Parguerol and Isoparguerol Isolated from the Sea Hare, Aplysia kurodai, Induce Neurite Outgrowth in PC-12 Cells
Abstract
:Introduction
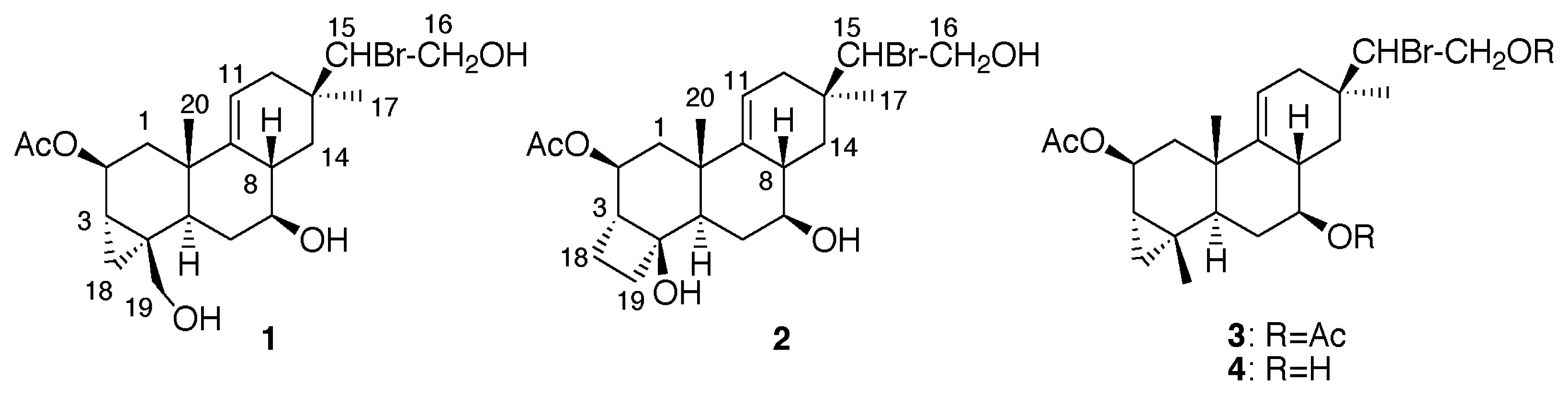
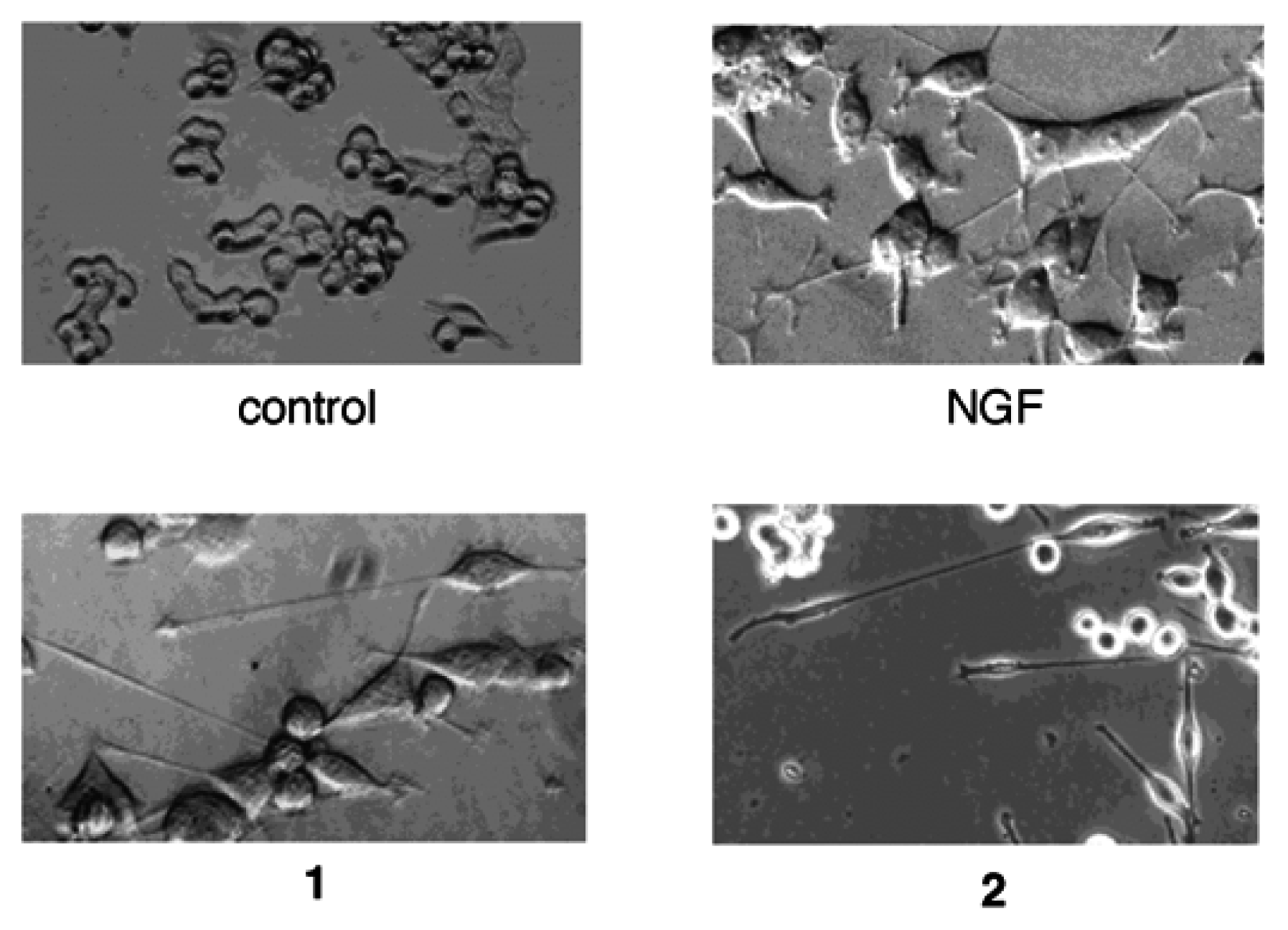
Results and Discussion
Conclusions
Experimental
General
Extraction and Isolation
Neurite Outgrowth Assay
Acknowledgments
- Samples Availability: Not available.
References and Notes
- Levi-Montalcini, R. The Nerve Growth Factor 35 Years Later. Science 1987, 237, 1154–1162. [Google Scholar]
- Barde, Y. A.; Edgar, D.; Thoenen, H. Purification of a New Neurotrophic Factor from Mammalian Brain. EMBO J 1982, 1, 549–553. [Google Scholar]
- Maisonpierre, P. C.; Belluscio, L.; Squinto, S.; Ip, N. Y.; Furth, M. E.; Lindsay, R. M.; Yancopoulos, G. D. Neurotrophin-3: a Neurotrophic Factor Related to NGF and BDNF. Science 1990, 247, 1446–1451. [Google Scholar]
- Lin, L.-F. H.; Doherty, D. H.; Lile, J. D.; Bektesh, S.; Collins, F. GDNF: A Glial Cell Line-Derived Neurotrophic Factor for Midbrain Dopaminergic Neurons. Science 1993, 260, 1130–1132. [Google Scholar]
- Greene, L. A.; Tischler, A. S. Establishment of a Noradrenergic Clonal Line of Rat Adrenal Pheochromocytoma Cells Which Respond to Nerve Growth Factor. Proc. Natl. Acad. Sci. USA 1976, 73, 2424–2428. [Google Scholar]
- Tsukamoto, S.; Macabalang, A. D.; Nakatani, K.; Obara, Y.; Nakahata, N.; Ohta, T. Tricholomalides A-C: New Neutrophic Diterpenes from the Mushroom Tricholoma sp. J. Nat. Prod 2003, 66, 1578–1581. [Google Scholar]
- Tsukamoto, S.; Miura, S.; Yamashita, Y.; Ohta, T. Aspermytin A: a New Neurotrophic Polyketide Isolated from a Marine-derived Fungus of the Genus Aspergillus. Bioorg. Med. Chem. Lett 2004, 14, 417–420. [Google Scholar]
- Schmitz, F. J.; Michaud, D. P.; Schmidt, P. G. Marine Natural Products: Parguerol, Deoxyparguerol, and Isoparguerol. New Brominated Diterpenes with Modified Pimarane Skeletons from the Sea Hare Aplysia dactylomela. J. Am. Chem. Soc 1982, 104, 6415–6423. [Google Scholar]
- Fenteany, G.; Schreiber, S. L. Specific Inhibition of the Chymotrypsin-like Activity of the Proteasome Induces a Bipolar Morphology in Neuroblastoma Cells. Chem. Biol 1996, 3, 905–912. [Google Scholar]
- Erickson, K. L. Constituents of Laurencia. In Marine Natural Products; Scheuer, P. J., Ed.; Academic: New York, 1983; Volume V, p. 132. [Google Scholar]
- Stallard, M. O.; Faulkner, D. J. Chemical Constituents of the Digestive Gland of the Sea Hare Aplysia californica. I. Importance of Diet. Comp. Biochem. Physiol. B 1974, 49B, 25–35. [Google Scholar]
- Stallard, M. O.; Faulkner, D. J. Chemical Constituents of the Digestive Gland of the Sea Hare Aplysia californica. II. Chemical Transformations. Comp. Biochem. Physiol. B 1974, 49B, 37–41. [Google Scholar]
- Ireland, C.; Stallard, M. O.; Faulkner, D. J. Some Chemical Constituents of the Digestive Gland of the Sea Hare Aplysia californica. J. Org. Chem 1976, 41, 2461–2465. [Google Scholar]
- Yamada, K.; Ojika, M.; Ishigaki, T.; Yoshida, Y.; Ekimoto, H.; Arakawa, M. Aplyronine A, a Potent Antitumor Substance, and the Congeners Aplyronines B and C Isolated from the Sea Hare Aplysia kurodai. J. Am. Chem. Soc 1993, 115, 11020–11021. [Google Scholar]
- Wessels, M.; Konig, G. M.; Wright, A. D. New Natural Product Isolation and Comparison of the Secondary Metabolite Content of Three Distinct Samples of the Sea Hare Aplysia dactylomela from Tenefife. J. Nat. Prod 2000, 63, 920–928. [Google Scholar]
- Ichiba, T.; Higa, T. New Cuparene-derived Sesquiterpenes with Unprecedented Oxygenation Patterns from the Sea Hare Aplysia dactylomela. J. Org. Chem 1986, 51, 3364–3366. [Google Scholar]
© 2004 by MDPI Reproduction is permitted for noncommercial purposes.
Share and Cite
Tsukamoto, S.; Yamashita, Y.; Yoshida, T.; Ohta, T. Parguerol and Isoparguerol Isolated from the Sea Hare, Aplysia kurodai, Induce Neurite Outgrowth in PC-12 Cells. Mar. Drugs 2004, 2, 170-175. https://doi.org/10.3390/md204170
Tsukamoto S, Yamashita Y, Yoshida T, Ohta T. Parguerol and Isoparguerol Isolated from the Sea Hare, Aplysia kurodai, Induce Neurite Outgrowth in PC-12 Cells. Marine Drugs. 2004; 2(4):170-175. https://doi.org/10.3390/md204170
Chicago/Turabian StyleTsukamoto, Sachiko, Yuko Yamashita, Takushi Yoshida, and Tomihisa Ohta. 2004. "Parguerol and Isoparguerol Isolated from the Sea Hare, Aplysia kurodai, Induce Neurite Outgrowth in PC-12 Cells" Marine Drugs 2, no. 4: 170-175. https://doi.org/10.3390/md204170



