Sporiolides A and B, New Cytotoxic Twelve-Membered Macrolides from a Marine-Derived Fungus Cladosporium Species
Abstract
:Introduction

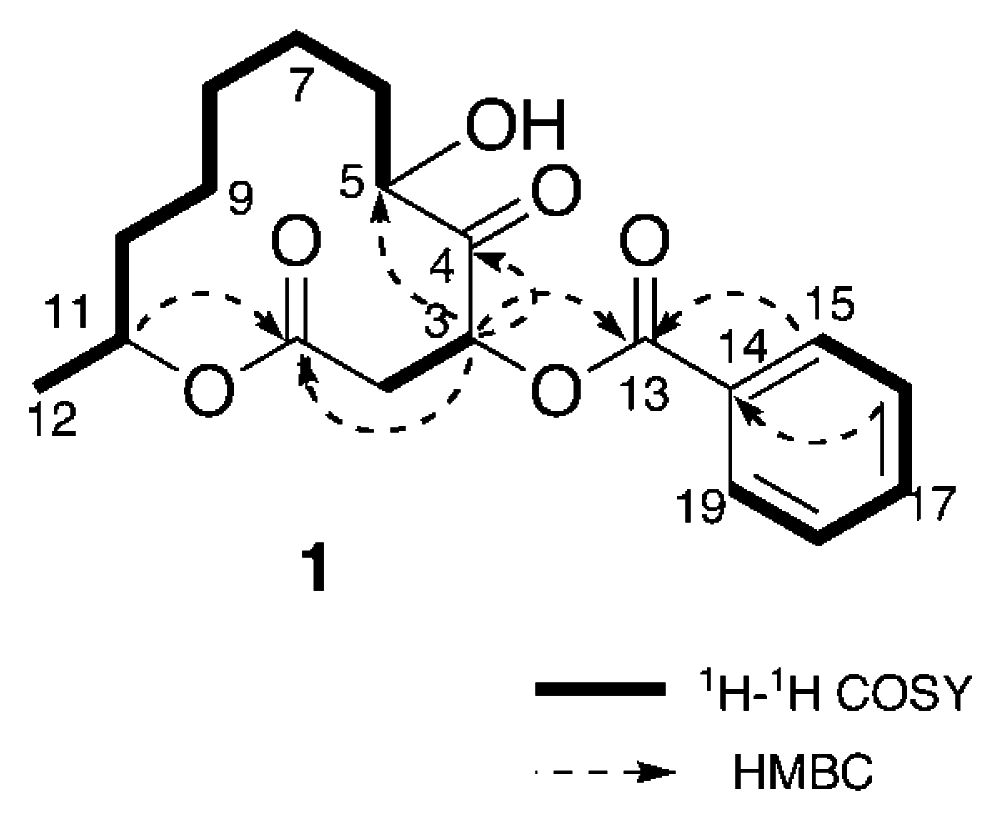
Results and Discussion
Experimental
General
Fungal Material and Fermentation
Extraction and Separation
Spectral Data
| 1 | 2 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| position | δHa | δCb | δHa | δCb | ||||||
| 1 | 168.8 | s | 171.5 | s | ||||||
| 2 | 3.52 | dd | 18.0, 9.8 | 40.5 | t | 3.30 | m | 42.2 | t | |
| 2.95 | d | 18.0 | 2.66 | m | ||||||
| 3 | 5.90 | d | 9.8 | 67.4 | d | 4.46 | dd | 9.0,2.0 | 74.1 | d |
| 4 | 207.1 | s | 207.8 | s | ||||||
| 5 | 4.40 | m | 76.0 | d | 4.36 | dd | 6.1,1.8 | 75.8 | d | |
| 6 | 2.02 | m | 30.5 | t | 1.99 | m | 30.5 | t | ||
| 1.77 | m | 1.69 | m | |||||||
| 7 | 1.34 | m | 19.0 | t | 1.47 | m | 22.8 | t | ||
| 1.17 | m | 1.05 | m | |||||||
| 8 | 1.50 | m | 26.6 | t | 1.37a | m | 26.6 | t | ||
| 1.12 | m | |||||||||
| 9 | 1.27 | m | 22.6 | t | 1.50 | m | 23.5 | t | ||
| 1.21 | m | 1.42 | m | |||||||
| 10 | 1.67 | m | 33.4 | t | 1.59 | m | 33.6 | t | ||
| 1.40 | m | 1.32 | m | |||||||
| 11 | 4.89 | m | 74.4 | d | 4.89 | m | 73.6 | d | ||
| 12 | 1.46b | d | 5.3 | 20.8 | q | 1.22b | d | 6.5 | 21.0 | q |
| 13 | 165.5 | s | 3.45b | s | 58.2 | q | ||||
| 14 | 129.2 | s | ||||||||
| 15, 19 | 8.05 | m | 129.9 | d | ||||||
| 16,18 | 7.43 | m | 128.4 | d | ||||||
| 17 | 7.56 | m | 133.5 | d | ||||||
| Test organisms | MIC (μg/ml) | |
|---|---|---|
| 1 | 2 | |
| Micrococcus luteus | 16.7 | 16.7 |
| Bacillus subtilis | >33.3 | >33.3 |
| Escherichia coli | >33.3 | >33.3 |
| Candida albicans | 16.7 | >33.3 |
| Cryptococcus neoformans | 8.4 | >33.3 |
| Paecilomyces variotii | >33.3 | >33.3 |
| Aspergillus niger | 16.7 | >33.3 |
| Neurospora crassa | 8.4 | >33.3 |
Acknowledgments
- Sample Availability: Samples are available from the authors.
Reference and Notes
- Blunt, J. W.; Copp, B. R.; Munro, M. H. G.; Northcote, P. T.; Prinsep, P. R. Nat. Prod. Rep 2004, 21, 1–49, and references cited therein.
- Tsuda, M.; Mugishima, T. K.; Komatsu, T.; Sone, T.; Tanaka, M.; Mikami, Y.; Shiro, M.; Hirai, M.; Ohizumi, Y.; Kobayashi, J. Tetrahedron 2003, 59, 3227–3230, and references cited therein.
- Zhang, H.; Tomoda, H.; Tabata, N.; Miura, H.; Namikoshi, M.; Yamaguchi, Y.; Masuma, R.; Omura, S. J. Antibiot 2001, 54, 635–641.
- Hirota, A.; Sakai, H.; Isogai, A. Agric. Biol. Chem 1985, 43, 731–735.
- Hirota, A.; Sakai, H.; Isogai, A.; Kitano, Y.; Ashida, T.; Hirota, H.; Takahashi, T. Agric. Biol. Chem 1985, 49, 903–904.
- Hirota, H.; Hirota, A.; Sakai, H.; Isogai, A.; Takahashi, T. Bull. Chem. Soc. Jpn 1985, 58, 2147–2148.
- Smith, C. J.; Abbanat, D.; Bernan, V. S.; Maiese, W. M.; Greenstein, M.; Jompa, J.; Tahir, A.; Ireland, C. M. J. Nat. Prod 2000, 63, 142–145.
- Since sporiolides A (1) and B (2) were unstable and were not obtained from the extracts of recultivation, stereochemistry of 1 and 2 was not determined.
- Jadulco, R.; Proksch, P.; Wray, V.; Sudarsono Berg, A.; Gräfe, U. J. Nat. Prod 2001, 64, 527–530.
© 2004 by MDPI Reproduction is permitted for noncommercial purposes.
Share and Cite
Shigemori, H.; Kasai, Y.; Komatsu, K.; Tsuda, M.; Mikami, Y.; Kobayashi, J. Sporiolides A and B, New Cytotoxic Twelve-Membered Macrolides from a Marine-Derived Fungus Cladosporium Species. Mar. Drugs 2004, 2, 164-169. https://doi.org/10.3390/md204164
Shigemori H, Kasai Y, Komatsu K, Tsuda M, Mikami Y, Kobayashi J. Sporiolides A and B, New Cytotoxic Twelve-Membered Macrolides from a Marine-Derived Fungus Cladosporium Species. Marine Drugs. 2004; 2(4):164-169. https://doi.org/10.3390/md204164
Chicago/Turabian StyleShigemori, Hideyuki, Yuu Kasai, Kazusei Komatsu, Masashi Tsuda, Yuzuru Mikami, and Jun’ichi Kobayashi. 2004. "Sporiolides A and B, New Cytotoxic Twelve-Membered Macrolides from a Marine-Derived Fungus Cladosporium Species" Marine Drugs 2, no. 4: 164-169. https://doi.org/10.3390/md204164




