Identification of Renieramycin A as an Antileishmanial Substance in a Marine Sponge Neopetrosia sp.
Abstract
:Introduction
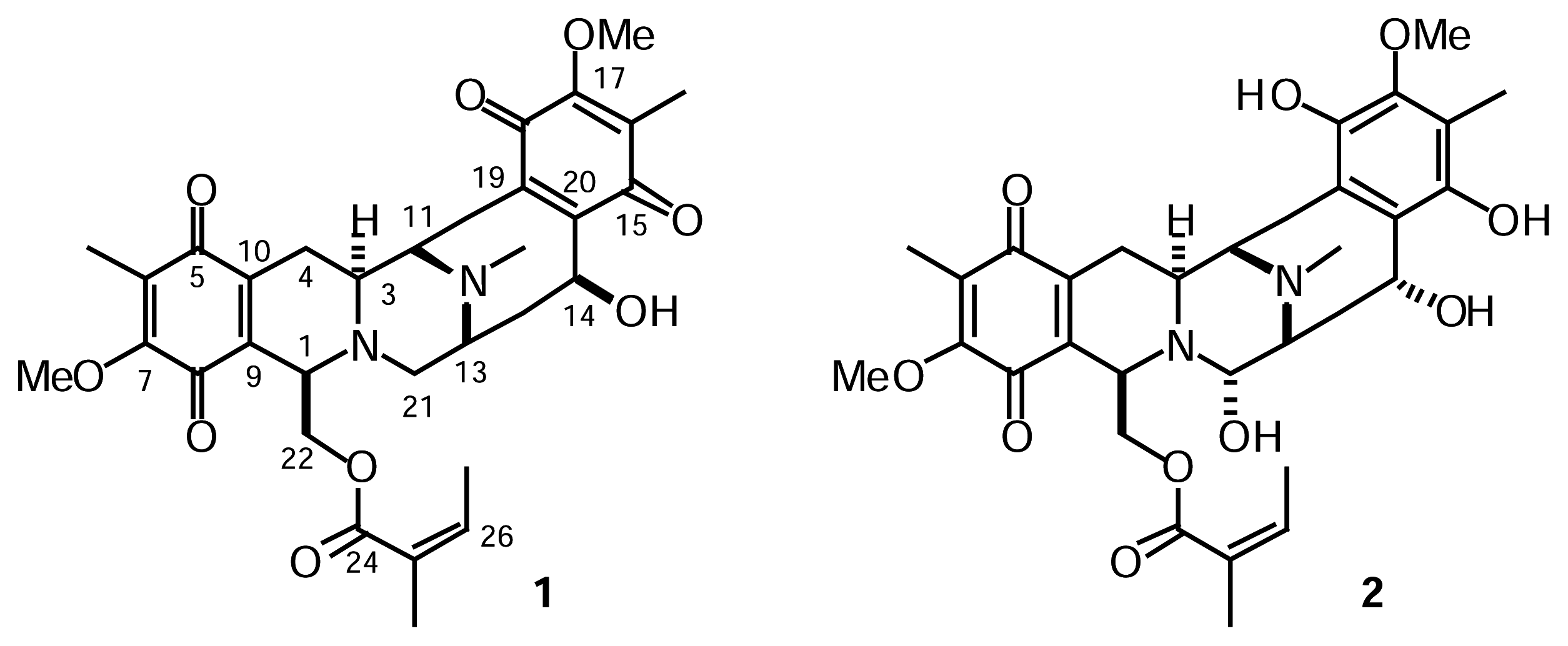
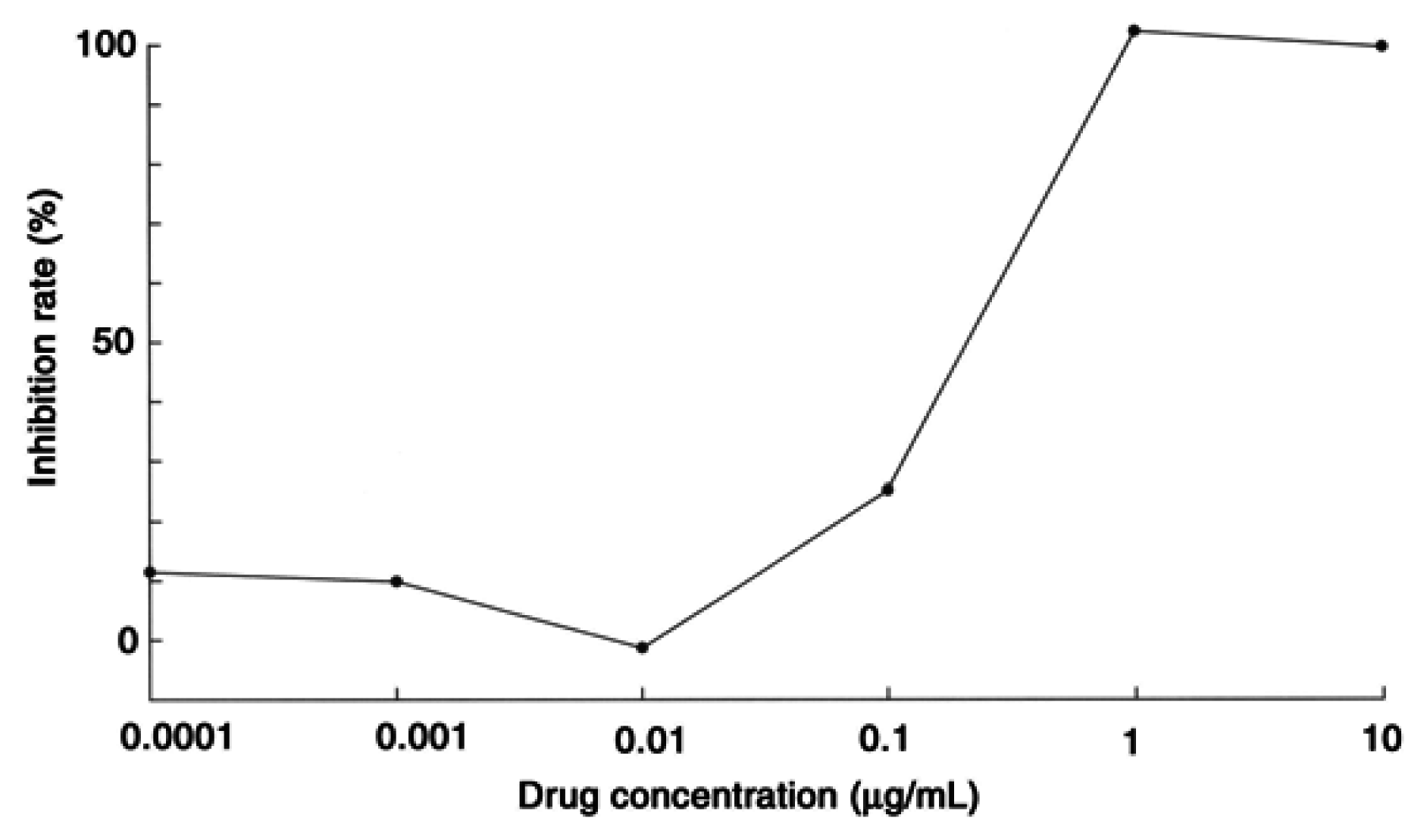
Results and Discussion
Conclusions
Experimental
General
Animal material
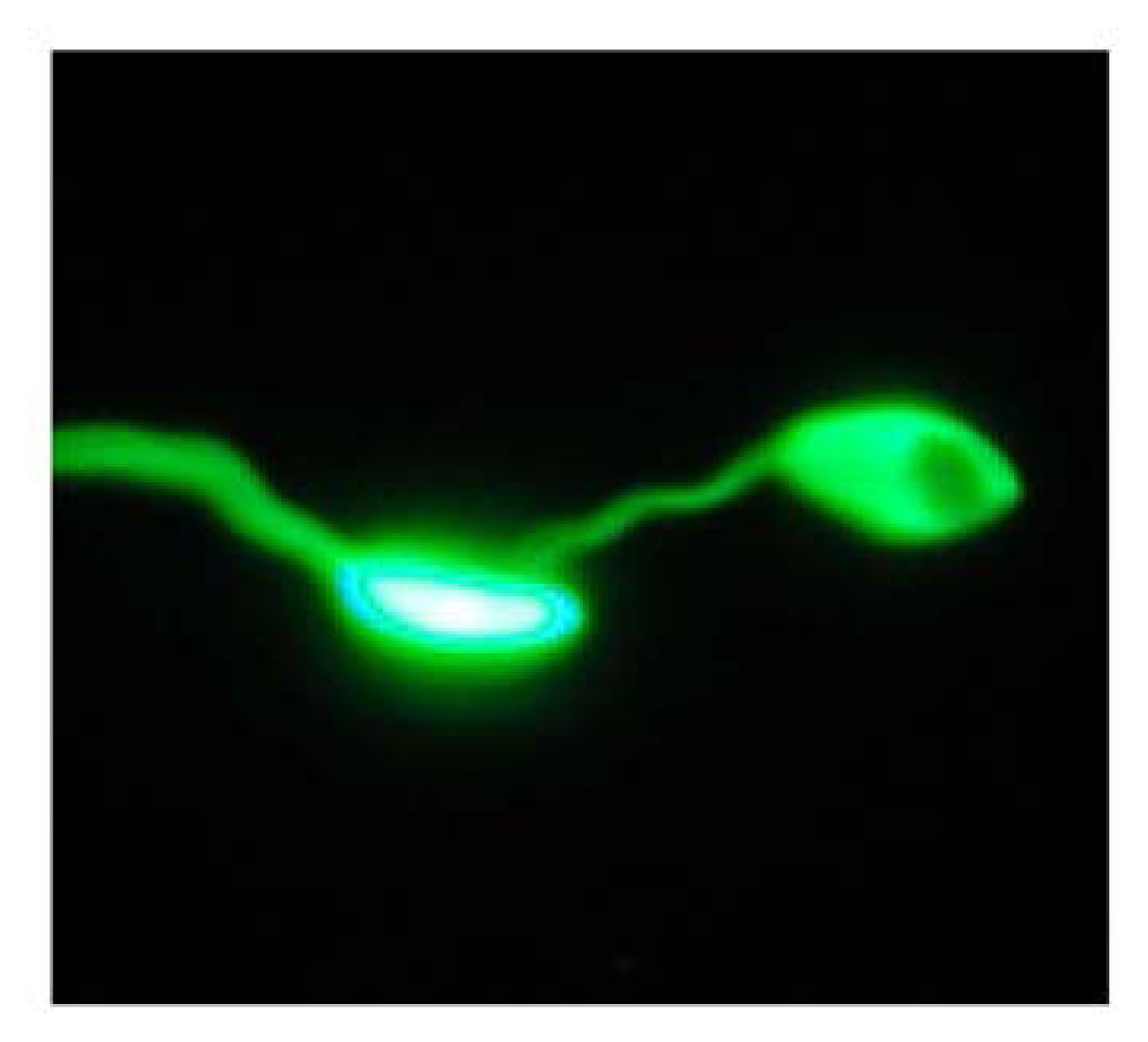
Antileishmanial assay
Isolation
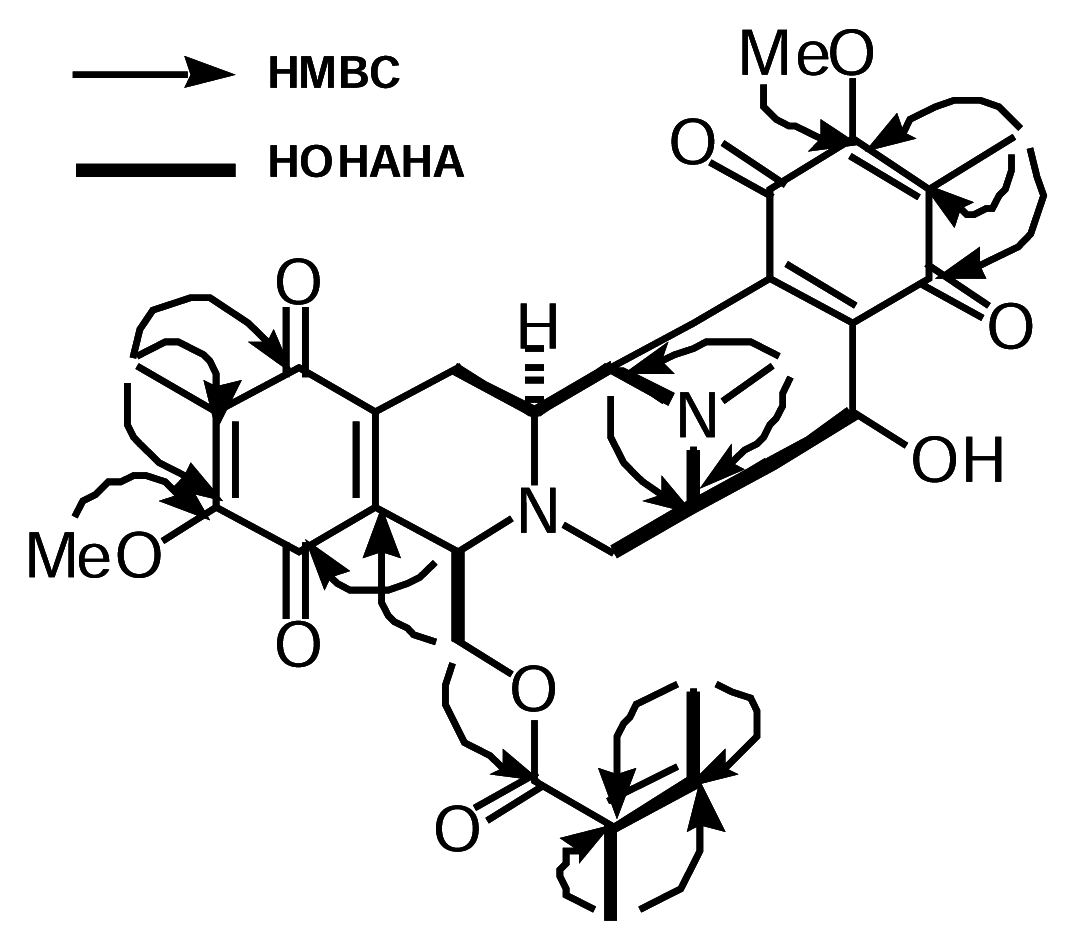
| #C | δCa | δHa | HMBC | δHb | δHb,c |
|---|---|---|---|---|---|
| 1 | 60.0 | 3.60 | C-8 | 3.62 | 3.60 |
| 3 | d | 2.65 | 2.64 | 2.64 | |
| 4 | d | 2.63 | 2.75 | 2.75 | |
| 1.2 | 1.26 | 1.26 | |||
| 5 | 187.1 | ||||
| 6 | 128.8 | ||||
| 7 | 157.7 | ||||
| 8 | 190.5 | ||||
| 9 | 144.0 | ||||
| 10 | d | ||||
| 11 | 56.9 | 4.04 | C-13 | 4.04 | 4.04 |
| 13 | 62.6 | 3.14 | 3.18 | 3.18 | |
| 14 | 71.5 | 3.62 | 4.43 | 4.44 | |
| 15 | 188.0 | ||||
| 16 | 130.2 | ||||
| 17 | 156.9 | ||||
| 18 | d | ||||
| 19 | d | ||||
| 20 | d | ||||
| 21 | 43.0 | 3.2 | 3.18 | 3.18 | |
| 2.7 | 2.71 | 2.71 | |||
| 22 | 64.0 | 4.45 | C-9 | 4.48 | 4.47 |
| 4.27 | C-24 | 4.19 | 4.19 | ||
| 24 | 168.9 | ||||
| 25 | 128.3 | ||||
| 26 | 140.2 | 5.94 | 5.92 | 5.92 | |
| 6-Me | 8.6 | 1.85 s | C-5, 6, 7 | 1.93 | 1.91 |
| 7-OMe | 61.2 | 3.95 s | C-7 | 4.00 | 4.00 |
| 12-NMe | 42.5 | 2.46 s | C-11, 13 | 2.43 | 2.43 |
| 16-Me | 8.3 | 1.91 s | C-15, 16, 17 | 1.93 | 1.92 |
| 17-OMe | 61.2 | 3.92 s | C-17 | 4.01 | 4.01 |
| 25-Me | 20.8 | 1.53 s | C-25, 26 | 1.57 | 1.55 |
| 26-Me | 15.8 | 1.72 d | C-25, 26 | 1.80 | 1.78 |
Acknowledgments
- Samples Availability: Not available.
References and Notes
- Herwaldt, B. L. Leishmaniasis. Lancet 1999, 354, 1191–1199. [Google Scholar]
- Aldina, B.; Diana, P. S.; Gabriel, G., Jr.; Hooman, M.; Diane, M. P.; Amelia, R. J.; Roque, A.; Robert, B.; Manoel, B. N.; Edgar, M. C.; Warren, D. J., Jr. Leishmaniasis in Bahia, Brazil: Evidence that Leishmania amazonensis Produces a Wide Spectrum of Clinical Disease. Am. J. Trop. Med. Hyg 1991, 44, 536–546. [Google Scholar]
- Barral, A.; Badaro, R.; Barral, N. M.; Grimaldi, G., Jr.; Momem, H.; Carvalho, E. M. Isolation of Leishmania mexicana amazonensis from the Bone Marrow in a Case of American Visceral Leishmaniasis. Am. J. Trop. Med. Hyg 1986, 35, 732–734. [Google Scholar]
- Grimaldi, G., Jr.; Tesh, R. B.; McMahon, P. D. A Review of the Geographic Distribution and Epidemiology of Leishmaniasis in the New World. Am. J. Trop. Med. Hyg 1989, 41, 687–725. [Google Scholar]
- Ashford, R. W.; Desjeux, P.; De Raadt, P. Estimation of Population at Risk of Infection and Number of Cases of Leishmaniasis. Parasitol. Today, Sect. B 1992, 8, 104–105. [Google Scholar]
- Balana-Fouce, R.; Reguera, R. M.; Cubria, J. C.; Ordonez, D. Review: The Pharmacology of Leishmaniasis. Gen. Pharmacol 1998, 30, 435–443. [Google Scholar]
- Jackson, J. E.; Tally, J. D.; Tang, D. B. An In Vitro Micromethod for Drug Sensitivity Testing of Leishmania. Am. J. Trop. Med. Hyg 1989, 41, 318–330. [Google Scholar]
- Brajtburg, J.; Bolard, J. Carrier Effects on Biological Activity of Amphotericin B. Clin. Microbiol. Rev 1996, 9, 512–531. [Google Scholar]
- Okuno, T.; Goto, Y.; Matsumoto, Y.; Otsuka, H.; Matsumoto, Y. Applications of Recombinant Leishmania amazonensis Expressing egfp or the β-Galactosidase Gene for Drug Screening and Histopathological Analysis. Exp. Anim 2003, 52, 109–118. [Google Scholar]
- Oku, N.; Matsunaga, S.; van Soest, R. W. M.; Fusetani, N. Renieramycin J, a Highly Cytotoxic Tetrahydroisoquinoline Alkaloid, from a marine Sponge Neopetrosia sp. J. Nat. Prod 2003, 66, 1136–1139. [Google Scholar]
- Renieramycin J isolated by Oku et al was renamed as renieramycin P. For renieramycin J, see ref [12].
- Suwanborirux, K.; Amnuoypol, S.; Plubrukarn, A.; Pummangura, S.; Kubo, A.; Tanaka, C.; Saito, N. Chemistry of Renieramycins. Part 3. Isolation and Structure of Stabilized Renieramycin Type Derivatives Possessing Antitumor Activity from Thai Sponge Xestospongia species, Pretreated with Potassium Cyanide. J. Nat. Prod 2003, 66, 1441. [Google Scholar]
- Kupchan, S. M.; Britton, R. W.; Ziegler, M. F.; Sigel, C. W. Tumor Inhibitors 82. Bruceantin, a New Potent Antileukemic Simaroubolide from Brucea antidysenterica. J. Org. Chem 1973, 38, 178–179. [Google Scholar]
- Frincke, J. M.; Faulkner, D. J. Antimicrobial Metabolites of the Sponge Reniera sp. J. Am. Chem. Soc 1982, 104, 265–269. [Google Scholar]
- Bax, A.; Summers, M. F. Proton and Carbon-13 Assignments from Sensitivity-enhanced Detection of Hheteronuclear Multiple-bond Connectivity by 2D Multiple Quantum NMR. J. Am. Chem. Soc 1986, 108, 2093–2094. [Google Scholar]
- Bax, A.; Aszalos, A.; Dinya, Z.; Sudo, K. Structure Elucidation of the Antibiotic Desertomycin through the Use of New Two-dimensional NMR Techniques. J. Am. Chem. Soc 1986, 108, 8056–8063. [Google Scholar]
- Compagnone, R. S.; Pina, I. C.; Rangel, H. R.; Dagger, F.; Suarez, A. I.; Reddy, M. V. R.; Faulkner, D. J. Antileishmanial Cyclic Peroxides from the Palauan Sponge Plakortis aff. angulospiculatus. Tetrahedron 1998, 54, 3057–3068. [Google Scholar]
- Copp, B. R.; Kayser, O.; Brun, R.; Kiderlen, A. F. Antiparasitic Activity of Marine Pyridoacridone alkaloids related to the ascididemnins. Planta Medica 2003, 69, 527–531. [Google Scholar]
- Rao, K. V.; Santarsiero, B. D.; Mesecar, A. D.; Schinazi, R. F.; Tekwani, B. L.; Hamann, M. T. New Manzamine Alkaloids with Activity against Infectious and Tropical Parasitic Diseases from an Indonesian Sponge. J. Nat. Prod 2003, 66, 823–828. [Google Scholar]
© 2004 by MDPI Reproduction is permitted for noncommercial purposes.
Share and Cite
Nakao, Y.; Shiroiwa, T.; Murayama, S.; Matsunaga, S.; Goto, Y.; Matsumoto, Y.; Fusetani, N. Identification of Renieramycin A as an Antileishmanial Substance in a Marine Sponge Neopetrosia sp. Mar. Drugs 2004, 2, 55-62. https://doi.org/10.3390/md202055
Nakao Y, Shiroiwa T, Murayama S, Matsunaga S, Goto Y, Matsumoto Y, Fusetani N. Identification of Renieramycin A as an Antileishmanial Substance in a Marine Sponge Neopetrosia sp. Marine Drugs. 2004; 2(2):55-62. https://doi.org/10.3390/md202055
Chicago/Turabian StyleNakao, Yoichi, Takeru Shiroiwa, Shuhei Murayama, Shigeki Matsunaga, Yasuyuki Goto, Yoshitsugu Matsumoto, and Nobuhiro Fusetani. 2004. "Identification of Renieramycin A as an Antileishmanial Substance in a Marine Sponge Neopetrosia sp." Marine Drugs 2, no. 2: 55-62. https://doi.org/10.3390/md202055




