eremophilanes (dendryphiellin B, C, and D), eremophilanes (dendryphiellins E, F, and G), and branched C9-carboxylic acids (dendryphiellic acid A and B) from the marine deuteromycete Dendryphiella salina (Sutherland) Pugh et Nicot. Helv. Chim. Acta 1989, 72, 438–446. [Google Scholar]
- Guerriero, A; Cuomo, V; Vanzanella, F; Pietra, F. A novel glyceryl ester (glyceryl dendryphiellate A), a trinor-eremophilane (dendryphiellin A1), and eremophilanes (dendryphiellins E1 and E2) from the marine deuteromycete Dendryphiella salina (Sutherland) Pugh et Nicot. Helv. Chim. Acta 1990, 73, 2090–2096. [Google Scholar]
- Huang, YF; Qiao, L; Lv, AL; Pei, YH; Tian, L. Eremophilane sesquiterenes from the marine fungus Penicillium sp. BL27-2. Chin. Chem. Lett 2008, 19, 562–564. [Google Scholar]
- Schneider, G; Anke, H; Sterner, O. New secondary metabolites from a mycophilic Hansfordia species. Nat. Prod. Lett 1997, 10, 133–138. [Google Scholar]
- Höller, U; König, GM; Wright, AD. Three new metabolites from marine-derived fungi of the genera Coniothyrium and Microsphaeropsis. J. Nat. Prod 1999, 62, 114–118. [Google Scholar]
- Shao, CL; Wang, CY; Li, CY; She, ZG; Gu, YC; Lin, YC. Microsphaeropsisin A, a new sesquiterpenoid isolated from the mangrove endophytic fungus (No. DZ39). Nat. Prod. Res 2009, 23, 1579–1583. [Google Scholar]
- McDonald, LA; Barbieri, LR; Bernan, VS; Janso, J; Lassota, P; Carter, GT. 07H239-A, a new cytotoxic eremophilane sesquiterpene from the marine-derived Xylariaceous fungus LL-07H239. J. Nat. Prod 2004, 67, 1565–1567. [Google Scholar]
- Singh, SB; Zink, D; Polishook, J; Valentino, D; Shafiee, A; Silverman, K; Felock, P; Teran, A; Vilella, D; Hazuda, DJ; Lingham, RB. Structure and absolute stereochemistry of HIV-1 integrase inhibitor integric acid. A novel eremophilane sesquiterpenoid produced by a Xylaria sp. Tetrahedron Lett 1999, 40, 8775–8779. [Google Scholar]
- Singh, SB; Felock, P; Hazuda, DJ. Chemical and enzymatic modifications of integric acid and HIV-1 integrase inhibitory activity. Bioorg. Med. Chem. Lett 2000, 10, 235–238. [Google Scholar]
- Puar, MS; Barrabee, E; Hallade, M; Patel, M. Sch 420789: a novel fungal metabolite with phospholipase D inhibitory activity. J. Antibiot 2000, 53, 837–838. [Google Scholar]
- Oh, H; Jensen, PR; Murphy, BT; Fiorilla, C; Sullivan, JF; Ramsey, T; Fenical, W. Cryptosphaerolide, a cytotoxic Mcl-1 inhibitor from a marine-derived ascomycete related to the genus Cryptosphaeria. J. Nat. Prod 2010, 73, 998–1001. [Google Scholar]
- Isaka, M; Srisanoh, U; Veeranondha, S; Choowong, W; Lumyong, S. Cytotoxic eremophilane sesquiterpenoids from the saprobic fungus Berkleasmium nigroapicale BCC 8220. Tetrahedron 2009, 65, 8808–8815. [Google Scholar]
- Yamada, T; Doi, M; Miura, A; Harada, W; Hiramura, M; Minoura, K; Tanaka, R; Numata, A. Absolute stereostructures of cell-adhesion inhibitors, peribysins A, E, F and G, produced by a sea hare-derived Periconia sp. J. Antibiot 2005, 58, 185–191. [Google Scholar]
- Yamada, T; Iritani, M; Minoura, K; Kawai, K; Numata, A. Peribysins A–D, potent cell-adhesion inhibitors from a sea hare-derived culture of Periconia species. Org. Biomol. Chem 2004, 2, 2131–2135. [Google Scholar]
- Yamada, T; Minoura, K; Tanaka, R; Numata, A. Cell-adhesion inhibitors produced by a sea hare-derived Periconia sp II - Absolute stereostructures of peribysins H and I. J. Antibiot 2006, 59, 345–350. [Google Scholar]
- Yamada, T; Minoura, K; Tanaka, R; Numata, A. Cell-adhesion inhibitors produced by a sea hare-derived Periconia sp - III absolute stereostructures of peribysin J and macrosphelide M. J. Antibiot 2007, 60, 370–375. [Google Scholar]
- Koshino, H; Satoh, H; Yamada, T; Esumi, Y. Structural revision of peribysins C and D. Tetrahedron Lett 2006, 47, 4623–4626. [Google Scholar]
- Angeles, AR; Dorn, DC; Kou, CA; Moore, MAS; Danishefsky, SJ. Total synthesis of peribysin E necessitates revision of the assignment of its absolute configuration. Angew. Chem. Int. Ed 2007, 46, 1451–1454. [Google Scholar]
- Angeles, AR; Waters, SP; Danishefsky, SJ. Total syntheses of (+)- and (−)-peribysin E. J. Am. Chem. Soc 2008, 130, 13765–13770. [Google Scholar]
- Cheng, XC; Varoglu, M; Abrell, L; Crews, P; Lobkovsky, E; Clardy, J. Chloriolins A–C, chlorinated sesquiterpenes produced by fungal cultures separated from a Jaspis marine sponge. J. Org. Chem 1994, 59, 6344–6348. [Google Scholar]
- Takahashi, S; Iinuma, H; Takita, T; Maeda, K; Umezawa, H. The structure of coriolin, a new sesquiterpene antibiotic. Tetrahedron Lett 1969, 53, 4663–4666. [Google Scholar]
- Takeuchi, T; Iinuma, H; Iwanaga, J; Takahash, S; Takita, T; Umezawa, H. Coriolin, a new basidiomycetes antibiotic. J. Antibiot 1969, 22, 215–217. [Google Scholar]
- Takahashi, S; Iinuma, H; Takita, T; Maeda, K; Umezawa, H. The structures of coriolin B and C. Tetrahedron Lett 1970, 19, 1637–1639. [Google Scholar]
- Wang, GYS; Abrell, LM; Avelar, A; Borgeson, BM; Crews, P. New hirsutane based sesquiterpenes from salt water cultures of a marine sponge-derived fungus and the terrestrial fungus Coriolus consors. Tetrahedron 1998, 54, 7335–7342. [Google Scholar]
- Gao, J; Yue, DC; Cheng, KD; Wang, SC; Yu, KB; Zheng, QT; Yang, JS. Gloeosteretriol a new sesquiterpene from the fermentation products of Gloeostereum incarnatum s. Ito et Imai. Yao Xue Xue Bao 1992, 27, 33–36. [Google Scholar]
- Amouzou, E; Ayer, WA; Browne, LM. Antifungal sesquiterpenoids from an arthroconidial fungus. J. Nat. Prod 1989, 52, 1042–1054. [Google Scholar]
- Belofsky, GN; Jensen, PR; Renner, MK; Fenical, W. New cytotoxic sesquiterpenoid nitrobenzoyl esters from a marine isolate of the fungus Aspergillus versicolor. Tetrahedron 1998, 54, 1715–1724. [Google Scholar]
- Rahbæk, L; Christophersen, C; Frisvad, J; Bengaard, HS; Larsen, S; Rassing, BR. Insulicolide A: A new nitrobenzoyloxy-substituted sesquiterpene from the marine fungus Aspergillus insulicola. J. Nat. Prod 1997, 60, 811–813. [Google Scholar]
- Liu, HB; Edrada-Ebel, R; Ebel, R; Wang, Y; Schulz, B; Draeger, S; Müller, WEG; Wray, V; Lin, WH; Proksch, P. Drimane sesquiterpenoids from the fungus Aspergillus ustus isolated from the marine sponge Suberites domuncula. J. Nat. Prod 2009, 72, 1585–1588. [Google Scholar]
- Proksch, P; Ebel, R; Edrada, R; Riebe, F; Liu, H; Diesel, A; Bayer, M; Li, X; Lin, WH; Grebenyuk, V; Müller, WEG; Draeger, S; Zuccaro, A; Schulz, B. Sponge-associated fungi and their bioactive compounds: the Suberites case. Bot. Mar 2008, 51, 209–218. [Google Scholar]
- Lu, ZY; Wang, Y; Miao, CD; Liu, PP; Hong, K; Zhu, WM. Sesquiterpenoids and benzofuranoids from the marine-derived fungus Aspergillus ustus 094102. J. Nat. Prod 2009, 72, 1761–1767. [Google Scholar]
- Ogawa, T; Ando, K; Tanaka, T; Uosaki, Y; Matsuda, Y. RES-1149-1 and -2, novel nonpeptidic endothelin type B receptor antagonists produced by Aspergillus sp. I. Taxonomy of producing strain, fermentation, isolation, and physico-chemical and biological properties. J. Antibiot 1996, 49, 1–5. [Google Scholar]
- Uosaki, Y; Yoshida, M; Ogawa, T; Saitoh, Y. RES-1149-1 and -2, novel non-peptidic endothelin type B receptor antagonists produced by Aspergillus sp. II. Structure determination and derivatization. J. Antibiot 1996, 49, 6–12. [Google Scholar]
- Ogawa, T; Uosaki, Y; Tanaka, T; Tsukuda, E; Mihara, A; Matsuda, Y. RES-1149-1 and-2, novel non-peptidic endothelin type B receptor antagonists produced by Aspergillus sp. III. Biochemical properties of RES-1149-1,-2 and structure-activity relationships. J. Antibiot 1996, 49, 168–172. [Google Scholar]
- Almeida, C; Eguereva, E; Kehraus, S; Siering, C; König, GM. Hydroxylated sclerosporin derivatives from the marine-derived fungus Cadophora malorum. J. Nat. Prod 2010, 73, 476–478. [Google Scholar]
- Katayama, M; Marumo, S. Structure of sclerosporin, a sporogenic substance of Sclerotinia fructicola. Tetrahedron Lett 1979, 20, 1773–1776. [Google Scholar]
- Katayama, M; Marumo, S; Hattori, H. The revised structure of sclerosporin, a sporogenic substance of Sclerotinia fructicola. The total synthesis of (±)-sclerosporin. Tetrahedron Lett 1983, 24, 1703–1706. [Google Scholar]
- Wei, MY; Wang, CY; Liu, QA; Shao, CL; She, ZG; Lin, YC. Five sesquiterpenoids from a marine-derived fungus Aspergillus sp. isolated from a gorgonian Dichotella gemmacea. Mar. Drugs 2010, 8, 941–949. [Google Scholar]
- Hamasaki, T; Sato, Y; Hatsuda, Y; Tanabe, M; Cary, LW. Sydowic acid, a new metabolite from Aspergillus sydowi. Tetrahedron Lett 1975, 9, 659–660. [Google Scholar]
- Hamasaki, T; Sato, Y; Hatsuda, Y. Isolation of new metabolites from Aspergillus sydowi and structure of sydowic acid. Agric. Biol. Chem 1975, 39, 2337–2340. [Google Scholar]
- Hamasaki, T; Nagayama, K; Hatsuda, Y. Two new metabolites, sydonic acid and hydroxysydonic acid, from Aspergillus sydowi. Agric. Biol. Chem 1978, 42, 37–40. [Google Scholar]
- Almeida, C; Elsaedi, S; Kehraus, S; König, GM. Novel bisabolane sesquiterpenes from the marine-derived fungus Verticillium tenerum. Nat. Prod. Commun 2010, 5, 507–510. [Google Scholar]
- Mülhaupt, T; Kaspar, H; Otto, S; Reichert, M; Bringmann, G; Lindel, T. Isolation, structural elucidation, and synthesis of curcutetraol. Eur. J. Org. Chem 2005, 2005, 334–341. [Google Scholar]
- Nukina, M; Sato, Y; Ikeda, M; Sassa, T. Sydonol, a new fungal morphogenic substance produced by an unidentified Aspergillus sp. Agric. Biol. Chem 1981, 45, 789–790. [Google Scholar]
- Henne, P; Thiericke, R; Grabley, S; Hutter, K; Wink, J; Jurkiewicz, E; Zeeck, A. Secondary metabolites by chemical screening, 23. Waraterpols, new Penicillium metabolites and their derivatives. Liebigs Ann. Chem 1993, 1993, 565–571. [Google Scholar]
- Laurent, D; Guella, G; Mancini, I; Roquebert, MF; Farinole, F; Pietra, F. A new cytotoxic tetralone derivative from Humicola grisea, a filamentous fungus from wood in the southeastern lagoon of New Caledonia. Tetrahedron 2002, 58, 9163–9167. [Google Scholar]
- Li, XF; Zhang, DH; Lee, U; Li, XG; Cheng, JG; Zhu, WL; Jung, JH; Choi, HD; Son, BW. Bromomyrothenone B and botrytinone, cyclopentenone derivatives from a marine isolate of the fungus Botrytis. J. Nat. Prod 2007, 70, 307–309. [Google Scholar]
- Li, XF; Kim, MK; Lee, U; Kim, SK; Kang, JS; Choi, HD; Son, BW. Myrothenones A and B, cyclopentenone derivatives with tyrosinase inhibitory activity from the marine-derived fungus Myrothecium sp. Chem. Pharm. Bull 2005, 53, 453–455. [Google Scholar]
- Li, XF; Kim, SK; Kang, JS; Choi, HD; Son, BW. Polyketide and sesquiterpenediol metabolites from a marine-derived fungus. Bull. Korean Chem. Soc 2004, 25, 607–608. [Google Scholar]
- Osterhage, C; König, GM; Höller, U; Wright, AD. Rare sesquiterpenes from the algicolous fungus Drechslera dematioidea. J. Nat. Prod 2002, 65, 306–313. [Google Scholar]
- Tamura, S; Sakurai, A; Kainuma, K; Takai, M. Isolation of helminthosporol as a natural plant growth regulator and its chemical structure. Agric. Biol. Chem 1963, 27, 738–739. [Google Scholar]
- Nukina, M; Hattori, H; Marumo, S. cis-Sativenediol, a plant growth promotor, produced by fungi. J. Am. Chem. Soc 1975, 97, 2542–2543. [Google Scholar]
- Dorn, F; Arigoni, D. Bicyclic derivative of (−)-longifolene from Helminthosporium sativum and H. victoriae. Experientia 1974, 30, 851–852. [Google Scholar]
- Alam, M; Jones, EBG; Hossain, MB; van der Helm, D. Isolation and structure of isoculmorin from the marine fungus Kallichroma tethys. J. Nat. Prod 1996, 59, 454–456. [Google Scholar]
- Langseth, W; Ghebremeskel, M; Kosiak, B; Kolsaker, P; Miller, D. Production of culmorin compounds and other secondary metabolites by Fusarium culmorum and F. graminearum strains isolated from Norwegian cereals. Mycopathologia 2001, 152, 23–34. [Google Scholar]
- Ghebremeskel, M; Langseth, W. The occurrence of culmorin and hydroxy-culmorins in cereals. Mycopathologia 2001, 152, 103–108. [Google Scholar]
- McCormick, SP; Alexander, NJ; Harris, LJ. CLM1 of Fusarium graminearum encodes a longiborneol synthase required for culmorin production. Appl. Environ. Microbiol 2010, 76, 136–141. [Google Scholar]
- Grove, JF. Macrocyclic trichothecenes. Nat. Prod. Rep 1993, 10, 429–448. [Google Scholar]
- Laurent, D; Guella, G; Roquebert, MF; Farinole, F; Mancini, I; Pietra, F. Cytotoxins, mycotoxins and drugs from a new deuteromycete, Acremonium neo-caledoniae, from the southwestern lagoon of New Caledonia. Planta Med 2000, 66, 63–66. [Google Scholar]
- Namikoshi, M; Akano, K; Meguro, S; Kasuga, I; Mine, Y; Takahashi, T; Kobayashi, H. A new macrocyclic trichothecene, 12,13-deoxyroridin E, produced by the marine-derived fungus Myrothecium roridum collected in Palau. J. Nat. Prod 2001, 64, 396–398. [Google Scholar]
- Xu, JZ; Takasaki, A; Kobayashi, H; Oda, T; Yamada, J; Mangindaan, REP; Ukai, K; Nagai, H; Namikoshi, M. Four new macrocyclic trichothecenes from two strains of marinederived fungi of the genus Myrothecium. J. Antibiot 2006, 59, 451–455. [Google Scholar]
- Amagata, T; Rath, C; Rigot, JF; Tarlov, N; Tenney, K; Valeriote, FA; Crews, P. Structures and cytotoxic properties of trichoverroids and their macrolide analogues produced by saltwater culture of Myrothecium verrucaria. J. Med. Chem 2003, 46, 4342–4350. [Google Scholar]
- Liu, JY; Huang, LL; Ye, YH; Zou, WX; Guo, ZJ; Tan, RX. Antifungal and new metabolites of Myrothecium sp. Z16, a fungus associated with white croaker Argyrosomus argentatus. J. Appl. Microbiol 2006, 100, 195–202. [Google Scholar]
- Xie, LW; Jiang, SM; Zhu, HH; Sun, W; Ouyang, YC; Dai, SK; Li, X. Potential inhibitors against Sclerotinia sclerotiorum, produced by the fungus Myrothecium sp. associated with the marine sponge Axinella sp. Eur. J. Plant Pathol 2008, 122, 571–578. [Google Scholar]
- Sugano, M; Sato, A; Iijima, Y; Oshima, T; Furuya, K; Kuwano, H; Hata, T; Hanzawa, H. Phomactin A: A novel PAF antagonist from a marine fungus Phoma sp. J. Am. Chem. Soc 1991, 113, 5463–5464. [Google Scholar]
- Sugano, M; Sato, A; Iijima, Y; Furuya, K; Haruyama, H; Yoda, K; Hata, T. Phomactins, novel PAF antagonists from marine fungus Phoma sp. J. Org. Chem 1994, 59, 564–569. [Google Scholar]
- Sugano, M; Sato, A; Iijima, Y; Furuya, K; Kuwano, H; Hata, T. Phomactin E, F, and G: new phomactin-group PAF antagonists from a marine fungus Phoma sp. J. Antibiot 1995, 48, 1188–1190. [Google Scholar]
- Chu, M; Patel, MG; Gullo, VP; Truumees, I; Puar, MS; McPhail, AT. Sch-47918: a novel PAF antagonist from the fungus Phoma sp. J. Org. Chem 1992, 57, 5817–5818. [Google Scholar]
- Chu, M; Truumees, I; Gunnarsson, I; Bishop, WR; Kreutner, W; Horan, AC; Patel, MG; Gullo, VP; Puar, MS. A novel class of platelet activating factor antagonists from Phoma sp. J. Antibiot 1993, 46, 554–563. [Google Scholar]
- Koyama, K; Ishino, M; Takatori, K; Sugita, T; Kinoshita, K; Takahashi, K. Phomactin H, a novel diterpene from an unidentified marine-derived fungus. Tetrahedron Lett 2004, 45, 6947–6948. [Google Scholar]
- Ishino, M; Kiyomichi, N; Takatori, K; Sugita, T; Shiro, M; Kinoshita, K; Takahashi, K; Koyama, K. Phomactin I, 13-epi-phomactin I, and phomactin J, three novel diterpenes from a marine-derived fungus. Tetrahedron 2010, 66, 2594–2597. [Google Scholar]
- Goldring, WPD; Pattenden, G. The phomactins. A novel group of terpenoid platelet activating factor antagonists related biogenetically to the taxanes. Acc. Chem. Res 2006, 39, 354–361. [Google Scholar]
- Chow, SY; Williams, HJ; Pennington, JD; Nanda, S; Reibenspies, JH; Scott, AI. Studies on taxadiene synthase: interception of the cyclization cascade at the verticillene stage and rearrangement to phomactatriene. Tetrahedron 2007, 63, 6204–6209. [Google Scholar]
- Tokiwano, T; Endo, T; Tsukagoshi, T; Goto, H; Fukushi, E; Oikawa, H. Proposed mechanism for diterpene synthases in the formation of phomactatriene and taxadiene. Org. Biomol. Chem 2005, 3, 2713–2722. [Google Scholar]
- Tokiwano, T; Fukushi, E; Endo, T; Oikawa, H. Biosynthesis of phomactins: common intermediate phomactatriene and taxadiene. Chem. Commun 2004, 1324–1325. [Google Scholar] [CrossRef]
- Oh, DC; Jensen, PR; Kauffman, CA; Fenical, W. Libertellenones A–D: Induction of cytotoxic diterpenoid biosynthesis by marine microbial competition. Bioorg. Med. Chem 2005, 13, 5267–5273. [Google Scholar]
- Cueto, M; Jensen, PR; Kauffman, C; Fenical, W; Lobkovsky, E; Clardy, J. Pestalone, a new antibiotic produced by a marine fungus in response to bacterial challenge. J. Nat. Prod 2001, 64, 1444–1446. [Google Scholar]
- Klemke, C; Kehraus, S; Wright, AD; König, GM. New secondary metabolites from the marine endophytic fungus Apiospora montagnei. J. Nat. Prod 2004, 67, 1058–1063. [Google Scholar]
- Hsu, YH; Nakagawa, M; Hirota, A; Shima, S; Nakayama, M. Structure of myrocin B, a new diterpene antibiotic produced by Myrothecium verrucaria. Agric. Biol. Chem 1988, 52, 1305–1307. [Google Scholar]
- Afiyatullov, SS; Kalinovsky, AI; Kuznetsova, TA; Isakov, VV; Pivkin, MV; Dmitrenok, PS; Elyakov, GB. New diterpene glycosides of the fungus Acremonium striatisporum isolated from a sea cucumber. J. Nat. Prod 2002, 65, 641–644. [Google Scholar]
- Afiyatullov, SS; Kalinovsky, AI; Kuznetsova, TA; Pivkin, MV; Prokof'eva, NG; Dmitrenok, PS; Elyakov, GB. New glycosides of the fungus Acremonium striatisporum isolated from a sea cucumber. J. Nat. Prod 2004, 67, 1047–1051. [Google Scholar]
- Afiyatullov, SS; Kalinovsky, AI; Pivkin, MV; Dmitrenok, PS; Kuznetsova, TA. New diterpene glycosides of the fungus Acremonium striatisporum isolated from a sea cucumber. Nat. Prod. Res 2006, 20, 902–908. [Google Scholar]
- Afiyatullov, SS; Kuznetsova, TA; Isakov, VV; Pivkin, MV; Prokof'eva, NG; Elyakov, GB. New diterpenic altrosides of the fungus Acremonium striatisporum isolated from a sea cucumber. J. Nat. Prod 2000, 63, 848–850. [Google Scholar]
- Daferner, M; Mensch, S; Anke, T; Sterner, O. Hypoxysordarin, a new sordarin derivative from Hypoxylon croceum. Z. Naturforsch., C J. Biosci 1999, 54, 474–480. [Google Scholar]
- Hauser, D; Sigg, HP. Isolierung und Abbau von Sordarin. Helv. Chim. Acta 1971, 54, 1178–1190. [Google Scholar]
- Ogita, T; Hayashi, A; Sato, S; Furaya, K. Antibiotic zofimarin. Jpn. Kokai Tokkyo Koho JP 6240292 1987. [Google Scholar]
- Vicente, F; Basilio, A; Platas, G; Collado, J; Bills, GF; Del Val, AG; Martin, J; Tormo, JR; Harris, GH; Zink, DL; Justice, M; Kahn, JN; Pelaez, F. Distribution of the antifungal agents sordarins across filamentous fungi. Mycol. Res 2009, 113, 754–770. [Google Scholar]
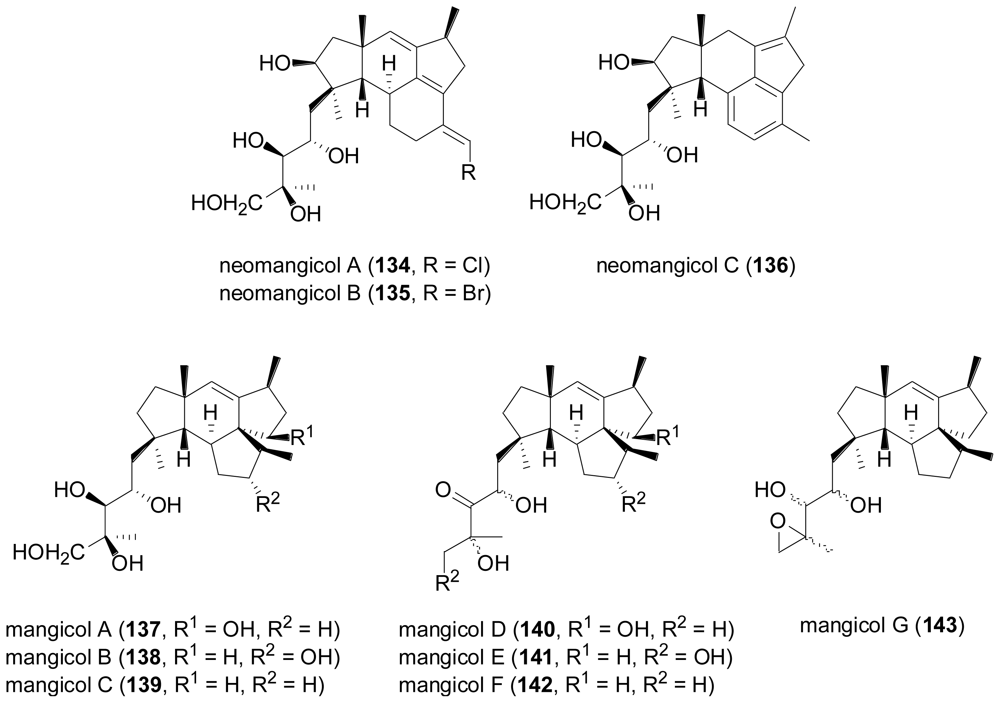
- Renner, MK; Jensen, PR; Fenical, W. Neomangicols: Structures and absolute stereochemistries of unprecedented halogenated sesterterpenes from a marine fungus of the genus Fusarium. J. Org. Chem 1998, 63, 8346–8354. [Google Scholar]
- Renner, MK; Jensen, PR; Fenical, W. Mangicols: Structures and biosynthesis of a new class of sesterterpene polyols from a marine fungus of the genus Fusarium. J. Org. Chem 2000, 65, 4843–4852. [Google Scholar]
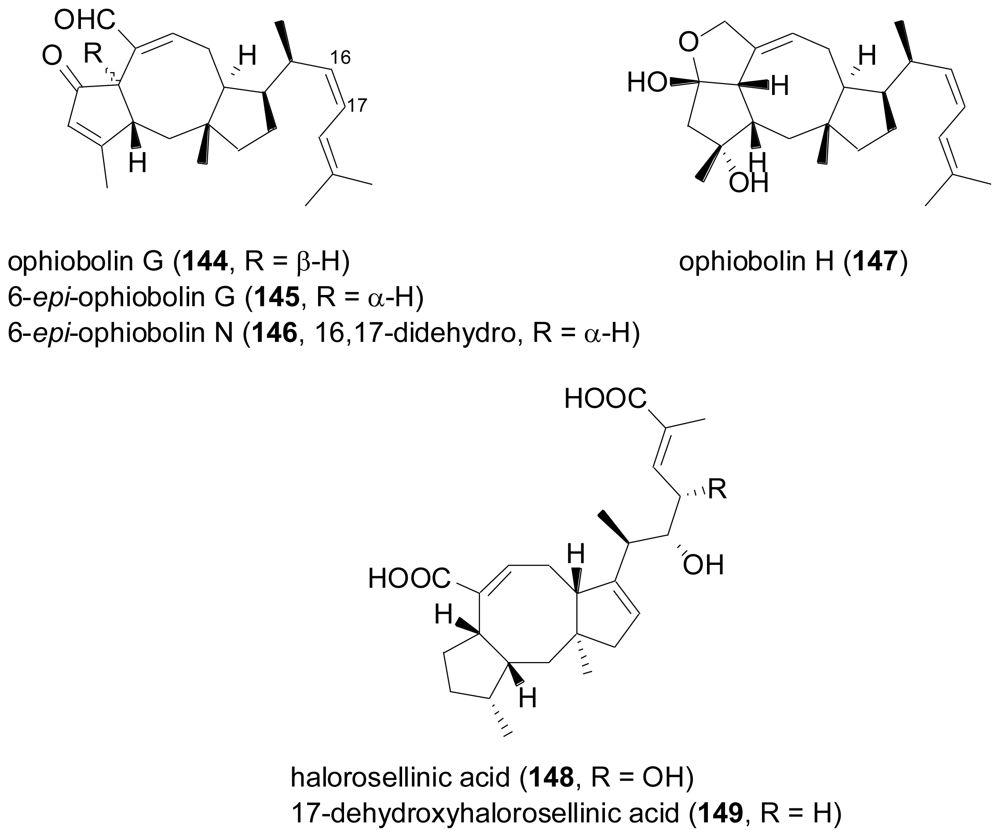
- Au, TK; Chick, WSH; Leung, PC. The biology of ophiobolins. Life Sci 2000, 67, 733–742. [Google Scholar]
- Wei, H; Itoh, T; Kinoshita, M; Nakai, Y; Kurotaki, M; Kobayashi, M. Cytotoxic sesterterpenes, 6-epi-ophiobolin G and 6-epi-ophiobolin N, from marine derived fungus Emericella variecolor GF10. Tetrahedron 2004, 60, 6015–6019. [Google Scholar]
- Chinworrungsee, M; Kittakoop, P; Isaka, M; Rungrod, A; Tanticharoen, M; Thebtaranonth, Y. Antimalarial halorosellinic acid from the marine fungus Halorosellinia oceanica. Bioorg. Med. Chem. Lett 2001, 11, 1965–1969. [Google Scholar]
- Chinworrungsee, M; Kittakoop, P; Isaka, M; Chanphen, R; Tanticharoen, M; Thebtaranonth, Y. Halorosellins A and B, unique isocoumarin glucosides from the marine fungus Halorosellinia oceanica. J. Chem. Soc. Perkin Trans. I 2002, 2473–2476. [Google Scholar] [CrossRef]
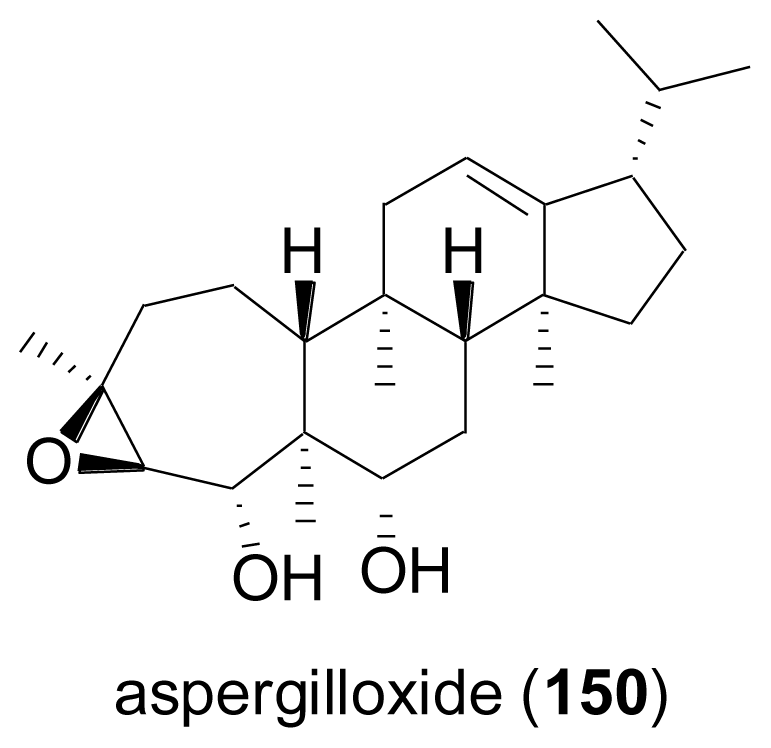
- Cueto, M; Jensen, PR; Fenical, W. Aspergilloxide, a novel sesterterpene epoxide from a marine-derived fungus of the genus Aspergillus. Org. Lett 2002, 4, 1583–1585. [Google Scholar]
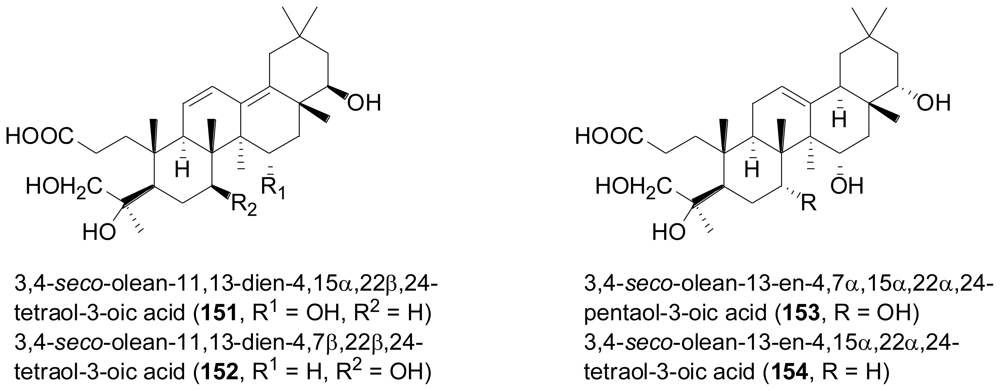
- Li, LY; Sattler, I; Deng, ZW; Groth, I; Walther, G; Menzel, KD; Peschel, G; Grabley, S; Lin, WH. A-seco-oleane-type triterpenes from Phomopsis sp (strain HK10458) isolated from the mangrove plant Hibiscus tiliaceus. Phytochemistry 2008, 69, 511–517. [Google Scholar]
- Shirane, N; Hashimoto, Y; Ueda, K; Takenaka, H; Katoh, K. Ring-A cleavage of 3-oxo-olean- 12-en-28-oic acid by the fungus Chaetomium longirostre. Phytochemistry 1996, 43, 99–104. [Google Scholar]
- Laskin, AI; Grabowich, P; De Lisle Meyers, C; Fried, J. Transformations of eburicoic acid. V. Cleavage of ring A by the fungus Glomerella fusarioides. J. Med. Chem 1964, 7, 406–409. [Google Scholar]
- Zhang, M; Wang, WL; Fang, YC; Zhu, TJ; Gu, QQ; Zhu, WM. Cytotoxic alkaloids and antibiotic nordammarane triterpenoids from the marine-derived fungus Aspergillus sydowi. J. Nat. Prod 2008, 71, 985–989. [Google Scholar]
- Chen, HY; Lin, CW; Chen, GY; Ou, GC. 3β-hydroxyfriedelan-17β-carboxylic acid. Acta Crystallogr. Sect. E: Struct. Rep. Online 2008, 64, O890–U2367. [Google Scholar]
- Smetanina, OF; Kuznetzova, TA; Denisenko, VA; Pivkin, MV; Khudyakova, YV; Gerasimenko, AV; Popov, DY; Il'in, SG; Elyakov, GB. 3β-Methoxyolean-18-ene (miliacin) from the marine fungus Chaetomium olivaceum. Russ. Chem. Bull 2001, 50, 2463–2465. [Google Scholar]
- Abe, S. Chemical structure of miliacin. Bull. Chem. Soc. Jpn 1960, 33, 271–272. [Google Scholar]
- Amagata, T; Tanaka, M; Yamada, T; Doi, M; Minoura, K; Ohishi, H; Yamori, T; Numata, A. Variation in cytostatic constituents of a sponge-derived Gymnascella dankaliensis by manipulating the carbon source. J. Nat. Prod 2007, 70, 1731–1740. [Google Scholar]
- Amagata, T; Doi, M; Tohgo, M; Minoura, K; Numata, A. Dankasterone, a new class of cytotoxic steroid produced by a Gymnascella species from a marine sponge. Chem. Commun 1999, 1321–1322. [Google Scholar] [CrossRef]
- Canonica, L; Danieli, B; Lesma, G; Palmisano, G; Mugnoli, A. Fe(II)-induced fragmentation reaction of γ-hydroperoxy-α,β-enones. Part 1. Synthesis of 13(14→8)-abeo-steroids. Helv. Chim. Acta 1987, 70, 701–716. [Google Scholar]
- Amagata, T; Amagata, A; Tenney, K; Valeriote, FA; Lobkovsky, E; Clardy, J; Crews, P. Unusual C25 steroids produced by a sponge-derived Penicillium citrinum. Org. Lett 2003, 5, 4393–4396. [Google Scholar]
- Kozlovsky, AG; Zhelifonova, VP; Ozerskaya, SM; Vinokurova, NG; Adanin, VM; Gräfe, U. Cyclocitrinol, a new fungal metabolite from Penicillium citrinum. Pharmazie 2000, 55, 470–471. [Google Scholar]
- Wang, F; Fang, Y; Zhang, M; Lin, A; Zhu, A; Gu, Q; Zhu, W. Six new ergosterols from the marine-derived fungus Rhizopus sp. Steroids 2008, 73, 19–26. [Google Scholar]
- Zhang, Y; Li, XM; Proksch, P; Wang, BG. Ergosterimide, a new natural Diels-Alder adduct of a steroid and maleimide in the fungus Aspergillus niger. Steroids 2007, 72, 723–727. [Google Scholar]
- Gao, H; Hong, K; Zhang, X; Liu, HW; Wang, NL; Zhuang, L; Yao, XS. New steryl esters of fatty acids from the mangrove fungus Aspergillus awamori. Helv. Chim. Acta 2007, 90, 1165–1178. [Google Scholar]
- Gao, H; Hong, K; Chen, GD; Wang, CX; Tang, JS; Yu, Y; Jiang, MM; Li, MM; Wang, NL; Yao, XS. New oxidized sterols from Aspergillus awamori and the endo-boat conformation adopted by the cyclohexene oxide system. Magn. Reson. Chem 2010, 48, 38–43. [Google Scholar]
- Sun, Y; Tian, L; Huang, J; Li, W; Pei, YH. Cytotoxic sterols from marine-derived fungus Penicillium sp. Nat. Prod. Res 2006, 20, 381–384. [Google Scholar]
- You, JL; Dai, HQ; Chen, ZH; Liu, GJ; He, ZX; Song, FH; Yang, X; Fu, HA; Zhang, LX; Chen, XP. Trichoderone, a novel cytotoxic cyclopentenone and cholesta-7,22-diene- 3β,5α,6β-triol, with new activities from the marine-derived fungus Trichoderma sp. J. Ind. Microbiol. Biotechnol 2010, 37, 245–252. [Google Scholar]
- Sakaki, H; Kaneno, H; Sumiya, Y; Tsushima, M; Miki, W; Kishimoto, N; Fujita, T; Matsumoto, S; Komemushi, S; Sawabe, A. A new carotenoid glycosyl ester isolated from a marine microorganism, Fusarium strain T-1. J. Nat. Prod 2002, 65, 1683–1684. [Google Scholar]
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ebel, R. Terpenes from Marine-Derived Fungi. Mar. Drugs 2010, 8, 2340-2368. https://doi.org/10.3390/md8082340
Ebel R. Terpenes from Marine-Derived Fungi. Marine Drugs. 2010; 8(8):2340-2368. https://doi.org/10.3390/md8082340
Chicago/Turabian StyleEbel, Rainer. 2010. "Terpenes from Marine-Derived Fungi" Marine Drugs 8, no. 8: 2340-2368. https://doi.org/10.3390/md8082340




