Algal Cell Factories: Approaches, Applications, and Potentials
Abstract
:1. Introduction
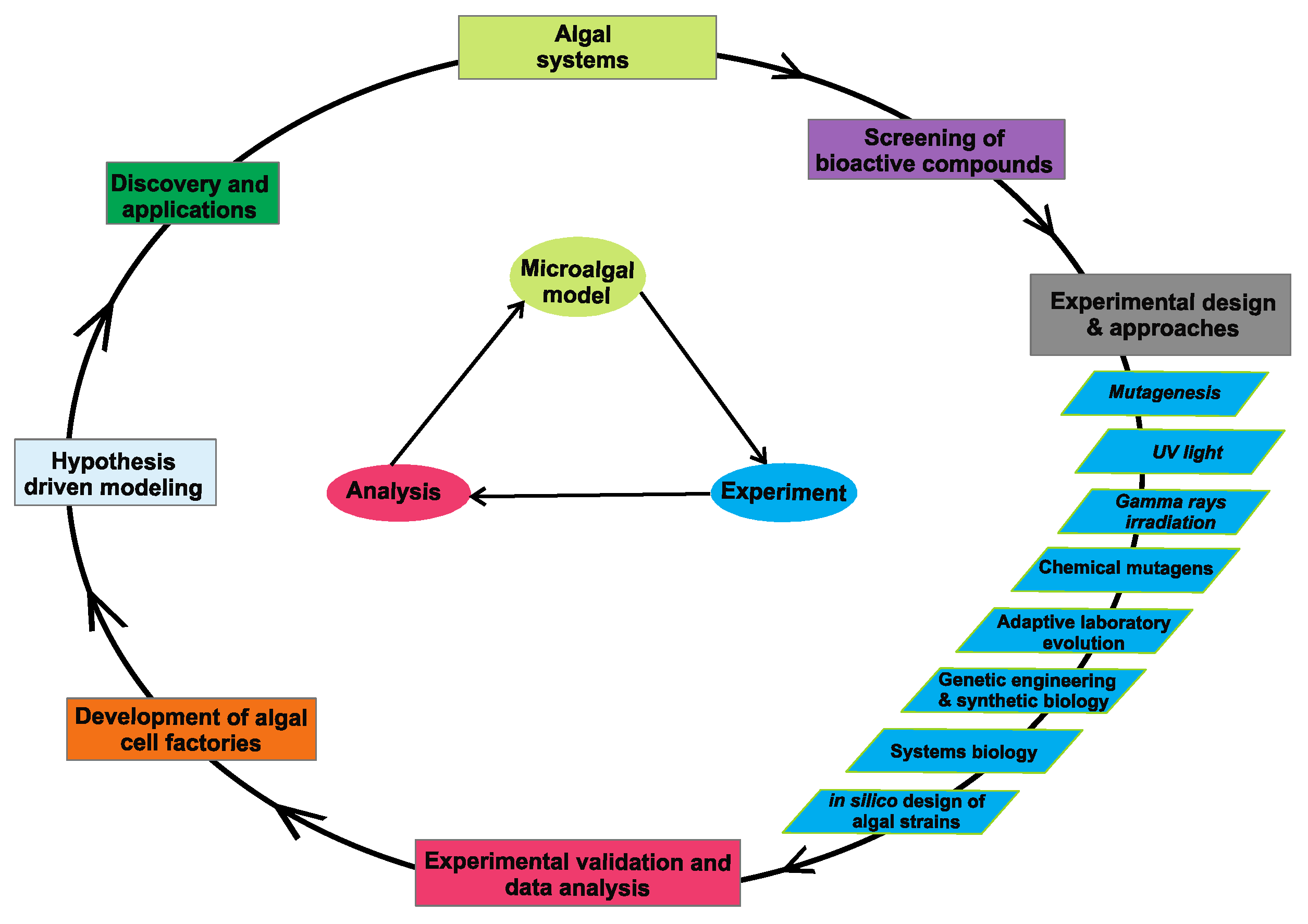
2. Approaches for Developing Algal Cell Factories
2.1. Mutagenesis
2.1.1. UV Light
2.1.2. Gamma Rays Irradiation
2.1.3. Chemical Mutagens
2.2. Adaptive Laboratory Evolution
2.3. Genetic Engineering
2.4. Systems Biology and In Silico Design of Algal Strains
3. Cell Factory Potentials in Macroalgae and Lower Plants
3.1. Macroalgal Species
3.2. The Moss P. patens
4. Concluding Remarks and Perspectives
Acknowledgments
Conflicts of Interest
References
- Raposo, M.F.D.; de Morais, R.M.S.C.; de Morais, A.M.M.B. Health applications of bioactive compounds from marine microalgae. Life Sci 2013, 93, 479–486. [Google Scholar] [CrossRef] [PubMed]
- Michalak, I.; Chojnacka, K. Algae as production systems of bioactive compounds. Eng. Life Sci. 2015, 15, 160–176. [Google Scholar] [CrossRef]
- DSM in Food, Baverages and Dietary Supplements. Nutritional Lipids. Available online: http://www.dsm.com/markets/foodandbeverages/en_US/products/nutritional-lipids.html (accessed on 25 November 2016).
- Rosenberg, J.N.; Oyler, G.A.; Wilkinson, L.; Betenbaugh, M.J. A green light for engineered algae: Redirecting metabolism to fuel a biotechnology revolution. Curr. Opin. Biotechnol. 2008, 19, 430–436. [Google Scholar] [CrossRef] [PubMed]
- Costa, J.A.V.; de Morais, M.G. 16 microalgae for food production. In Fermentation Processes Engineering in the Food Industry; CRC Press, Taylor & Francis Group: Boca Raton, FL, USA, 2013; p. 405. [Google Scholar]
- Fu, W.Q.; Wichuk, K.; Brynjolfsson, S. Developing diatoms for value-added products: Challenges and opportunities. New Biotechnol. 2015, 32, 547–551. [Google Scholar] [CrossRef] [PubMed]
- Wichuk, K.; Brynjolfsson, S.; Fu, W.Q. Biotechnological production of value-added carotenoids from microalgae. Bioengineered 2014, 5, 204–208. [Google Scholar] [CrossRef] [PubMed]
- Salehi-Ashtiani, K.; Koussa, J.; Dohai, B.S.; Chaiboonchoe, A.; Cai, H.; Dougherty, K.A.; Nelson, D.R.; Jijakli, K.; Khraiwesh, B. Toward applications of genomics and metabolic modeling to improve algal biomass productivity. In Biomass and Biofuels from Microalgae; Springer: Basel, Switzerland, 2015; pp. 173–189. [Google Scholar]
- Gov, E.; Arga, K.Y. Systems biology solutions to challenges in marine biotechnology. Front. Mar. Sci. 2014, 1, 14. [Google Scholar] [CrossRef]
- Xu, C.; Liu, L.; Zhang, Z.; Jin, D.; Qiu, J.; Chen, M. Genome-scale metabolic model in guiding metabolic engineering of microbial improvement. Appl. Microbiol. Biotechnol. 2013, 97, 519–539. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, D.; Shimizu, K. An overview on biofuel and biochemical production by photosynthetic microorganisms with understanding of the metabolism and by metabolic engineering together with efficient cultivation and downstream processing. Bioresour. Bioprocess. 2015, 2, 17. [Google Scholar] [CrossRef]
- Mimouni, V.; Ulmann, L.; Pasquet, V.; Mathieu, M.; Picot, L.; Bougaran, G.; Cadoret, J.-P.; Morant-Manceau, A.; Schoefs, B. The potential of microalgae for the production of bioactive molecules of pharmaceutical interest. Curr. Pharm. Biotechnol. 2012, 13, 2733–2750. [Google Scholar] [CrossRef] [PubMed]
- Kodym, A.; Afza, R. Physical and chemical mutagenesis. In Plant Functional Genomics; Humana Press, Inc.: Totowa, NJ, USA, 2003; pp. 189–203. [Google Scholar]
- Pasquet, V.; Cherouvrier, J.R.; Farhat, F.; Thiery, V.; Piot, J.M.; Berard, J.B.; Kaas, R.; Serive, B.; Patrice, T.; Cadoret, J.P.; et al. Study on the microalgal pigments extraction process: Performance of microwave assisted extraction. Process. Biochem. 2011, 46, 59–67. [Google Scholar] [CrossRef]
- Ikehata, H.; Ono, T. The mechanisms of UV mutagenesis. J. Radiat. Res. 2011, 52, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Depauw, F.A.; Rogato, A.; Ribera d’Alcala, M.; Falciatore, A. Exploring the molecular basis of responses to light in marine diatoms. J. Exp. Bot. 2012, 63, 1575–1591. [Google Scholar] [CrossRef] [PubMed]
- Klanchui, A.; Khannapho, C.; Phodee, A.; Cheevadhanarak, S.; Meechai, A. iAK692: A genome-scale metabolic model of Spirulina platensis c1. BMC Syst. Biol. 2012, 6, 71. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Zhao, Y.; Liu, L.; Ao, X.; Ma, L.; Wu, M.; Ma, F. Improving cell growth and lipid accumulation in green microalgae Chlorella sp. Via UV irradiation. Appl. Biochem. Biotechnol. 2015, 175, 3507–3518. [Google Scholar] [CrossRef] [PubMed]
- Abdrabu, R.; Sharma, S.K.; Khraiwesh, B.; Jijakli, K.; Nelson, D.R.; Alzahmi, A.; Koussa, J.; Sultana, M.; Khapli, S.; Jagannathan, R.; et al. Single-cell characterization of microalgal lipid contents with confocal raman microscopy. In Essentials of Single-Cell Analysis; Springer: Berlin, Germany, 2016; pp. 363–382. [Google Scholar]
- Sharma, S.K.; Nelson, D.R.; Abdrabu, R.; Khraiwesh, B.; Jijakli, K.; Arnoux, M.; O’Connor, M.J.; Bahmani, T.; Cai, H.; Khapli, S.; et al. An integrative raman microscopy-based workflow for rapid in situ analysis of microalgal lipid bodies. Biotechnol. Biofuels 2015, 8, 164. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.-I.; Yoon, M.; Joe, M.; Park, H.; Lee, S.G.; Han, S.J.; Lee, P.C. Development of microalga Scenedesmus dimorphus mutant with higher lipid content by radiation breeding. Bioprocess Biosyst. Eng. 2014, 37, 2437–2444. [Google Scholar] [CrossRef] [PubMed]
- Harper, M.; Lee, C.J. Genome-wide analysis of mutagenesis bias and context sensitivity of N-methyl-N′-nitro-N-nitrosoguanidine (NTG). Mutat. Res./Fundam. Mol. Mech. Mutagen. 2012, 731, 64–67. [Google Scholar] [CrossRef] [PubMed]
- Kamath, B.S.; Vidhyavathi, R.; Sarada, R.; Ravishankar, G. Enhancement of carotenoids by mutation and stress induced carotenogenic genes in Haematococcus pluvialis mutants. Bioresour. Technol. 2008, 99, 8667–8673. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.; Choi, G.-G.; Choi, Y.-E.; Sung, M.; Park, M.S.; Yang, J.-W. Enhancement of lipid productivity by ethyl methane sulfonate-mediated random mutagenesis and proteomic analysis in Chlamydomonas reinhardtii. Korean J. Chem. Eng. 2014, 31, 1036–1042. [Google Scholar] [CrossRef]
- Applebee, M.K.; Joyce, A.R.; Conrad, T.M.; Pettigrew, D.W.; Palsson, B.O. Functional and metabolic effects of adaptive glycerol kinase (Glpk) mutants in Escherichia coli. J. Biol. Chem. 2011, 286, 23150–23159. [Google Scholar] [CrossRef] [PubMed]
- Dragosits, M.; Mattanovich, D. Adaptive laboratory evolution—Principles and applications for biotechnology. Microb. Cell Fact. 2013, 12, 64. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, D.H.; Feist, A.M.; Barrett, C.L.; Palsson, B.O. Cumulative number of cell divisions as a meaningful timescale for adaptive laboratory evolution of Escherichia coli. PLoS ONE 2011, 6, e26172. [Google Scholar] [CrossRef] [PubMed]
- Fu, W.; Guethmundsson, O.; Paglia, G.; Herjolfsson, G.; Andresson, O.S.; Palsson, B.O.; Brynjolfsson, S. Enhancement of carotenoid biosynthesis in the green microalga Dunaliella salina with light-emitting diodes and adaptive laboratory evolution. Appl. Microbiol. Biotechnol. 2013, 97, 2395–2403. [Google Scholar] [CrossRef] [PubMed]
- Yi, Z.; Xu, M.; Magnusdottir, M.; Zhang, Y.; Brynjolfsson, S.; Fu, W. Photo-oxidative stress-driven mutagenesis and adaptive evolution on the marine diatom Phaeodactylum tricornutum for enhanced carotenoid accumulation. Mar. Drugs 2015, 13, 6138–6151. [Google Scholar] [CrossRef] [PubMed]
- Tillich, U.M.; Wolter, N.; Franke, P.; Duhring, U.; Frohme, M. Screening and genetic characterization of thermo-tolerant Synechocystis sp. PCC6803 strains created by adaptive evolution. BMC Biotechnol. 2014, 14, 66. [Google Scholar] [CrossRef] [PubMed]
- Uchiyama, J.; Kanesaki, Y.; Iwata, N.; Asakura, R.; Funamizu, K.; Tasaki, R.; Agatsuma, M.; Tahara, H.; Matsuhashi, A.; Yoshikawa, H.; et al. Genomic analysis of parallel-evolved cyanobacterium Synechocystis sp. PCC6803 under acid stress. Photosynth. Res. 2015, 125, 243–254. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Shi, M.; Niu, X.; Zhang, X.; Gao, L.; Chen, L.; Wang, J.; Zhang, W. Metabolomic basis of laboratory evolution of butanol tolerance in photosynthetic Synechocystis sp. PCC6803. Microb. Cell Fact. 2014, 13, 151. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Zhao, Q.; Miao, X.; Shi, J. Enhancement of lipid production in low-starch mutants Chlamydomonas reinhardtii. by adaptive laboratory evolution. Bioresour. Technol. 2013, 147, 499–507. [Google Scholar] [CrossRef] [PubMed]
- Velmurugan, N.; Sung, M.; Yim, S.S.; Park, M.S.; Yang, J.W.; Jeong, K.J. Systematically programmed adaptive evolution reveals potential role of carbon and nitrogen pathways during lipid accumulation in Chlamydomonas reinhardtii. Biotechnol. Biofuels 2014, 7, 117. [Google Scholar] [CrossRef] [PubMed]
- Flowers, J.M.; Hazzouri, K.M.; Pham, G.M.; Rosas, U.; Bahmani, T.; Khraiwesh, B.; Nelson, D.R.; Jijakli, K.; Abdrabu, R.; Harris, E.H.; et al. Whole-genome resequencing reveals extensive natural variation in the model green alga Chlamydomonas reinhardtii. Plant Cell 2015, 27, 2353–2369. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Wang, L.; Zhao, Q.; Wei, W.; Sun, Y. Improving high carbon dioxide tolerance and carbon dioxide fixation capability of Chlorella sp. By adaptive laboratory evolution. Bioresour. Technol. 2015, 185, 269–275. [Google Scholar] [CrossRef] [PubMed]
- Bowler, C.; Allen, A.E.; Badger, J.H.; Grimwood, J.; Jabbari, K.; Kuo, A.; Maheswari, U.; Martens, C.; Maumus, F.; Otillar, R.P.; et al. The phaeodactylum genome reveals the evolutionary history of diatom genomes. Nature 2008, 456, 239–244. [Google Scholar] [CrossRef] [PubMed]
- Coesel, S.; Obornik, M.; Varela, J.; Falciatore, A.; Bowler, C. Evolutionary origins and functions of the carotenoid biosynthetic pathway in marine diatoms. PLoS ONE 2008, 3, e2896. [Google Scholar] [CrossRef] [PubMed]
- Schellenberger Costa, B.; Jungandreas, A.; Jakob, T.; Weisheit, W.; Mittag, M.; Wilhelm, C. Blue light is essential for high light acclimation and photoprotection in the diatom Phaeodactylum tricornutum. J. Exp. Bot. 2013, 64, 483–493. [Google Scholar] [CrossRef] [PubMed]
- Lepetit, B.; Sturm, S.; Rogato, A.; Gruber, A.; Sachse, M.; Falciatore, A.; Kroth, P.G.; Lavaud, J. High light acclimation in the secondary plastids containing diatom Phaeodactylum tricornutum is triggered by the redox state of the plastoquinone pool. Plant Physiol. 2013, 161, 853–865. [Google Scholar] [CrossRef] [PubMed]
- Lohbeck, K.T.; Riebesell, U.; Reusch, T.B.H. Adaptive evolution of a key phytoplankton species to ocean acidification (vol 5, pg 346, 2012). Nat. Geosci. 2012, 5, 917. [Google Scholar] [CrossRef]
- Hempel, F.; Maier, U.G. An engineered diatom acting like a plasma cell secreting human IGG antibodies with high efficiency. Microb. Cell Fact. 2012, 11, 126. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Kim, J.H.; Kim, S.W. Synthetic biology and metabolic engineering for marine carotenoids: New opportunities and future prospects. Mar. Drugs 2014, 12, 4810–4832. [Google Scholar] [CrossRef] [PubMed]
- Jijakli, K.; Abdrabu, R.; Khraiwesh, B.; Nelson, D.R.; Koussa, J.; Salehi-Ashtiani, K. Molecular genetic techniques for algal bioengineering. In Biomass and Biofuels from Microalgae; Springer: Basel, Switzerland, 2015; pp. 155–171. [Google Scholar]
- Leon, R.; Couso, I.; Fernandez, E. Metabolic engineering of ketocarotenoids biosynthesis in the unicelullar microalga Chlamydomonas reinhardtii. J. Biotechnol. 2007, 130, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Cordero, B.F.; Obraztsova, I.; Couso, I.; Leon, R.; Vargas, M.A.; Rodriguez, H. Enhancement of lutein production in Chlorella sorokiniana (chorophyta) by improvement of culture conditions and random mutagenesis. Mar. Drugs 2011, 9, 1607–1624. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Couso, I.; Cordero, B.F.; Vargas, M.A.; Rodriguez, H. Efficient heterologous transformation of Chlamydomonas reinhardtii npq2 mutant with the zeaxanthin epoxidase gene isolated and characterized from C hlorella zofingiensis. Mar. Drugs 2012, 10, 1955–1976. [Google Scholar] [CrossRef] [PubMed]
- Apt, K.E.; Kroth-Pancic, P.G.; Grossman, A.R. Stable nuclear transformation of the diatom Phaeodactylum tricornutum. Mol. Gen. Genet. 1996, 252, 572–579. [Google Scholar] [PubMed]
- Miyahara, M.; Aoi, M.; Inoue-Kashino, N.; Kashino, Y.; Ifuku, K. Highly efficient transformation of the diatom Phaeodactylum tricornutum by multi-pulse electroporation. Biosci. Biotechnol. Biochem. 2013, 77, 874–876. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, M.L.; Haslam, R.P.; Napier, J.A.; Sayanova, O. Metabolic engineering of Phaeodactylum tricornutum for the enhanced accumulation of omega-3 long chain polyunsaturated fatty acids. Metab. Eng. 2014, 22, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.V.; Misquitta, R.W.; Reddy, V.S.; Rao, B.J.; Rajam, M.V. Genetic transformation of the green alga—Chlamydomonas reinhardtii by agrobacterium tumefaciens. Plant Sci. 2004, 166, 731–738. [Google Scholar] [CrossRef]
- Rathod, J.; Prakash, G.; Pandit, R.; Lali, A. Agrobacterium-mediated transformation of promising oil-bearing marine algae Parachlorella kessleri. Photosynth. Res. 2013, 118, 141–146. [Google Scholar] [CrossRef] [PubMed]
- Cheng, R.B.; Ma, R.J.; Li, K.; Rong, H.; Lin, X.Z.; Wang, Z.K.; Yang, S.J.; Ma, Y. Agrobacterium tumefaciens mediated transformation of marine microalgae schizochytrium. Microbiol. Res. 2012, 167, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Hlavova, M.; Turoczy, Z.; Bisova, K. Improving microalgae for biotechnology—from genetics to synthetic biology. Biotechnol. Adv. 2015, 33, 1194–1203. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.; Brueggeman, A.J.; Horken, K.M.; Plucinak, T.M.; Weeks, D.P. Successful transient expression of Cas9 and single guide RNA genes in Chlamydomonas reinhardtii. Eukaryot. Cell 2014, 13, 1465–1469. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.-E.; Lim, J.-M.; Koh, H.G.; Kim, E.K.; Kang, N.K.; Jeon, S.; Kwon, S.; Shin, W.-S.; Lee, B.; Hwangbo, K.; et al. CRISPR/Cas9-induced knockout and knock-in mutations in Chlamydomonas reinhardtii. Sci. Rep. 2016, 6, 27810. [Google Scholar] [CrossRef] [PubMed]
- Nymark, M.; Sharma, A.K.; Sparstad, T.; Bones, A.M.; Winge, P. A crispr/cas9 system adapted for gene editing in marine algae. Sci. Rep. 2016, 6, 24951. [Google Scholar] [CrossRef] [PubMed]
- Khraiwesh, B.; Jijakli, K.; Swift, J.; Chaiboonchoe, A.; Abdrabu, R.; Chao, P.-W.; Yen, L.; Salehi-Ashtiani, K. Prospective applications of synthetic biology for algal bioproduct optimization. In Biomass and Biofuels from Microalgae; Springer International Publishing: Basel, Switzerland, 2015; Volume 2, pp. 137–154. [Google Scholar]
- Karas, B.J.; Molparia, B.; Jablanovic, J.; Hermann, W.J.; Lin, Y.C.; Dupont, C.L.; Tagwerker, C.; Yonemoto, I.T.; Noskov, V.N.; Chuang, R.Y.; et al. Assembly of eukaryotic algal chromosomes in yeast. J. Biol. Eng. 2013, 7, 30. [Google Scholar] [CrossRef] [PubMed]
- Chaiboonchoe, A.; Ghamsari, L.; Dohai, B.; Ng, P.; Khraiwesh, B.; Jaiswal, A.; Jijakli, K.; Koussa, J.; Nelson, D.R.; Cai, H.; et al. Systems level analysis of the Chlamydomonas reinhardtii metabolic network reveals variability in evolutionary co-conservation. Mol. BioSyst. 2016, 12, 2394–2407. [Google Scholar] [CrossRef] [PubMed]
- Dai, Z.; Nielsen, J. Advancing metabolic engineering through systems biology of industrial microorganisms. Curr. Opin. Biotechnol. 2015, 36, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Guarnieri, M.T.; Pienkos, P.T. Algal omics: Unlocking bioproduct diversity in algae cell factories. Photosynth. Res. 2015, 123, 255–263. [Google Scholar] [CrossRef] [PubMed]
- Schellenberger, J.; Que, R.; Fleming, R.M.; Thiele, I.; Orth, J.D.; Feist, A.M.; Zielinski, D.C.; Bordbar, A.; Lewis, N.E.; Rahmanian, S. Quantitative prediction of cellular metabolism with constraint-based models: The cobra toolbox v2. 0. Nat. Protoc. 2011, 6, 1290–1307. [Google Scholar] [CrossRef] [PubMed]
- Koussa, J.; Chaiboonchoe, A.; Salehi-Ashtiani, K. Computational approaches for microalgal biofuel optimization: A review. BioMed Res. Int. 2014, 649453. [Google Scholar] [CrossRef] [PubMed]
- Karp, P.D.; Latendresse, M.; Paley, S.M.; Krummenacker, M.; Ong, Q.D.; Billington, R.; Kothari, A.; Weaver, D.; Lee, T.; Subhraveti, P. Pathway tools version 19.0 update: Software for pathway/genome informatics and systems biology. Brief. Bioinform. 2015. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Reed, J.L. Optorf: Optimal metabolic and regulatory perturbations for metabolic engineering of microbial strains. BMC Syst. Biol. 2010, 4, 53. [Google Scholar] [CrossRef] [PubMed]
- Wagner, A.; Zarecki, R.; Reshef, L.; Gochev, C.; Sorek, R.; Gophna, U.; Ruppin, E. Computational evaluation of cellular metabolic costs successfully predicts genes whose expression is deleterious. Proc. Natl. Acad. Sci. USA 2013, 110, 19166–19171. [Google Scholar] [CrossRef] [PubMed]
- Burgard, A.P.; Pharkya, P.; Maranas, C.D. Optknock: A bilevel programming framework for identifying gene knockout strategies for microbial strain optimization. Biotechnol. Bioeng. 2003, 84, 647–657. [Google Scholar] [CrossRef] [PubMed]
- Imam, S.; Schäuble, S.; Valenzuela, J.; López García de Lomana, A.; Carter, W.; Price, N.D.; Baliga, N.S. A refined genome-scale reconstruction of chlamydomonas metabolism provides a platform for systems-level analyses. Plant J. 2015, 1239–1256. [Google Scholar] [CrossRef] [PubMed]
- Chaiboonchoe, A.; Dohai, B.S.; Cai, H.; Nelson, D.R.; Jijakli, K.; Salehi-Ashtiani, K. Microalgal metabolic network model refinement through high-throughput functional metabolic profiling. Front. Bioeng. Biotechnol. 2014, 2, 68. [Google Scholar] [CrossRef] [PubMed]
- Krumholz, E.W.; Yang, H.; Weisenhorn, P.; Henry, C.S.; Libourel, I.G. Genome-wide metabolic network reconstruction of the Picoalga ostreococcus. J. Exp. Bot. 2012, 63, 2353–2362. [Google Scholar] [CrossRef] [PubMed]
- Hunt, K.A.; Folsom, J.P.; Taffs, R.L.; Carlson, R.P. Complete enumeration of elementary flux modes through scalable, demand-based subnetwork definition. Bioinformatics 2014, 30, 1569–1578. [Google Scholar] [CrossRef] [PubMed]
- Levering, J.; Broddrick, J.; Dupont, C.L.; Peers, G.; Beeri, K.; Mayers, J.; Gallina, A.A.; Allen, A.E.; Palsson, B.O.; Zengler, K. Genome-scale model reveals metabolic basis of biomass partitioning in a model diatom. PLoS ONE 2016, 11, e0155038. [Google Scholar] [CrossRef] [PubMed]
- Singh, D.; Carlson, R.; Fell, D.; Poolman, M. Modelling metabolism of the diatom Phaeodactylum tricornutum. Biochem. Soc. Trans. 2015, 43, 1182–1186. [Google Scholar] [CrossRef] [PubMed]
- Gao, C.; Wang, Y.; Shen, Y.; Yan, D.; He, X.; Dai, J.; Wu, Q. Oil accumulation mechanisms of the oleaginous microalga Chlorella protothecoides revealed through its genome, transcriptomes, and proteomes. BMC Genom. 2014, 15, 582. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Xiong, W.; Dai, J.; Wu, Q. Genome-based metabolic mapping and 13c flux analysis reveal systematic properties of an oleaginous microalga Chlorella protothecoides. Plant Physiol. 2015, 167, 586–599. [Google Scholar] [CrossRef] [PubMed]
- Juneja, A.; Chaplen, F.W.; Murthy, G.S. Genome scale metabolic reconstruction of Chlorella variabilis for exploring its metabolic potential for biofuels. Bioresour. Technol. 2016, 213, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Zuñiga, C.; Li, C.-T.; Huelsman, T.; Levering, J.; Zielinski, D.C.; McConnell, B.O.; Long, C.P.; Knoshaug, E.P.; Guarnieri, M.T.; Antoniewicz, M.R. Genome-scale metabolic model for the green alga Chlorella vulgaris utex 395 accurately predicts phenotypes under autotrophic, heterotrophic, and mixotrophic growth conditions. Plant Physiol. 2016, 589–602. [Google Scholar] [CrossRef] [PubMed]
- Bochner, B.R.; Gadzinski, P.; Panomitros, E. Phenotype microarrays for high-throughput phenotypic testing and assay of gene function. Genome Res. 2001, 11, 1246–1255. [Google Scholar] [CrossRef] [PubMed]
- Knoop, H.; Gründel, M.; Zilliges, Y.; Lehmann, R.; Hoffmann, S.; Lockau, W.; Steuer, R. Flux balance analysis of cyanobacterial metabolism: The metabolic network of Synechocystis sp. PCC6803. PLoS Comput. Biol. 2013, 9, e1003081. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, T.; Kajihata, S.; Yoshikawa, K.; Matsuda, F.; Furusawa, C.; Hirasawa, T.; Shimizu, H. Integrated metabolic flux and omics analysis of Synechocystis sp. PCC6803 under mixotrophic and photoheterotrophic conditions. Plant Cell Physiol. 2014, 55, 1605–1612. [Google Scholar] [CrossRef] [PubMed]
- Triana, J.; Montagud, A.; Siurana, M.; Fuente, D.; Urchueguía, A.; Gamermann, D.; Torres, J.; Tena, J.; de Córdoba, P.F.; Urchueguía, J.F. Generation and evaluation of a genome-scale metabolic network model of Synechococcus elongatus PCC7942. Metabolites 2014, 4, 680–698. [Google Scholar] [CrossRef] [PubMed]
- Montagud, A.; Navarro, E.; de Córdoba, P.F.; Urchueguía, J.F.; Patil, K.R. Reconstruction and analysis of genome-scale metabolic model of a photosynthetic bacterium. BMC Syst. Biol. 2010, 4, 156. [Google Scholar] [CrossRef] [PubMed]
- Nogales, J.; Gudmundsson, S.; Knight, E.M.; Palsson, B.O.; Thiele, I. Detailing the optimality of photosynthesis in cyanobacteria through systems biology analysis. Proc. Natl. Acad. Sci. USA 2012, 109, 2678–2683. [Google Scholar] [CrossRef] [PubMed]
- Vitkin, E.; Shlomi, T. Mirage: A functional genomics-based approach for metabolic network model reconstruction and its application to cyanobacteria networks. Genome Biol. 2012, 13, R111. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Prieto, M.A.; Semeniuk, T.A.; Futschik, M.E. Toward a systems-level understanding of gene regulatory, protein interaction, and metabolic networks in cyanobacteria. Front. Genet. 2014, 5, 191. [Google Scholar] [CrossRef] [PubMed]
- Lau, N.-S.; Matsui, M.; Abdullah, A.A.-A. Cyanobacteria: Photoautotrophic microbial factories for the sustainable synthesis of industrial products. BioMed Res. Int. 2015, 754934. [Google Scholar] [CrossRef] [PubMed]
- Johnson, T.J.; Gibbons, J.L.; Gu, L.; Zhou, R.; Gibbons, W.R. Molecular genetic improvements of cyanobacteria to enhance the industrial potential of the microbe: A review. Biotechnol. Prog. 2016. [Google Scholar] [CrossRef] [PubMed]
- Ye, C.; Qiao, W.; Yu, X.; Ji, X.; Huang, H.; Collier, J.; Liu, L. Reconstruction and analysis of the genome-scale metabolic model of schizochytrium limacinum sr21 for docosahexaenoic acid production. BMC Genom. 2015, 16, 799. [Google Scholar] [CrossRef] [PubMed]
- Hao, T.; Han, B.; Ma, H.; Fu, J.; Wang, H.; Wang, Z.; Tang, B.; Chen, T.; Zhao, X. In silico metabolic engineering of Bacillus subtilis for improved production of riboflavin, Egl-237,(R,R)-2, 3-butanediol and isobutanol. Mol. BioSyst. 2013, 9, 2034–2044. [Google Scholar] [CrossRef] [PubMed]
- Rensing, S.A.; Lang, D.; Zimmer, A.D.; Terry, A.; Salamov, A.; Shapiro, H.; Nishiyama, T.; Perroud, P.-F.; Lindquist, E.A.; Kamisugi, Y. The physcomitrella genome reveals evolutionary insights into the conquest of land by plants. Science 2008, 319, 64–69. [Google Scholar] [CrossRef] [PubMed]
- Frank, W.; Decker, E.; Reski, R. Molecular tools to study Physcomitrella patens. Plant Biol. 2005, 7, 220–227. [Google Scholar] [CrossRef] [PubMed]
- Reski, R. Development, genetics and molecular biology of mosses. Bot. Acta 1998, 111, 1–15. [Google Scholar] [CrossRef]
- Trosset, J.-Y.; Carbonell, P. Synthetic biology for pharmaceutical drug discovery. Drug Des. Dev. Ther. 2015, 9, 6285–6302. [Google Scholar] [CrossRef] [PubMed]
- Rorrer, G.L. Bioprocess engineering of phototrophic marine organisms. In Springer Handbook of Marine Biotechnology; Kim, S.-K., Ed.; Springer: Berlin/Heidelberg, Germany, 2015; pp. 257–294. [Google Scholar]
- Holdt, S.L.; Kraan, S. Bioactive compounds in seaweed: Functional food applications and legislation. J. Appl. Phycol. 2011, 23, 543–597. [Google Scholar] [CrossRef]
- Rorrer, G.L.; Mullikin, R.; Huang, B.; Gerwick, W.H.; Maliakal, S.; Cheney, D.P. Production of bioactive metabolites by cell and tissue cultures of marine macroalgae in bioreactor systems. In Plant Cell and Tissue Culture for the Production of Food Ingredients; Fu, T.-J., Singh, G., Curtis, W.R., Eds.; Springer: Boston, MA, USA, 1999; pp. 165–184. [Google Scholar]
- Armin, H. Algae biotechnology–green cell-factories on the rise. Curr. Biotechnol. 2015, 4, 389–415. [Google Scholar]
- Lin, H.; Qin, S. Tipping points in seaweed genetic engineering: Scaling up opportunities in the next decade. Mar. Drugs 2014, 12, 3025–3045. [Google Scholar] [CrossRef] [PubMed]
- Food and Agriculture Organization of the United Nations. Available online: http://www.fao.org/home/en/ (accessed on 30 November 2016).
- Khraiwesh, B.; Qudeimat, E.; Thimma, M.; Chaiboonchoe, A.; Jijakli, K.; Alzahmi, A.; Arnoux, M.; Salehi-Ashtiani, K. Genome-wide expression analysis offers new insights into the origin and evolution of Physcomitrella patens stress response. Sci. Rep. 2015, 5, 17434. [Google Scholar] [CrossRef] [PubMed]
- Reski, R.; Parsons, J.; Decker, E.L. Moss-made pharmaceuticals: From bench to bedside. Plant Biotechnol. J. 2015, 13, 1191–1198. [Google Scholar] [CrossRef] [PubMed]
- Huether, C.M.; Lienhart, O.; Baur, A.; Stemmer, C.; Gorr, G.; Reski, R.; Decker, E.L. Glyco-engineering of moss lacking plant-specific sugar residues. Plant Biol. 2005, 7, 292–299. [Google Scholar] [CrossRef] [PubMed]
- Parsons, J.; Altmann, F.; Graf, M.; Stadlmann, J.; Reski, R.; Decker, E.L. A gene responsible for prolyl-hydroxylation of moss-produced recombinant human erythropoietin. Sci. Rep. 2013, 3, 3019. [Google Scholar] [CrossRef] [PubMed]
- Baur, A.; Reski, R.; Gorr, G. Enhanced recovery of a secreted recombinant human growth factor using stabilizing additives and by co-expression of human serum albumin in the moss Physcomitrella patens. Plant Biotechnol. J. 2005, 3, 331–340. [Google Scholar] [CrossRef] [PubMed]
- Goel, H.L.; Mercurio, A.M. Vegf targets the tumour cell. Nat. Rev. Cancer 2013, 13, 871–882. [Google Scholar] [CrossRef] [PubMed]
- Niederkrüger, H.; Dabrowska-Schlepp, P.; Schaaf, A. Suspension culture of plant cells under phototrophic conditions. In Industrial Scale Suspension Culture of Living Cells; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2014; pp. 259–292. [Google Scholar]
- Rosales-Mendoza, S.; Orellana-Escobedo, L.; Romero-Maldonado, A.; Decker, E.L.; Reski, R. The potential of Physcomitrella patens as a platform for the production of plant-based vaccines. Expert Rev. Vaccines 2014, 13, 203–212. [Google Scholar] [CrossRef] [PubMed]
- Orellana-Escobedo, L.; Rosales-Mendoza, S.; Romero-Maldonado, A.; Parsons, J.; Decker, E.L.; Monreal-Escalante, E.; Moreno-Fierros, L.; Reski, R. An env-derived multi-epitope hiv chimeric protein produced in the moss Physcomitrella patens is immunogenic in mice. Plant Cell Rep. 2015, 34, 425–433. [Google Scholar] [CrossRef] [PubMed]
- Schulte, J.; Reski, R. High throughput cryopreservation of 140 000 Physcomitrella patens mutants. Plant Biol. 2004, 6, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Guiry, M.; Guiry, G. World-Wide Electronic Publication. National University of Ireland, Galway. Available online: http://www.algaebase.org (accessed on 31 October 2016).
- Suzuki, S.; Shirato, S.; Hirakawa, Y.; Ishida, K.-I. Nucleomorph genome sequences of two chlorarachniophytes, Amorphochlora amoebiformis and Lotharella vacuolata. Genome Biol. Evol. 2015, 7, 1533–1545. [Google Scholar] [CrossRef] [PubMed]
- Curtis, B.A.; Tanifuji, G.; Burki, F.; Gruber, A.; Irimia, M.; Maruyama, S.; Arias, M.C.; Ball, S.G.; Gile, G.H.; Hirakawa, Y. Algal genomes reveal evolutionary mosaicism and the fate of nucleomorphs. Nature 2012, 492, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Blanc, G.; Gallot-Lavallée, L.; Maumus, F. Provirophages in the bigelowiella genome bear testimony to past encounters with giant viruses. Proc. Natl. Acad. Sci. USA 2015, 112, E5318–E5326. [Google Scholar] [CrossRef] [PubMed]
- Rasala, B.A.; Mayfield, S.P. Photosynthetic biomanufacturing in green algae; production of recombinant proteins for industrial, nutritional, and medical uses. Photosynth. Res. 2015, 123, 227–239. [Google Scholar] [CrossRef] [PubMed]
- Yamada, T.; Chuchird, N.; Kawasaki, T.; Nishida, K.; Hiramatsu, S. Chlorella viruses as a source of novel enzymes. J. Biosci. Bioeng. 1999, 88, 353–361. [Google Scholar] [CrossRef]
- Hawkins, R.L.; Nakamura, M. Expression of human growth hormone by the eukaryotic alga, chlorella. Curr. Microbiol. 1999, 38, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Misra, N.; Patra, M.C.; Panda, P.K.; Sukla, L.B.; Mishra, B.K. Homology modeling and docking studies of fabh (β-ketoacyl-ACP synthase III) enzyme involved in type II fatty acid biosynthesis of Chlorella variabilis: A potential algal feedstock for biofuel production. J. Biomol. Struct. Dyn. 2013, 31, 241–257. [Google Scholar] [CrossRef] [PubMed]
- Hu, L.; Li, H.; Qin, R.; Xu, R.; Li, J.; Li, L.; Wei, P.; Yang, J. Plant phosphomannose isomerase as a selectable marker for rice transformation. Sci. Rep. 2016, 6, 25921. [Google Scholar] [CrossRef] [PubMed]
- Vogt, A.; Guo, Y.; Tsunoda, S.; Kateriya, S.; Elstner, M.; Hegemann, P. Conversion of a light-driven proton pump into a light-gated ion channel. Sci. Rep. 2015, 5, 16450. [Google Scholar] [CrossRef] [PubMed]
- Botebol, H.; Lesuisse, E.; Šuták, R.; Six, C.; Lozano, J.-C.; Schatt, P.; Vergé, V.; Kirilovsky, A.; Morrissey, J.; Léger, T. Central role for ferritin in the day/night regulation of iron homeostasis in marine phytoplankton. Proc. Natl. Acad. Sci. USA 2015, 112, 14652–14657. [Google Scholar] [CrossRef] [PubMed]
- Blatt, A.; Bauch, M.E.; Pörschke, Y.; Lohr, M. A lycopene β-cyclase/lycopene ε-cyclase/light-harvesting complex-fusion protein from the green alga Ostreococcus lucimarinus can be modified to produce α-carotene and β-carotene at different ratios. Plant J. 2015, 82, 582–595. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.; Chen, H.; Gu, Z.; Zhang, H.; Chen, W.; Chen, Y.Q. Application of a delta-6 desaturase with α-linolenic acid preference on eicosapentaenoic acid production in Mortierella alpina. Microb. Cell Fact. 2016, 15, 117. [Google Scholar] [CrossRef] [PubMed]
- von Der Heyde, E.L.; Klein, B.; Abram, L.; Hallmann, A. The inducible nita promoter provides a powerful molecular switch for transgene expression in Volvox carteri. BMC Biotechnol. 2015, 15, 5. [Google Scholar] [CrossRef] [PubMed]
- Adarme-Vega, T.C.; Lim, D.K.Y.; Timmins, M.; Vernen, F.; Li, Y.; Schenk, P.M. Microalgal biofactories: A promising approach towards sustainable omega-3 fatty acid production. Microb. Cell Fact. 2012, 11, 96. [Google Scholar] [CrossRef] [PubMed]
- Chlamydomonas Resource Center. Methods. Available online: http://www.chlamycollection.org/methods/ (accessed on 30 November 2016).
- Quinn, P.; Bowers, R.M.; Zhang, X.; Wahlund, T.M.; Fanelli, M.A.; Olszova, D.; Read, B.A. cDna microarrays as a tool for identification of biomineralization proteins in the Coccolithophorid Emiliania huxleyi (haptophyta). Appl. Environ. Microbiol. 2006, 72, 5512–5526. [Google Scholar] [CrossRef] [PubMed]
- Endo, H.; Yoshida, M.; Uji, T.; Saga, N.; Inoue, K.; Nagasawa, H. Stable nuclear transformation system for the Coccolithophorid alga Pleurochrysis carterae. Sci. Rep. 2016, 6, 22252. [Google Scholar] [CrossRef] [PubMed]
- Sineshchekov, O.A.; Govorunova, E.G.; Jung, K.-H.; Zauner, S.; Maier, U.-G.; Spudich, J.L. Rhodopsin-mediated photoreception in cryptophyte flagellates. Biophys. J. 2005, 89, 4310–4319. [Google Scholar] [CrossRef] [PubMed]
- Vieler, A.; Wu, G.; Tsai, C.-H.; Bullard, B.; Cornish, A.J.; Harvey, C.; Reca, I.-B.; Thornburg, C.; Achawanantakun, R.; Buehl, C.J. Genome, functional gene annotation, and nuclear transformation of the heterokont oleaginous alga Nannochloropsis oceanica CCMP1779. PLoS Genet. 2012, 8, e1003064. [Google Scholar] [CrossRef] [PubMed]
- Iwai, M.; Hori, K.; Sasaki-Sekimoto, Y.; Shimojima, M.; Ohta, H. Manipulation of oil synthesis in nannochloropsis strain nies-2145 with a phosphorus starvation–inducible promoter from Chlamydomonas reinhardtii. Front. Microbiol. 2015, 6, 912. [Google Scholar] [CrossRef] [PubMed]
- Charrier, B.; Rolland, E.; Gupta, V.; Reddy, C. Production of genetically and developmentally modified seaweeds: Exploiting the potential of artificial selection techniques. Front. Plant Sci. 2015, 6, 127. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.; Zhang, L.; Tao, Y.; Guo, L.; Sun, J.; Li, X.; Zhao, N.; Peng, J.; Li, X.; Zeng, L. Construction of a high density snp linkage map of kelp (Saccharina japonica) by sequencing taq I site associated DNA and mapping of a sex determining locus. BMC Genom. 2015, 16, 189. [Google Scholar] [CrossRef] [PubMed]
- Trentacoste, E.M.; Shrestha, R.P.; Smith, S.R.; Glé, C.; Hartmann, A.C.; Hildebrand, M.; Gerwick, W.H. Metabolic engineering of lipid catabolism increases microalgal lipid accumulation without compromising growth. Proc. Natl. Acad. Sci. USA 2013, 110, 19748–19753. [Google Scholar] [CrossRef] [PubMed]
- Moog, D.; Stork, S.; Zauner, S.; Maier, U.-G. In silico and in vivo investigations of proteins of a minimized eukaryotic cytoplasm. Genome Biol. Evol. 2011, 3, 375–382. [Google Scholar] [CrossRef] [PubMed]
- Karas, B.J.; Diner, R.E.; Lefebvre, S.C.; McQuaid, J.; Phillips, A.P.; Noddings, C.M.; Brunson, J.K.; Valas, R.E.; Deerinck, T.J.; Jablanovic, J. Designer diatom episomes delivered by bacterial conjugation. Nat. Commun. 2015, 6, 6925. [Google Scholar] [CrossRef] [PubMed]
- Sumiya, N.; Fujiwara, T.; Kobayashi, Y.; Misumi, O.; Miyagishima, S.-Y. Development of a heat-shock inducible gene expression system in the red alga Cyanidioschyzon merolae. PLoS ONE 2014, 9, e111261. [Google Scholar] [CrossRef] [PubMed]
- Takusagawa, M.; Nakajima, Y.; Saito, T.; Misumi, O. Primitive red alga Cyanidioschyzon merolae accumulates storage glucan and triacylglycerol under nitrogen depletion. J. Gen. Appl. Microbiol. 2016, 62, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Rulong, L. Pyropia conchocelis: Potential as an algal source for carotenoid extraction. Am. J. BioSci. 2015, 3, 121. [Google Scholar] [CrossRef]
- Takenaka, S.; Sugiyama, S.; Ebara, S.; Miyamoto, E.; Abe, K.; Tamura, Y.; Watanabe, F.; Tsuyama, S.; Nakano, Y. Feeding dried purple laver (nori) to vitamin b12-deficient rats significantly improves vitamin b12 status. Br. J. Nutr. 2007, 85, 699. [Google Scholar] [CrossRef]
- Cian, R.E.; Drago, S.R.; de Medina, F.S.; Martinez-Augustin, O. Proteins and carbohydrates from red seaweeds: Evidence for beneficial effects on gut function and microbiota. Mar. Drugs 2015, 13, 5358–5383. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.H.; Kim, E.-Y.; Mikami, K.; Nam, T.J. Chemoprotective effects of a recombinant protein from pyropia yezoensis and synthetic peptide against acetaminophen-induced chang liver cell death. Int. J. Mol. Med. 2015, 36, 369–376. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, M.; Valentao, P.; Andrade, P.B. Biologically active oxylipins from enzymatic and nonenzymatic routes in macroalgae. Mar. Drugs 2016, 14, 23. [Google Scholar] [CrossRef] [PubMed]
- Thornton, D.C.; Chen, J. Exopolymer production as a function of cell permeability and death in a diatom (Thalassiosira weissflogii) and a cyanobacterium (Synechococcus elongatus). J. Phycol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Takaichi, S. Carotenoids in algae: Distributions, biosyntheses and functions. Mar. Drugs 2011, 9, 1101–1118. [Google Scholar] [CrossRef] [PubMed]
- Chénard, C.; Wirth, J.F.; Suttle, C.A. Viruses infecting a freshwater filamentous cyanobacterium (Nostoc sp.) encode a functional crispr array and a proteobacterial DNA polymerase B. mBio 2016, 7, e00667-00616. [Google Scholar] [CrossRef] [PubMed]
- Heidorn, T.; Camsund, D.; Huang, H.-H.; Lindberg, P.; Oliveira, P.; Stensjö, K.; Lindblad, P. Synthetic biology in cyanobacteria engineering and analyzing novel functions. Methods Enzymol. 2010, 497, 539–579. [Google Scholar]
- Shishido, T.K.; Humisto, A.; Jokela, J.; Liu, L.; Wahlsten, M.; Tamrakar, A.; Fewer, D.P.; Permi, P.; Andreote, A.P.; Fiore, M.F.; et al. Antifungal compounds from cyanobacteria. Mar. Drugs 2015, 13, 2124–2140. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Xu, X.; Gu, L.; Hildreth, M.; Zhou, R. Simultaneous gene inactivation and promoter reporting in cyanobacteria. Appl. Microbiol. Biotechnol. 2015, 99, 1779–1793. [Google Scholar] [CrossRef] [PubMed]
- Gangl, D.; Zedler, J.A.Z.; Rajakumar, P.D.; Martinez, E.M.R.; Riseley, A.; Wlodarczyk, A.; Purton, S.; Sakuragi, Y.; Howe, C.J.; Jensen, P.E.; et al. Biotechnological exploitation of microalgae. J. Exp. Bot. 2015, 66, 6975–6990. [Google Scholar] [CrossRef] [PubMed]
- Ledford, H. Gene-editing surges as us rethinks regulations. Nat. News 2016, 532, 158–159. [Google Scholar] [CrossRef] [PubMed]
- U.S Food & Drug Administration: Statement of Policy-Foods Derived from New Plant Varieties. Available online: http://www.fda.gov/Food/GuidanceRegulation/GuidanceDocumentsRegulatoryInformation/Biotechnology/ucm096095.htm (accessed on 30 November 2016).
- Greenwell, H.C.; Laurens, L.M.L.; Shields, R.J.; Lovitt, R.W.; Flynn, K.J. Placing microalgae on the biofuels priority list: A review of the technological challenges. J. R. Soc. Interface 2010, 7, 703–726. [Google Scholar] [CrossRef] [PubMed]
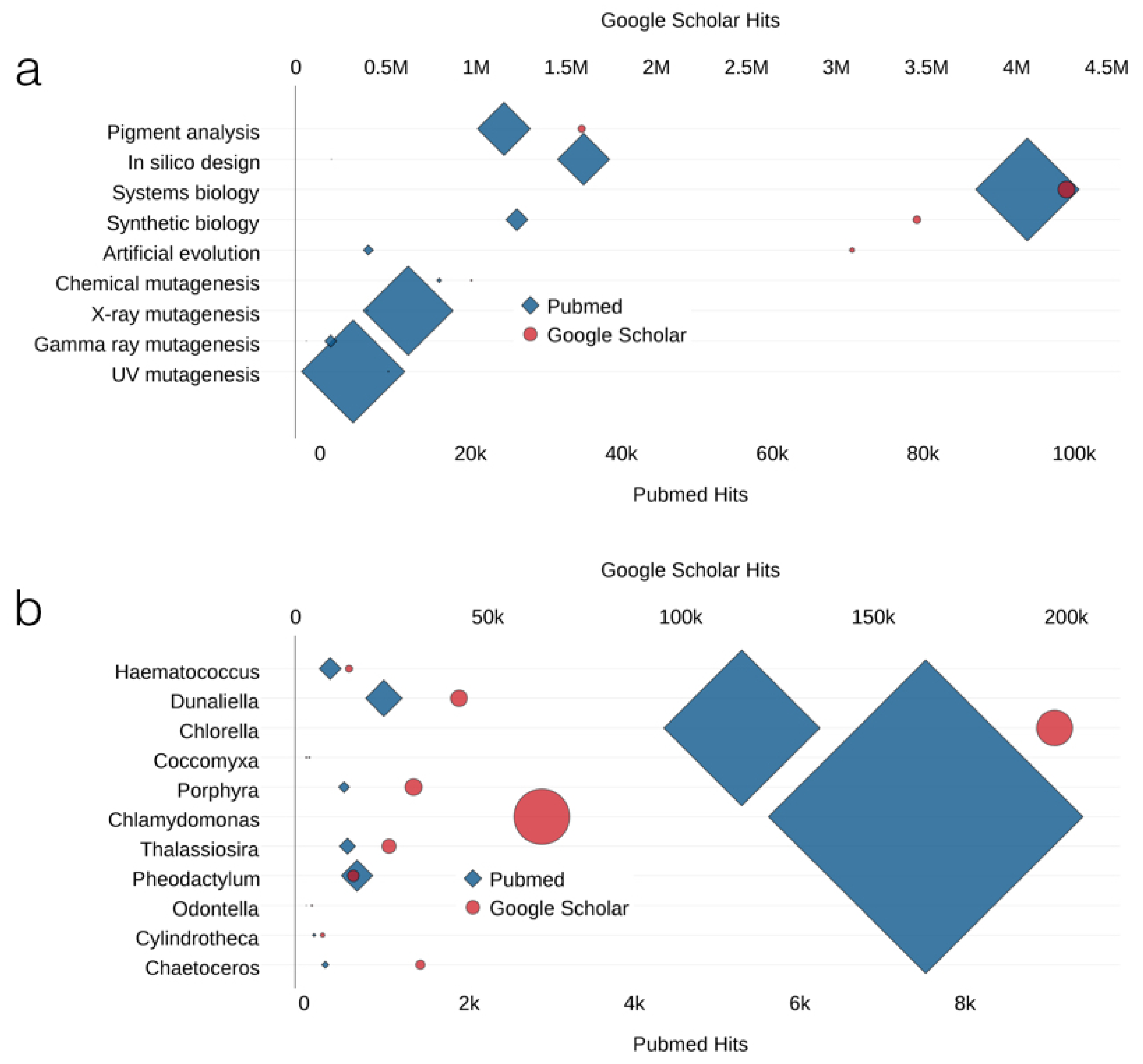
| Species | Group | Environment | Bioactive Compounds | Properties | Genetic Tools | Ref. 1 |
|---|---|---|---|---|---|---|
| Bigelowiella natans | Cercozoa | marine | Genome; transcriptome under diurnal cycle; Viral elements known | [112,113,114] | ||
| Chlorella sp. | Chlorophyta | freshwater | Lipids, human growth hormone | Human nutrition, biofuels, medicines | Genes for active enzymes cloned; in silico models | [115,116,117,118,119] |
| Coccomyxa subellipsoidea | Chlorophyta | freshwater | Flounder growth hormone | Flounder fry exposed to Chlorella-expressed fGH for 30 days exhibited a 25% increase in both total length and width | Trans gene expression | [115,120] |
| Ostreococcus lucimarinus | Chlorophyta | marine | asymmetric carotenoids | Antioxidant molecules, human nutrition | Trans gene expression | [121,122] |
| Micromonas pusilla | Chlorophyta | marine | Trans gene expression, cDNA libraries | [123] | ||
| Volvox carteri f. nagariensis | Chlorophyta | freshwater | Trans gene expression, cDNA libraries | [124] | ||
| Chlamydomonas reinhardtii | Chlorophyta | freshwater | Human nutrition, biofuels | Trans gene expression, cDNA libraries | [125,126] | |
| Emiliania huxleyi | Haptophyta | marine | Calcium carbonate, dimethyl sulfoxide | Human nutrition, weather influence | cDNA libraries | [127,128] |
| Guillardia theta | Cryptophyta | marine | Functional genes cloned for trans-expression in E. coli | [129] | ||
| Nannochloropsis gaditana | Chrysophyta | marine | lipids | Human nutrition, biofuels | Trans gene expression | [130,131] |
| Ectocarpus siliculosus | Phaeophyta | marine | Trans gene expression | [132] | ||
| Saccharina japonica | Phaeophyta | marine | porphyrin derivatives (pheophorbide a, pheophytin a) | anti-inflammatory activity | SNP linkage map | [2,133] |
| Thalassiosira oceanica | Phaeophyta | marine | cDNA libraries, RNAi, cloning of functional genes | [134] | ||
| Thalassiosira pseudonana | Phaeophyta | marine | lipids | Human nutrition | [125] | |
| Phaeodactylum tricornutum | Phaeophyta | marine | lipids | Human nutrition, biofuels | cDNA libraries, Trans gene expression, in silico models | [125,135,136] |
| Cyanidioschyzon merolae | Rhodophyta | freshwater | lipids | Human nutrition, biofuels | Trans gene expression | [137,138] |
| Pyropia yezoensis | Rhodophyta | marine | carotenoids, vitamin B12, PGP glycoprotein, Usujilene—kind of mycosporine-glycine like amino acid | food applications (nori in suschi), anti-inflammatory activity, antioxidative activity | Functional genes cloned | [2,139,140,141,142] |
| Gracilariopsis lemaneiformis | Rhodophyta | marine | fatty acid (12S-hydroxyeicopentaenoic acid-12S-HEPE) | Human nutrition | Protoplast fusion | [143] |
| Chondrus crispus | Rhodophyta | marine | carrageenan | Food applications | Established qPCR references | [96] |
| Synechococcus elongatus | Cyanophyta | freshwater | exopolymers | Carbon cycling, materials applications | Trans gene expression, Tn5 mutagenesis, fusion PCR, CRISPR | [144] |
| Anabaena variabilis | Cyanophyta | freshwater | β-Carotene hydroxylase | Trans gene expression | [145,146,147] | |
| Anabaena cylindric | Cyanophyta | freshwater | scytophycin | antifungal activity | [148,149] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fu, W.; Chaiboonchoe, A.; Khraiwesh, B.; Nelson, D.R.; Al-Khairy, D.; Mystikou, A.; Alzahmi, A.; Salehi-Ashtiani, K. Algal Cell Factories: Approaches, Applications, and Potentials. Mar. Drugs 2016, 14, 225. https://doi.org/10.3390/md14120225
Fu W, Chaiboonchoe A, Khraiwesh B, Nelson DR, Al-Khairy D, Mystikou A, Alzahmi A, Salehi-Ashtiani K. Algal Cell Factories: Approaches, Applications, and Potentials. Marine Drugs. 2016; 14(12):225. https://doi.org/10.3390/md14120225
Chicago/Turabian StyleFu, Weiqi, Amphun Chaiboonchoe, Basel Khraiwesh, David R. Nelson, Dina Al-Khairy, Alexandra Mystikou, Amnah Alzahmi, and Kourosh Salehi-Ashtiani. 2016. "Algal Cell Factories: Approaches, Applications, and Potentials" Marine Drugs 14, no. 12: 225. https://doi.org/10.3390/md14120225
APA StyleFu, W., Chaiboonchoe, A., Khraiwesh, B., Nelson, D. R., Al-Khairy, D., Mystikou, A., Alzahmi, A., & Salehi-Ashtiani, K. (2016). Algal Cell Factories: Approaches, Applications, and Potentials. Marine Drugs, 14(12), 225. https://doi.org/10.3390/md14120225







