Ceramide as a Target of Marine Triterpene Glycosides for Treatment of Human Myeloid Leukemia
Abstract
:1. Introduction
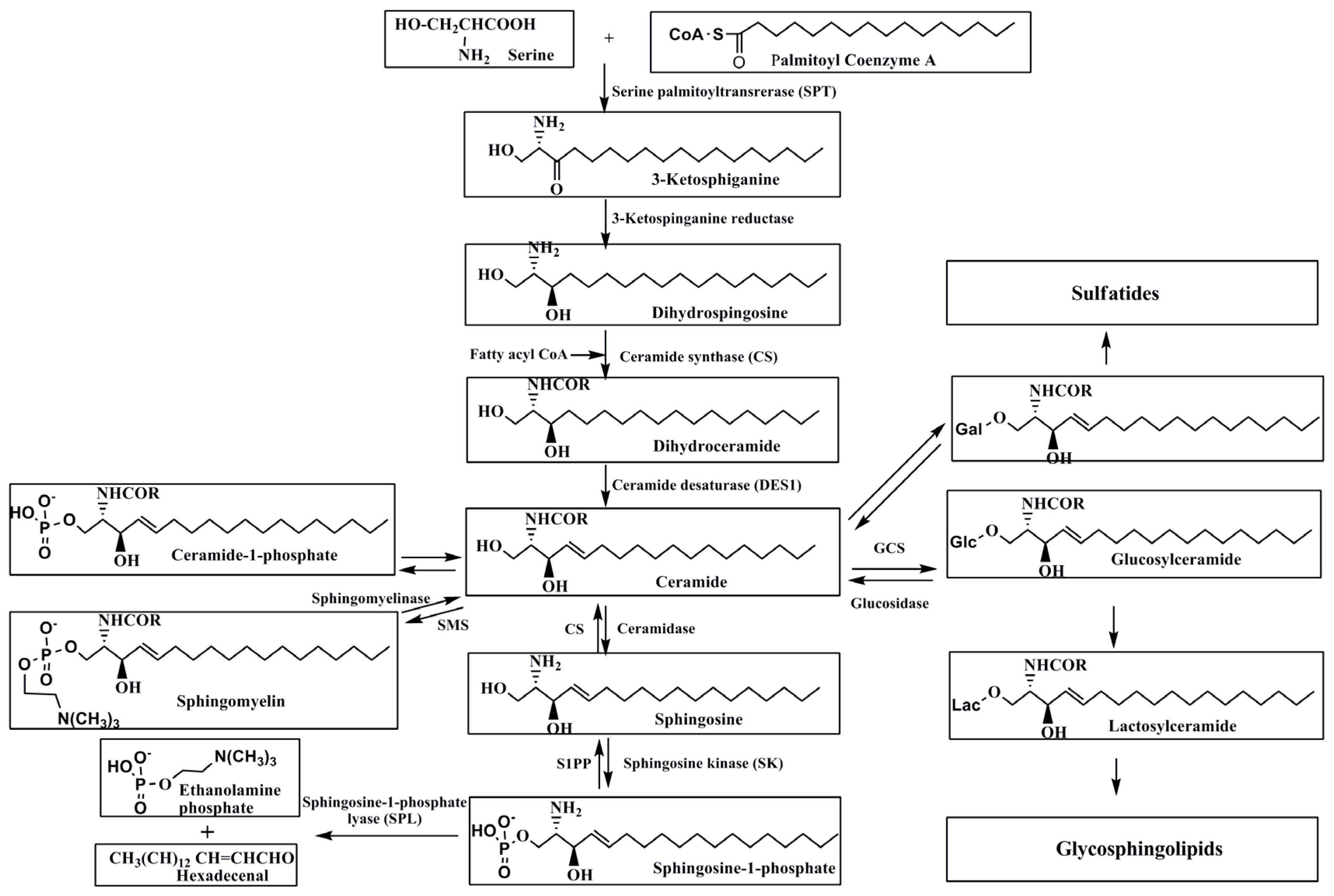
2. Metabolism of Sphingolipids
2.1. De Novo Synthesis of Sphingolipids
2.2. Degradation of Sphingolipids
3. Role of Ceramide in Leukemia
3.1. Induction of Cell Differentiation
3.2. Induction of Apoptosis
3.3. Induction of Autophagy
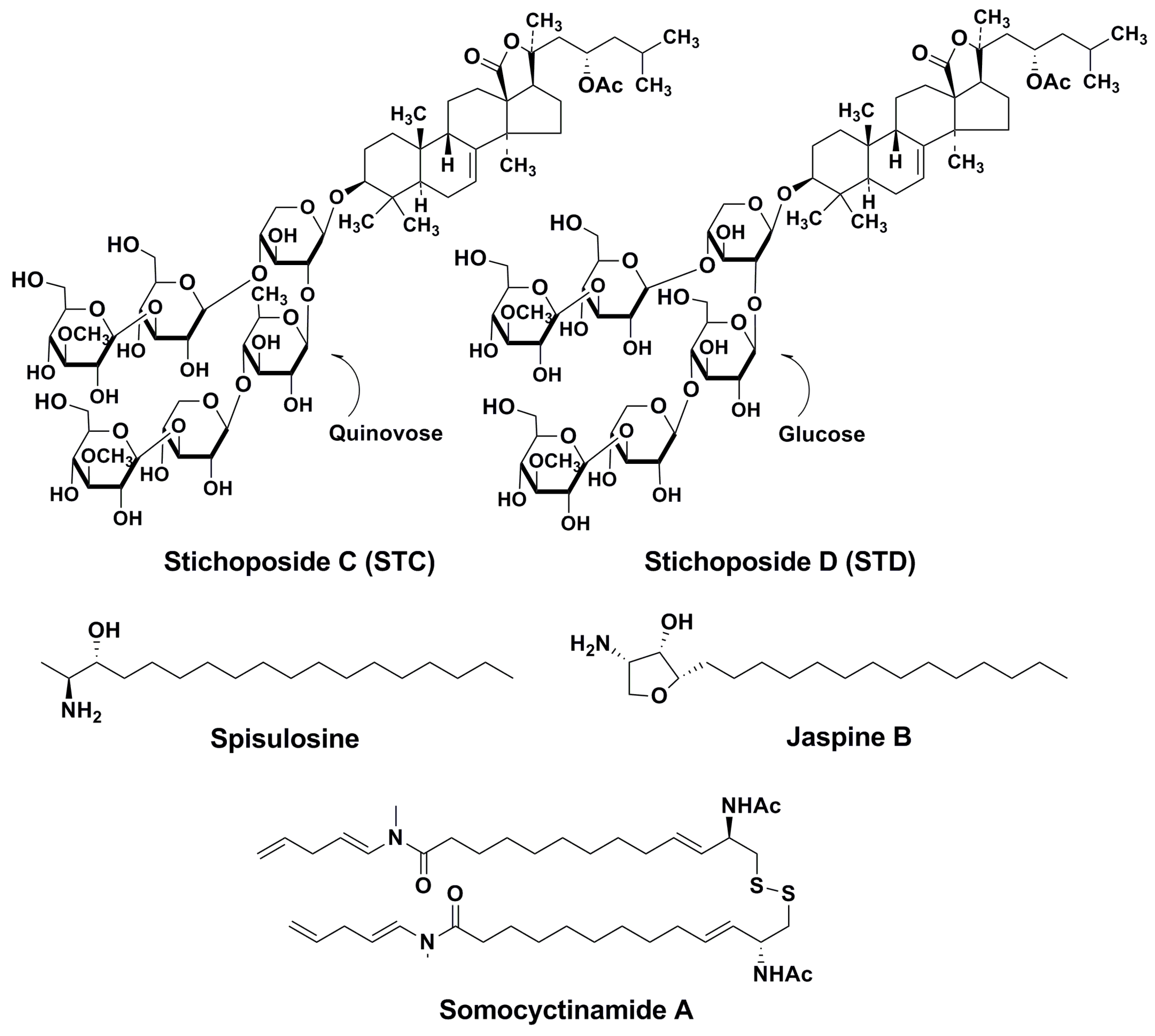
4. Action Mechanisms of Stichoposides Related to Ceramide Generation. Some Other Marine Natural Products with Similar Action Mechanisms
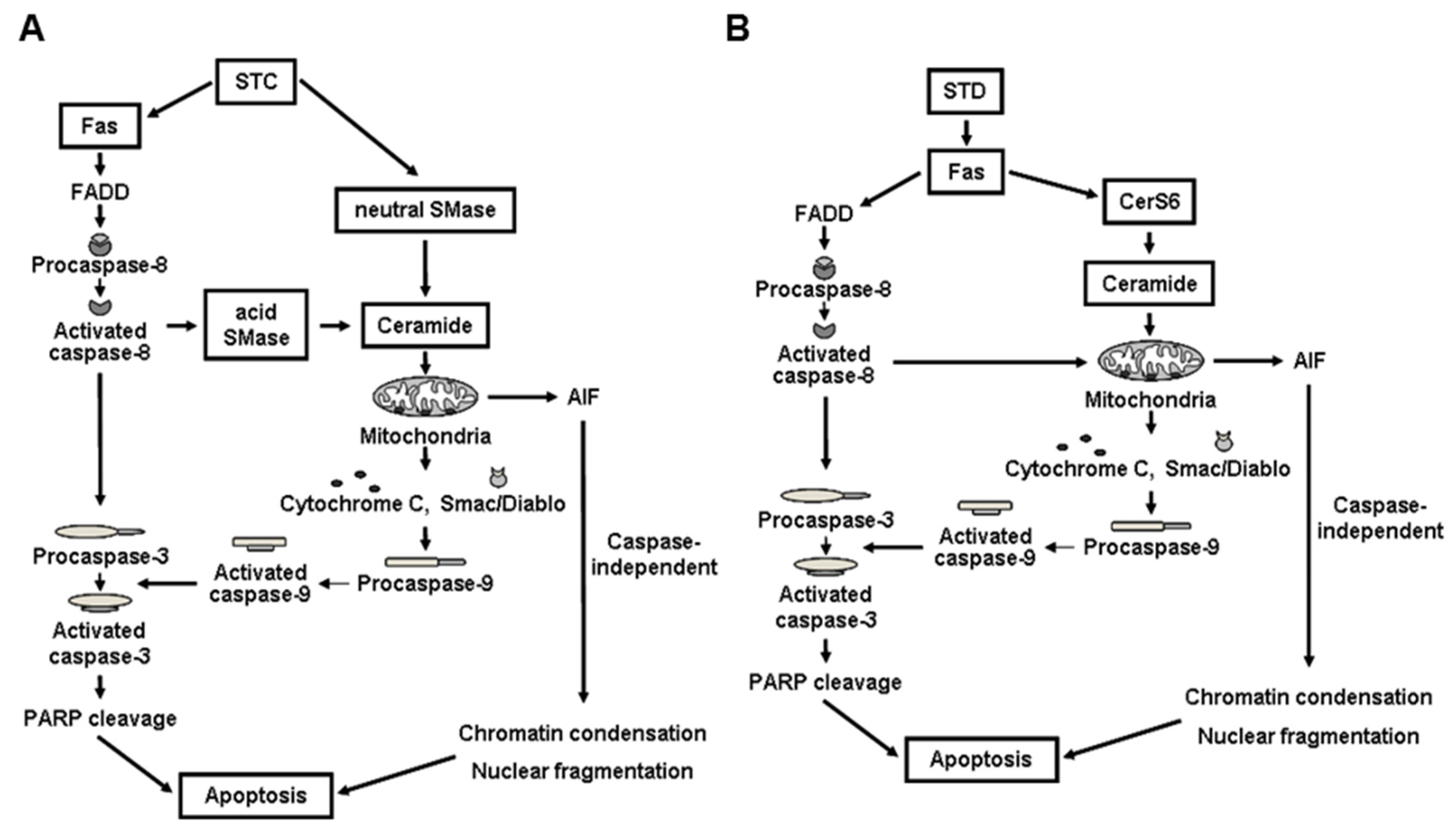
4.1. STC
4.2. STD
4.3. Some Other Marine Inducers of Ceramide Accumulation
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Adada, M.; Canals, D.; Hannun, Y.A.; Obeid, L.M. Sphingolipid regulation of ezrin, radixin, and moesin proteins family: Implications for cell dynamics. Biochim. Biophys. Acta 2014, 1841, 727–737. [Google Scholar] [CrossRef] [PubMed]
- Maceyka, M.; Spiegel, S. Sphingolipid metabolites in inflammatory disease. Nature 2014, 510, 58–67. [Google Scholar] [CrossRef] [PubMed]
- Truman, J.P.; Garcia-Barros, M.; Obeid, L.M.; Hannun, Y.A. Evolving concepts in cancer therapy through targeting sphingolipid metabolism. Biochim. Biophys. Acta 2014, 1841, 1174–1188. [Google Scholar] [CrossRef] [PubMed]
- Obeid, L.M.; Linardic, C.M.; Karolak, L.A.; Hannun, Y.A. Programmed cell death induced by ceramide. Science 1993, 259, 1769–1771. [Google Scholar] [CrossRef] [PubMed]
- Okazaki, T.; Bell, R.M.; Hannun, Y.A. Sphingomyelin turnover induced by vitamin D3 in HL-60 cells. Role in cell differentiation. J. Biol. Chem. 1989, 264, 19076–19080. [Google Scholar] [PubMed]
- Gault, C.R.; Obeid, L.M.; Hannun, Y.A. An overview of sphingolipid metabolism: From synthesis to breakdown. Adv. Exp. Med. Biol. 2010, 688, 1–23. [Google Scholar] [PubMed]
- Park, J.W.; Park, W.J.; Futerman, A.H. Ceramide synthases as potential targets for therapeutic intervention in human diseases. Biochim. Biophys. Acta 2014, 1841, 671–681. [Google Scholar] [CrossRef] [PubMed]
- Kalinin, V.I.; Aminin, D.L.; Avilov, S.A.; Silchenko, A.S.; Stonik, V.A. Triterpene glycosides from sea cucumbers (Holothurioidae, Echinodermata), biological activities and functions. In Studies in Natural Product Chemistry (Bioactive Natural Products); Atta-ur-Rahman, Ed.; Elsevier Science Publisher: Amsterdam, The Netherlands, 2008; pp. 135–196. [Google Scholar]
- Kitagawa, I. Research of biologically active marine natural products. Yakugaku Zasshi 1988, 108, 398–416. [Google Scholar] [PubMed]
- Stonik, V.A.; Maltsev, I.I.; Kalinovsky, A.I.; Conde, K.; Elyakov, G.B. Glycosides of marine-invertebrates. XI. The two novel triterpene glycosides from holothurian of Stichopodidae family. Chem. Nat. Prod. 1982, 18, 177–182. [Google Scholar]
- Stonik, V.A.; Maltsev, I.I.; Elyakov, G.B. Structures of thelenoside-A and thelenoside-B from the sea cucumber Thelenota ananas. Chem. Nat. Prod. 1982, 18, 182–186. [Google Scholar]
- Yun, S.H.; Park, E.S.; Shin, S.W.; Na, W.Y.; Han, J.Y.; Jeong, J.S.; Shastina, X.V.; Stonik, V.A.; Park, J.I.; Kwak, J.Y. Stichoposide C induces apoptosis through the generation of ceramide in leukemia and colorectal cancer cells and shows in vivo antitumor activity. Clin. Cancer Res. 2012, 18, 5934–5948. [Google Scholar] [CrossRef] [PubMed]
- Kitatani, K.; Taniguchi, M.; Okazaki, T. Role of sphingolipids and metabolizing enzymes in hematological malignancies. Mol. Cells 2015, 38, 482–495. [Google Scholar] [CrossRef] [PubMed]
- Hanada, K. Serine palmitoyltransferase, a key enzyme of sphingolipid metabolism. Biochim. Biophys. Acta 2003, 1632, 16–30. [Google Scholar] [CrossRef]
- Hanada, K.; Hara, T.; Nishijima, M. Purification of the serine palmitoyltransferase complex responsible for sphingoid base synthesis by using affinity peptide chromatography techniques. J. Biol. Chem. 2000, 275, 8409–8415. [Google Scholar] [CrossRef] [PubMed]
- Stiban, J.; Tidhar, R.; Futerman, A.H. Ceramide synthases: Roles in cell physiology and signaling. Adv. Exp. Med. Biol. 2010, 688, 60–71. [Google Scholar] [PubMed]
- Hannun, Y.A.; Obeid, L.M. Many ceramides. J. Biol. Chem. 2011, 286, 27855–27862. [Google Scholar] [CrossRef] [PubMed]
- Laviad, E.L.; Albee, L.; Pankova-Kholmyansky, I.; Epstein, S.; Park, H.; Merrill, A.H., Jr.; Futermann, A.H. Characterization of ceramide synthase 2: Tissue distribution, substrate specificity, and inhibition by sphingosine 1-phosphate. J. Biol. Chem. 2008, 283, 5677–5684. [Google Scholar] [CrossRef] [PubMed]
- Mesicek, J.; Lee, H.; Feldman, T.; Jiang, X.; Skobeleva, A.; Berdyshev, E.V.; Haimovitz-Friedman, A.; Fuks, Z.; Kolesnick, R. Ceramide synthases 2, 5, and 6 confer distinct roles in radiation-induced apoptosis in HeLa cells. Cell. Signal. 2010, 22, 1300–1307. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Cuenca, S.; Barbarroja, N.; Vidal-Puig, A. Dihydroceramide desaturase 1, the gatekeeper of ceramide induced lipotoxicity. Biochim. Biophys. Acta 2015, 1851, 40–50. [Google Scholar] [CrossRef] [PubMed]
- Yamaji, T.; Hanada, K. Sphingolipid metabolism and interorganellar transport: Localization of sphingolipid enzymes and lipid transfer proteins. Traffic 2015, 16, 101–122. [Google Scholar] [CrossRef] [PubMed]
- Futerman, A.H.; Hannun, Y.A. The complex life of simple sphingolipids. EMBO Rep. 2004, 5, 777–782. [Google Scholar] [CrossRef] [PubMed]
- Futerman, A.H.; Riezman, H. The ins and outs of sphingolipid synthesis. Trends Cell Biol. 2005, 15, 312–318. [Google Scholar] [CrossRef] [PubMed]
- Dinur, T.; Osiecki, K.M.; Legler, G.; Gatt, S.; Desnick, R.J.; Grabowski, G.A. Human acid β-glucosidase: Isolation and amino acid sequence of a peptide containing the catalytic site. Proc. Natl. Acad. Sci. USA 1986, 83, 1660–1664. [Google Scholar] [CrossRef] [PubMed]
- Grabowski, G.A. Gaucher disease. Enzymology, genetics, and treatment. Adv. Hum. Genet. 1993, 21, 377–441. [Google Scholar] [PubMed]
- Park, J.H.; Schuchman, E.H. Acid ceramidase and human disease. Biochim. Biophys. Acta 2006, 1758, 2133–2138. [Google Scholar] [CrossRef] [PubMed]
- Kitatani, K.; Idkowiak-Baldys, J.; Hannun, Y.A. The sphingolipid salvage pathway in ceramide metabolism and signaling. Cell. Signal. 2008, 20, 1010–1018. [Google Scholar] [CrossRef] [PubMed]
- Tettamanti, G.; Bassi, R.; Viani, P.; Riboni, L. Salvage pathways in glycosphingolipid metabolism. Biochimie 2003, 85, 423–437. [Google Scholar] [CrossRef]
- Pitson, S.M. Regulation of sphingosine kinase and sphingolipid signaling. Trends Biochem. Sci. 2011, 36, 97–107. [Google Scholar] [CrossRef] [PubMed]
- Saba, J.D.; de la Garza-Rodea, A.S. S1P lyase in skeletal muscle regeneration and satellite cell activation: Exposing the hidden lyase. Biochim. Biophys. Acta 2013, 1831, 167–175. [Google Scholar] [CrossRef] [PubMed]
- Pyne, S.; Lee, S.C.; Long, J.; Pyne, N.J. Role of sphingosine kinase and lipid phosphate phosphatases in regulating spatial sphingosine 1-phosphate signaling in health and disease. Cell. Signal. 2009, 21, 14–21. [Google Scholar] [CrossRef] [PubMed]
- James, S.Y.; Williams, M.A.; Kelsey, S.M.; Newland, A.C.; Colston, K.W. The role of vitamin D derivatives and retinoids in the differentiation of human leukaemia cells. Biochem. Pharmcol. 1997, 54, 625–634. [Google Scholar] [CrossRef]
- Okazaki, T.; Bielawska, A.; Bell, R.M.; Hannun, Y.A. Role of ceramide as a lipid mediator of 1α,25-dihydroxyvitamin D3-induced cell differentiation. J. Biol. Chem. 1990, 265, 15823–15831. [Google Scholar] [PubMed]
- Okazaki, T.; Bielawska, A.; Domae, N.; Bell, R.M.; Hannun, Y.A. Characteristics and partial purification of a novel cytosolic, magnesium-independent, neutral sphingomyelinase activated in the early signal transduction of 1α,25-dihydroxyvitamin D3-induced HL-60 cell differentiation. J. Biol. Chem. 1994, 269, 4070–4077. [Google Scholar] [PubMed]
- Langmann, T.; Buechler, C.; Ries, S.; Schaeffler, A.; Aslanidis, C.; Schuierer, M.; Weiler, M.; Sandhoff, K.; de Jong, P.J.; Schmitz, G. Transcription factors Sp1 and AP-2 mediate induction of acid sphingomyelinase during monocytic differentiation. J. Lipid Res. 1999, 40, 870–880. [Google Scholar] [PubMed]
- Kim, D.S.; Kim, S.H.; Song, J.H.; Chang, Y.T.; Hwang, S.Y.; Kim, T.S. Enhancing effects of ceramide derivatives on 1,25-dihydrocyvitamin D3-induced differentiation of human HL-60 leukemia cells. Life Sci. 2007, 81, 1638–1644. [Google Scholar] [CrossRef] [PubMed]
- Ogretmen, B.; Hannun, Y.A. Biologically active sphingolipids in cancer pathogenesis and treatment. Nat. Rev. Cancer 2004, 4, 604–616. [Google Scholar] [CrossRef] [PubMed]
- Segui, B.; Andrieu-Abadie, N.; Jaffrezou, J.P.; Benoist, H.; Levade, T. Sphingolipids as modulators of cancer cell death: Potential therapeutic targets. Biochim. Biophys. Acta 2006, 758, 2104–2120. [Google Scholar] [CrossRef] [PubMed]
- Macchia, M.; Barontini, S.; Bertini, S.; Di Bussolo, V.; Fogli, S.; Giovannetti, E.; Grossi, E.; Minutolo, F.; Danesi, R. Design, synthesis, and characterization of the antitumor activity of novel ceramide analogues. J. Med. Chem. 2001, 44, 3994–4000. [Google Scholar] [CrossRef] [PubMed]
- Granot, T.; Milhas, D.; Carpentier, S.; Dagan, A.; Sequi, B.; Gratt, S.; Levade, T. Caspase-dependent and -independent cell death of Jurkat human leukemia cells induced by novel synthetic ceramide analogs. Leukemia 2006, 20, 392–399. [Google Scholar] [CrossRef] [PubMed]
- Dagan, A.; Wang, C.; Fibach, E.; Gatt, S. Synthetic, non-natural sphingolipid analogs inhibit the biosynthesis of cellular sphingolipids, elevate ceramide and induce apoptotic cell death. Biochim. Biophys. Acta 2003, 1633, 161–169. [Google Scholar] [CrossRef]
- Bieberich, E.; Kawaguchi, T.; Yu, R.K. N-Acylatedserinol is a novel ceramide mimic inducing apoptosis in neuroblastoma cells. J. Biol. Chem. 2000, 275, 177–181. [Google Scholar] [CrossRef] [PubMed]
- Struckhoff, A.P.; Bittman, R.; Burow, M.E.; Clejan, S.; Elliott, S.; Hammond, T.; Tang, Y.; Beckman, B.S. Novel ceramide analogs as potential chemotherapeutic agents in breast cancer. J. Pharmacol. Exp. Ther. 2004, 309, 523–532. [Google Scholar] [CrossRef] [PubMed]
- Samsel, L.; Zaidel, G.; Drumgoole, H.M.; Jelovac, D.; Drachenberg, C.; Rhee, J.G.; Brodie, A.M.; Bielawska, A.; Smyth, M.J. The ceramide analog, B13, induces apoptosis in prostate cancer cell lines and inhibits tumor growth in prostate cancer xenografts. Prostate 2004, 58, 382–393. [Google Scholar] [CrossRef] [PubMed]
- Novgorodov, S.A.; Szulc, Z.M.; Luberto, C.; Jones, J.A.; Bielawski, J.; Bielawska, A.; Hannun, Y.A.; Obeid, L.M. Positively charged ceramide is a potent inducer of mitochondrial permeabilization. J. Biol. Chem. 2005, 280, 16096–16105. [Google Scholar] [CrossRef] [PubMed]
- Hannun, Y.A. Functions of ceramide in coordinating cellular response to stress. Science 1996, 274, 1855–1859. [Google Scholar] [CrossRef] [PubMed]
- Hannun, Y.A.; Obeid, L.M. The ceramide-centric universe of lipid-mediated cell regulation: Stress encounters of the lipid kind. J. Biol. Chem. 2002, 277, 25847–25850. [Google Scholar] [CrossRef] [PubMed]
- Ogretmen, B.; Hannun, Y.A. Updates on functions of ceramide in chemotherapy-induced cell death and in multidrug resistance. Drug Resist. Updates 2001, 4, 368–377. [Google Scholar] [CrossRef] [PubMed]
- Chalfant, C.E.; Szuic, Z.; Roddy, P.; Bielawska, A.; Hannun, Y.A. The structural requirements for ceramide activation of serine-threonine protein phosphatases. J. Lipid Res. 2004, 45, 496–506. [Google Scholar] [CrossRef] [PubMed]
- Heinrich, M.; Neumeyer, J.; Jakob, M.; Hallas, C.; Tchikov, V.; Winoto-Morbach, S.; Wickel, M.; Schneider-Brachert, W.; Trauzold, A.; Hethke, A.; et al. Cathepsin D links TNF-induced acid sphingomyelinase to Bid-mediated caspase-9 and -3 activation. Cell Death Differ. 2004, 11, 550–563. [Google Scholar] [CrossRef] [PubMed]
- Nica, A.F.; Tsan, C.C.; Watt, J.C.; Jiffar, T.; Kurinna, S.; Jurasz, P.; Konopleva, M.; Andreeff, M.; Radomski, M.W.; Ruvolo, P.P. Ceramide promotes apoptosis in chronic myelogenous leukemia-derived K562 cells by a mechanism involving caspase-8 and JNK. Cell Cycle 2008, 7, 3362–3370. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Mun, J.Y.; Chun, Y.J.; Choi, K.H.; Kim, M.Y. Bax-dependent apoptosis induced by ceramide in HL-60 cells. FEBS Lett. 2001, 505, 264–268. [Google Scholar] [CrossRef]
- Kim, H.J.; Oh, J.E.; Kim, S.W.; Chun, Y.J.; Kim, M.Y. Ceramide induces p38 MAPK-dependent apoptosis and Bax translocation via inhibition of Akt in HL-60 cells. Cancer Lett. 2008, 260, 88–95. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Ryland, L.; Yang, J.; Liao, A.; Aliaga, C.; Watts, R.; Tan, S.F.; Kaiser, J.; Shanmugavelandy, S.S.; Rogers, A.; et al. Targeting of survivin by nanoliposomal ceramide induces complete remission in a rat model of NK-LGL leukemia. Blood 2010, 116, 4192–4201. [Google Scholar] [CrossRef] [PubMed]
- Iwai, K.; Kondo, T.; Watanabe, M.; Yabu, T.; Taguchi, Y.; Umehara, H.; Takahashi, A.; Uchiyama, T.; Okazaki, T. Ceramide increases oxidative damage due to inhibition of catalase by caspase-3-dependent proteolysis in HL-60 cell apoptosis. J. Biol. Chem. 2003, 278, 9813–9822. [Google Scholar] [CrossRef] [PubMed]
- Herr, I.; Wilhelm, D.; Bohler, T.; Angel, P.; Debatin, K.-M. Activation of CD95 (APO-1/Fas) signaling by ceramide mediates cancer therapy-induced apoptosis. EMBO J. 1997, 20, 6200–6206. [Google Scholar] [CrossRef] [PubMed]
- Scarlatti, F.; Bauvy, C.; Ventruti, A.; Sala, G.; Cluzeaud, F.; Vandewalle, A.; Ghidoni, R.; Codongno, P. Ceramide-mediated macroautophagy involves inhibition of protein kinase B and up-regulation of beclin 1. J. Biol. Chem. 2004, 279, 18384–18391. [Google Scholar] [CrossRef] [PubMed]
- Pattingre, S.; Bauvy, C.; Capentier, S.; Levade, T.; Levine, B.; Codogno, P. Role of JNK1-dependent Bcl-2 phosphorylation in ceramide-induced macroautophagy. J. Biol. Chem. 2009, 284, 2719–2728. [Google Scholar] [CrossRef] [PubMed]
- Taha, T.A.; Mullen, T.D.; Obeid, L.M. A house divided: Ceramide, sphingosine, and sphingosine-1-phosphate in programmed cell death. Biochim. Biophys. Acta 2006, 1758, 2027–2036. [Google Scholar] [CrossRef] [PubMed]
- Strum, J.C.; Ghosh, S.; Bell, R.M. Lipid second messengers. A role in cell growth regulation and cell cycle progression. Adv. Exp. Mol. Biol. 1997, 407, 421–431. [Google Scholar]
- Brown, D.A.; London, E. Functions of lipid rafts in biological membranes. Annu. Rev. Cell Dev. Biol. 1998, 14, 111–136. [Google Scholar] [CrossRef] [PubMed]
- Kolesnick, R.N.; Goni, F.M.; Alonso, A. Compartmentalization of ceramide signaling: Physical foundations and biological effects. J. Cell. Physiol. 2000, 184, 285–300. [Google Scholar] [CrossRef]
- Levade, T.; Jaffrezou, J.P. Signalling sphingomyelinases: Which, where, how and why? Biochim. Biophys. Acta 1999, 1438, 1–17. [Google Scholar] [CrossRef]
- Goni, F.M.; Alonso, A. Sphingomyelinases: Enzymology and membrane activity. FEBS Lett. 2002, 531, 38–46. [Google Scholar] [CrossRef]
- Gulbins, E.; Kolesnick, R. Acid sphingomyelinase-derived ceramide signaling in apoptosis. Subcell. Biochem. 2002, 36, 229–244. [Google Scholar] [PubMed]
- Park, E.S.; Yun, S.H.; Shin, S.W.; Kwak, J.Y.; Park, J.I. Induction of apoptosis and antitumor activity by stichoposide D through the generation of ceramide in human leukemia cells. J. Life Sci. 2012, 22, 760–771. [Google Scholar] [CrossRef]
- Yun, S.H.; Park, E.S.; Shin, S.W.; Ju, M.H.; Han, J.Y.; Jeong, J.S.; Kim, S.H.; Stonik, V.A.; Kwak, J.Y.; Park, J.I. By activating Fas/ceramide synthase 6/p38 kinase in lipid rafts, stichoposide D inhibits growth of leukemia xenografts. Oncotarget 2015, 6, 27596–27612. [Google Scholar] [CrossRef] [PubMed]
- Park, J.I.; Bae, H.R.; Kim, C.G.; Stonik, V.A.; Kwak, J.Y. Relationships between chemical structures and functions of triterpene glycosides isolated from sea cucumbers. Front. Chem. 2014, 2, 77. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, A.M.; Malagarie-Cazenave, S.; Olea, N.; Vara, D.; Cuevas, C.; Diaz-Laviada, I. Spisulosine (ES-285) induces prostate tumor PC-3 and LNCaP cell death by de novo synthesis of ceramide and PKCzeta activation. Eur. J. Pharmacol. 2008, 584, 237–245. [Google Scholar] [CrossRef] [PubMed]
- Vilar, E.; Grunwald, V.; Schoffski, P.; Singer, H.; Salazar, R.; Iglesias, J.L.; Casado, E.; Cullel-Young, M.; Baselga, J.; Tabernero, J. A phase I dose-escalating study of ES-285, a marine sphingolipid-derived compound, with repeat dose administration in patients with advanced solid tumors. Investig. New Drugs 2012, 30, 299–305. [Google Scholar] [CrossRef] [PubMed]
- Salma, J.; Lafont, E.; Therville, N.; Carpentier, S.; Bonnafe, M.J.; Levade, T.; Genisson, Y.; Andrieu-Abadie, N. The natural marine anhydrophytosphingosine, Jaspine B, induces apoptosis in melanoma cells by interfering with ceramide metabolism. Biochem. Pharm. 2009, 78, 477–485. [Google Scholar] [CrossRef] [PubMed]
- Wrasidlo, W.; Mielgo, A.; Torres, V.A.; Barbero, S.; Stoletov, K.; Suyama, T.L.; Klemke, R.L.; Gerwick, W.H.; Carson, D.A.; Stupack, D.G. The marine lipopeptide somocystinamide a triggers apoptosis via caspase 8. Proc. Natl. Acad. Sci. USA 2008, 105, 2313–2318. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yun, S.-H.; Shin, S.-W.; Stonik, V.A.; Park, J.-I. Ceramide as a Target of Marine Triterpene Glycosides for Treatment of Human Myeloid Leukemia. Mar. Drugs 2016, 14, 205. https://doi.org/10.3390/md14110205
Yun S-H, Shin S-W, Stonik VA, Park J-I. Ceramide as a Target of Marine Triterpene Glycosides for Treatment of Human Myeloid Leukemia. Marine Drugs. 2016; 14(11):205. https://doi.org/10.3390/md14110205
Chicago/Turabian StyleYun, Seong-Hoon, Sung-Won Shin, Valentin A. Stonik, and Joo-In Park. 2016. "Ceramide as a Target of Marine Triterpene Glycosides for Treatment of Human Myeloid Leukemia" Marine Drugs 14, no. 11: 205. https://doi.org/10.3390/md14110205
APA StyleYun, S.-H., Shin, S.-W., Stonik, V. A., & Park, J.-I. (2016). Ceramide as a Target of Marine Triterpene Glycosides for Treatment of Human Myeloid Leukemia. Marine Drugs, 14(11), 205. https://doi.org/10.3390/md14110205






