Absorption and Transport of Sea Cucumber Saponins from Apostichopus japonicus
Abstract
:1. Introduction
2. Results
2.1. Characterization of Echinoside A
2.2. Method Validation
2.2.1. Linearity and Sensitivity
2.2.2. Precision and Accuracy
2.2.3. Recovery and Stability
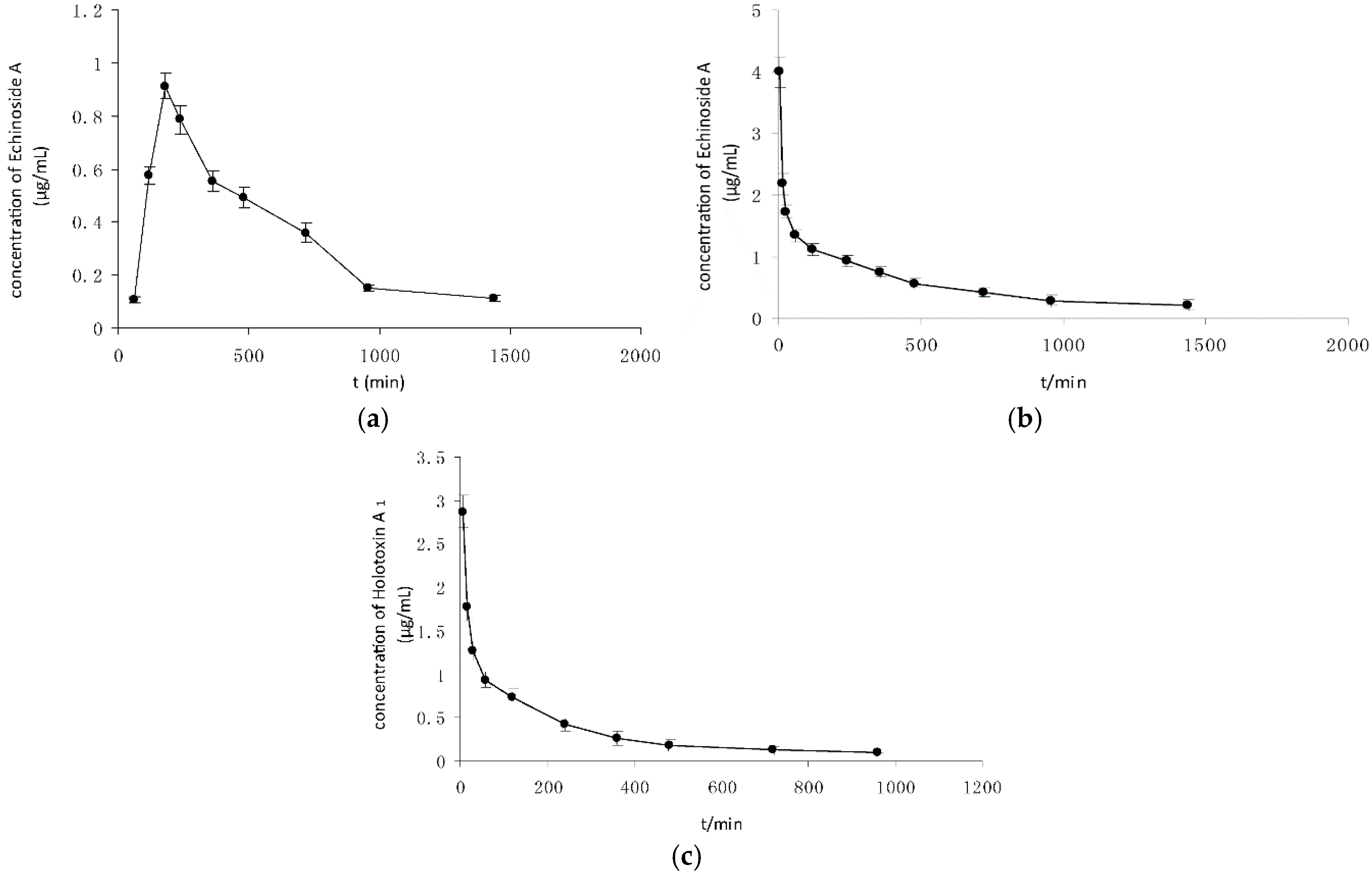
2.3. Pharmacokinetic Analysis of Sea Cucumber Saponins
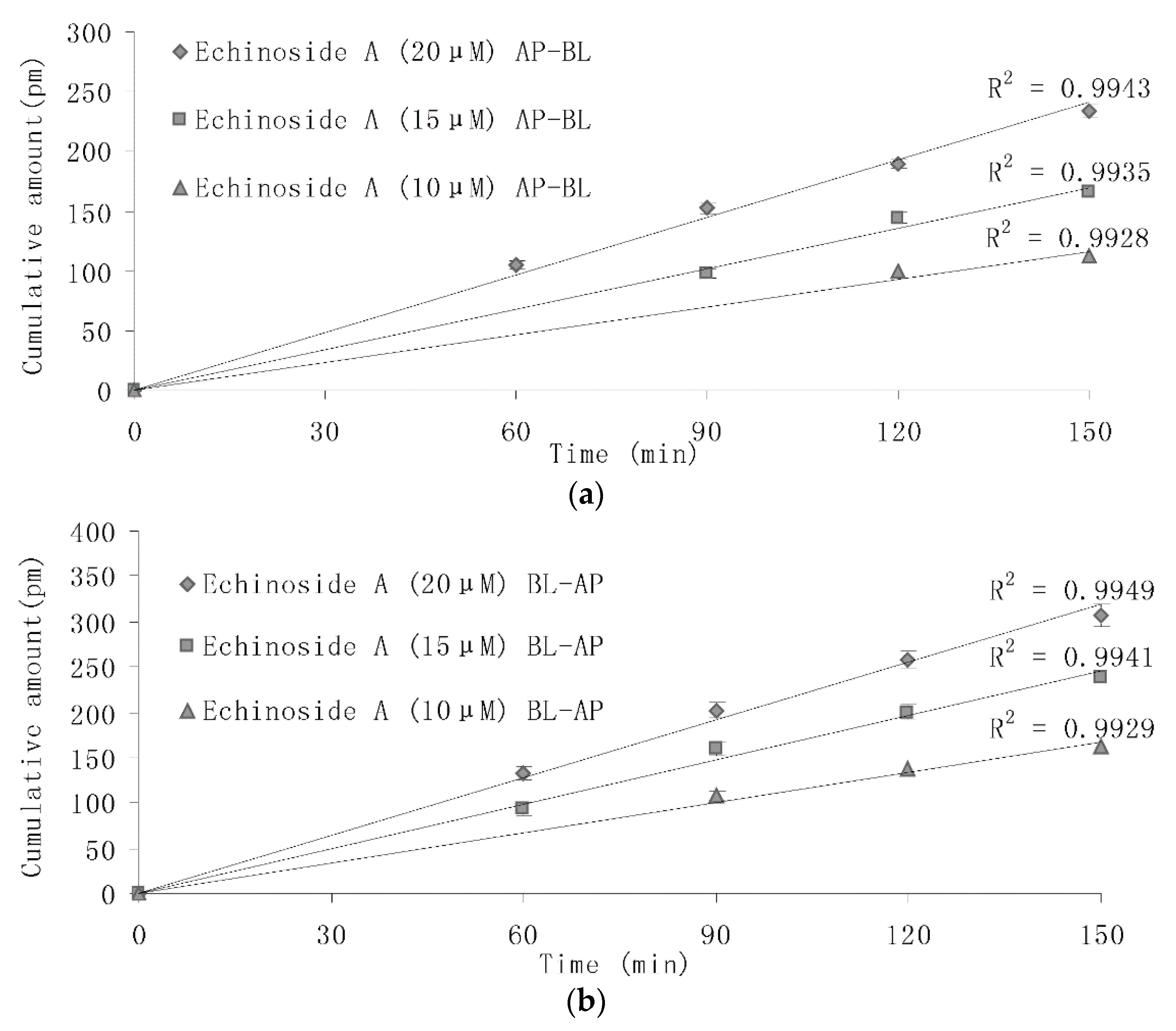
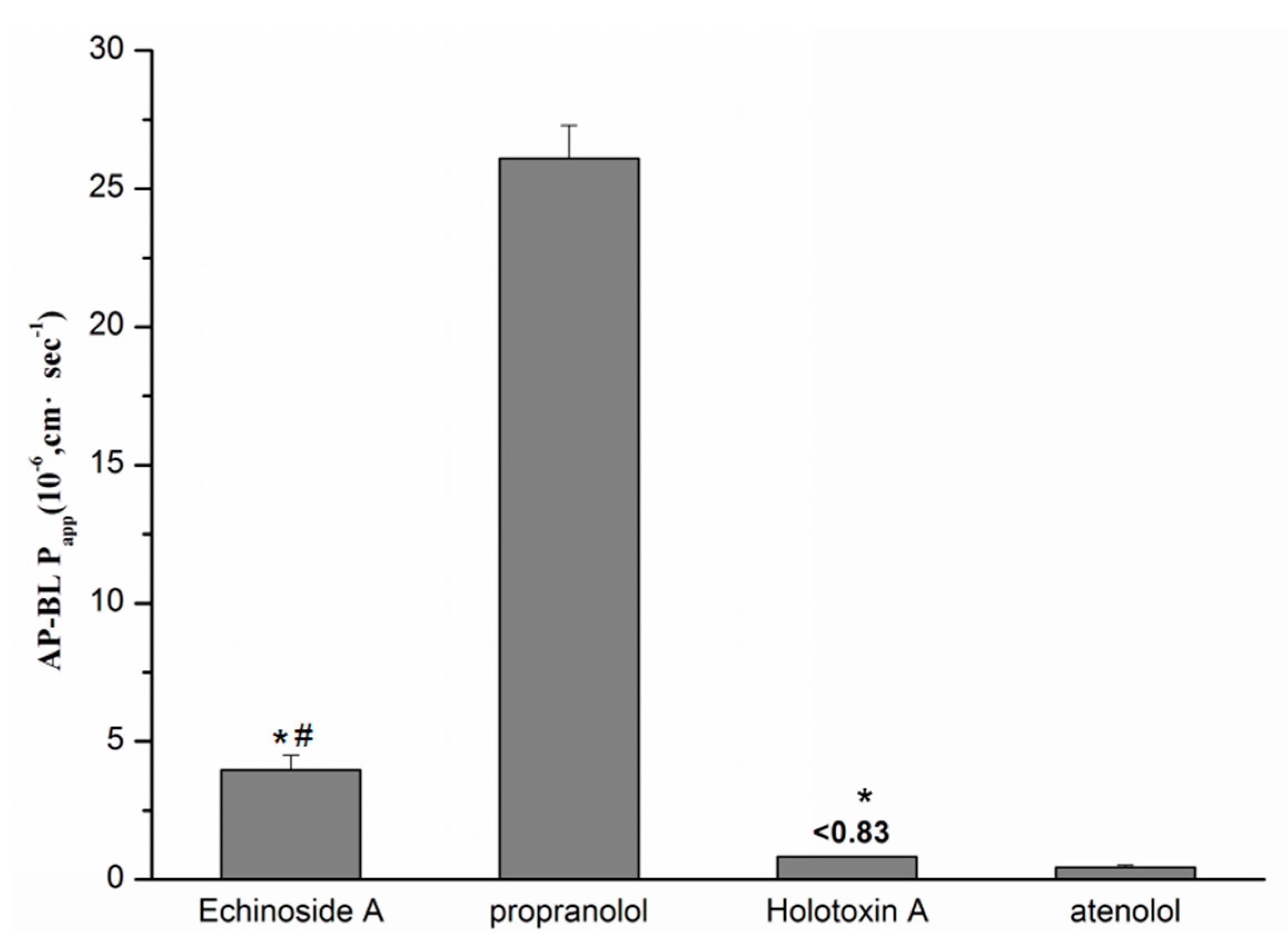
2.4. Transport Experiments of the Caco-2 Cell Model
2.5. Single-Pass Intestinal Perfusion of Sea Cucumber Saponins
2.6. Formatting of Mathematical Components
3. Discussion
4. Materials and Methods
4.1. Chemicals and Materials
4.2. Animals
4.3. Chromatographic Conditions
4.4. Method Validation
4.4.1. Linearity
4.4.2. Precision and Accuracy
4.4.3. Recovery and Stability
4.5. In Vivo Pharmacokinetics of Sea Cucumber Saponins in Rats
4.5.1. Oral Administration of Sea Cucumber Saponins
4.5.2. Intravenous Administration of Sea Cucumber Saponins
4.6. Caco-2 Cells Model
4.6.1. Cell Culture
4.6.2. Transepithelial Transport Experiments
4.7. Single-Pass Intestinal Perfusion Model
4.8. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Caulier, G.; Dyck, S.V.; Gerbaux, P.; Eeckhaut, I.; Flammang, P. Review of saponin diversity in sea cucumbers belonging to the family Holothuriidae. SPC Beche-de-mer Inf. Bull. 2011, 31, 48–54. [Google Scholar]
- Kalinin, V.I.; Silchenko, A.S.; Avilov, S.A.; Stonik, V.A.; Smirnov, A.V. Sea cucumbers triterpene glycosides, the recent progress in structural elucidation and chemotaxonomy. Phytochem. Rev. 2005, 4, 221–236. [Google Scholar] [CrossRef]
- Sparg, S.G.; Light, M.E.; Staden, J.V. Biological activities and distribution of plant saponins. J. Ethnopharmacol. 2004, 94, 219–243. [Google Scholar] [CrossRef] [PubMed]
- Park, J.I.; Bae, H.R.; Kim, C.G.; Stonik, V.A.; Kwak, J.Y. Relationships between chemical structures and functions of triterpene glycosides isolated from sea cucumbers. Front. Chem. 2014, 2, 77–90. [Google Scholar] [CrossRef] [PubMed]
- Han, H.; Yi, Y.H.; Li, L.; Wang, X.H.; Liu, B.S.; Sun, P.; Pan, M.X. A new triterpene glycoside from sea cucumber Holothuria leucospilotal. Chin. Chem. Lett. 2007, 18, 161–164. [Google Scholar] [CrossRef]
- Bordbar, S.; Anwar, F.; Saari, N. High-Value Components and Bioactives from Sea Cucumbers for Functional Foods—A Review. Mar. Drugs 2011, 9, 1761–1805. [Google Scholar] [CrossRef] [PubMed]
- Kitagawa, I.; Sugawara, T.; Yosioka, I.; Kuriyama, K. Saponin and sapogenol. XV. Antifungal glycosides from the sea cucumber Stichopus japonicus selenka (2). Structures of holotoxin A and holotoxin B. Chem. Pharm. Bull. 1976, 24, 275–284. [Google Scholar] [CrossRef] [PubMed]
- Zou, Z.R.; Yi, Y.H.; Wu, H.M.; Wu, J.H.; Liaw, C.C.; Lee, K.H. Intercedensides AC, three new cytotoxic triterpene glycosides from the sea cucumber Mensamaria intercedens Lampert. J. Nat. Prod. 2003, 66, 1055–1060. [Google Scholar] [CrossRef] [PubMed]
- Kalinin, V.I.; Prokofieva, N.G.; Likhatskaya, G.N.; Schentsova, E.B.; Agafonova, I.G.; Avilov, S.A.; Drozdova, O.A. Hemolytic activities of triterpene glycosides from the holothurian order Dendrochirotida: Some trends in the evolution of this group of toxi. Toxicon 1996, 34, 475–483. [Google Scholar] [CrossRef]
- Han, H.; Yi, Y.H.; Li, L.; Liu, B.S.; La, M.P.; Zhang, H.W. Antifungal active triterpene glycosides from sea cucumber Holothuria scabra. Acta Pharm. Sin. 2009, 44, 620–624. [Google Scholar]
- Aminin, D.L.; Agafonova, I.G.; Berdyshev, E.V.; Isachenko, E.G.; Avilov, S.A.; Stonik, V.A. Immunomodulatory properties of cucumariosides from the edible Far-Eastern holothurian Cucumaria japonica. J. Med. Food 2001, 4, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Kerr, R.G.; Chen, Z. In vivo and in vitro biosynthesis of saponins in sea cucumbers. J. Nat. Prod. 1995, 58, 172–176. [Google Scholar] [CrossRef] [PubMed]
- Prokof'eva, N.G.; Chaikina, E.L.; Kicha, A.A.; Ivanchina, N.V. Biological activities of steroid glycosides from starfish. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2003, 134, 695–701. [Google Scholar] [CrossRef]
- Popov, A.M. Comparative study of the hemolytic and cytotoxic activities of Triterpenoids isolated from Ginseng and Sea Cucumbers. Biolchemisty 2002, 29, 120–128. [Google Scholar]
- Zakeri-Milani, P.; Valizadeh, H.; Tajerzadeh, H. Predicting human intestinal permeability using single-pass intestinal perfusion in rat. J. Pharm. Sci. 2007, 10, 368–379. [Google Scholar]
- Hillgren, K.M.; Kato, A.; Borechard, R.T. In vitro systems for studying intestinal drug absorption. Med. Res. Rev. 1995, 15, 83–109. [Google Scholar] [CrossRef] [PubMed]
- Salphati, L.; Childers, K.; Pan, L.; Tsutsui, K.; Takahashi, L. Evaluation of a single-pass intestinal perfusion method in rat for the prediction of absorption in man. J. Pharm. Pharmacol. 2001, 53, 1007–1013. [Google Scholar] [CrossRef] [PubMed]
- Cook, T.J.; Shenoy, S.S. Intestinal permeability of chlorpyrifos using a single-pass intestinal perfusion method in the rat. Toxicology 2003, 184, 125–133. [Google Scholar] [CrossRef]
- Murota, K.; Shimizu, S.; Miyamoto, S.; Izumi, T.; Obata, A.; Kikuchi, M.; Terao, J. Unique uptake and transport of isoflavone aglycones by human intestinal Caco-2 cells: Comparison of isoflavonoids and flavonoids. J. Nutr. 2002, 132, 1956–1961. [Google Scholar] [PubMed]
- Walter, E.; Kissel, T.; Raddatz, P. Transport of peptidomimetic renin inhibitors across monolayers of a human intestinal cell line (Caco-2): Evidence for self-enhancement of paracellular transport route. Pharm. Res. 1995, 12, 1801–1805. [Google Scholar] [CrossRef] [PubMed]
- Fossati, L.; Dechaume, R.; Hardillier, E.; Chevillon, D.; Prevost, C.; Bolze, S.; Maubon, N. Use of simulated intestinal fluid for Caco-2 permeability assay of lipophilic drugs. Int. J. Pharm. 2008, 360, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Borlak, J.; Zwadlo, C. Expression of drug-metabolizing enzymes, nuclear transcription factors and ABC transporters in Caco-2 cells. Xenobiotica 2003, 33, 927–943. [Google Scholar] [CrossRef] [PubMed]
- Maubon, N.; Vee, M.L.; Fossati, L.; Audry, M.; Ferrec, E.L.; Bolze, S.; Fardel, O. Analysis of drug transporter expression in human intestinal Caco-2 cells by real-time PCR. Fundam. Clin. Pharm. 2007, 21, 659–663. [Google Scholar] [CrossRef] [PubMed]
- Taylor, D.C. Animal models for oral drug delivery in man: In situ and in vivo approaches. J. Pharm. Sci. 1984, 73, 1676. [Google Scholar] [CrossRef]
- Sutton, S.C.; Rinaldi, M.T.S.; Vukovinsky, K.E. Comparison of the Gravimetric, Phenol Red, and 14C-PEG-3350 Methods to Determine Water Absorption in the Rat Single-Pass Intestinal Perfusion Model. AAPS Pharm. Sci. 2001, 3, 93–97. [Google Scholar] [CrossRef]
- Hu, J.; Reddy, M.B.; Hendrich, S.; Murphy, P.A. Soyasaponin I and sapongenol B have limited absorption by Caco-2 intestinal cells and limited bioavailability in women. J. Nutr. 2004, 134, 1867–1873. [Google Scholar] [PubMed]
- Han, M.; Han, L.M.; Wang, Q.S.; Bai, Z.H.; Fang, X.L. Mechanism of oral absorption of panaxnotoginseng saponins. Acta Pharm. Sin. 2006, 41, 498–505. [Google Scholar]
- Hill, R.A.; Connolly, J.D. Triterpenoids. Nat. Prod. Rep. 2012, 29, 780–818. [Google Scholar] [CrossRef] [PubMed]
- Walgren, R.A.; Walle, U.K.; Walle, T. Transport of quercetin and its glucosides across human intestinal epithelial Caco-2 cells. Biochem. Pharmacol. 1998, 55, 1721–1727. [Google Scholar] [CrossRef]
- Artursson, P. Epithelial transport of drugs in cell culture. I: A model for studying the passive diffusion of drugs over intestinal absorbtive (Caco2) cells. J. Pharm. Sci. 1990, 79, 476–482. [Google Scholar] [CrossRef] [PubMed]
- Lu, S.; Gough, A.W.; Bobrowski, W.F.; Stewart, B.H. Transport properties are not altered across Caco-2 cells with heightened TEER despite underlying physiological and ultrastructural changes. J. Pharm. Sci. 1996, 85, 270–273. [Google Scholar] [CrossRef] [PubMed]
- Levitt, M.D.; Kneip, J.M.; Levitt, D.G. Use of laminar flow and unstirred layer models to predict intestinal absorption in the rat. J. Clin. Investig. 1988, 81, 1365–1369. [Google Scholar] [CrossRef] [PubMed]
- Artursson, P.; Karlsson, J. Correlation between oral drug absorption in humans and apparent drug permeability coefficients in human intestinal epithelial (Caco-2) cells. Biochem. Biophys. Res. Commun. 1991, 175, 880–885. [Google Scholar] [CrossRef]
- Lau, Y.Y.; Chen, Y.H.; Liu, T.T. Evaluation of a novel in vitro Caco-2 hepatocyte hybrid system for predicting in vivo oral bioavailability. Drug Metab. Dispos. 2004, 32, 937–942. [Google Scholar] [PubMed]
- Caulier, G.; Flammang, P.; Rakotoarisoa, P. Preservation of the bioactive saponins of Holothuria scabra through the processing of trepang. Cah. Biol. Mar. 2013, 54, 685–690. [Google Scholar]
- Zhang, R.; Wang, Y.H.; Liu, Y.F.; Yang, J.; Jiang, T.F.; Lv, Z.H. Isolation, preparation and analysis of triterpene glycoside Holotoxin A1 in Apostichopus japonicus. Chin. J. Mar. Drugs 2013, 32, 8–14. [Google Scholar]
- Li, M.F.; Zhao, Z.X.; Lin, C.Z. Pharmacokinetic Studies of llexsaponin A1 in Rats. Tradit. Chin. Drug Res. Clin. Pharmacol. 2011, 22, 187–189. [Google Scholar]
- Odani, T.; Tanizawa, H.; Takino, Y. Studies on the absorption, distribution, excretion and metabolism of ginseng saponins. II. The absorption, distribution and excretion of ginsenoside Rg1 in the rat. Chem. Pharm. Bull. 1964, 14, 189–192. [Google Scholar] [CrossRef]
- Lin, L.; Liu, J.X.; Zhang, Y. Pharmacokinetic Studies of Ginsenoside Rg1, Re, Rb1 and Rd in Rats by LC-MS/MS Method. Chin. Pharm. J. 2009, 373–377. [Google Scholar]
- Cattoor, K.; Bracke, M.; Deforce, D. Transport of Hop Bitter Acids across Intestinal Caco-2 Cell Monolayers. J. Agric. Food Chem. 2010, 58, 4132–4140. [Google Scholar] [CrossRef] [PubMed]
- Palmgrén, J.J.; Mönkkönen, J.; Jukkola, E. Characterization of Caco-2 cell monolayer drug transport properties by cassette dosing using UV/fluorescence HPLC. Eur. J. Pharm. Biopharm. 2004, 57, 319–328. [Google Scholar] [CrossRef] [PubMed]
- Laurent, C.; Besançon, P.; Caporiccio, B. Flavonoids from a grape seed extract interact with digestive secretions and intestinal cells as assessed in an in vitro digestion/Caco-2 cell culture model. Food Chem. 2007, 100, 1704–1712. [Google Scholar] [CrossRef]
- Lennernäs, H.; Palm, K. Comparison between active and passive drug transport in human intestinal epithelial (caco-2) cells in vitro and human jejunum in vivo. Int. J. Pharm. 1996, 127, 103–107. [Google Scholar] [CrossRef]
- Song, N.N.; Li, Q.S.; Liu, C.X. Intestinal permeability of metformin using single-pass intestinal perfusion in rats. World J. Gastroenterol. 2006, 12, 4064–4070. [Google Scholar] [CrossRef] [PubMed]
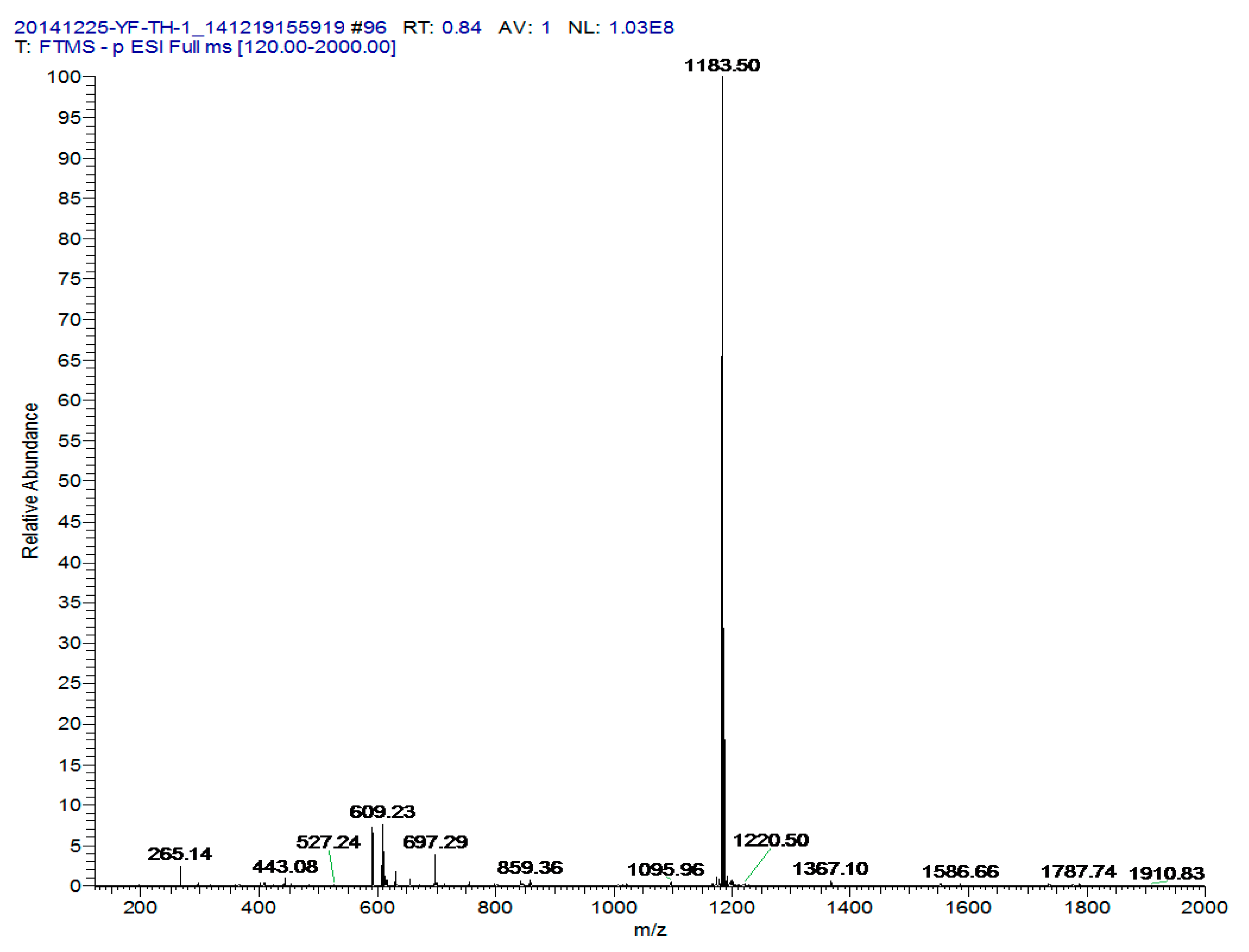
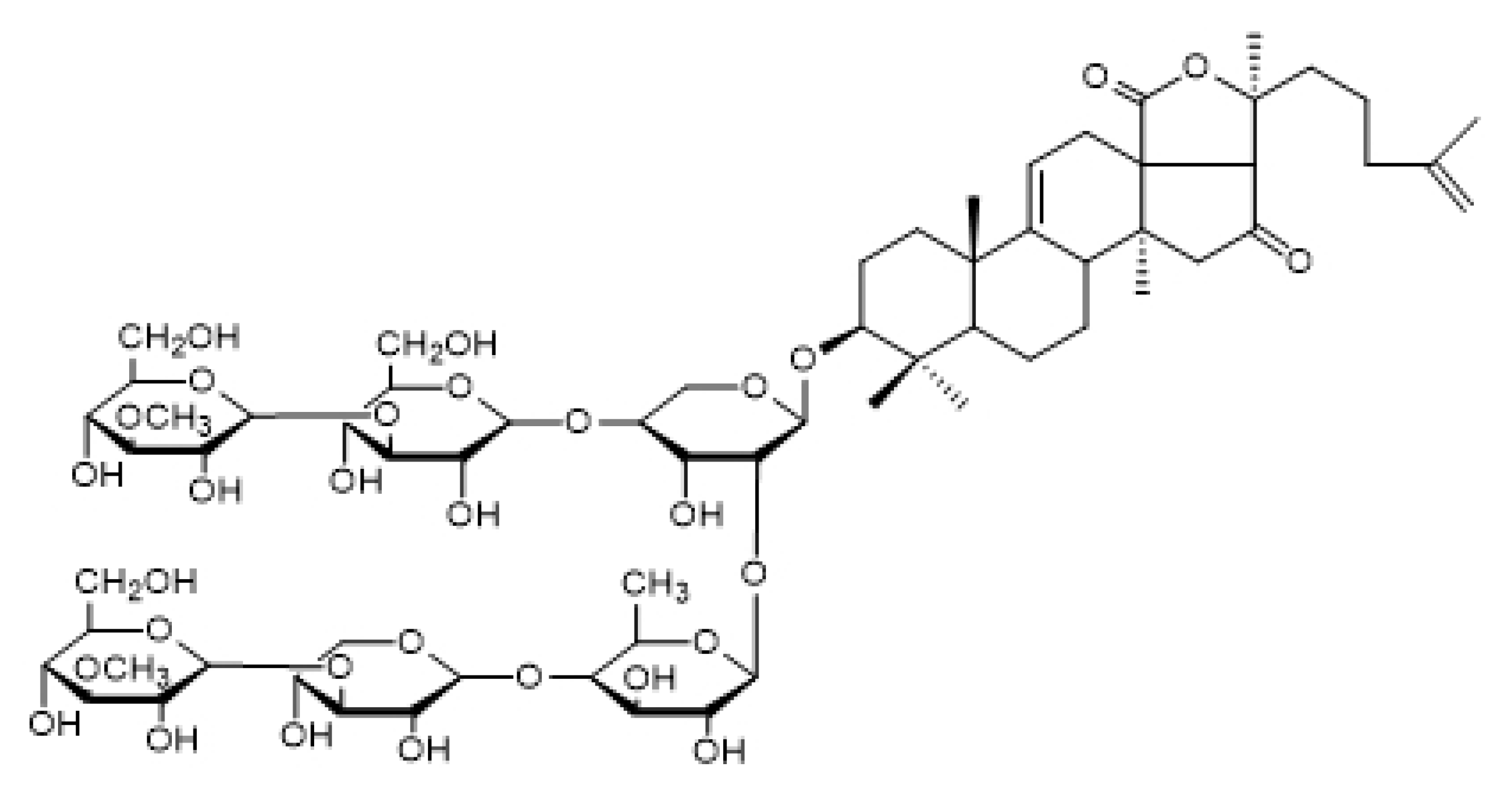
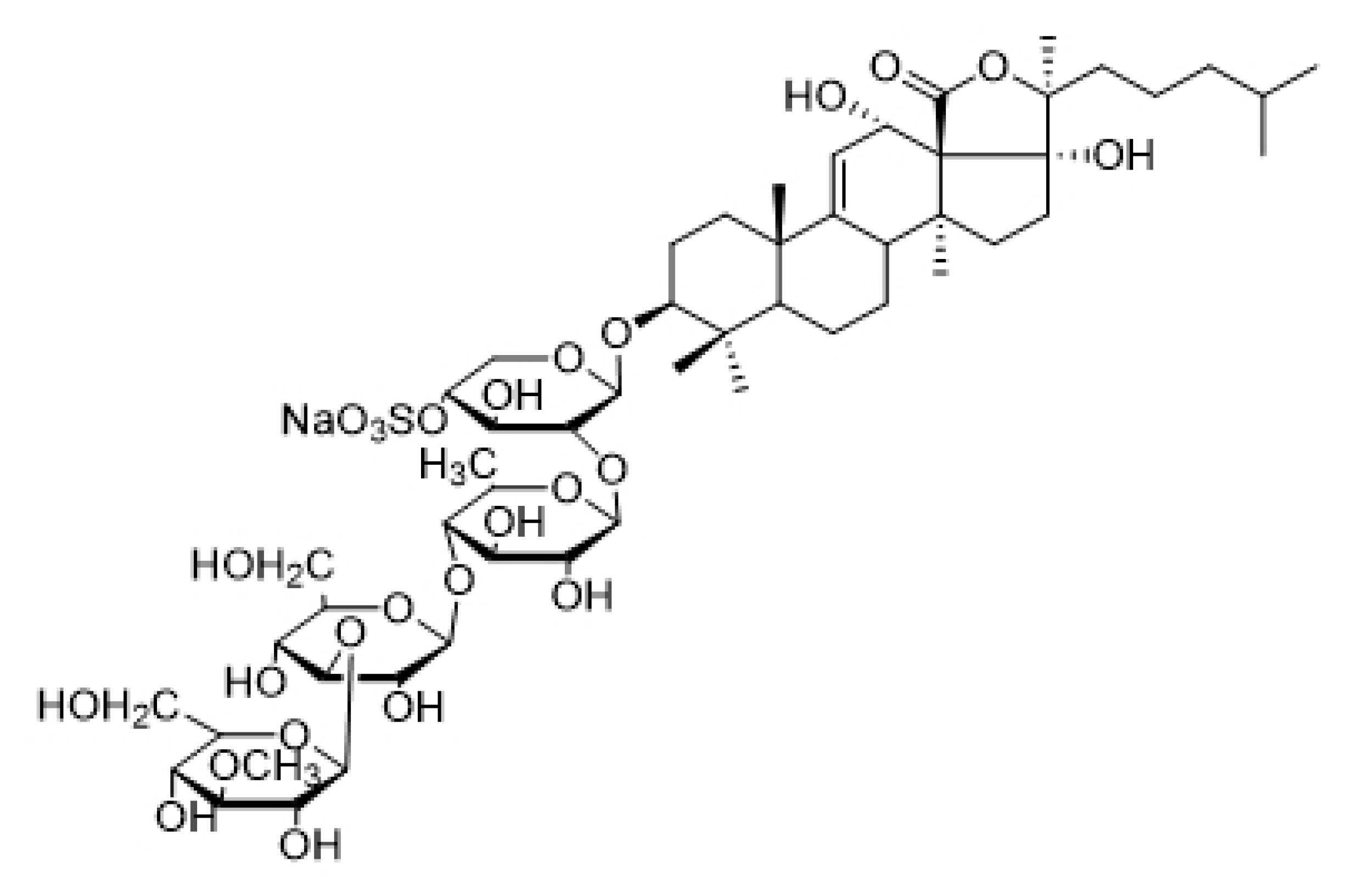


| Position | δC | δH (J in Hz) | Position | δC | δH (J in Hz) |
|---|---|---|---|---|---|
| 1 | 36.9 t | 1.54 m | 15 | 37.2 t | 1.89 m |
| 1.88 m | 1.42 m | ||||
| 2 | 27.6 t | 1.90 m | 16 | 36.4 t | 2.37 d (11.7) |
| 2.14 m | 2.70 m | ||||
| 3 | 89.2 d | 3.39 d (10.9) | 17 | 89.8 s | - |
| 4 | 40.5s | - | 18 | 175.3 s | - |
| 5 | 53.2 d | 1.10 s | 19 | 23.5 q | 1.28 s |
| 6 | 21.7 t | 1.36 m | 20 | 87.7 s | - |
| 1.54 m | 21 | 23.6 q | 1.76 s | ||
| 7 | 28.8 t | 1.54 m | 22 | 39.5 t | 1.85 m |
| 1.76 m | 23 | 22.8 t | 1.80 m | ||
| 8 | 41.4 d | 3.39 d (10.9) | 24 | 40.2 t | 1.18 m |
| 9 | 154.5 s | - | 25 | 28.5 d | 1.54 m |
| 10 | 40.2 s | - | 26 | 23.2 q | 0.86 s |
| 11 | 116.1 d | 5.63 d (4.2) | 27 | 23.3 q | 0.87 m |
| 12 | 71.8 d | 5.05 m | 30 | 28.5 q | 1.28 s |
| 13 | 59.1 s | - | 31 | 17.2 q | 1.13 s |
| 14 | 46.9 s | - | 32 | 20.6 q | 1.70 s |
| Position | δC | δH (J in Hz) | Position | δC | δH (J in Hz) |
|---|---|---|---|---|---|
| Xyl | - | - | Glc | - | - |
| 1 | 105.8 | 4.71 | 1 | 105.5 | 4.96 |
| 2 | 83.8 | 4.02 | 2 | 72.2 | 4.02 |
| 3 | 76.2 | 4.26 | 3 | 88.4 | 3.88 |
| 4 | 74.4 | 5.12 | 4 | 65.3 | 4.23 |
| 5 | 62.8 t | 3.67 | 5 | 76.9 | 4.26 |
| 4.40 | 6 | 61.3 t | 4.30 | ||
| - | - | - | 4.55 | ||
| Qui | - | - | MeGlc | - | - |
| 1 | 105.9 | 5.05 | 1 | 106.2 | 5.32 |
| 2 | 74.5 | 4.02 | 2 | 76.3 | 4.02 |
| 3 | 75.6 | 4.17 | 3 | 89.8 | 4.23 |
| 4 | 87.9 | 3.75 | 4 | 70.4 | 4.19 |
| 5 | 71.9 | 3.75 | 5 | 78.5 | 3.88 |
| 6 | 18.7 | 1.72 | 6 | 62.7 | 4.26 |
| - | - | - | OMe | 61.3 | 3.86 |
| Compounds | Nominal Conc. (ng/mL) | Intra-Day | Inter-Day | ||||
|---|---|---|---|---|---|---|---|
| Measured Conc. (mean ± S.D., ng/mL) | Accuracy (R.E.%) | Precision (R.S.D.%) | Measured Conc. (mean ± S.D., ng/mL) | Accuracy (R.E.%) | Precision (R.S.D.%) | ||
| Echinoside A | 100.00 | 95.60 ± 4.90 | −4.4 | 5.1 | 96.20 ± 5.08 | −3.2 | 5.3 |
| 1000.00 | 985.92 ± 27.51 | −1.4 | 2.8 | 1023.65 ± 40.33 | 2.4 | 3.9 | |
| 5000.00 | 4931.22 ± 94.00 | −1.4 | 1.9 | 5112.43 ± 71.88 | 2.2 | 1.4 | |
| Holotoxin A1 | 100.00 | 104.89 ± 7.60 | 4.9 | 7.2 | 105.75 ± 4.09 | 5.8 | 3.9 |
| 1000.00 | 1029.80 ± 39.89 | 3.0 | 3.9 | 1030.41 ± 31.51 | 3.0 | 3.6 | |
| 5000.00 | 4902.63 ± 114.78 | −2.0 | 2.3 | 4826.33 ± 87.15 | −3.5 | 1.8 | |
| Compounds | Concentration (ng/mL) | Absolute Recovery (mean ± SD %) | CV% |
|---|---|---|---|
| Echinoside A | 100 | 91.30 ± 5.92 | 8.70 |
| 1000 | 94.74 ± 5.51 | 5.26 | |
| 5000 | 95.66 ± 4.08 | 4.34 | |
| Holotoxin A1 | 100 | 90.58 ± 6.72 | 9.42 |
| 1000 | 96.13 ± 4.55 | 3.87 | |
| 5000 | 95.96 ± 4.80 | 4.04 |
| Compounds | Nominal Conc. (ng/mL) | Accuracy (%) | |||||
|---|---|---|---|---|---|---|---|
| Freeze-Thaw Stability | Short-Term Stability | Long-Term Stability | |||||
| Perfusate | Plasma | Perfusate | Plasma | Perfusate | Plasma | ||
| Echinoside A | 100.00 | 91.6 | 94.4 | 90.8 | 95.4 | 89.2 | 97.2 |
| 1000.00 | 91.9 | 96.7 | 91.3 | 96.0 | 92.6 | 102.1 | |
| 5000.00 | 93.5 | 98.9 | 94.4 | 102.4 | 91.1 | 100.3 | |
| Holotoxin A1 | 100.00 | 88.2 | 93.4 | 92.7 | 96.2 | 92.4 | 94.7 |
| 1000.00 | 92.1 | 101.4 | 90.9 | 99.3 | 91.3 | 98.6 | |
| 5000.00 | 94.3 | 98.0 | 93.8 | 96.5 | 94.8 | 99.6 | |
| Compounds | Administration | Pharmacokinetic Parameters | |||
|---|---|---|---|---|---|
| T1/2 (h) | Tmax (h) | Cmax (µg/mL) | AUC (h·μg/mL) | ||
| Echinoside A | oral | 6.9 ± 0.32 | 3.0 | 0.91 ± 0.02 | 9.27 ± 0.39 |
| intravenous | 8.52 ± 0.32 | 0.08 | 4.0 ± 0.44 * | 16.43 ± 0.45 * | |
| Holotoxin A1 | intravenous | 4.4 ± 0.06 | 0.08 | 2.87 ± 0.17 | 6.53 ± 0.15 |
| Time (min) | Echinoside A | Recovery | Holotoxin A1 | ||||
|---|---|---|---|---|---|---|---|
| AP Chamber (%) | BL Chamber (%) | Cell Fraction (%) | AP Chamber (%) | BL Chamber (%) | Cell Fraction (%) | ||
| 60 | 82.14 ± 2.85 | 1.04 ± 0.12 | 14.22 ± 1.14 | 97.40 ± 2.76 | 97.66 ± 2.02 | - | - |
| 90 | 81.63 ± 2.04 | 1.51 ± 0.11 | 15.05 ± 1.29 | 98.19 ± 1.97 | 96.56 ± 1.89 | - | - |
| 120 | 80.25 ± 2.67 | 1.90 ± 0.20 | 14.43 ± 1.24 | 96.58 ± 2.33 | 95.46 ± 1.72 | - | - |
| 150 | 79.38 ± 2.21 | 2.36 ± 0.21 | 14.78 ± 0.92 | 96.52 ± 2.04 | 95.90 ± 2.27 | <0.2 | <0.2 |
| Compounds | Papp (× 10−6, cm·s−1) | Efflux Ratio | |
|---|---|---|---|
| AP-BL | BL-AP | ||
| Echinoside A (20 μM) | 3.96 ± 0.55 | 5.38 ± 0.81 | 1.36 ± 0.17 |
| Echinoside A (20 μM) + verapamil | 4.04 ± 0.39 | 5.33 ± 0.70 | 1.32 ± 0.16 |
| Holotoxin A1 (18 μM) | <0.83 | <0.83 | - |
| Holotoxin A1 (18 μM) + verapamil | <0.83 | <0.83 | - |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, S.; Wang, Y.; Jiang, T.; Wang, H.; Yang, S.; Lv, Z. Absorption and Transport of Sea Cucumber Saponins from Apostichopus japonicus. Mar. Drugs 2016, 14, 114. https://doi.org/10.3390/md14060114
Li S, Wang Y, Jiang T, Wang H, Yang S, Lv Z. Absorption and Transport of Sea Cucumber Saponins from Apostichopus japonicus. Marine Drugs. 2016; 14(6):114. https://doi.org/10.3390/md14060114
Chicago/Turabian StyleLi, Shuai, Yuanhong Wang, Tingfu Jiang, Han Wang, Shuang Yang, and Zhihua Lv. 2016. "Absorption and Transport of Sea Cucumber Saponins from Apostichopus japonicus" Marine Drugs 14, no. 6: 114. https://doi.org/10.3390/md14060114






