LC/MS Analysis of Tetrodotoxin and Its Deoxy Analogs in the Marine Puffer Fish Fugu niphobles from the Southern Coast of Korea, and in the Brackishwater Puffer Fishes Tetraodon nigroviridis and Tetraodon biocellatus from Southeast Asia
Abstract
:1. Introduction
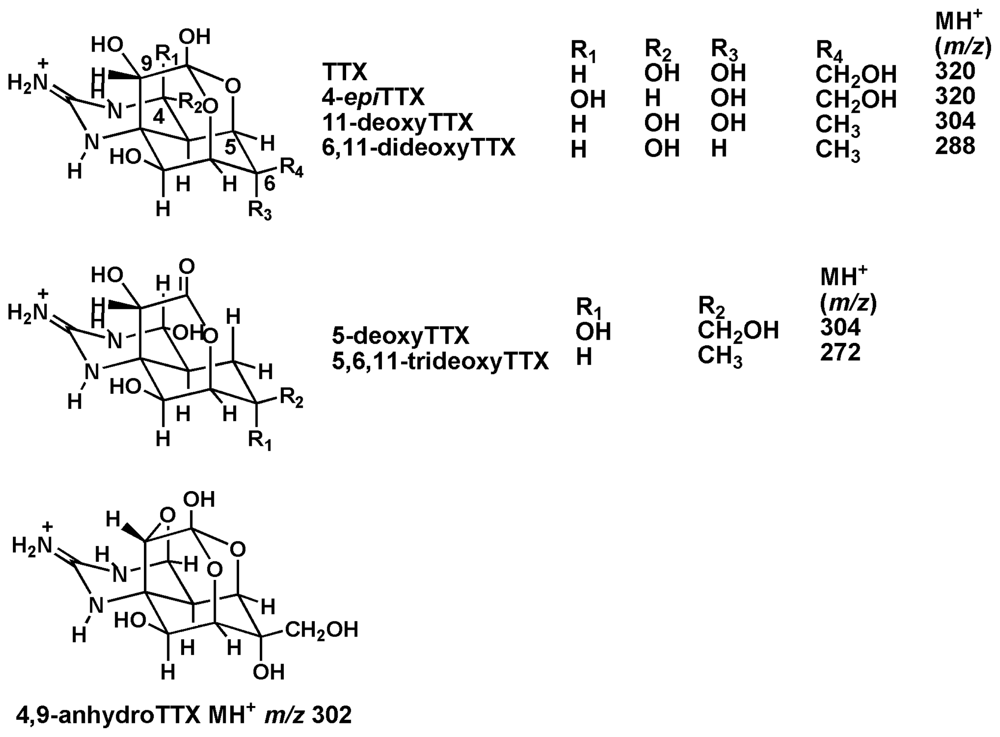
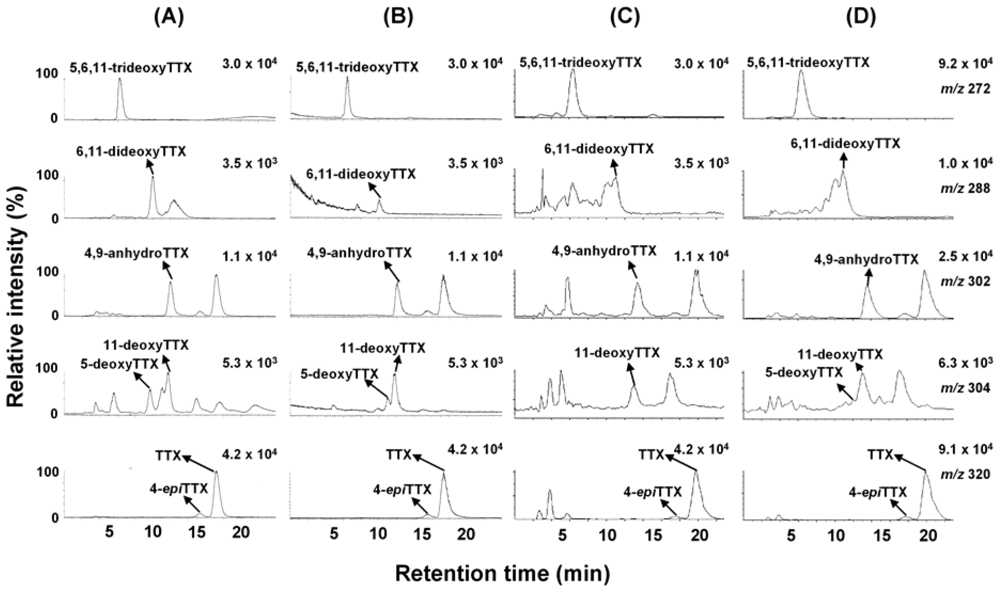
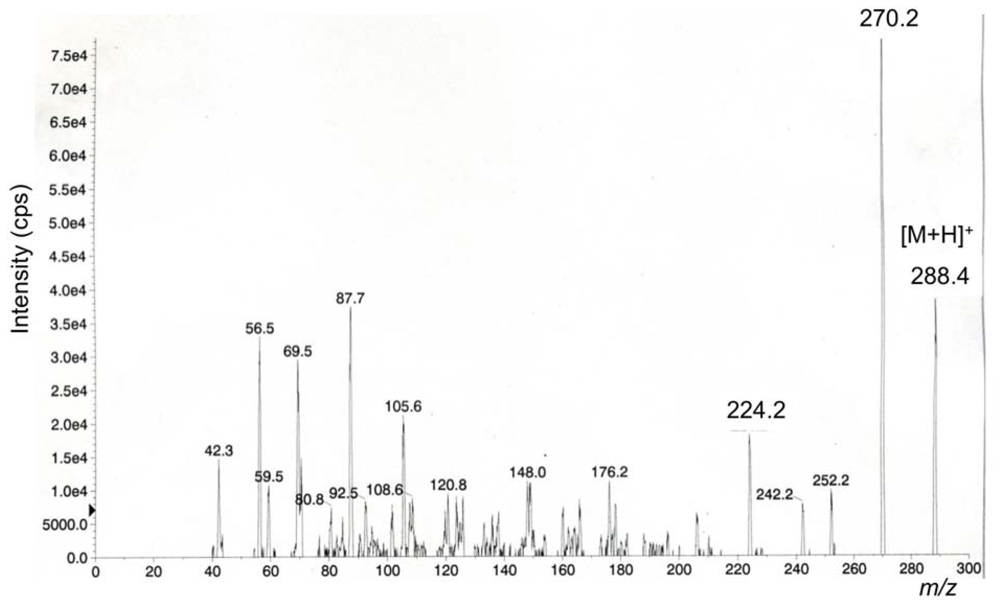
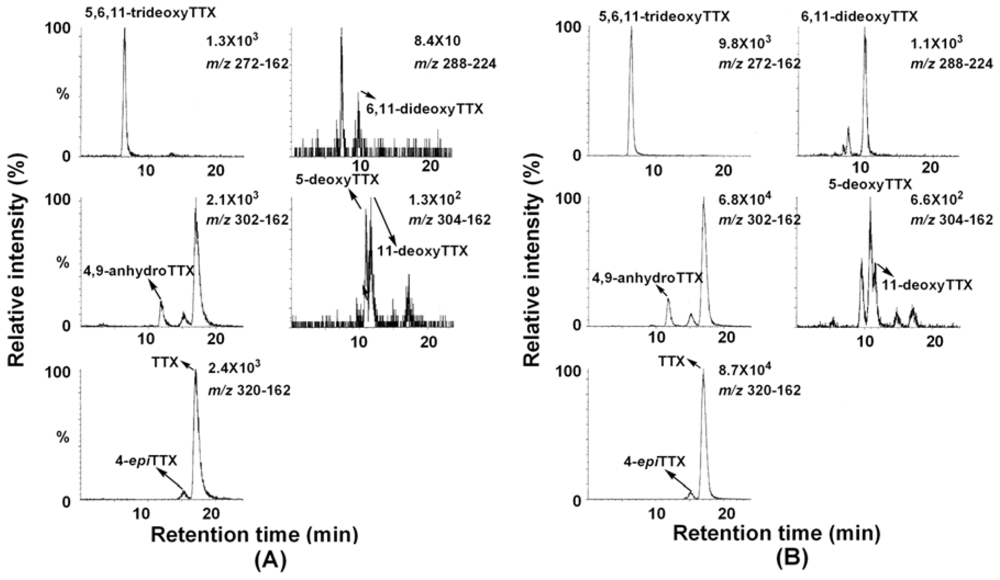
2. Results and Discussion
3. Experimental Section
3.1. Puffer fishes specimens
3.2. Preparation of sample solutions
3.3. Preparation of semi-purified TTXs mixture as the standard for LC/MS
3.4. LC/MS, MS/MS, and LC/MS/MS
4. Conclusions
Acknowledgements
References and Notes
- Goto, T; Kishi, Y; Takahashi, S; Hirata, Y. Tetrodotoxin. Tetrahedron 1965, 21, 2059–2088. [Google Scholar]
- Nakamura, M; Yasumoto, T. Tetrodotoxin derivatives in puffer fish. Toxicon 1985, 23, 271–276. [Google Scholar]
- Yotsu-Yamashita, M; Schimmele, B; Yasumoto, T. Isolation and structural assignment of 5-deoxytetrodotoxin from the puffer fish Fugu poecilonotus. Biosci Biotechnol Biochem 1999, 63, 961–963. [Google Scholar]
- Yasumoto, T; Yotsu, M; Murata, M; Naoki, H. New tetrodotoxin analogue from the newt Cynops ensicauda. J Am Chem Soc 1988, 110, 2344–2345. [Google Scholar]
- Jang, JH; Yotsu-Yamashita, M. 6,11-DideoxyTTX from the puffer fish, Fugu pardalis. Toxicon 2007, 50, 947–951. [Google Scholar]
- Yotsu-Yamashita, M; Yamagishi, Y; Yasumoto, T. 5,6,11-Trideoxytetrodotoxin from the puffer fish, Fugu poecilonotus. Tetrahedron Lett 1995, 36, 9329–9332. [Google Scholar]
- Kao, CY; Furman, F. A Pharmacological studies on tetrodotoxin, a potent neurotoxin. J Pharmacol Exp Ther 1963, 140, 31–40. [Google Scholar]
- Shoji, Y; Yotsu-Yamashita, M; Miyazawa, T; Yasumoto, T. Electrospray ionization mass spectrometry of tetrodotoxin and its analogs: liquid mass spectrometry, and liquid chromatography/tandem mass spectrometry. Anal Biochem 2001, 290, 10–17. [Google Scholar]
- Nakagawa, T; Jang, J; Yotsu-Yamashita, M. Hydrophilic interaction liquid chromatography-electrospray ionization mass spectrometry of tetrodotoxin and its analogs. Anal Biochem 2006, 352, 142–144. [Google Scholar]
- Jang, J; Yotsu-Yamashita, M. Distribution of tetrodotoxin, saxitoxin, and their analogs among tissues of the puffer fish Fugu pardalis. Toxicon 2006, 48, 980–987. [Google Scholar]
- Yasumoto, T; Michishita, T. Fluorometric determination of tetrodotoxin by high performance liquid chromatography. Agric Biol Chem 1985, 49, 3077–3080. [Google Scholar]
- Yotsu, M; Endo, A; Yasumoto, T. An improved tetrodotoxin analyzer. Agric Biol Chem 1989, 53, 895–898. [Google Scholar]
- Rodriguez, P; Alfonso, A; Vale, C; Alfonso, C; Vale, P; Tellez, A; Botana, LM. First toxicity report of tetrodotoxin and 5,6,11-trideoxyTTX in the trumpet shell Charonia lampas lampas in Europe. Anal Chem 2008, 80, 5622–5629. [Google Scholar]
- Yasumoto, T; Yasumura, D; Yotsu, M; Michishita, T; Endo, A; Kotaki, Y. Bacterial production of tetrodotoxin and anhydrotetrodotoxin. Agric Biol Chem 1986, 50, 793–795. [Google Scholar]
- Noguchi, T; Jeo, J; Arakawa, O; Sugita, H; Deguchi, Y; Shida, Y; Hashimoto, K. Occurrence of tetrodotoxin and anhydrotetrodotoxin in Vibrio sp. Isolated from the intestines of a xanthid crab, Atergatis floridus. J Biochem 1986, 99, 311–314. [Google Scholar]
- Yasumoto, T; Yotsu-Yamashita, M. Chemical and etiological studies on tetrodotoxin and its analogs. J Toxicol Toxin Rev 1996, 15, 81–90. [Google Scholar]
- Yotsu-Yamashita, M. Chemistry of puffer fish toxin. J Toxicol Toxin Rev 2001, 20, 51–66. [Google Scholar]
- Jang, JH; Yotsu-Yamashita, M. Tetrodotoxin and 5,6,11-trideoxytetrodotoxin in Tetraodon nigroviridis and T. biocellatus collected from Southeast Asia. Proceedings of the 5th World Fisheries Congress, Preliminary data of Tetraodon nigroviridis and Tetraodon biocellatus has been reported in the proceedings, Yokohama, Japan, 20–24 October 2008.
- Ryu, CH; Kim, DG; Kim, JH; Jang, JH; Lee, JS. Toxicity of the Grass puffer, Takifugu niphobles (Bogseom). J Korean Soc Food Sci Nutr 2003, 32, 986–990. (in Korean). [Google Scholar]
- Kono, M; Matsui, T; Furukawa, K; Yotsu-Yamashita, M; Yamamori, K. Accumulation of tetrodotoxin and 4,9-anhydrotetrodotoxin in cultured juvenile kusafugu Fugu niphobles by dietary administration of natural toxic komonfugu Fugu poecilonotus liver. Toxicon 2008, 51, 1269–1273. [Google Scholar]
- Ikeda, K; Emoto, Y; Tatsuno, R; Wang, JJ; Ngy, L; Taniyama, S; Takatani, T; Arakawa, O. Maturation-associated changes in toxicity of the pufferfish Takifugu poecilonotus. Toxicon 2010, 55, 289–297. [Google Scholar]
- Shin-Jung, L; Liao, CF; Arakawa, O; Noguchi, T; Hwang, DF. Toxicities of two freshwater puffers in Taiwan. J Nat Toxins 2002, 11, 103–110. [Google Scholar]
- Yamamori, K; Furukawa, K; Kono, M; Matsui, T. The toxification of juvenile cultured kusafugu Takifugu niphobles by oral administration of crystalline tetrodotoxin. J Food Hyg Soc Jpn 2004, 45, 73–75. (in Japanese). [Google Scholar]
- Kono, M; Matsui, T; Furukawa, K; Takase, T; Yamamori, K; Kaneda, H; Aoki, D; Jang, JH; Yotsu-Yamashita, M. Examination of transformation among tetrodotoxin and its analogs in the living cultured juvenile puffer fish, kusafugu, Fugu niphobles by intramuscular administration. Toxicon 2008, 52, 714–720. [Google Scholar]
| Tissue | Sex | Toxin content (nmol/g) | ||||||
|---|---|---|---|---|---|---|---|---|
| TTX | 4-epiTTX | 4,9-anhydro TTX | 5-deoxy TTX | 11-deoxy TTX | 6,11-dideoxy TTX | 5,6,11-trideoxy TTX | ||
| Ovary | F | 129–263 | 10–26 | 30–83 | 4–22 | 8–39 | 77–310 | 289–603 |
| Testicle | M | 1–11 | <0.5–1 | <0.5–3 | <0.5 | <0.5 | <0.5–9 | 1–13 |
| Liver | F | 4–24 | <0.5–6 | 5–39 | <0.5–3 | <0.5–1 | 1–24 | 3–16 |
| M | 9–31 | 1–3 | 9–15 | <0.5–2 | <0.5–1 | <0.5–59 | 10–18 | |
| Skin | F | 78–93 | 8–10 | 21–30 | 3–10 | 3–8 | 6–13 | 110–155 |
| M | 38–197 | 2–9 | 6–26 | 1–7 | 1–9 | 2–28 | 39–153 | |
| Muscle | F | 3–8 | <0.5–1 | <0.5–1 | <0.5 | <0.5 | 1–6 | 5–7 |
| M | 8–16 | 1–3 | <0.5–1 | <0.5 | <0.5 | <0.5–5 | <0.5–14 | |
| Intestine | F | 8–67 | 1–16 | 7–110 | 1–4 | <0.5–12 | 11–145 | 25–71 |
| M | 3–7 | <0.5–1 | 2–6 | <0.5 | <0.5 | 2–28 | 5–7 | |
| Species | Toxin content (nmol/g) | ||||||
|---|---|---|---|---|---|---|---|
| TTX | 4-epiTTX | 4,9-anhydro TTX | 5-deoxy TTX | 11-deoxy TTX | 6,11-dideoxy TTX | 5,6,11-trideoxy TTX | |
| T. nigroviridis | 0.6–294 | <0.5–5 | <0.5–47 | <0.5–7 | <0.5–7 | <0.5–8 | <0.5–151 |
| T. biocellatus | 27–190 | 2–16 | 4–41 | <0.5–2 | <0.5–17 | <0.5–18 | 21–202 |
| Precursor (m/z) | Product (m/z) | Retention time (min) | |
|---|---|---|---|
| TTX | 320 | 162 | 17.04 |
| 4-epiTTX | 320 | 162 | 15.15 |
| 4,9-anhydroTTX | 302 | 162 | 11.75 |
| 5-deoxyTTX | 304 | 162 | 10.82 |
| 11-deoxyTTX | 304 | 162 | 11.50 |
| 6,11-dideoxyTTX | 288 | 224 | 10.04 |
| 5,6,11-trideoxyTTX | 272 | 162 | 6.24 |
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Jang, J.-H.; Lee, J.-S.; Yotsu-Yamashita, M. LC/MS Analysis of Tetrodotoxin and Its Deoxy Analogs in the Marine Puffer Fish Fugu niphobles from the Southern Coast of Korea, and in the Brackishwater Puffer Fishes Tetraodon nigroviridis and Tetraodon biocellatus from Southeast Asia. Mar. Drugs 2010, 8, 1049-1058. https://doi.org/10.3390/md8041049
Jang J-H, Lee J-S, Yotsu-Yamashita M. LC/MS Analysis of Tetrodotoxin and Its Deoxy Analogs in the Marine Puffer Fish Fugu niphobles from the Southern Coast of Korea, and in the Brackishwater Puffer Fishes Tetraodon nigroviridis and Tetraodon biocellatus from Southeast Asia. Marine Drugs. 2010; 8(4):1049-1058. https://doi.org/10.3390/md8041049
Chicago/Turabian StyleJang, Jun-Ho, Jong-Soo Lee, and Mari Yotsu-Yamashita. 2010. "LC/MS Analysis of Tetrodotoxin and Its Deoxy Analogs in the Marine Puffer Fish Fugu niphobles from the Southern Coast of Korea, and in the Brackishwater Puffer Fishes Tetraodon nigroviridis and Tetraodon biocellatus from Southeast Asia" Marine Drugs 8, no. 4: 1049-1058. https://doi.org/10.3390/md8041049




