Chitin and Chitosan as Multipurpose Natural Polymers for Groundwater Arsenic Removal and As2O3 Delivery in Tumor Therapy
Abstract
:1. Introduction
2. Chitin and Chitosan for Arsenic Species Complexation and Removal
3. Chitosan for As2O3 Incorporation and Delivery
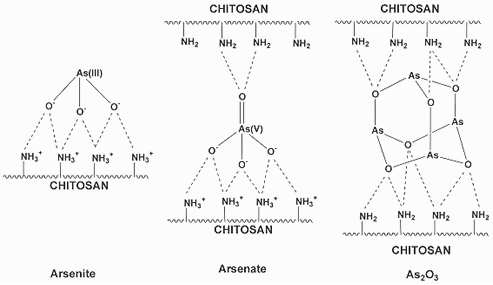
4. Interactions of Arsenite, Arsenate and Arsenic Trioxide (As2O3) with Chitosan
5. Conclusions
Acknowledgements
- Samples Availability: Available from the authors.
References and Notes
- Muzzarelli, RAA. Gupta, SN, Ed.; Chitin nanostructures in living organisms. In Chitin Formation and Diagenesis; Springer: New York, NY, USA, 2010. [Google Scholar]
- Roberts, GAF. Chitin Chemistry; Macmillan: London, UK, 1992. [Google Scholar]
- Muzzarelli, RAA (Ed.) Chitin Enzymology; Atec Edizioni: Grottammare, Italy, 1996; Volume 2.
- Muzzarelli, RAA. Natural Chelating Polymers; Pergamon Press: Oxford, UK, 1973. [Google Scholar]
- Yusof, NL; Lim, LY; Khor, E. Flexible chitin films: structural studies. Carbohydr Res 2004, 339, 2701–2711. [Google Scholar]
- Acosta, N; Aranaz, I; Peniche, C; Heras, A. Tramadol Release from a Delivery System Based on Alginate-Chitosan Microcapsules. Macromol Biosci 2003, 3, 546–551. [Google Scholar]
- Peniche, C; Argüelles-Monal, W; Peniche, H; Acosta, N. Chitosan: An Attractive Biocompatible Polymer for Microencapsulation. Macromol Biosci 2003, 3, 511–520. [Google Scholar]
- Bhatnagar, A; Sillanpaa, M. Applications of chitin- and chitosan-derivatives for the detoxification of water and wastewater--a short review. Adv Colloid Interface Sci 2009, 152, 26–38. [Google Scholar]
- Jayakumar, R; Selvamurugan, N; Nair, SV; Tokura, S; Tamura, H. Preparative methods of phosphorylated chitin and chitosan--an overview. Int J Biol Macromol 2008, 43, 221–225. [Google Scholar]
- Shi, C; Zhu, Y; Ran, X; Wang, M; Su, Y; Cheng, T. Therapeutic potential of chitosan and its derivatives in regenerative medicine. J Surg Res 2006, 133, 185–192. [Google Scholar]
- Chandy, T; Sharma, CP. Chitosan--as a biomaterial. Biomater Artif Cells Artif Organs 1990, 18, 1–24. [Google Scholar]
- Garelick, H; Jones, H; Dybowska, A; Valsami-Jones, E. Arsenic pollution sources. Rev.
- IARC Working Group on the Evaluation of Carcinogenic Risks to Humans. Overall Evaluations of Carcinogenicity: An Updating of IARC Monographs Volumes 1 to 42, Lyon, France, 10–18 March 1987.
- Nickson, R; Sengupta, C; Mitra, P; Dave, SN; Banerjee, AK; Bhattacharya, A; Basu, S; Kakoti, N; Moorthy, NS; Wasuja, M; Kumar, M; Mishra, DS; Ghosh, A; Vaish, DP; Srivastava, AK; Tripathi, RM; Singh, SN; Prasad, R; Bhattacharya, S; Deverill, P. Current knowledge on the distribution of arsenic in groundwater in five states of India. J Environ Sci Health A Tox Hazard Subst Environ Eng 2007, 42, 1707–1718. [Google Scholar]
- World Health Organization. Regional Office for Europe, Drinking-Water Quality: Guidelines for Selected Herbicides; World Health Organization, Regional Office for Europe: Copenhagen, Denmark, 1987; p. 23.
- Signes, A; Mitra, K; Burlo, F; Carbonell-Barrachina, AA. Effect of cooking method and rice type on arsenic concentration in cooked rice and the estimation of arsenic dietary intake in a rural village in West Bengal, India. Food Addit Contam Part A Chem Anal Control Expo Risk Assess 2008, 25, 1345–1352. [Google Scholar]
- Zhu, YG; Williams, PN; Meharg, AA. Exposure to inorganic arsenic from rice: a global health issue? Environ Pollut 2008, 154, 169–171. [Google Scholar]
- Masotti, A; Da Sacco, L; Bottazzo, GF; Sturchio, E. Risk assessment of inorganic arsenic pollution on human health. Environ Pollut 2009, 157, 1771–1772. [Google Scholar]
- Sen Gupta, B; Chatterjee, S; Rott, U; Kauffman, H; Bandopadhyay, A; DeGroot, W; Nag, NK; Carbonell-Barrachina, AA; Mukherjee, S. A simple chemical free arsenic removal method for community water supply--a case study from West Bengal, India. Environ Pollut 2009, 157, 3351–3353. [Google Scholar]
- Mohan, D; Pittman, CU, Jr. Arsenic removal from water/wastewater using adsorbents--A critical review. J Hazard Mater 2007, 142, 1–53. [Google Scholar]
- Lin, TF; Wu, JK. Adsorption of arsenite and arsenate within activated alumina grains: equilibrium and kinetics. Water Res 2001, 35, 2049–2057. [Google Scholar]
- Reed, BE; Vaughan, R; Jiang, L. As(III), As(V), Hg, and Pb Removal by Fe-Oxide Impregnated Activated Carbon. J Environ Eng 2000, 126, 869–873. [Google Scholar]
- Genc-Fuhrman, H; Tjell, JC; McConchie, D. Increasing the arsenate adsorption capacity of neutralized red mud (Bauxsol). J Colloid Interface Sci 2004, 271, 313–320. [Google Scholar]
- Gimenez, J; Martinez, M; de Pablo, J; Rovira, M; Duro, L. Arsenic sorption onto natural hematite, magnetite, and goethite. J Hazard Mater 2007, 141, 575–580. [Google Scholar]
- Thirunavukkarasu, OS; Viraraghavan, T; Subramanian, KS; Chaalal, O; Islam, MR. Arsenic Removal in Drinking Water—Impacts and Novel Removal Technologies. Energy Sources Part A 2005, 27, 209. [Google Scholar]
- Raven, KP; Jain, A; Loeppert, RH. Arsenite and Arsenate Adsorption on Ferrihydrite: Kinetics, Equilibrium, and Adsorption Envelopes. Environ Sci Technol 1998, 32, 344–349. [Google Scholar]
- Katsoyiannis, IA; Zouboulis, AI. Removal of arsenic from contaminated water sources by sorption onto iron-oxide-coated polymeric materials. Water Res 2002, 36, 5141–5155. [Google Scholar]
- Hasan, K; Halim, MA; Hossain, MK; Rana, MS; Waliul Hoque, SAM. Bioadsorbtion of Arsenic by Prepared and Commercial Crab Shell Chitosan. Biotechnology 2009, 8, 160–165. [Google Scholar]
- Yadanaparthi, SK; Graybill, D; von Wandruszka, R. Adsorbents for the removal of arsenic, cadmium, and lead from contaminated waters. J Hazard Mater 2009, 171, 1–15. [Google Scholar]
- Guibal, E. Interactions of metal ions with chitosan-based sorbents: A review. Sep Purif Technol 2004, 38, 43–74. [Google Scholar]
- Sun, B; Mi, Z; An, G; Liu, G; Zou, J. Preparation of Biomimetic Materials Made from Polyaspartyl Polymer and Chitosan for Heavy-Metal Removal. Ind Eng Chem Res 2009, 48, 9823–9829. [Google Scholar]
- Elson, CM; Davies, DH; Hayes, ER. Removal of arsenic from contaminated drinking water by a chitosan/chitin mixture. Water Res 1980, 14, 1307–1311. [Google Scholar]
- Boddu, VM; Abburi, K; Talbott, JL; Smith, ED; Haasch, R. Removal of arsenic (III) and arsenic (V) from aqueous medium using chitosan-coated biosorbent. Water Res 2008, 42, 633–642. [Google Scholar]
- Chen, CY; Chang, TH; Kuo, JT; Chen, YF; Chung, YC. Characteristics of molybdate-impregnated chitosan beads (MICB) in terms of arsenic removal from water and the application of a MICB-packed column to remove arsenic from wastewater. Bioresour Technol 2008, 99, 7487–7494. [Google Scholar]
- Kartal, SN; Imamura, Y. Removal of copper, chromium, and arsenic from CCA-treated wood onto chitin and chitosan. Bioresour Technol 2005, 96, 389–392. [Google Scholar]
- Aranaz, I; Mengíbar, M; Harris, R; Paños, I; Miralles, B; Acosta, N; Galed, G; Heras, Á. Functional Characterization of Chitin and Chitosan. Curr Chem Biol 2009, 3, 203–230. [Google Scholar]
- Mcafee, BJ; Gould, WD; Nadeau, JC; Costa, ACA. Biosorption of metal ions using chitosan, chitin and biomass of Rhizopus oryzae. Sep Sci Technol 2001, 36, 3207. [Google Scholar]
- Chen, GQ; Shi, XG; Tang, W; Xiong, SM; Zhu, J; Cai, X; Han, ZG; Ni, JH; Shi, GY; Jia, PM; Liu, MM; He, KL; Niu, C; Ma, J; Zhang, P; Zhang, TD; Paul, P; Naoe, T; Kitamura, K; Miller, W; Waxman, S; Wang, ZY; de The, H; Chen, SJ; Chen, Z. Use of arsenic trioxide (As2O3) in the treatment of acute promyelocytic leukemia (APL): I. As2O3 exerts dose-dependent dual effects on APL cells. Blood 1997, 89, 3345–3353. [Google Scholar]
- Zhang, P. The use of arsenic trioxide (As2O3) in the treatment of acute promyelocytic leukemia. J Biol Regul Homeost Agents 1999, 13, 195–200. [Google Scholar]
- Cui, X; Kobayashi, Y; Akashi, M; Okayasu, R. Metabolism and the paradoxical effects of arsenic: carcinogenesis and anticancer. Curr Med Chem 2008, 15, 2293–2304. [Google Scholar]
- Gochi, A; Orita, K; Fuchimoto, S; Tanaka, N; Ogawa, N. The prognostic advantage of preoperative intratumoral injection of OK-432 for gastric cancer patients. Br J Cancer 2001, 84, 443–451. [Google Scholar]
- Yu, J; Qian, H; Li, Y; Wang, Y; Zhang, X; Liang, X; Fu, M; Lin, C. Arsenic trioxide (As2O3) reduces the invasive and metastatic properties of cervical cancer cells in vitro and in vivo. Gynecol Oncol 2007, 106, 400–406. [Google Scholar]
- You-Jin, J; Se-Kwon, K. Antitumor activity of chitosan oligosaccharides produced in ultrafiltration membrane reactor system. J Microbiol Biotechnol 2002, 12, 503–507. [Google Scholar]
- Kim, S; Rajapakse, N. Enzymatic production and biological activities of chitosan oligosaccharides (COS): A review. Carbohydr Polym 2005, 62, 357–368. [Google Scholar]
- Gabizon, AA; Shmeeda, H; Zalipsky, S. Pros and cons of the liposome platform in cancer drug targeting. J Liposome Res 2006, 16, 175–183. [Google Scholar]
- Maeda, H; Bharate, GY; Daruwalla, J. Polymeric drugs for efficient tumor-targeted drug delivery based on EPR-effect. Eur J Pharm Biopharm 2009, 71, 409–419. [Google Scholar]
- Masotti, A; Bordi, F; Ortaggi, G; Marino, F; Palocci, C. A novel method to obtain chitosan/DNA nanospheres and a study of their release properties. Nanotechnology 2008, 19, 055302. [Google Scholar]
- Masotti, A; Ortaggi, G. Chitosan micro- and nanospheres: fabrication and applications for drug and DNA delivery. Mini Rev Med Chem 2009, 9, 463–469. [Google Scholar]
- Soane, RJ; Frier, M; Perkins, AC; Jones, NS; Davis, SS; Illum, L. Evaluation of the clearance characteristics of bioadhesive systems in humans. Int J Pharm 1999, 178, 55–65. [Google Scholar]
- Varma, AJ; Deshpande, SV; Kennedy, JF. Metal complexation by chitosan and its derivatives: a review. Carbohydr Polym 2004, 55, 77–93. [Google Scholar]
- Crini, G; Badot, P. Application of chitosan, a natural aminopolysaccharide, for dye removal from aqueous solutions by adsorption processes using batch studies: A review of recent literature. Prog Polym Sci 2008, 33, 399–447. [Google Scholar]
- Stefancich, S; Delben, F; Muzzarelli, RAA. Interaction of soluble chitosans with dyes in water. I. Optical evidence. Carbohydr Polym 1994, 24, 17–23. [Google Scholar]
- Delben, F; Gabrielli, P; Muzzarelli, RAA; Stefancich, S. Interaction of soluble chitosans with dyes in water. II. Thermodynamic data. Carbohydr Polym 1994, 24, 25–30. [Google Scholar]
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Da Sacco, L.; Masotti, A. Chitin and Chitosan as Multipurpose Natural Polymers for Groundwater Arsenic Removal and As2O3 Delivery in Tumor Therapy. Mar. Drugs 2010, 8, 1518-1525. https://doi.org/10.3390/md8051518
Da Sacco L, Masotti A. Chitin and Chitosan as Multipurpose Natural Polymers for Groundwater Arsenic Removal and As2O3 Delivery in Tumor Therapy. Marine Drugs. 2010; 8(5):1518-1525. https://doi.org/10.3390/md8051518
Chicago/Turabian StyleDa Sacco, Letizia, and Andrea Masotti. 2010. "Chitin and Chitosan as Multipurpose Natural Polymers for Groundwater Arsenic Removal and As2O3 Delivery in Tumor Therapy" Marine Drugs 8, no. 5: 1518-1525. https://doi.org/10.3390/md8051518