Violacein-Producing Collimonas sp. from the Sea Surface Microlayer of Costal Waters in Trøndelag, Norway
Abstract
:1. Introduction
2. Results and Discussion
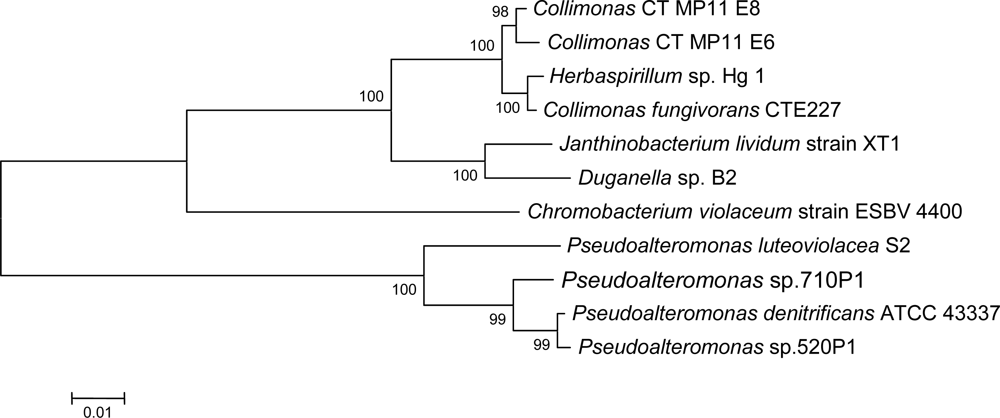
2.1. Isolation of Collimonas CT
2.2. Identification and characterization of antimicrobial compound and pigment
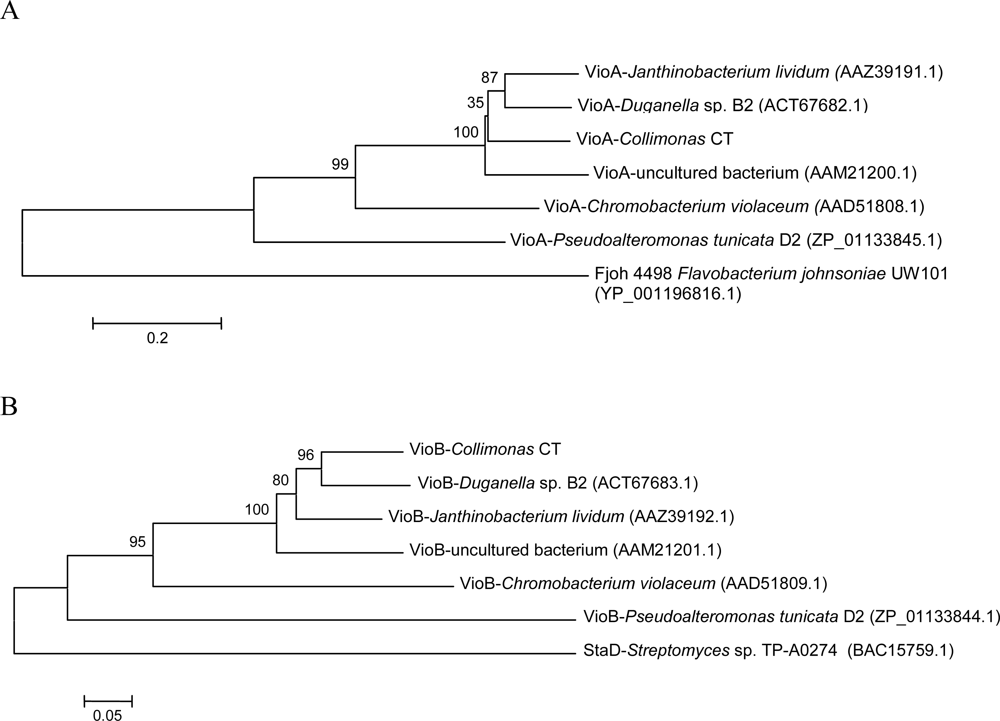
2.3. Violacein biosynthesis genes in Collimonas CT
3. Experimental Section
3.1. Sampling sites and sample collection
3.2. Preparation of bacterial inoculums
3.3. Culture condition for production and extraction of secondary metabolites
3.4. Antimicrobial assay
3.5. Fractionation and LC-MS-TOF analysis of bacterial extracts
3.6. Cloning, sequencing and phylogenetic analysis
Acknowledgments
- Samples Availability: Available from the authors.
References and Notes
- de Boer, W; Leveau, JHJ; Kowalchuk, GA; Klein Gunnewiek, PJA; Ablen, ECA; Figge, MJ; Sjollema, K; Janse, JD; van Veen, JA. Collimonas fungivorans gen. nov., sp. nov., a chitinolytic soil bacterium with the ability to grow on living fungal hyphae. Int J Sys Evol Microbiol 2004, 54, 857–864. [Google Scholar]
- DeMoss, RD; Evans, NR. Incorporation of C14-labeled substrates into violacein. J Bacteriol 1960, 79, 729–733. [Google Scholar]
- Momen, AZMR; Hoshino, T. Biosynthesis of Violacein: Intact incorporation of the tryptophan molecule on the oxindole side with intramolecular rearrangement of the indole ring on the 5-hydroxyindole side. Biosci Biotechnol Biochem 2000, 64, 539–549. [Google Scholar]
- DeMoss, RD; Evans, NR. Physiological aspects of violacein biosynthesis in nonproliferating cells. J Bacteriol 1959, 78, 583–586. [Google Scholar]
- Matz, C; Webb, J; Schupp, PJ; Phang, SY; Penesyan, A; Egan, S; Steinberg, P; Kjelleberg, S. Marine biofilm bacteria evade eukaryotic predation by targeted chemical defense. PLoS ONE 2008, 3(7), e2744. [Google Scholar]
- Antônio, RV; Creczynski-Pasa, TB. Genetic analysis of violacein biosynthesis by Chromobacterium violaceum. Genet Mol Res 2004, 3, 85–91. [Google Scholar]
- Rettori, D; Durán, N. Production, extraction and purification of violacein: an antibiotic pigment produced by Chromobacterium violaceum. World J Microbiol Biotechnol 1998, 14, 685–688. [Google Scholar]
- Pantanella, F; Berlutti, F; Passariello, C; Sarli, S; Morea, C; Schippa, S. Violacein and biofilm production in Janthinobacterium lividum. J Appl Microbiol 2007, 102, 992–999. [Google Scholar]
- Yada, S; Wang, Y; Zou, Y; Nagasaki, K; Hosokawa, K; Osaka, I; Arakawa, R; Enomoto, K. Isolation and characterization of two groups of novel marine bacteria producing violacein. Mar Biotechnol 2008, 10, 128–132. [Google Scholar]
- Wang, H-S; Jiang, P-X; Jiang, R-B; Xing, X-H. Reconstruction of violacein biosynthesis pathway from Duganella sp. B2. (GI:25455448). . Unpublished. Submitted to GenBank 27.07.09.
- August, PR; Grossman, TH; Minor, C; Draper, MP; MacNeil, LA; Pemberton, JM; Call, KM; Holt, D; Osburne, MS. Sequence analysis and functional characterization of the violacein biosynthetic pathway from Chromobacterium violaceum. J Mol Microbiol Biotechnol 2000, 2, 513–519. [Google Scholar]
- Brady, SF; Chao, CJ; Handelsman, J; Clardy, J. Cloning and heterologous expression of a natural product biosynthetic gene cluster from eDNA. Org Lett 2001, 3, 1981–1984. [Google Scholar]
- Sánchez, C; Braña, AF; Méndez, C; Salas, JA. Reevaluation of the violacein biosynthetic pathway and its relationship to indolocarbazole biosynthesis. ChemBioChem 2006, 7, 1231–1240. [Google Scholar]
- Matz, C; Deines, P; Boenigk, J; Arndt, H; Eberl, L; Kjelleberg, S; Jürgens, K. Impact of violacein-producing bacteria on survival and feeding of bacterivorous nanoflagellates. Appl Environ Microbiol 2004, 70, 1593–1599. [Google Scholar]
- Leon, LL; Miranda, CC; De Souza, AO; Duràn, N. Antileishmanial activity of the violacein extracted from Chromobacterium violaceum. J Antimicrob Chemother 2001, 48, 449–450. [Google Scholar]
- Ferreira, CV; Bos, CL; Versteeg, HH; Justo, GZ; Durán, N; Peppelenbosch, MP. Molecular mechanism of violacein-mediated human leukemia cell death. Blood 2004, 104, 1459–1467. [Google Scholar]
- Kodach, LL; Bos, CL; Durán, N; Peppelenbosch, MP; Ferreira, CV; Hardwick, JCH. Violacein synergistically increases 5-fluorouracil cytotoxity, induces apoptosis and inhibits Akt-mediated signal transduction in human colorectal cancer cells. Carcinogenesis 2006, 27, 508–516. [Google Scholar]
- Andrighetti-Fröhner, CR; Antonio, RV; Creczynski-Pasa, TV; Barardi, CRM; Simões, CMO. Cytotoxicity and potential antiviral evaluation of violacein produced by Chromobacterium violaceum. Mem Inst Oswaldo Cruz 2003, 98, 843–848. [Google Scholar]
- Lichstein, HC; van De Sand, VF. The antibiotic activity of violacein, prodigiosin, and phthiocol. J Bacteriol 1946, 52, 145–146. [Google Scholar]
- Nakamura, Y; Asada, C; Sawada, T. Production of antibacterial violet pigment by psychrotropic bacterium RT102 Strain. Biotechnol Bioprocess Eng 2003, 8, 37–40. [Google Scholar]
- Konzen, M; de Marco, D; Cordova, CAS; Vieira, TO; Antônio, RV; Creczynski-Pasa, TB. Antioxidant properties of violacein: Possible relation on its biological function. Bioorg Med Chem 2006, 14, 8307–8313. [Google Scholar]
- Andrighetti-Fröhner, CR; Kratz, JM; Antonio, RV; Creczynski-Pasa, TB; Barardi, CRM; Simões, CMO. In vitro testing for genotoxicity of violacein assessed by Comet and Micronucleus assays. Mutation Res 2006, 603, 97–103. [Google Scholar]
- Richard, C. Chromobacterium violaceum, opportunist pathogenic bacteria in tropical and subtropical regions. Bull Soc Pathol Axot 1993, 86, 169–173. [Google Scholar]
- Höppener-Ogawa, S; de Boer, W; Leveau, JHJ; van Veen, JA; de Brandt, E; Vanlaere, E; Sutton, H; Dare, DJ; Vandamme, P. Collimonas arenae sp. nov. and Collimonas pratensis sp. nov., isolated from (semi-) natural grassland soils. Int J Syst Evol Microbiol 2008, 58, 414–419. [Google Scholar]
- Schmidt, M; Priemé, A; Stougaard, P. Bacterial diversity in permanently cold and alkaline ikaite columns from Greenland. Extremophiles 2006, 10, 551–562. [Google Scholar]
- Männistö, MK; Häggbloma, MM. Characterization of psychrotolerant heterotrophic bacteria from Finnish Lapland. System Appl Microbiol 2006, 29, 229–243. [Google Scholar]
- Shivaji, S; Ray, MK; Seshu Kumar, G; Reddy, GSN; Saisree, L; Wynn-Williams, DD. Identification of Janthinobacterium lividum from the soils of the islands of Scotia Ridge and from Antarctic Peninsula. Polar Biol 1991, 11, 267–271. [Google Scholar]
- Fantinatti-Garboggini, F; de Almeida, R; Portillo, VdA; Barbosa, TAP; Trevilato, PB; Neto, CER; Coêlho, RD; Silva, DW; Bartoleti, LA; Hanna, ES; Brocchi, M; Manfio, GP. Drug resistance in Chromobacterium violaceum. Genet Mol Res 2004, 3, 134–147. [Google Scholar]
- de Azevedo, MBM; Alderete, J; Rodriguez, JA; Souza, AO; Rettori, D; Torsoni, MA; Faljoni-Alario, A; Haun, M; Durán, N. Biological activities of violacein, a new antitumoral indole derivative, in an inclusion complex with β-cyclodextrin. J Incl Phenom Macro 2000, 37, 93–101. [Google Scholar]
- Yang, LH; Xiong, H; Lee, OO; Qi, S-H; Qian, P-Y. Effect of agitation on violacein production in Pseudoalteromonas luteoviolacea isolated from a marine sponge. Lett Appl Microbiol 2007, 44, 625–630. [Google Scholar]
- Corpe, WA. Variation in pigmentation and morphology of colonies of gelatinous strains of Chromobacterium species from soil. J Bacteriol 1953, 66, 470–477. [Google Scholar]
- Sivendra, R; Lo, HS. Identification of Chromobacterium violaceum: pigmented and non-pigmented strains. J Gen Microbiol 1975, 90, 21–31. [Google Scholar]
- Sneath, PHA. Genus Janthinobacterium De Ley, Seegers and Gillis 1978, 164AL. In Bergey’s Manual of Systematic Bacteriology; Krieg, NR, Holt, JG, Eds.; The Williams & Wilkins Co: Baltimore, MD, USA, 1984; Volume 1, pp. 376–377. [Google Scholar]
- Lincoln, SP; Fermor, TR; Tindall, BJ. Janthinobacterium agaricidamnosum sp. nov., a soft rot pathogen of Agaricus bisporus. Int J Syst Bacteriol 1999, 49, 1577–1589. [Google Scholar]
- Hakvåg, S; Fjærvik, E; Josefsen, KD; Ian, E; Ellingsen, TE; Zotchev, SB. Characterization of Streptomyces spp. isolated from the sea surface microlayer in the Trondheim fjord, Norway. Mar Drugs 2008, 6, 620–635. [Google Scholar]
- Jørgensen, H; Fjærvik, E; Hakvåg, S; Bruheim, P; Bredholt, H; Klinkenberg, G; Ellingsen, TE; Zotchev, SB. Candicidin biosynthetic gene cluster is widely distributed among Streptomyces spp. isolated from the sediments and the neuston layer in the Trondheim fjord, Norway. Appl Environ Microbiol 2009, 75, 3296–303. [Google Scholar]
- Lane, DJ. 16S/23S rRNA sequencing. In Nucleic Acid Techniques in Bacterial Systematic; Stackebrandt, E, Goodfellow, M, Eds.; John Wiley & Sons Ltd: Chichester, UK, 1991; pp. 115–175. [Google Scholar]
- Tamura, K; Dudley, J; Nei, M; Kumar, S. MEGA4: Molecular Evolutionary Genetics Analysis (MEGA) software version 4.0. Mol Biol Evol 2007, 24, 1596–1599. [Google Scholar]
| Strain or plasmid | Description | Source or reference |
|---|---|---|
| Candida albicans | Cyhr AmBs, Hps, Nyss † | ATCC• (strain 10231) |
| Micrococcus luteus | Amoxs, Amps, Cms, Ccls, Nbs, Oles, Pens, Pcns, Rifs, Tys £ | ATCC• (strain 9341) |
| E. coli K12 | ||
| Enterococcus faecium | Ampr, Ctcr, Eryr, Lcmr, Vcmr, Amr, Bacr, Csr, Spr § | CCUG* (strain 37832) |
| Enterococcus faecium Collimonas CT | Amr, Bacr, Csr, Spr | CTC (strain 492) This study |
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hakvåg, S.; Fjærvik, E.; Klinkenberg, G.; Borgos, S.E.F.; Josefsen, K.D.; Ellingsen, T.E.; Zotchev, S.B. Violacein-Producing Collimonas sp. from the Sea Surface Microlayer of Costal Waters in Trøndelag, Norway. Mar. Drugs 2009, 7, 576-588. https://doi.org/10.3390/md7040576
Hakvåg S, Fjærvik E, Klinkenberg G, Borgos SEF, Josefsen KD, Ellingsen TE, Zotchev SB. Violacein-Producing Collimonas sp. from the Sea Surface Microlayer of Costal Waters in Trøndelag, Norway. Marine Drugs. 2009; 7(4):576-588. https://doi.org/10.3390/md7040576
Chicago/Turabian StyleHakvåg, Sigrid, Espen Fjærvik, Geir Klinkenberg, Sven Even F. Borgos, Kjell D. Josefsen, Trond E. Ellingsen, and Sergey B. Zotchev. 2009. "Violacein-Producing Collimonas sp. from the Sea Surface Microlayer of Costal Waters in Trøndelag, Norway" Marine Drugs 7, no. 4: 576-588. https://doi.org/10.3390/md7040576
APA StyleHakvåg, S., Fjærvik, E., Klinkenberg, G., Borgos, S. E. F., Josefsen, K. D., Ellingsen, T. E., & Zotchev, S. B. (2009). Violacein-Producing Collimonas sp. from the Sea Surface Microlayer of Costal Waters in Trøndelag, Norway. Marine Drugs, 7(4), 576-588. https://doi.org/10.3390/md7040576




