Antifungal Activity of (+)-Curcuphenol, a Metabolite from the Marine Sponge Didiscus oxeata
Abstract
:Introduction
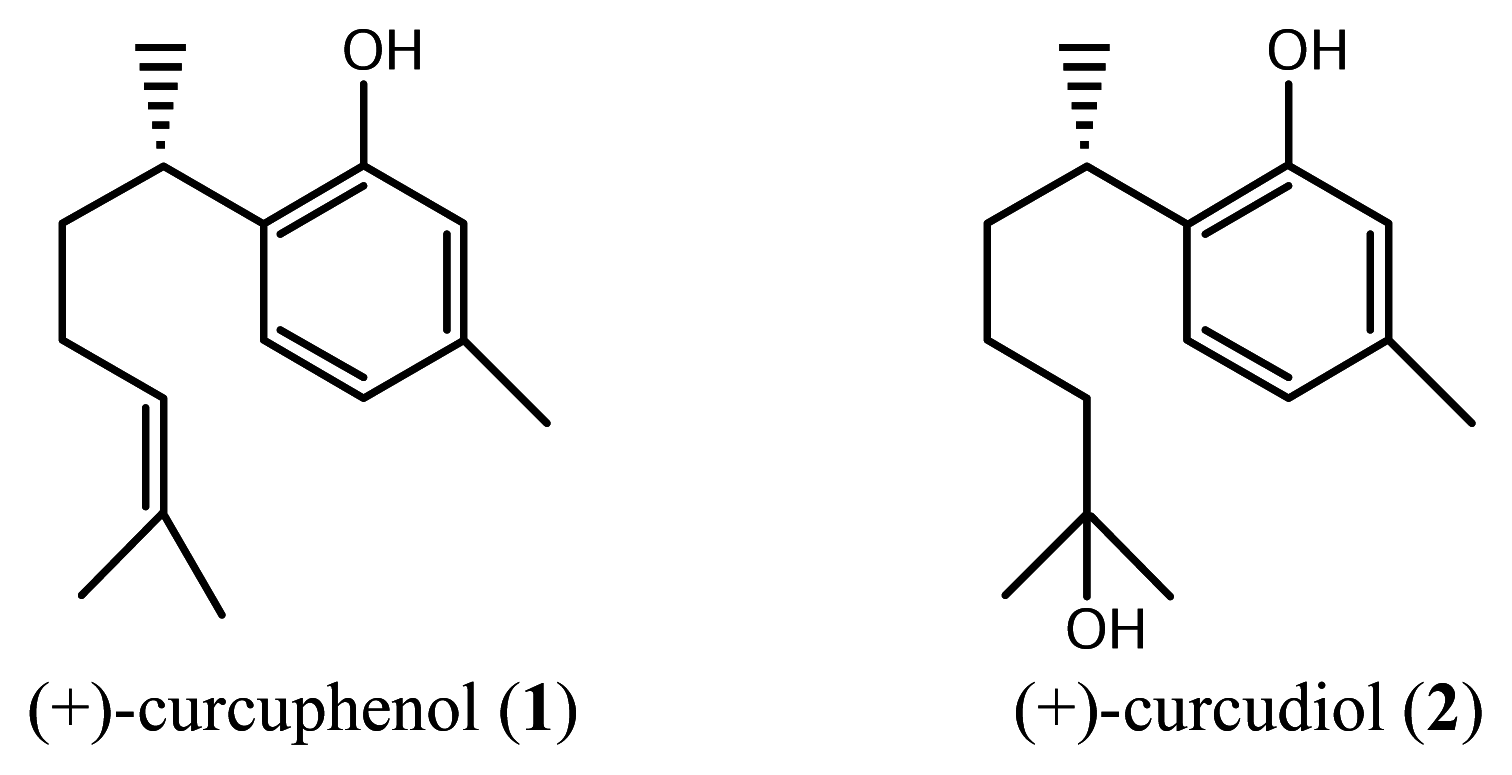
Results and Discussion
Conclusion
Experimental
Biological material
Isolation of sesquiterpenes
Biological activity
| Organism | (+)-Curcuphenol | Carbendazim® | Amphotericin B |
|---|---|---|---|
| Filamentous Fungi | |||
| Absidia ramosa | 4.5 cm | 6.0 cm | - |
| Aspergillus niger | 1.0 cm | 3.5 cm | - |
| Botrytis cinera | ± | 2.0 cm | - |
| Cladosporium cucumerinum | 2.0 cm | 3.7 cm | - |
| Fusarium oxysporum | ± | 2.0 cm | - |
| Penicillium expansum | ± | 4.0 cm | - |
| Rhizopus oryzae | 1.6 cm | 3.5 cm | - |
| Trichoderma harzianum | ± | 4.0 cm | - |
| Trichoderma koningii | 1.0 cm | 4.2 cm | - |
| Trichophyton mentagrophytes | 1.0 cm | - | 5.0 cm |
Acknowledgements
- Sample Availability: Samples are available from the authors.
References
- Blunt, J. W.; Copp, B. R.; Munro, M. H. G.; Northcote, P. T.; Prinsep, M. R. Marine Natural Products. Nat. Prod. Rep 2003, 20, 1–48 a, nd previous reviews by Faulkner.. [Google Scholar]
- Wright, A. M.; Pomponi, S. A.; McConnell, O. J.; Kohmoto, S.; McCarthy. (+)-Curcuphenol and (+)-Curcudiol, Sesquiterpene Phenols from Shallow and Deep Water Collections of the Marine Sponge Didiscus oxeata. J. Nat. Prod 1987, 50, 976–978. [Google Scholar]
- Braekman, J. C.; Daloze, D.; Moussiaux, B.; Stoller, C.; Deneubourg, F. Sponge Secondary Metabolites: New Results. Pure & Appl. Chem 1989, 61, 509–512. [Google Scholar]
- Duque, C.; Zea, S.; De Silvestri; Calderon, A.; Medina, A. Biological Activity vs. Chemical Composition of the Chloroform Extract of the Marine Sponge Didiscus oxeata. Rev. Colomb. Quim 1988, 17, 39–46, CA: 113, 208508y.. [Google Scholar]
- Butler, M. S.; Capon, R. J. Aromatic Bisabolenes from an Australian Marine Sponge, Arenochalina sp. J Nat Prod 1991, 54, 619–623. [Google Scholar]
- Tsukamoto, S.; Kato, H.; Hirota, H.; Fusetani, N. Antifouling Terpenes and Steroids Against Barnacle Larvae from Marine Sponges. Biofouling 1997, 11, 283–291. [Google Scholar]
- Rodríguez, D. A.; Vera, B. New Diterpenes from the Caribbean Sponge Epipolasis reiswigi. J. Org. Chem 2001, 66, 6364–6368. [Google Scholar]
- Fusetani, N.; Sugano, M.; Matsunaga, S.; Hashimoto, K. (+)-Curcuphenol and Dehydrocurcuphenol, Novel Sesquiterpenes wich Inhibit H, K-ATPase, from Sponge Epipolasis sp. Experentia 1987, 43, 1234–1235. [Google Scholar]
- Podile, A.; Prakash, A. Lysis and Biological Control of Aspergillus niger by Bacillus subtilis AF 1. Can. J. Microbiol 1996, 42, 533–538. [Google Scholar]
- Gadhi, C.; Benharref, A.; Jana, M.; Basile, A.; Contet-Audonneau, N.; Fortier, B. Antidermatophytic Properties of Extracts from the Leaves of Aristolochia paucinervis Pomel. Phytother. Res 2001, 15, 79–81. [Google Scholar]
- Ono, M.; Yamamoto, Y.; Akita, H. Reactions of Methyl 4,5-Epoxy-(2E)-pentenoate with Arenes. II. Application to the Synthesis of (±)-Curcudiol, (±)-Curcuphenol, (±)-Curcuhydroquinone, and (±)-Curcuquinone. Chem. Pharm. Bull 1995, 43, 553–558. [Google Scholar]
- Ono, M.; Ogura, Y.; Hatogai, K.; Akita, H. Total Synthesis of (S)-(+)-Curcudiol, and (S)-(+) and (R)-(−)-Curcuphenol. Chem. Pharm. Bull 2001, 49, 1581–1585. [Google Scholar]
- Amade, P.; Pesando, D.; Chevolot, L. Antimicrobial Activities of Marine Sponges from French Polynesia and Brittany. Marine Biology 1982, 70, 223–228. [Google Scholar]
© 2004 by MDPI Reproduction is permitted for noncommercial purposes.
Share and Cite
Gaspar, H.; Feio, S.S.; Rodrigues, A.I.; Van Soest, R. Antifungal Activity of (+)-Curcuphenol, a Metabolite from the Marine Sponge Didiscus oxeata. Mar. Drugs 2004, 2, 8-13. https://doi.org/10.3390/md201008
Gaspar H, Feio SS, Rodrigues AI, Van Soest R. Antifungal Activity of (+)-Curcuphenol, a Metabolite from the Marine Sponge Didiscus oxeata. Marine Drugs. 2004; 2(1):8-13. https://doi.org/10.3390/md201008
Chicago/Turabian StyleGaspar, Helena, Sonia Savluchinske Feio, Ana Isabel Rodrigues, and Rob Van Soest. 2004. "Antifungal Activity of (+)-Curcuphenol, a Metabolite from the Marine Sponge Didiscus oxeata" Marine Drugs 2, no. 1: 8-13. https://doi.org/10.3390/md201008




