Bioactive Bromotyrosine-Derived Alkaloids from the Polynesian Sponge Suberea ianthelliformis
Abstract
1. Introduction
2. Results and Discussion
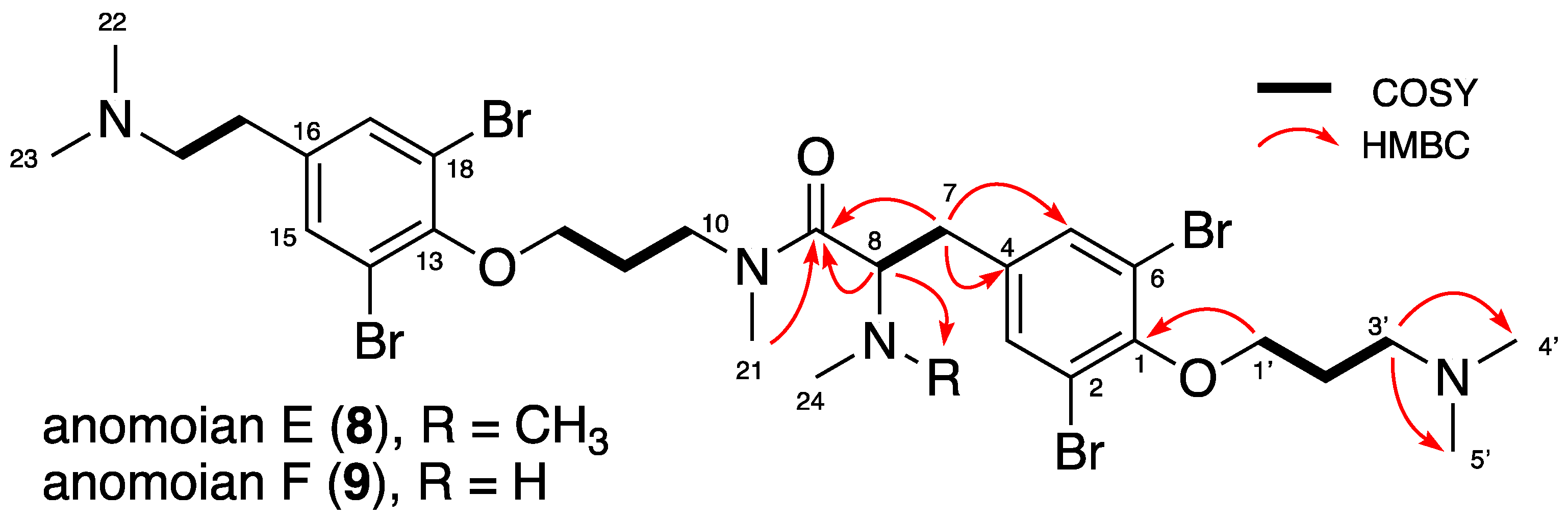
2.1. Isolation and Structure Elucidation
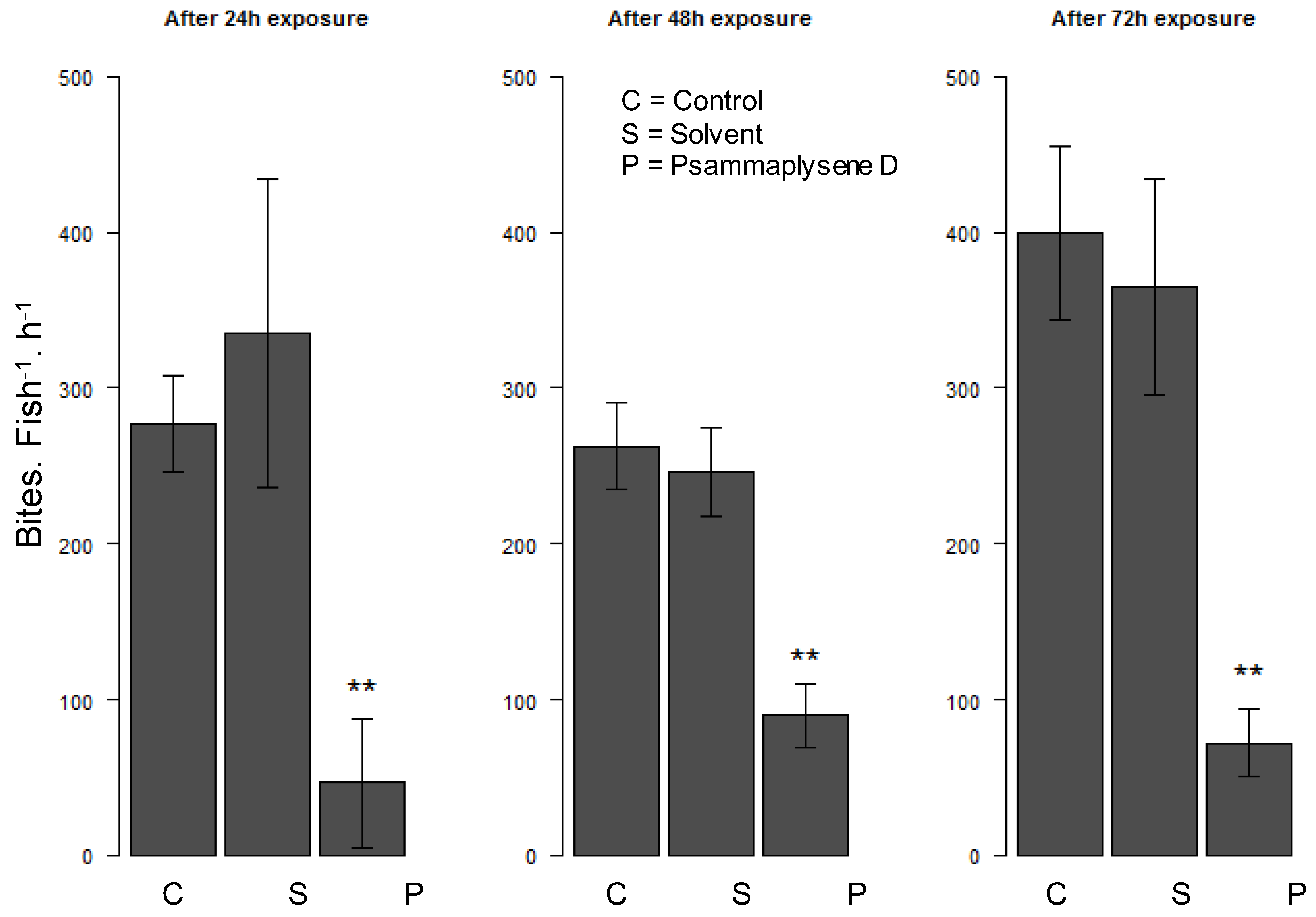
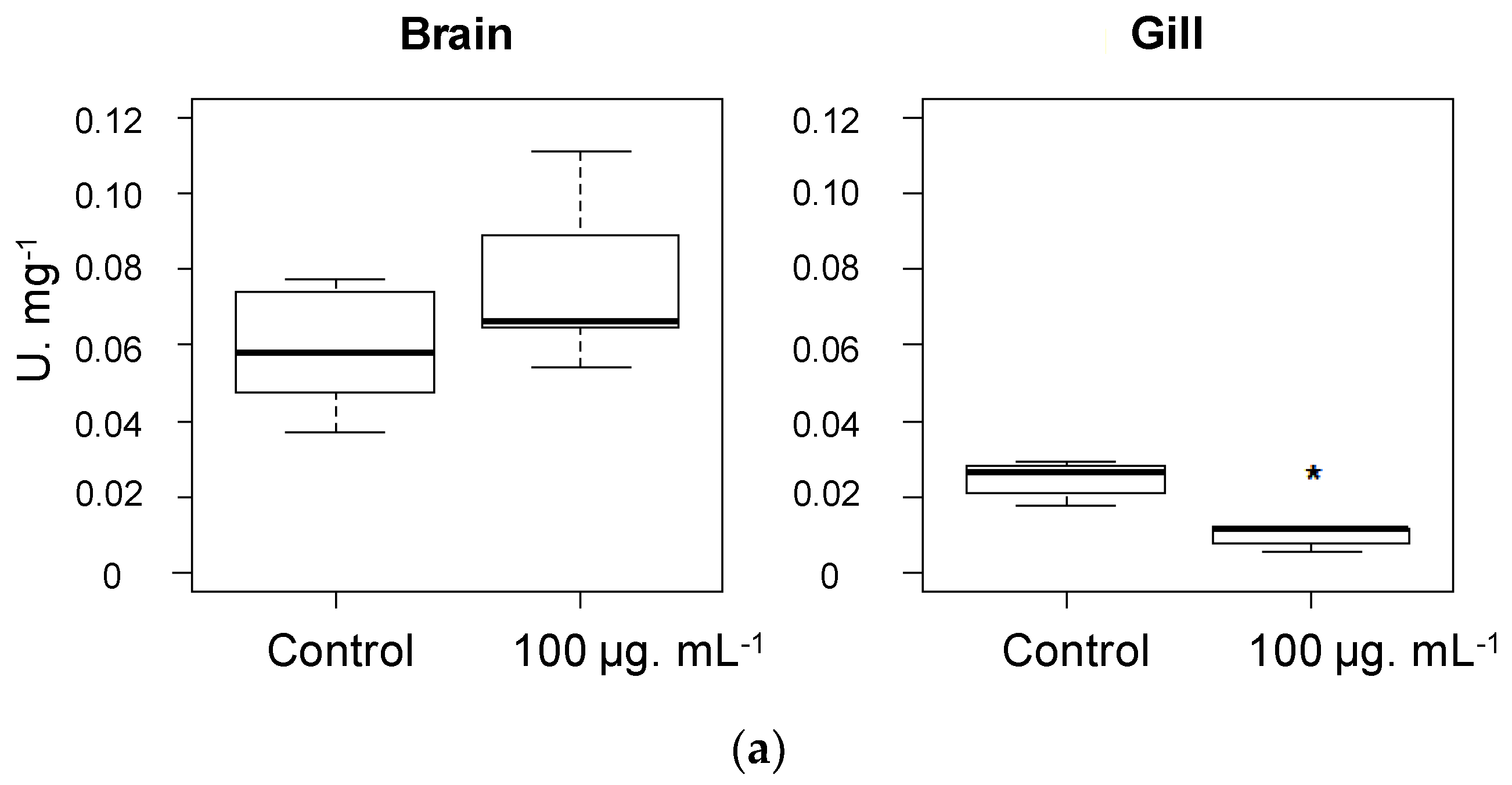
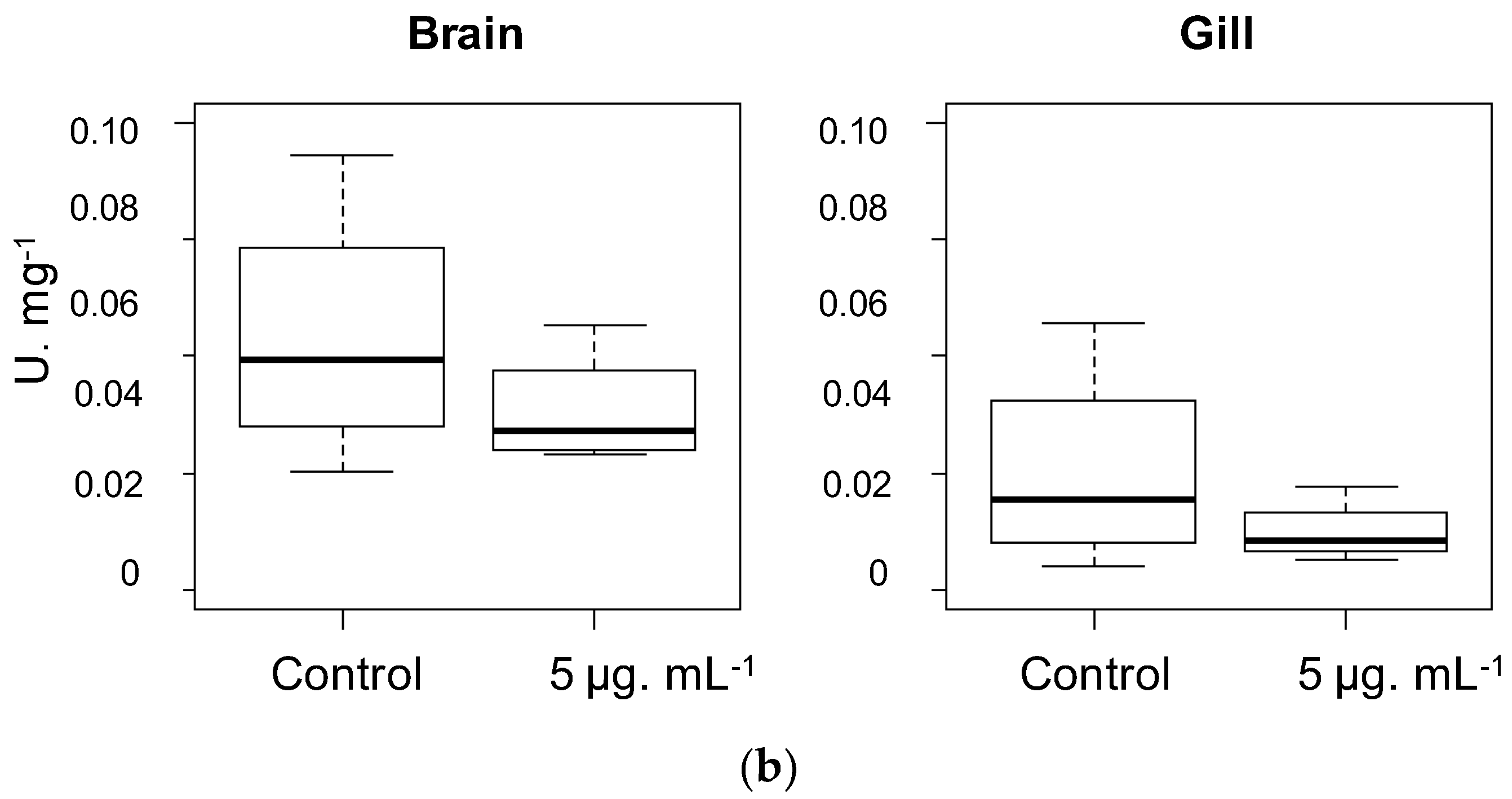
2.2. Biological Activities
3. Material and Methods
3.1. General Procedures
3.2. Animal Material
3.3. Isolation of Bioactive COMPOUNDS
3.4. Cell Culture and Cell Proliferation Assay
3.5. Acetylcholinesterase Inhibition Assay
3.5.1. Kinetic Study
3.5.2. Guppy Bioassay
3.5.3. Reef Fish Feeding Behavior Experiment
3.5.4. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Rahboek, L.; Christophersen, C. The Alkaloids: Chemistry and Biology; Academic Press: Cambridge, MA, USA, 2001; Volume 57, p. 185. [Google Scholar]
- Nuñez, C.V.; Almeida, E.V.R.; Granato, A.C.; Marques, S.O.; Santos, K.O.; Pereira, F.R.; Macedo, M.L.; Ferreira, A.G.; Hajdu, E.; Pinheiro, U.S.; et al. Chemical variability within the marine sponge Aplysina fulva. Biochem. Syst. Ecol. 2008, 36, 283–296. [Google Scholar] [CrossRef]
- Puyana, M.; Pawlik, J.; Blum, J.; Fenical, W. Metabolite variability in Caribbean sponges of the genus Aplysina. Rev. Bras. Farmacogn. 2015, 25, 592–599. [Google Scholar] [CrossRef][Green Version]
- König, G.M.; Wright, A.D. Agelorins A and B, and 11-Epi-fistularin-3, Three New Antibacterial Fistularin-3 Derivatives from the Tropical Marine Sponge Agelas oroides. Heterocycles 1993, 36, 1351–1358. [Google Scholar] [CrossRef]
- Nicacio, K.J.; Ióca, L.P.; Fróes, A.M.; Leomil, L.; Appolinario, L.R.; Thompson, C.C.; Thompson, F.L.; Ferreira, A.G.; Williams, D.E.; Andersen, R.J.; et al. Cultures of the Marine Bacterium Pseudovibrio denitrificans Ab134 Produce Bromotyrosine-Derived Alkaloids Previously Only Isolated from Marine Sponges. J. Nat. Prod. 2017, 80, 235–240. [Google Scholar] [CrossRef] [PubMed]
- Erpenbeck, D.; Van Soest, R.W.M. Status and Perspective of Sponge Chemosystematics. Mar. Biotechnol. 2007, 9, 2–19. [Google Scholar] [CrossRef] [PubMed]
- Shaker, K.H.; Zinecker, H.; Ghani, M.A.; Imhoff, J.F.; Schneider, B. Bioactive metabolites from the sponge Suberea sp. Chem. Biodivers. 2010, 7, 2880–2887. [Google Scholar] [CrossRef] [PubMed]
- Hirano, K.; Kubota, T.; Tsuda, M.; Watanabe, K.; Fromont, J.; Kobayashi, J. Ma’edamines A and B, cytotoxic bromotyrosine alkaloids with a unique 2 (1H) pyrazinone ring from Sponge Suberea sp. Tetrahedron 2002, 56, 8107–8110. [Google Scholar] [CrossRef]
- Tsuda, M.; Sakuma, Y.; Kobayashi, J. Suberedamines A and B, new bromotyrosine alkaloids from a sponge Suberea species. J. Nat. Prod. 2001, 64, 950–982. [Google Scholar] [CrossRef]
- Kijjoa, A.; Watanadilok, R.; Sonchaeng, P.; Puchakarn, S.; Sawangwong, P.; Herz, W. Bromotyrosine derivatives from the marine sponge Suberea aff. praetensa. Boll. Mus. Ist. Biol. Univ. Genova 2004, 68, 391–397. [Google Scholar]
- Florean, C.; Schnekenburger, M.; Lee, J.; Kim, K.R.; Mazumder, A.; Song, S.; Kim, J.; Grandjenette, C.; Kim, J.; Yoon, A.; et al. Discovery and characterization of Isofistularin-3, a marine brominated alkaloid, as a new DNA demethylating agent inducing cell cycle arrest and sensitization to TRAIL in cancer cells. Oncotarget 2016, 7, 24027–24049. [Google Scholar] [CrossRef] [PubMed]
- Shaala, L.A.; Youssef, D.T.A.; Badr, J.M.; Sulaiman, M.; Kherd, A. Bioactive secondary metabolites from the Red Sea marine Verongid sponge Suberea species. Mar. Drugs 2015, 13, 1621–1631. [Google Scholar] [CrossRef] [PubMed]
- Shaala, L.A.; Khalifa, S.I.; Mesbah, M.K.; van Soest, R.W.M.; Youssef, D.T.A. Subereaphenol A, a new cytotoxic and antimicrobial dibrominated phenol from the Red Sea sponge Suberea mollis. Nat. Prod. Commun. 2008, 3, 219–222. [Google Scholar] [CrossRef]
- Abou-Ashour, M.I.; Shaala, L.A.; Youssef, D.T.A.; Bader, J.M.; Habib, A.M. Bioactive brominated metabolites from the Red Sea sponge Suberea mollis. J. Nat. Prod. 2008, 71, 1464–1467. [Google Scholar] [CrossRef] [PubMed]
- Shaala, L.A.; Bamane, F.H.; Badr, J.M.; Youssef, D.T.A. Brominated arginine-derived alkaloids from the Red Sea sponge Suberea mollis. J. Nat. Prod. 2001, 74, 1517–1520. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.; Davis, R.A.; Feng, Y.; Sykes, M.L.; Shelper, T.; Avery, V.M.; Camp, D.; Quinn, R.J. Ianthelliformisamines A-C, antibacterial bromotyrosine-derived metabolites from the marine sponge Suberea ianthelliformis. J. Nat. Prod. 2012, 75, 1001–1005. [Google Scholar] [CrossRef] [PubMed]
- Mani, L.; Jullian, V.; Mourkazel, B.; Valentin, A.; Dubois, J.; Cresteil, T.; Folcher, E.; Hooper, J.N.A.; Erpenbeck, D.; Aalbersberg, W.; et al. New antiplasmodial bromotyrosine derivatives from Suberea ianthelliformis (Lendenfeld, 1888). Chem. Biodivers. 2012, 9, 1436–1451. [Google Scholar] [CrossRef] [PubMed]
- El-Demerdash, A.; Moriou, C.; Martin, M.T.; Rodrigues-Stien, A.; Petek, S.; Demoy-Schnider, M.; Hall, K.; Hooper, J.N.A.; Debitus, C.; Al-Mourabit, A. Cytotoxic guanidine alkaloids from a French Polynesian Monanchora n. sp. sponge. J. Nat. Prod. 2016, 79, 1929–1937. [Google Scholar] [CrossRef] [PubMed]
- Kashman, Y.; Groweiss, A.; Shmueli, U. Latrunculin, A new 2-thiazolidinone macrolide from the marine Sponge Latrunculia magnifica. Tetrahedron Lett. 1980, 21, 3629–3632. [Google Scholar] [CrossRef]
- Neeman, I.; Fishelson, L.; Kashman, Y. Isolation of a new toxin from the sponge Latrunculia magnifica in the Gulf of Aquaba (Red Sea). Mar. Biol. 1975, 30, 293–296. [Google Scholar] [CrossRef]
- Buchanan, M.S.; Carroll, A.R.; Addepalli, R.; Avery, V.M.; Hooper, J.N.A.; Quinn, R.J. Psammaplysenes C and D, Cytotoxic Alkaloids from Psammoclemma sp. J. Nat. Prod. 2007, 70, 1827–1829. [Google Scholar] [CrossRef] [PubMed]
- Yin, L.W.; Feng, Y.; Shimizu, Y.; Pfeifer, T.; Wellington, C.; Hooper, J.N.A.; Quinn, R. Aplysinellamides A–C, Bromotyrosine-Derived Metabolites from an Australian Aplysinella sp. Marine Sponge. J. Nat. Prod. 2014, 77, 1210–1214. [Google Scholar] [CrossRef]
- Molinski, T.F.; Faulkner, D.J. An antibacterial pigment from the sponge Dendrilla membranosa. Tetrahedron Lett. 1988, 29, 2137–2138. [Google Scholar] [CrossRef]
- Spectral Data for Xanthurenic Acid Are Available on Line at the Spectral Database for Organic Compounds. Available online: http://sdbs.db.aist.go.jp/sdbs/cgi-bin/cre_index.cgi (accessed on 26 April 2018).
- Schroeder, F.C.; Kau, T.R.; Silver, P.A.; Clardy, J. The Psammaplysenes, Specific Inhibitors of FOXO1a Nuclear Export. J. Nat. Prod. 2005, 68, 574–576. [Google Scholar] [CrossRef] [PubMed]
- Kernan, M.R.; Cambie, R.C.; Bergquist, P.R. Chemistry of Sponges, VIII. Anomoian A, a Bromotyrosine Derivative from Anomoiantbella popeae. J. Nat. Prod. 1990, 53, 720–723. [Google Scholar] [CrossRef]
- Tarazone, G.; Santamaria, G.; Cruz, P.G.; Ferandez, R.; Pérez, M.; Martinez-Leal, J.F.; Rodriguez, J.; Jiménez, C.; Cuevas, C. Cytotoxic Anomoian B and Aplyzanzine B, New Bromotyrosine Alkaloids from Indonesian Sponges. ACS Omega 2017, 2, 3494–3501. [Google Scholar] [CrossRef]
- Kottakota, S.K.; Evangelopoulos, D.; Alnimr, A.; Bhakta, S.; McHugh, T.D.; Gray, G.; Groundwater, P.W.; Marrs, E.C.L.; Perry, J.D.; Spilling, C.D.; et al. Synthesis and Biological Evaluation of Purpurealidin E-Derived Marine Sponge Metabolites: Aplysamine-2, Aplyzanzine A, and Suberedamines A and B. J. Nat. Prod. 2012, 75, 1090–1101. [Google Scholar] [CrossRef] [PubMed]
- Bocquené, G.; Galgani, F. Biological Effects of Contaminants: Cholinesterase Inhibition by Organophosphate and Carbamate Compounds; ICES Techniques in Marine Environmental Sciences, No. 22; International Council for the Exploration of the Sea: Copenhagen, Denmark, 1998. [Google Scholar]
- Silva, T.; Reis, J.; Teixeira, J.; Borges, F. Alzheimer’s disease, enzyme targets and drug discovery struggles: From natural products to drug prototypes. Ageing Res. Rev. 2014, 15, 116–145. [Google Scholar] [CrossRef] [PubMed]
- Olatunji, O.J.; Ogundajo, A.L.; Oladosu, I.A.; Changwichit, K.; Ingkanina, K.; Yuenyongsawad, S.; Plubrukarn, A. Non-competitive inhibition of acetylcholinesterase by bromotyrosine alkaloids. Nat. Prod. Commun. 2014, 9, 1559–1561. [Google Scholar] [PubMed]
- Sirimangkalakitti, N.; Olatunji, O.J.; Changwichit, K.; Saesong, T.; Chamni, S.; Chanvorachote, P.; Ingkaninan, K.; Plubrukarn, A.; Suwanborirux, K. Bromotyrosine Alkaloids with Acetylcholinesterase activity from the Thai sponge Aconthodendrilla sp. Nat. Prod. Commun. 2015, 10, 1945–1949. [Google Scholar] [PubMed]
- Sepcic, K.; Mancini, I.; Vidic, I.; Jovan, U.; Frassanito, R.; Pietra, F.; Macek, P.; Turk, T. Antibacterial and anticholinesterase activities of aplysamine-4, a bromotyrosine-derived metabolite of a Red Sea marine sponge. J. Nat. Toxins 2001, 10, 181–191. [Google Scholar] [PubMed]
- Schneider, S.L. A critical review of cholinesterase inhibitors as a treatment modality in Alzheimer’s disease. Dialogues Clin. Neurosci. 2000, 2, 111–128. [Google Scholar]
- Aagaard, L. Chapter 1-Central nervous system stimulants and drugs that suppress appetite. Side Effects Drugs Annu. 2014, 35, 1–25. [Google Scholar]
- Zou, Y.; Lu, D.; Liu, L.; Zhang, H.; Zhou, Y. Olfactory dysfunction in Alzheimer’s disease. Neuropsychiatr. Dis. Treat. 2016, 12, 869–875. [Google Scholar] [CrossRef] [PubMed]
- Thompson, J.E.; Barrow, K.D.; Faulkner, D.J. Localization of Two Brominated Metabolites, Aerothionin and Homoaerothinin, in Spherulous cells of the Marine Sponge Aplysina fistularis (=Verongia thiona). Acta Zool. 1983, 64, 199–210. [Google Scholar] [CrossRef]
- Lane, J.Q.; Roddam, C.M.; Langlois, G.W.; Kudela, R.M. Application of Solid Phase Adsorption Toxin Tracking (SPATT) for field detection of the hydrophilic phycotoxins domoic acid and saxitoxin in coastal California. Limnol. Oceanogr. Methods 2010, 8, 645–660. [Google Scholar] [CrossRef]
- Derby, C.D.; Aggio, J.F. The Neuroecology of Chemical Defenses. Integr. Comp. Biol. 2011, 51, 771–780. [Google Scholar] [CrossRef] [PubMed]
- Debitus, C. BSMPF-1 Cruise, RV Alis, 2009. Available online: http://dx.doi.org/10.17600/9100030 (accessed on 26 April 2018).
- Ellman, G.L.; Courtney, D.; Andres, V.; Featherstone, R.M. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharmacol. 1961, 7, 88–95. [Google Scholar] [CrossRef]
- Lecchini, D.; Dufour, V.; Carleton, J.; Strand, S.; Galzin, R. Estimating the Patch size of larval fishes during colonization on coral reefs. J. Fish Biol. 2004, 65, 1142–1146. [Google Scholar] [CrossRef]
- Georgiades, S.N.; Clardy, J. Total Synthesis of Psammaplysenes A and B, Naturally Occurring Inhibitors of FOXO1a Nuclear Export. Org. Lett. 2005, 7, 4091–4094. [Google Scholar] [CrossRef] [PubMed]
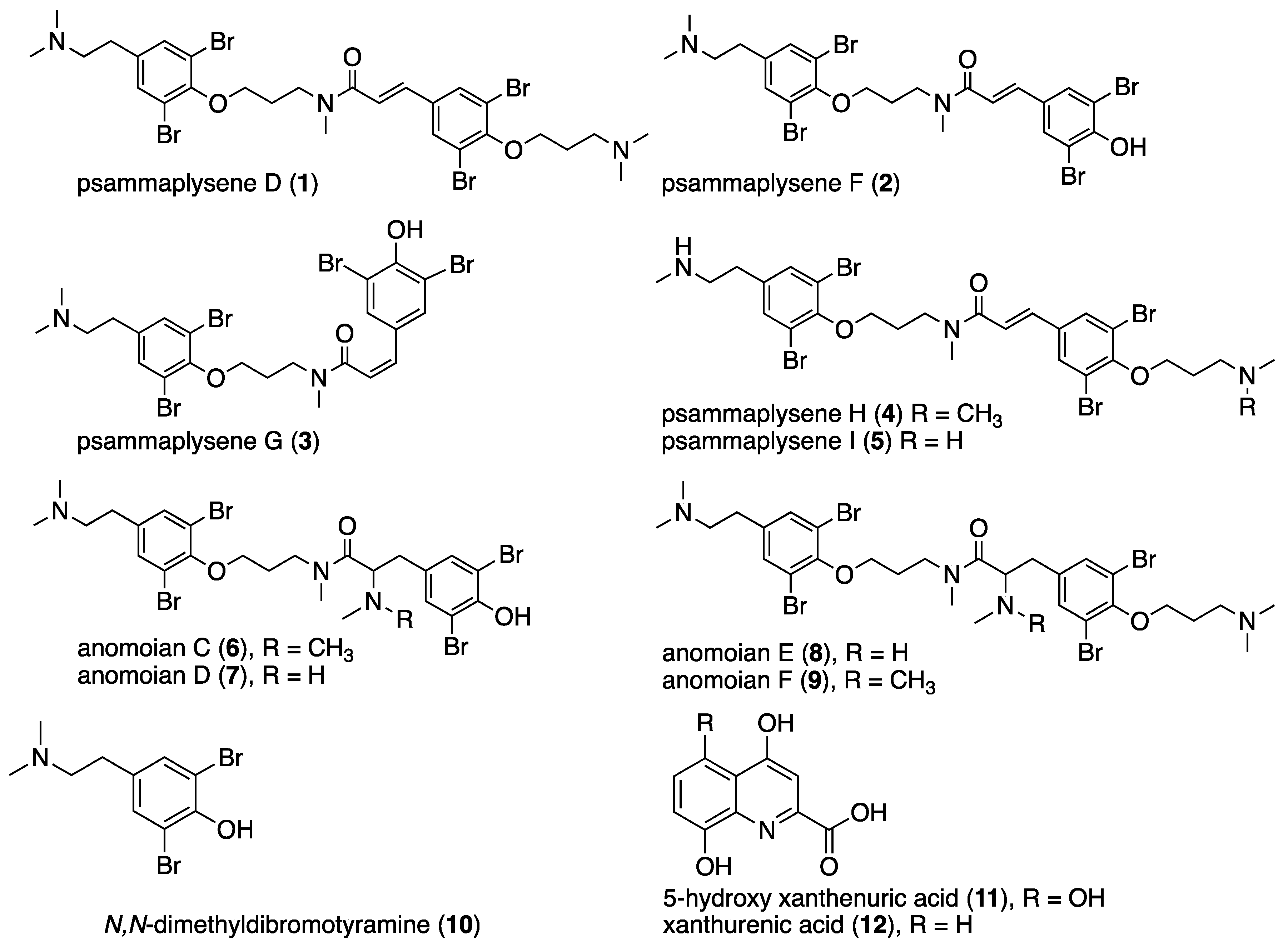
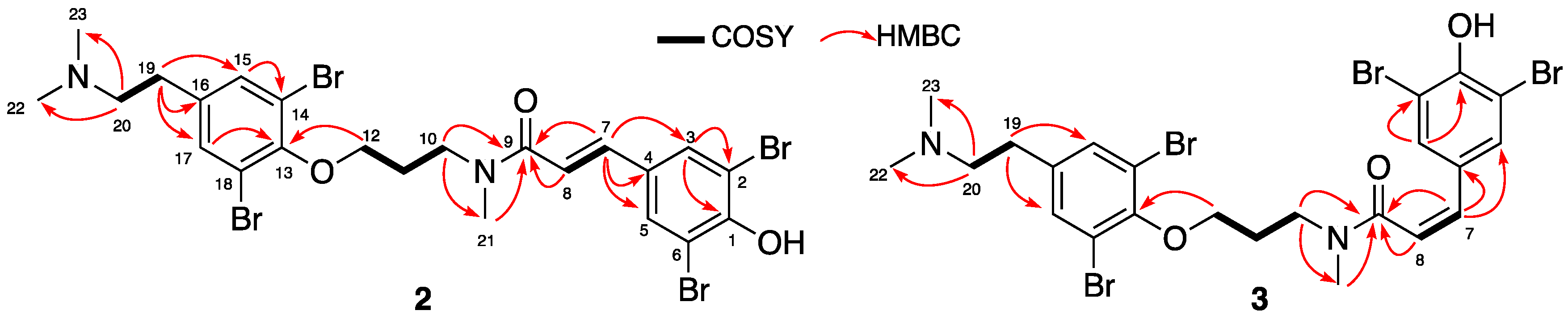
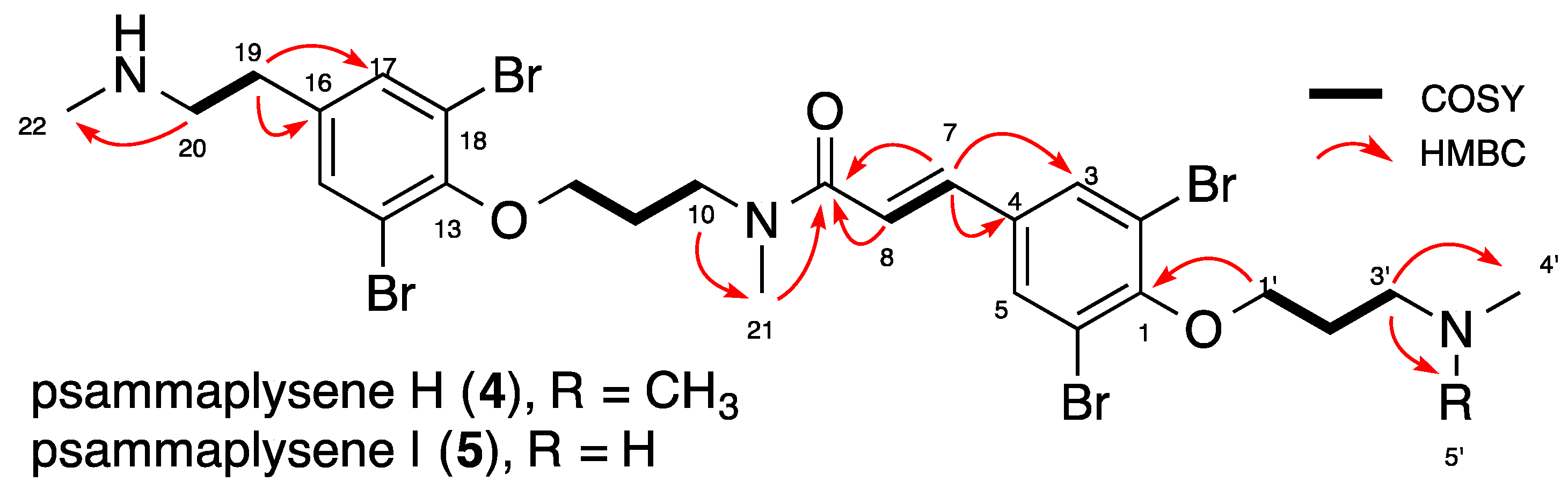
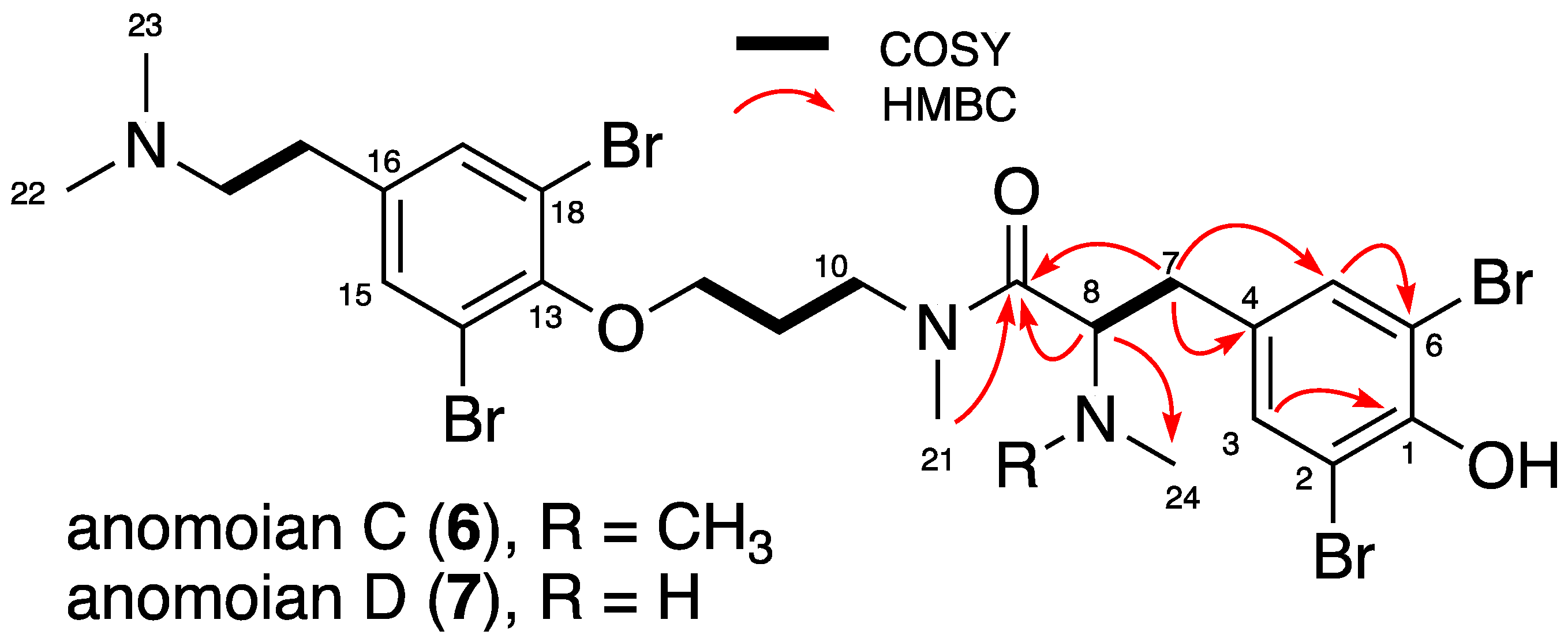
| Position | Psammaplysene F (2) | Psammaplysene G (3) | ||||
|---|---|---|---|---|---|---|
| No. | δC, Type a | δH mult, (J in Hz) a | HMBC | δC, Type a | δH mult, (J in Hz) a | HMBC |
| 1 | 154.10, C | - | - | 153.29/153.04, C | - | - |
| 2 | 112.80, C | - | - | 112.49, C | - | - |
| 3 | 133.09/133.01, CH | 7.78/7.70, s | 1, 2, 5, 6, 7 | 133.70/133.42, CH | 7.53/7.54, s | 1, 2, 5, 6, 7 |
| 4 | 130.98, C | - | - | 130.70/131.16, C | - | - |
| 5 | 133.09/133.01, CH | 7.78/7.70, s | 1, 2, 3, 6, 7 | 133.70/133.42, CH | 7.53/7.54, s | 1, 2, 3, 6, 7 |
| 6 | 112.80, C | - | - | 112.49, C | - | - |
| 7 | 140.89/141.19, CH | 7.36–7.39/7.35–7.38, d (15.5) | 3, 4, 5, 9 | 132.24/132.49, CH | 6.49–6.52/6.56–6.58, d (12.5) | 3, 4, 5, 9 |
| 8 | 118.90/118.72, CH | 7.11–7.15/6.99–7.02, d (15.5) | 4, 7, 9 | 123.90/124.59, CH | 6.13–6.16/6.09–6.12, d (12.5) | 4, 7, 9 |
| 9 | 168.96/168.80, C | - | - | 171.13/171.01, C | - | - |
| 10 | 48.20/47.14, CH2 | 3.89/3.74, t (7.2) | 9, 11, 12, 21 | 48.97/45.75, CH2 | 3.65/3.73, t (7.5) | 9, 11, 12, 21 |
| 11 | 30.86/29.16, CH2 | 2.22/2.16, m | 10, 12 | 29.32/28.68, CH2 | 2.02/2.18, m | 10,12 |
| 12 | 71.94/72.64, CH2 | 4.11/4.05, t (6.4) | 10, 11, 13 | 72.03/72.32, CH2 | 3.78/4.04, t (6.5) | 10, 11, 13 |
| 13 | 153.88/153.94, C | - | - | 153.55/153.86, C | - | - |
| 14 | 119.57, C | - | - | 119.36/119.59, C | - | - |
| 15 | 134.63/134.56, CH | 7.57, br s | 13, 14, 17, 18, 19 | 134.51/134.57, CH | 7.51/7.56, s | 13, 14, 17, 18, 19 |
| 16 | 136.81/136.99, C | - | - | 136.85, C | - | - |
| 17 | 134.63/134.56, CH | 7.57, br s | 13, 14, 15, 18, 19 | 134.51/134.57, CH | 7.51/7.56, s | 13, 14, 15, 18, 19 |
| 18 | 119.57, C | - | - | 119.36/119.59, C | - | - |
| 19 | 30.45, CH2 | 3.00, m | 15, 16, 17, 20, 22, 23 | 30.51/30.56, CH2 | 3.00, m | 15, 16, 17, 20, 22, 23 |
| 20 | 59.24, CH2 | 3.30, m | 16, 19, 22, 23 | 59.32/59.24, CH2 | 3.34, m | 16, 19, 22, 23 |
| 21 | 34.74/36.55, CH3 | 3.09/3.28, s | 9, 10 | 32.59/36.67, CH3 | 3.06/3.03, s | 9, 10 |
| 22 | 43.62, CH3 | 2.91, s | 20, 23 | 43.70/43.73, CH3 | 2.91, s | 20, 23 |
| 23 | 43.62, CH3 | 2.91, s | 20, 22 | 43.70/43.73, CH3 | 2.91, s | 20, 22 |
| Position | Psammaplysene H (4) | Psammaplysene I (5) | ||||
|---|---|---|---|---|---|---|
| No | δC, Type a | δH mult, (J in Hz) a | HMBC | δC, Type a | δH mult, (J in Hz) a | HMBC |
| 1 | 154.7, C | - | - | 154.7, C | - | - |
| 2 | 119.6, C | - | - | 119.6, C | - | - |
| 3 | 133.5/133.4, CH | 7.92/7.85, d | 1, 2, 5, 6, 7 | 133.5/133.4, CH | 7.92/7.85, d | 1, 2, 5, 6, 7 |
| 4 | 136.2, C | - | - | 136.2, C | - | - |
| 5 | 133.5/133.4, CH | 7.92/7.85, d | 1, 2, 3, 6, 7 | 133.5/133.4, CH | 7.92/7.85, d | 1, 2, 3, 6, 7 |
| 6 | 119.6, C | - | - | 119.6, C | - | - |
| 7 | 140.2/140.0, CH | 7.40–7.44, m | 3, 4, 5, 9 | 140.2/140.0, CH | 7.40–7.44, m | 3, 4, 5, 9 |
| 8 | 121.6/121.7, CH | 7.14–7.27, dd (15.0) | 4, 7, 9 | 121.6/121.7, CH | 7.14–7.27, dd (15.0) | 4, 7, 9 |
| 9 | 168.5, C a | - | - | 168.5, C a | - | - |
| 10 | 48.4/47.3, CH2 | 3.91/3.76, t (7.0) | 9, 11, 12, 21 | 48.4/47.3, CH2 | 3.91/3.76, t (7.0) | 9, 11, 12, 21 |
| 11 | 30.6/29.1, CH2 | 2.23/2.17, m | 10, 12 | 30.6/29.1, CH2 | 2.23/2.17, m | 10, 12 |
| 12 | 72.6/71.9, CH2 | 4.12/4.07, m | 10, 11, 13 | 72.6/71.9, CH2 | 4.12/4.07, m | 10, 11, 13 |
| 13 | 154.7, C | - | - | 154.7, C | - | - |
| 14 | 119.6, C | - | - | 119.6, C | - | - |
| 15 | 134.5, CH | 7.56/7.53, m | 13, 14, 17, 18, 19 | 134.5, CH | 7.56/7.53, m | 13, 14, 17, 18, 19 |
| 16 | 137.8, C b | - | - | 137.8, C b | - | - |
| 17 | 134.5, CH | 7.56/7.53, m | 13, 14, 15, 18, 19 | 134.5, CH | 7.56/7.53, m | 13, 14, 15, 18, 19 |
| 18 | 119.6, C | - | - | 119.6, C | - | - |
| 19 | 32.2, CH2 | 2.93, t (8.0) | 16, 20, 22 | 32.2, CH2 | 2.93, t (8.0) | 16, 20, 22 |
| 20 | 51.1, CH2 | 3.21, m | 16, 19, 22 | 51.1, CH2 | 3.21, m | 16, 19, 22 |
| 21 | 34.7/36.6, CH3 | 3.10/3.29, s | 9, 10 | 34.7/36.6, CH3 | 3.10/3.29, s | 9, 10 |
| 22 | 34.0, CH3 | 2.70, s | 20 | 34.0, CH3 | 2.70, s | 20 |
| 1′ | 57.2, CH2 | 3.27, m | 1, 2′, 3′ | 48.7, CH2 | 3.35, m | 1, 2′, 3′ |
| 2′ | 27.6, CH2 | 2.23, m | 1′, 3′ | 30.9 CH2 | 2.23, m | 1′, 3′ |
| 3′ | 71.9, CH2 | 4.18, m | 1′, 2′, 4′, 5′ | 71.9, CH2 | 4.18, m | 1′, 2′, 4′ |
| 4′ | 44.3, CH3 | 2.78/2.74, s | 3′, 5′ | 34.0, CH3 | 2.77/2.76, s | 3′ |
| 5′ | 44.3, CH3 | 2.79/2.74, s | 3′, 4′ | - | - | - |
| Position | Anomoian C (6) | Anomoian D (7) | ||||
|---|---|---|---|---|---|---|
| No | δC, Type a | δH mult, (J in Hz) a | HMBC | δC, Type a | δH mult, (J in Hz) a | HMBC |
| 1 | 151.65/151.47, C | - | - | 152.7, C | - | - |
| 2 | 112.36, C | - | - | 112.69, C | - | - |
| 3 | 134.47/134.41, CH | 7.34/7.32, s | 1, 2, 4, 5, 6, 7 | 134.40, CH | 7.37/7.35, s | 1, 2, 4, 5, 6, 7 |
| 4 | 132.77/133.49, C | - | - | 130.53/129.90, C | - | - |
| 5 | 134.47/134.41, CH | 7.34/7.32, s | 1, 2, 3, 4, 6, 7 | 134.40, CH | 7.37/7.35, s | 1, 2, 3, 4, 6, 7 |
| 6 | 112.36, C | - | - | 112.69, C | - | - |
| 7 | 33.14/32.14, CH2 | 2.90/2.83–2.99, m | 3, 4, 5, 8, 9 | 37.40/38.02, CH2 | 2.83–3.04/2.92, m | 3, 4, 5, 8, 9 |
| 8 | 66.86, CH | 3.82, m | 7, 9, 24, 25 | 60.79/61.29, CH | 4.26/4.17, m | 7, 9, 24 |
| 9 | 172.36/172.63, C | - | - | 171.40/172.00, C | - | - |
| 10 | 47.14/48.24, CH2 | 3.40–3.72, m | 9, 11, 21 | 47.67/47.78, CH2 | 3.48–3.80/3.26–3.51, m | 9, 11, 21 |
| 11 | 29.09/30.45, CH2 | 1.92–2.00/1.73–1.92, m | 10, 12 | 29.15/30.50, CH2 | 1.95–2.05/1.95, m | 10, 12 |
| 12 | 72.31/71.72, CH2 | 3.85–3.93/3.88, m | 11, 13 | 72.35/71.61, CH2 | 3.97/3.99, m | 11, 13 |
| 13 | 153.61/153.30, C | - | - | 153.72, C | - | - |
| 14 | 119.43, C | - | - | 119.47, C | - | - |
| 15 | 134.47/134.50, CH | 7.52/7.53, s | 13, 14, 16, 17, 18, 19 | 134.51/134.53, CH | 7.54/7.55, s | 13, 14, 16, 17, 18, 19 |
| 16 | 137.97/138.24, C | - | - | 137.72, C | - | - |
| 17 | 134.47/134.50, CH | 7.52/7.53, s | 13, 14, 15, 16, 18, 19 | 134.51/134.53, CH | 7.54/7.55, s | 13, 14, 15, 16, 18, 19 |
| 18 | 119.43, C | - | - | 119.47, C | - | - |
| 19 | 31.40/31.46, CH2 | 2.92, m | 15, 16, 17, 20 | 31.22, CH2 | 2.93, m | 15, 16, 17, 20 |
| 20 | 60.12/60.14, CH2 | 3.08, m | 16, 19, 22, 23 | 59.96, CH2 | 3.21, m | 16, 19, 22, 23 |
| 21 | 36.64/34.26, CH3 | 2.92/2.93, s | 9, 10 | 36.25/34.29, CH3 | 2.50/2.47, s | 9, 10 |
| 22 | 44.30/44.34, CH3 | 2.72/2.71, s | 20, 23 | 44.18/44.21, CH3 | 2.74/2.74, s | 20, 23 |
| 23 | 44.30/44.34, CH3 | 2.72/2.71, s | 20, 22 | 44.18/44.21, CH3 | 2.74/2.74, s | 20, 22 |
| 24 | 42.31/41.19, CH3 | 2.45/2.42, s | 8, 25 | 33.26/33.68, CH3 | 2.82/3.00, s | 8 |
| 25 | 42.31/41.19, CH3 | 2.45/2.42, s | 8, 24 | - | - | - |
| Position | Anomoian E (8) | Anomoian F (9) | ||||
|---|---|---|---|---|---|---|
| No | δC, Type a | δH mult, (J in Hz) a | HMBC | δC, Type a | δH mult, (J in Hz) a | HMBC |
| 1 | 153.29/153.15, C | - | - | 152.76/152.57, C | - | - |
| 2 | 119.08.119.14, C | - | - | 118.92, C | - | - |
| 3 | 135.35/135.19, CH | 7.51/7.48, s | 1, 2, 5, 6, 7 | 135.32/135.22, CH | 7.51/7.48, s | 1, 2, 5, 6, 7 |
| 4 | 137.14/138.15, C | - | - | 138.66/137.72, C | - | - |
| 5 | 135.35/135.19, CH | 7.51/7.48, s | 1, 2, 3, 6, 7 | 135.32/135.22, CH | 7.51/7.48, s | 1, 2, 3, 6, 7 |
| 6 | 119.08.119.14, C | - | - | 118.92, C | - | - |
| 7 | 38.03/38.65, CH2 | 2.84–3.03/2.93, m | 3, 4, 5, 8, 9 | 33.05/31.91, CH2 | 3.00/2.92–3.07, m | 3, 4, 5, 8, 9 |
| 8 | 60.72/61.23, CH | 4.15/4.06, m | 4, 7, 9, 24 | 66.58/66.79, CH | 3.97/3.89, m | 4, 7, 9, 24, 25 |
| 9 | 172.73/173.46, C | - | - | 172.10/171.65, C | - | - |
| 10 | 47.57/47.82, CH2 | 3.51–3.76/3.26–3.60, m | 9, 11, 12, 21 | 48.26/47.36, CH2 | 3.41–3.74/3.51–3.64, m | 9, 11, 12, 21 |
| 11 | 29.16/30.64, CH2 | 1.95–2.03/1.95, m | 10, 12 | 30.71/29.08, CH2 | 1.69–1.98/1.90–1.98, m | 10, 12 |
| 12 | 72.36/71.71, CH2 | 3.99, m | 10, 11, 13 | 71.82/72.40, CH2 | 3.94/3.95, m | 10, 11, 13 |
| 13 | 153.54, C | - | - | 153.77/153.51, C | - | - |
| 14 | 119.42, C | - | - | 119.52, C | - | - |
| 15 | 134.54/134.52, CH | 7.55/7.56, s | 13, 14, 17, 18, 19 | 134.57/134.60, CH | 7.57/7.58, s | 13, 14, 17, 18, 19 |
| 16 | 137.74/138.52, C | - | - | 137.28, C | - | - |
| 17 | 134.54/134.52, CH | 7.55/7.56, s | 13, 14, 15, 18, 19 | 134.57/134.60, CH | 7.57/7.58, s | 13, 14, 15, 18, 19 |
| 18 | 119.42, C | - | - | 119.52, C | - | - |
| 19 | 31.31/31.41, CH2 | 2.93, m | 15, 16, 17, 20 | 30.67/30.59, CH2 | 2.99, m | 15, 16, 17, 20 |
| 20 | 60.01/60.08, CH2 | 3.09, m | 16, 19, 22, 23 | 59.43, CH2 | 3.26, m | 16, 19, 22, 23 |
| 21 | 36.24/34.33, CH3 | 2.88/2.99, s | 9, 10 | 34.31/36.64, CH3 | 2.92/2.94, s | 9, 10 |
| 22 | 44.19/44.24, CH3 | 2.73/2.71, s | 20, 23 | 43.75, CH3 | 2.85/2.85, s | 20, 23 |
| 23 | 44.19/44.24, CH3 | 2.73/2.71, s | 20, 22 | 43.75, CH3 | 2.85/2.85, s | 20, 22 |
| 24 | 33.71/34.11, CH3 | 2.42/2.39, s | 4, 8 | 42.23/42.07, CH3 | 2.51/2.45, s | 4, 8, 25 |
| 25 | - | - | - | 42.23/42.07, CH3 | 2.51/2.45, s | 4, 8, 24 |
| 1′ | 71.66/71.62, CH2 | 4.09, m | 1, 2′, 3′ | 71.44, CH2 | 4.01, t (5.5) | 1, 2′, 3′ |
| 2′ | 26.95, CH2 | 2.25, m | 1′, 3′ | 26.62, CH2 | 2.27, m | 1′, 3′ |
| 3′ | 57.15, CH2 | 3.36, m | 1′, 2′, 4′, 5′ | 57.07, CH2 | 3.45, m | 1′, 2′, 4′, 5′ |
| 4′ | 44.05, CH3 | 2.85, s | 3′, 5′ | 43.79, CH3 | 2.92/2.91, s | 3′, 5′ |
| 5′ | 44.05, CH3 | 2.85, s | 3′, 4′ | 43.79, CH3 | 2.92/2.91, s | 3′, 4′ |
| Compounds | 10 μM a | 1 μM a |
|---|---|---|
| Psammaplysene D (1) | 100 ± 0.2 | 95 ± 0.5 |
| Psammaplysene F (2) | 73 ± 2 | 20 ± 0.2 |
| Psammaplysene G (3) | 75 ± 0.4 | 17 ± 1 |
| Anomoian C (6) | 28 ± 5 | 15 ± 3 |
| Anomoian D (7) | 29 ± 2 | 17 ± 2 |
| Anomoian E (8) | 82 ± 1 | 6 ± 2 |
| Anomoian F (9) | 100 ± 0.5 | 20 ± 0.6 |
| N,N-dimethyldibromotyramine (10) | 100 ± 1 | 89 ± 1 |
| 5-hydroxy xanthenuric acid (11) | 25 ± 1 | 6 ± 0.5 |
| Sponge/Compounds | AChE Origin Activities | Ref. | |
|---|---|---|---|
| Pseudoceratina purpurea Purealidine Q Isoanomoian A Aplysanzine A | Eel IC50 = 1.2 µM (NCI a) IC50 = 70 µM (NCI) IC50 = 104 µM (NCI) | Olatunji et al. 2014 [31] | |
| Acanthodendrilla sp. Homoaerothionin Fistularin 1 | Human recombinant AchE IC50 = 4.5 µM (CI b) IC50 = 47.5 µM (ND c) | Sirimangkalakitti et al. 2015 [32] | |
| Unidentified Verongida Aplysamine-4 | Eel KI = 16 µM (NCI) | Insect recombinant KI = 2 µM (NCI) | Sepcic et al. 2001 [33] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
El-Demerdash, A.; Moriou, C.; Toullec, J.; Besson, M.; Soulet, S.; Schmitt, N.; Petek, S.; Lecchini, D.; Debitus, C.; Al-Mourabit, A. Bioactive Bromotyrosine-Derived Alkaloids from the Polynesian Sponge Suberea ianthelliformis. Mar. Drugs 2018, 16, 146. https://doi.org/10.3390/md16050146
El-Demerdash A, Moriou C, Toullec J, Besson M, Soulet S, Schmitt N, Petek S, Lecchini D, Debitus C, Al-Mourabit A. Bioactive Bromotyrosine-Derived Alkaloids from the Polynesian Sponge Suberea ianthelliformis. Marine Drugs. 2018; 16(5):146. https://doi.org/10.3390/md16050146
Chicago/Turabian StyleEl-Demerdash, Amr, Céline Moriou, Jordan Toullec, Marc Besson, Stéphanie Soulet, Nelly Schmitt, Sylvain Petek, David Lecchini, Cécile Debitus, and Ali Al-Mourabit. 2018. "Bioactive Bromotyrosine-Derived Alkaloids from the Polynesian Sponge Suberea ianthelliformis" Marine Drugs 16, no. 5: 146. https://doi.org/10.3390/md16050146
APA StyleEl-Demerdash, A., Moriou, C., Toullec, J., Besson, M., Soulet, S., Schmitt, N., Petek, S., Lecchini, D., Debitus, C., & Al-Mourabit, A. (2018). Bioactive Bromotyrosine-Derived Alkaloids from the Polynesian Sponge Suberea ianthelliformis. Marine Drugs, 16(5), 146. https://doi.org/10.3390/md16050146







