Chitin and Chitosan Preparation from Marine Sources. Structure, Properties and Applications
Abstract
:1. Introduction
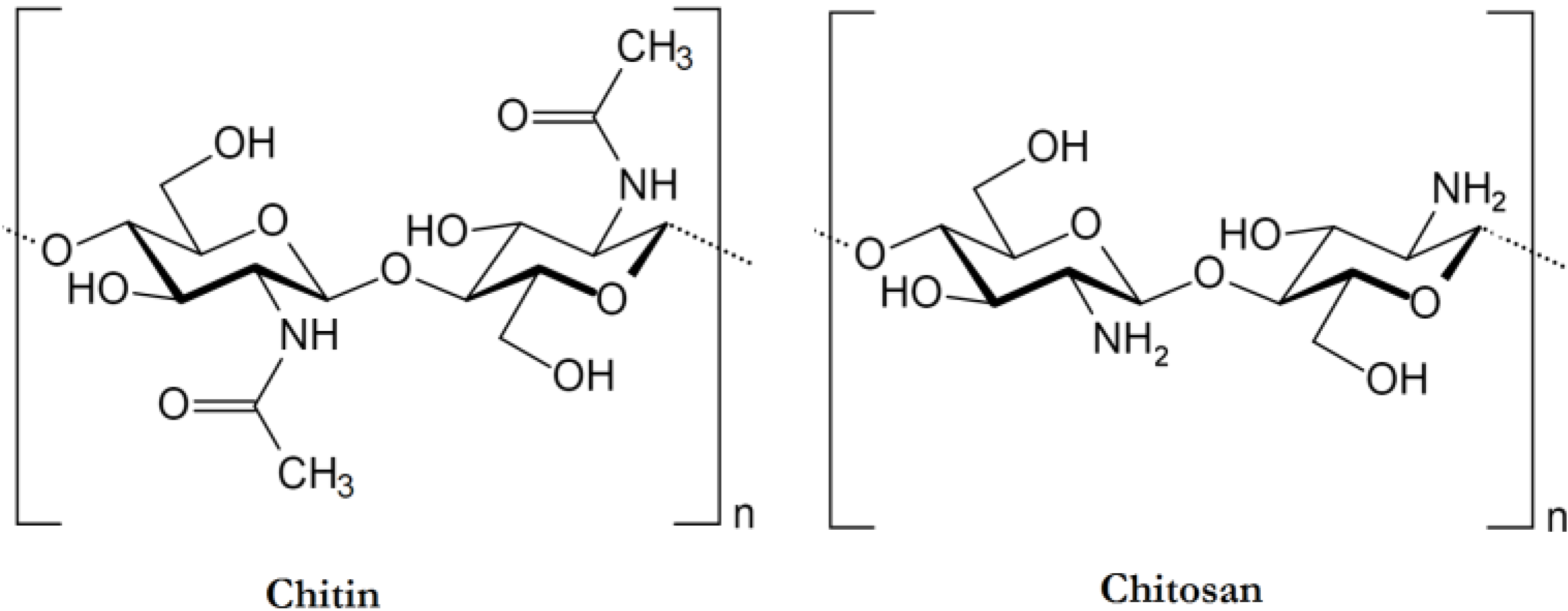
2. Chitin Preparation and Characterization
2.1. Morphology of Chitin
2.2. Chitin Extraction
2.2.1. Chemical Extraction
2.2.1.1. Chemical Deproteinization
2.2.1.2. Chemical Demineralization
| Source | Deproteinization | Demineralization | References | |||||
|---|---|---|---|---|---|---|---|---|
| NaOH Concentration * | Temperature (°C) | Number of Baths | Duration (h) | HCl Concentration* | Temperature (°C) | Duration (h) | ||
| 12 species of crustaceous and cephalopods | 0.3 M | 80–85 | From 2 to 7 according to the source | 1 h for each bath | 0.55 M | Room | 15 mn to 1 h by bath repeated 2–5 times according to the source | [21] |
| Shrimp | 0.125 M | 100 | 1 | 0.5 | 1.25 M | Room | 1 | [36] |
| Shrimp | 1.25 M | 100 | 1 | 0.5 | 1.57 M | 20–22 | 1–3 | [37] |
| Crab | 0.5 M | 65 | 1 | 2 | 1.57 M | Room | 5 | [22] |
| Crab | 1 M | 80 | 1 | 3 | 1 M | Room | 12 | [38] |
| Crab | 1 M | 100 | 1 | 36 | 2 M | Room | 48 | [32] |
| Crab | 1 M | 100 | 3 | 72 | 1 M | Room | - | [23] |
| Crab | 1.25 M | 85–90 | 3 | 24 | 1.37 M | Room | 24 | [39] |
| Crab/Lobster | 2.5 M | Room | 3 | 72 | 11 M | −20 | 4 | [40] |
| Krill | 0.875 M | 90–95 | 1 | 2 | 0.6 M | Room | 2 | [25] |
| Lobster | 1 M | 100 | 5 | 12 | 2 M | Room | 5 | [24] |
| Squid | 2 M | Room | 2 | One night | 1 M | Room | One night | [41] |
| 2 M | 100 | 4 | ||||||
| Lobster | 10% | 100 | 1 | 2.5 | 10% HCl 90% formic | Room | 18 | [26] |
| Krill | 3.5% | 25 | 1 | 2 | 3.5% | 20 | 1.5 | [42] |
| Lobster | 5% | 80–85 | 2 | 0.5 | 5% | 70 | 4 | [43] |
| Crawfish | 3.5% | 65 | 1 | 2 | 1 M | Room | 0.5 | [44] |
| Crab | 1 M | 50 | 1 | 6 | 1 M | 20 | 3 | [30] |
| Shrimp | 1% | 65 | 1 | 1 | 0.5 M | Room | - | [45] |
| Shrimp | 3% | 100 | 1 | 1 | 1 M | Room | 0.5 | [46] |
| Shrimp | 4% | 100 | 1 | 1 | 5% | Room | - | [47] |
2.2.1.3. Processes Preserving Chitin Structure
| Source | Number of Deproteinization Baths | Number of Demineralization Baths | DA | |
|---|---|---|---|---|
| 0.3 M; NaOH 80 °C; 1 h | 0.55 M HCl; 25 °C; 2 h | |||
| Cirripedia | Anatife | 4 | 2 | 100 |
| Reptantia Brachyura | Red crab | 3 | 5 | 97 |
| Marbled crab | 3 | 3 | 99 | |
| Spider crab | 3 | 3 | 96 | |
| Reptantia Macrura | Lobster | 3 | 3 | - |
| Crayfish | 7 | 3 | 100 | |
| slipper lobster | 3 | 2 | - | |
| Freshwater crayfish | 3 | 2 | - | |
| Natantia | Pink shrimp | 3 | 3 | 100 |
| Grey Shrimp | 2 | 2 | 100 | |
| Stomatopoda | Squilla | 3 | 3 | 100 |
| Cephalopoda | Squid | 2 | 2 | 100 |
2.2.2. Biological Extraction of Chitin
2.2.2.1. Enzymatic Deproteinization
2.2.2.2. Fermentation
(a) Lactic Acid Fermentation
(b) Non Lactic-Acid Fermentation
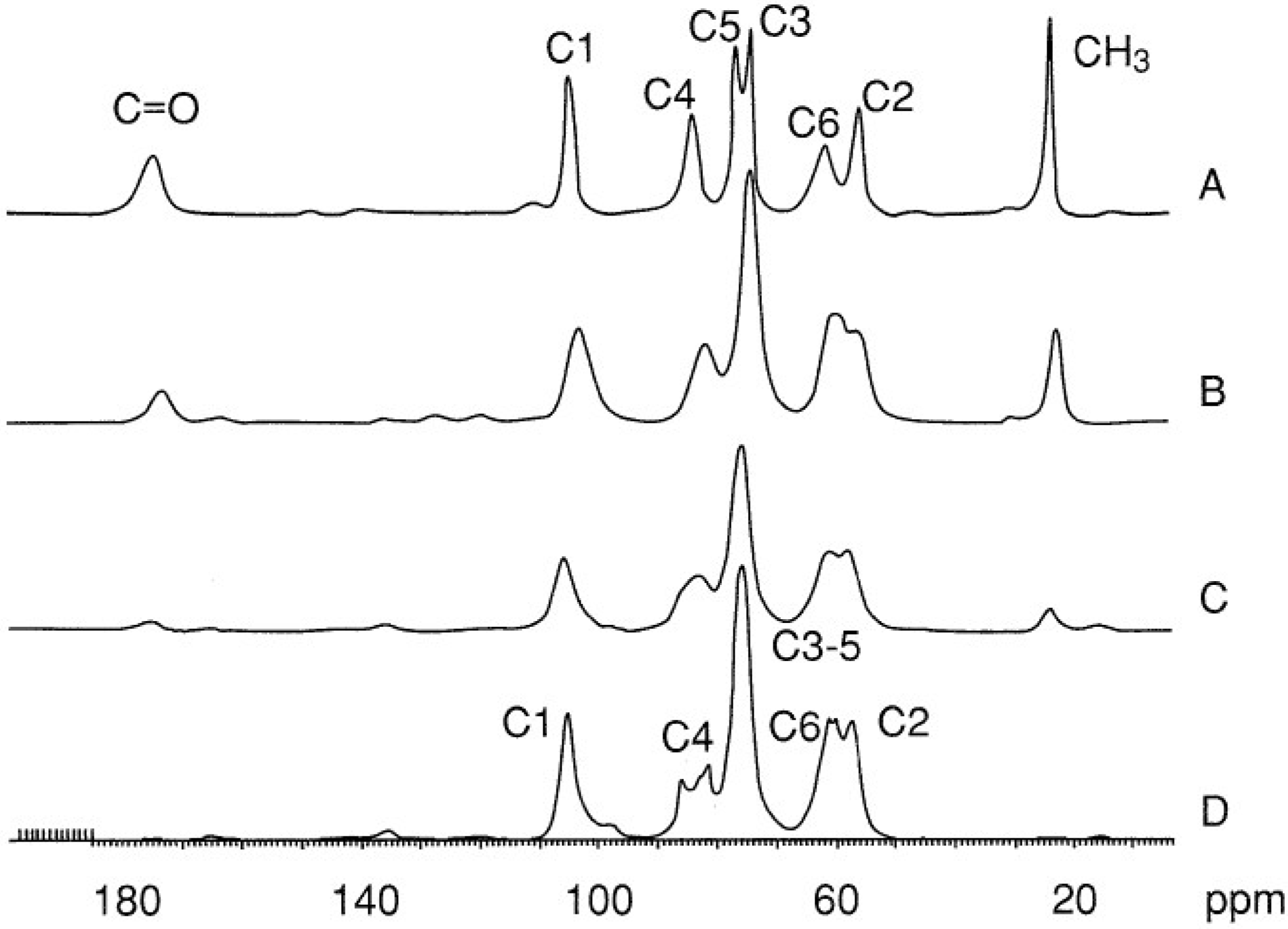
2.3. Chitin Characterization and Solubility
3. Chitosan Preparation and Characterization
3.1. Chitosan Preparation
3.1.1. Chemical Deacetylation
3.1.2. Enzymatic Deacetylation
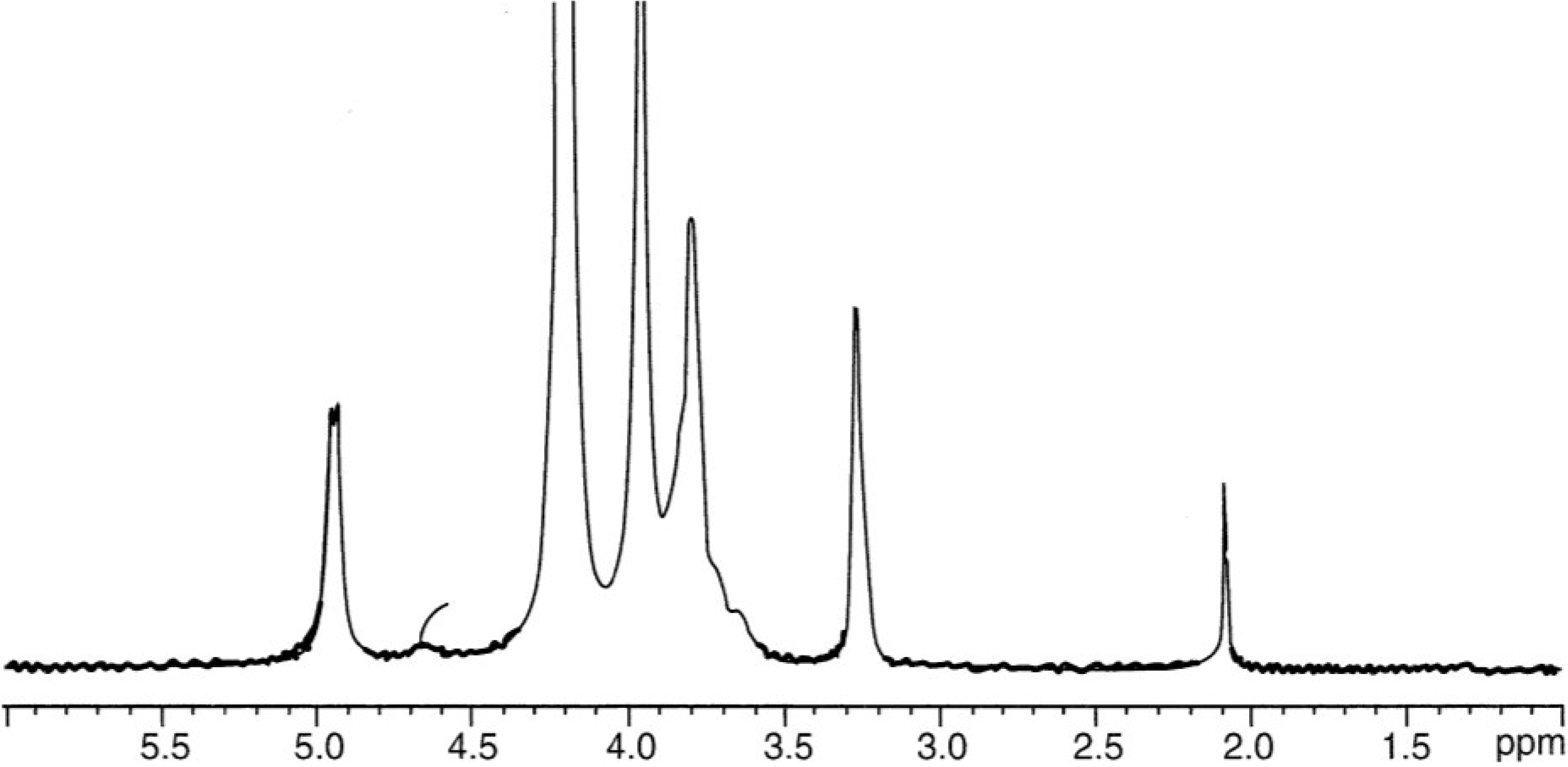
3.2. Chitosan Characterization and Solubility
3.3. Processing and Main Properties of Chitosan-Based Materials
4. Relation between Chemical Structure and Biological Activities
4.1. Antimicrobial Activity
4.2. Antioxidant Activity
4.3. Antitumor Activity
5. Pharmaceutical and Biomedical Applications of Chitin and Chitosan
| Forms | Applications |
|---|---|
| Beads | Drug delivery [266] |
| Microspheres [265] | Enzyme immobilization |
| Gene delivery vehicle [267] | |
| Nanoparticles | Encapsulation of sensitive drugs [172] |
| Coatings | Surface modification |
| Textile finishes | |
| Fibers | Medical textiles |
| Suture | |
| Nanofibers [268] | Guided bone regeneration |
| Scaffold for nerve tissue regeneration | |
| Nonwonen bioactive fibers [269] | Wound healing |
| Films | Wound care |
| Dialysis membrane | |
| Antitumoral [270] | |
| Semi-permeable film for wound dressing [271] | |
| Powder | Adsorbent for pharmaceutical and medical devices |
| Surgical glove powder | |
| Enzyme immobilization | |
| Sponge [272] | Mucosomal hemostatic dressing |
| Wound dressing | |
| Drug delivery [272] | |
| Enzyme entrapment | |
| Artificial skin [271] | |
| Shaped objects | Orthopedics |
| Contact lenses | |
| Solutions | Cosmetics |
| Bacteriostatic agent | |
| Hemostatic agent | |
| Anticoagulants | |
| Antitumor agent | |
| Gene delivery [267] | |
| Spermicide [245] | |
| Gels | Delivery vehicle |
| Implants, coating | |
| Tissue engineering | |
| Wound dressing for wet treatment [271] | |
| Tablets | Compressed diluent |
| Disintegrating agent | |
| Excipient [273] | |
| Capsules | Delivery vehicle |
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Rinaudo, M. Chitin and chitosan: Properties and applications. Prog. Polym. Sci. 2006, 31, 603–632. [Google Scholar] [CrossRef]
- Rinaudo, M. Main properties and current applications of some polysaccharides as biomaterials. Polym. Int. 2008, 57, 397–430. [Google Scholar]
- Rinaudo, M. Physical properties of chitosan and derivatives in sol and gel states. In Chitosan-Based Systems for Biopharmaceuticals: Delivery, Targeting and Polymer Therapeutics; Sarmento, B., das Neves, J., Eds.; John Wiley & Sons: Chichester, UK, 2012; pp. 23–44. [Google Scholar]
- Rinaudo, M. Materials based on chitin and chitosan. In Bio-Based Plastics: Materials and Applications; Kabasci, S., Ed.; John Wiley & Sons: Chichester, UK, 2014; pp. 63–80. [Google Scholar]
- Rudall, K.M.; Kenchington, W. The chitin system. Biol. Rev. 1973, 40, 597–636. [Google Scholar] [CrossRef]
- Blackwell, J. Chitin. In Biopolymers; Walton, A.G., Blackwell, J., Eds.; Academic Press: New York, NY, USA, 1973; pp. 474–489. [Google Scholar]
- Persson, J.E.; Domard, A.; Chanzy, H. Single crystals of a-chitin. Int. J. Biol. Macromol. 1990, 13, 221–224. [Google Scholar]
- Helbert, W.; Sugiyama, J. High-resolution electron microscopy on cellulose II and α-chitin single crystals. Cellulose 1998, 5, 113–122. [Google Scholar] [CrossRef]
- Ruiz-Herrera, J.; Sing, V.O.; van der Woude, W.J.; Bartnicki-Garcia, S. Microfibril assembly by granules of chitin synthetase. Proc. Natl. Acad. Sci. USA 1975, 72, 2706–2710. [Google Scholar] [CrossRef] [PubMed]
- Bartnicki-Garcia, S.; Persson, J.; Chanzy, H. An electron microscope and electron diffraction study of the effect of calcofluor and congo red on the biosynthesis of chitin in vitro. Arch. Biochem. Biophys. 1994, 310, 6–15. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, J.; Sugiyama, J.; Kimura, S.; Imai, T.; Itoh, T.; Watanabe, T.; Kobayashi, S. Artificial chitin spherulites composed of single crystalline ribbons of α-chitin via enzymatic polymerization. Macromolecules 2000, 33, 4155–4160. [Google Scholar] [CrossRef]
- Rudall, K.M. Chitin and its association with other molecules. J. Polym. Sci. Part C 1969, 28, 83–102. [Google Scholar] [CrossRef]
- Blackwell, J.; Parker, K.D.; Rudall, K.M. Chitin in pogonophore tubes. J. Mar. Biol. Assoc. UK 1965, 45, 659–661. [Google Scholar] [CrossRef]
- Gaill, F.; Persson, J.; Sugiyama, P.; Vuong, R.; Chanzy, H. The chitin system in the tubes of deep sea hydrothermal vent worms. J. Struct. Biol. 1992, 109, 116–128. [Google Scholar] [CrossRef]
- Kurita, K.; Tomita, K.; Ishi, S.; Nishimura, S-I.; Shimoda, K. β-chitin as a convenient starting material for acetolysis for efficient preparation of N-acetylchitooligosaccharides. J. Polym. Sci. A Polym. Chem. 1993, 31, 2393–2395. [Google Scholar] [CrossRef]
- Horst, M.N.; Walker, A.N.; Klar, E. The pathway of crustacean chitin synthesis. In The Crustacean Integument: Morphology and Biochemistry; Horst, M.N., Freeman, J.A., Eds.; CRC: Boca Raton, FL, USA, 1993; pp. 113–149. [Google Scholar]
- No, H.K.; Hur, E.Y. Control of foam formation by antifoam during demineralization of crustacean shell in preparation of chitin. J. Agric. Food. Chem. 1998, 46, 3844–3846. [Google Scholar] [CrossRef]
- Percot, A.; Viton, C.; Domard, A. Characterization of shrimp shell deproteinization. Biomacromolecules 2003, 4, 1380–1385. [Google Scholar] [CrossRef] [PubMed]
- Johnson, E.L.; Peniston, Q.P. Utilization of shellfish waste for chitin and chitosan production. In Chemistry & Biochemistry of Marine Food Products; Martin, R.E., Flick, G.J., Hebard, C.E., Ward, D.R., Eds.; AVI Publishing Co.: Westport, CT, USA, 1982; Chapter 19; p. 415. [Google Scholar]
- Shahidi, F.; Synowiecki, J. Isolation and characterization of nutrients and value-added products from snow crab (Chifroeceles opilio) and shrimp (Panda- 111sb orealis) processing discards. J. Agric. Food Chem. 1991, 39, 1527–1532. [Google Scholar] [CrossRef]
- Tolaimate, A.; Desbrieres, J.; Rhazi, M.; Alagui, A. Contribution to the preparation of chitins and chitosans with controlled physico-chemical properties. Polymer 2003, 44, 7939–7952. [Google Scholar] [CrossRef]
- Muzzarelli, R.A.A.; Tanfani, F.; Emanuelli, M.; Gentile, S. The chelation of cupric ions by chitosan membranes [Callinectes sapidus, blue crab shell]. J. Appl. Biochem. 1980, 2, 380–389. [Google Scholar]
- Hackman, R.H. Studies on chitin. I. Enzymatic degradation of chitin and chitin esters. Aust. J. Biol. Sci. 1954, 7, 168–178. [Google Scholar] [PubMed]
- Hakman, R.H.; Goldberg, M. Light-scattering and infrared-spectrophotometric studies of chitin and chitin derivatives. Carbohydr. Res. 1974, 38, 35–45. [Google Scholar] [CrossRef]
- Anderson, G.G.; de Pablo, N.; Romo, C. Antartic krill (Euphausia superba) as a source of chitin and chitosan. In Proceedings of First International Conference on Chitin and Chitosan; Muzzarelli, R.A.A., Priser, E.R., Eds.; Mit Sea Grant Program: Cambridge, MA, USA, 1978; pp. 54–63. [Google Scholar]
- Horowitz, S.T.; Roseman, S.; Blumental, H.J. Preparation of glucosamine oligosaccharides. 1. Separation. J. Am. Chem. Soc. 1957, 79, 5046–5049. [Google Scholar] [CrossRef]
- Synowiecki, J.; Sikorski, Z.E.; Naczk, M. Immobilization of invertase on krill chitin. Biotechnol. Bioeng. 1981, 23, 231–233. [Google Scholar] [CrossRef]
- Foster, A.B.; Webber, J.M. Chitin. Adv. Carbohydr. Chem. 1960, 15, 371–393. [Google Scholar] [PubMed]
- Austin, P.R.; Brine, C.J.; Castle, J.E.; Zikakis, J.P. Chitin: New facets of research. Science 1981, 212, 749–753. [Google Scholar] [CrossRef] [PubMed]
- Brine, C.J.; Austin, P.R. Chitin variability with species and method of preparation. Comp. Biochem. Physiol. 1981, 69B, 283–286. [Google Scholar]
- Peniston, Q.P.; Lohnson, E.L. Process for Demineralization of Crustacea Shells. U.S. Patent 4,066,735, 3 January 1978. [Google Scholar]
- Shimahara, K.; Ohkouchi, K.; Ikeda, M. Chitin Chemistry; Roberts, G.A.F., Ed.; Macmillan Press: London, UK, 1992; p. 56. [Google Scholar]
- Okafor, N. Isolation of chitin from the shell of the cuttlefish, Sepia oficirralis L. Biochim. Biophys. Acta 1965, 101, 193–200. [Google Scholar] [CrossRef] [PubMed]
- Truong, T.; Hausler, R.; Monette, F.; Niquette, P. Fishery industrial waste valorization for the transformation of chitosan by hydrothermo-chemical method. Rev. Sci. Eau 2007, 20, 253–262. [Google Scholar]
- Marquis-Duval, F.O. Isolation et valorisation des constituants de la carapace de la crevette nordique. Ph.D. Dissertation, Laval University, Quebec, Canada, 2008. [Google Scholar]
- Madhavan, P.; Nair, K.G.R. Utilisation of prawn waste-isolation of chitin and its conversion to chitosan. Fish. Technol. 1974, 11, 50–53. [Google Scholar]
- Moorjani, M.N.; Achutha, V.; Khasim, D.I. Parameters affecting the viscosity of chitosan from prawn waste. J. Food Sci. Technol. 1975, 12, 187–189. [Google Scholar]
- Mima, S.; Miya, M.; Iwamoto, R.; Yoshikawa, S. Highly deacetylated chitosan and its properties. J. Appl. Polym. Sci. 1983, 28, 1909–1917. [Google Scholar] [CrossRef]
- Broussignac, P. Un haut polymère naturel peu connu dans l’industrie, Le chitosane. Chim. Ind. Genie Chim. 1968, 99, 1241–1247. [Google Scholar]
- BeMiller, J.N.; Whistler, R.L. Alkaline degradation of amino sugars. J. Org. Chem. 1963, 27, 1161–1164. [Google Scholar] [CrossRef]
- Kurita, K.; Tomita, K.; Tada, T.; Ishii, S.; Nishimura, S.I.; Shimoda, K. Squid chitin as a potential alternative chitin source: Deacetylation behavior and characteristic properties. J. Polym. Sci. Pol. Chem. 1993, 31, 485–491. [Google Scholar] [CrossRef]
- Brzeski, M.M. Concept of chitin chitosan isolation from Antartic Krill (Euphausia superba) shells on a technical scale. In Proceedings of the Second International Conference on Chitin and Chitosan; Hirano, S., Tokura, S., Eds.; The Japan Society of Chitin and Chitosan: Sapporo, Japan, 1982; pp. 15–29. [Google Scholar]
- Blumberg, R.; Southall, C.L.; van Rensburg, N.J.; Volckman, O.B. South African fish products. XXXII—The rock lobster: A study of chitin production from processing wastes. J. Sci. Food Agric. 1951, 2, 571–576. [Google Scholar] [CrossRef]
- No, H.K.; Meyers, S.P.; Lee, K.S. Isolation and characterization of chitin from crawfish shell waste. J. Agric. Food Chem. 1989, 37, 575–579. [Google Scholar] [CrossRef]
- Wu, A.C.M.; Bough, W.A. A study of variables in the chitosan manufacturing process in relation to molecular-weight distribution, chemical characteristics and waste-treatment effectiveness. In Proceedings of the 1st International Conference on Chitin/Chitosan, Boston, USA, 11–13 April 1977; Muzzarelli, R.A.A., Pariser, E.R., Eds.; MIT Sea Grant Program, Massachusetts Institute of Technology: Cambridge, MA, USA, 1978; pp. 88–102. [Google Scholar]
- Bough, W.A.; Salter, W.L.; Wu, A.C.M.; Perkins, B.E. Influence of manufacturing variables on the characteristics and effectiveness of chitosan products 1. Chemical composition, viscosity, and molecular-weight distribution of chitosan products. Biotechnol. Bioeng. 1978, 20, 1931–1943. [Google Scholar] [CrossRef]
- Sluyanarayana Rao, S.V.; Yashodha, K.P.; Mahendrakar, N.S. Puttarajappa. Deacetylation of chitin at low temperature by a novel alkali impregnation technique. Indian J. Technol. 1987, 25, 194–196. [Google Scholar]
- Tolaimate, A. Exploration des gisements chitineux de la faune marine marocaine. Procédé d’extraction de chitines fortement acétylées. Préparation de chitosanes à caractéristiques contrôlées. Ph.D. Dissertation, Cadi Ayyad University, Marrakech, Maroc, 2000. [Google Scholar]
- Khanafari, A.; Marandi, R.; Sanatei, S. Recovery of chitin and chitosan from shrimp waste by chemical and microbial methods. Iran. J. Environ. Health Sci. Eng. 2008, 5, 1–24. [Google Scholar]
- Bustos, R.O.; Healy, M.G. Microbial deproteinization of waste prawn shell. In Proceedings of the Second International Symposium on Environmental Biotechnology; Biotechnology’ 94: Brighton, UK, 1994; pp. 15–25. [Google Scholar]
- Arbia, W.; Arbia, L.; Adour, L.; Amrane, A. Chitin extraction from crustacean shells using biological methods—A review. Food Technol. Biotech. 2013, 51, 12–25. [Google Scholar]
- Gortari, M.C.; Hours, R.A. Biotechnological processes for chitin recovery out of crustacean waste: A mini-review. Electron. J. Biotechnol. 2013, 16, 14–14. [Google Scholar]
- Guerrero Legarreta, I.; Zakaria, Z.; Hall, G.M. Lactic fermentation of prawn waste: Comparison of commercial and isolated starter cultures. In Advances in Chitin Science; Domard, A., Jeuniaux, C., Muzzarelli, R., Roberts, G., Eds.; Jacques Andre publishers: Lyon, France, 1996; Volume I, pp. 399–406. [Google Scholar]
- Cira, L.A.; Huerta, S.; Guerrero, I.; Rosas, R.; Shirai, K. Scaling up of lactic acid fermentation of prawn wastes in packed-bed column reactor for chitin recovery. In Advances in Chitin Science; Peter, M.G., Domard, A., Muzzarelli, R.A.A., Eds.; Potsdam University: Postdam, Germany, 2000; Volume IV, pp. 2–27. [Google Scholar]
- Rao, M.B.; Tanksale, A.M.; Ghatge, M.S.; Deshpande, V.V. Molecular and biotechnological aspects of microbial proteases. Microbiol. Mol. Biol. Rev. 1998, 62, 597–635. [Google Scholar] [PubMed]
- Synowiecki, J.; Al-Khateeb, N.A.A.Q. The recovery of protein hydrolysate during enzymatic isolation of chitin from shrimp Crangon crangon processing discards. Food Chem. 2000, 68, 147–152. [Google Scholar] [CrossRef]
- Gildberg, A.; Stenberg, E. A new process for advanced utilisation of shrimp waste. Process Biochem. 2001, 36, 809–812. [Google Scholar] [CrossRef]
- Manni, L.; Ghorbel-Bellaaj, O.; Jellouli, K.; Younes, I.; Nasri, M. Extraction and characterization of chitin, chitosan, and protein hydrolysates prepared from shrimp waste by treatment with crude protease from Bacillus cereus SV1. Appl. Biochem. Biotechnol. 2010, 162, 345–357. [Google Scholar] [CrossRef] [PubMed]
- Younes, I.; Ghorbel-Bellaaj, O.; Nasri, R.; Chaabouni, M.; Rinaudo, M.; Nasri, M. Chitin and chitosan preparation from shrimp shells using optimized enzymatic deproteinization. Process Biochem. 2012, 47, 2032–2039. [Google Scholar] [CrossRef]
- Mukhin, V.A.; Novikov, V.Y. Enzymatic hydrolysis of proteins from crustaceans of the Barents Sea. Appl. Biochem. Micro+ 2001, 37, 538–542. [Google Scholar] [CrossRef]
- Younes, I.; Nasri, R.; Bkahiria, I.; Jellouli, K.; Nasri, M. New proteases extracted from red scorpionfish (Scorpaena scrofa) viscera: Characterization and application as a detergent additive and for shrimp waste deproteinization. Food Bioprod. Process. 2014. [Google Scholar] [CrossRef]
- Kaur, S.; Dhillon, G.S. Recent trends in biological extraction of chitin from marine shell wastes: A review. Crit. Rev. Biotechnol. 2015, 35, 44–61. [Google Scholar]
- Prameela, K.; Murali Mohan, C.; Smitha, P.V.; Hemablatha, K.P.J. Bioremediation of shrimp biowaste by using natural probiotic for chitin and carotenoid production an alternative method to hazardous chemical method. Int. J. Appl. Biol. Pharm. Technol. 2010, 1, 903–910. [Google Scholar]
- Rao, M.S.; Muñoz, J.; Stevens, W.F. Critical factors in chitin production by fermentation of shrimp biowaste. Appl. Microbiol. Biotechnol. 2000, 54, 808–813. [Google Scholar] [CrossRef] [PubMed]
- Oh, K.T.; Kim, Y.J.; Nguyen, V.N.; Jung, W.J.; Park, R.D. Demineralization of crab shell waste by Pseudomonas aeruginosa F722. Process Biochem. 2007, 42, 1069–1074. [Google Scholar] [CrossRef]
- Choorit, W.; Patthanamanee, W.; Manurakchinakorn, S. Use of response surface method for the determination of demineralization efficiency in fermented shrimp shells. Bioresour. Technol. 2008, 99, 6168–6173. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.K.; Shih, I.L.; Tzeng, Y.M.; Wang, S.L. Production and purification of protease from a Bacillus subtilis that can deproteinize crustacean wastes. Enzyme Microb. Technol. 2000, 26, 406–413. [Google Scholar] [CrossRef] [PubMed]
- Sini, T.K.; Santhosh, S.; Mathew, P.T. Study on the production of chitin and chitosan from shrimp shell by using Bacillus subtilis fermentation. Carbohydr. Res. 2007, 342, 2423–2429. [Google Scholar] [CrossRef] [PubMed]
- Ghorbel-Bellaaj, O.; Younes, I.; Maalej, H.; Hajji, S.; Nasri, M. Chitin extraction from shrimp shell waste using Bacillus bacteria. Int. J. Biol. Macromol. 2012, 51, 1196–1201. [Google Scholar] [CrossRef] [PubMed]
- Ghorbel-Bellaaj, O.; Jellouli, K.; Younes, I.; Manni, L.; Ouled Salem, M.; Nasri, M. A solvent-stable metalloprotease produced by Pseudomonas aeruginosa A2 grown on shrimp shell waste and its application in chitin extraction. Appl. Biochem. Biotechnol. 2011, 164, 410–425. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.L.; Chio, S.H. Deproteination of shrimp and crab shell with the protease of Pseudomonas aeruginosa K-1. Enzy. Microb. Technol. 1998, 22, 629–633. [Google Scholar] [CrossRef]
- Mahmoud, N.S.; Ghaly, A.E.; Arab, F. Unconventional approach for demineralization of deproteinized crustacean shells for chitin production. Am. J. Biochem. Biotechnol. 2007, 3, 1–9. [Google Scholar] [CrossRef]
- Jung, W.J.; Kuk, J.H.; Kim, K.Y.; Park, R.D. Demineralization of red crab shell waste by lactic acid fermentation. Appl. Microbiol. Biotechnol. 2005, 67, 851–854. [Google Scholar] [CrossRef] [PubMed]
- Teng, W.L.; Khor, E.; Tan, T.K.; Lim, L.Y.; Tan, S.C. Concurrent production of chitin from shrimp shells and fungi. Carbohydr. Res. 2001, 332, 305–316. [Google Scholar] [CrossRef] [PubMed]
- Atkins, E.D.T. Conformation in polysaccharides and complex carbohydrates. J. Biosci. 1985, 8, 375–387. [Google Scholar] [CrossRef]
- Gonell, H.W. Roëtgenographische studien an chitin. Z. Physiol. Chem. 1926, 152, 18–30. [Google Scholar] [CrossRef]
- Clark, G.L.; Smith, A.F. X-ray studies of chitin, chitosan, and derivatives. J. Phys. Chem. 1936, 40, 863–879. [Google Scholar] [CrossRef]
- Gardner, K.H.; Blackwell, J. Refinement of the structure of β-chitin. Biopolymers 1975, 14, 1581–1595. [Google Scholar] [CrossRef] [PubMed]
- Minke, R.; Blackwell, J. The structure of α-chitin. J. Mol. Biol. 1978, 120, 167–181. [Google Scholar] [CrossRef] [PubMed]
- Darmon, S.E.; Rudall, K.M. Infra-red and X-ray studies of chitin. Disc. Faraday. Soc. 1950, 9, 251–260. [Google Scholar] [CrossRef]
- Pearson, F.G.; Marchessault, R.H.; Liang, C.Y. Infrared spectra of crystalline polysaccharides. V. Chitin. J. Polym. Sci. 1960, 13, 101–116. [Google Scholar]
- Falk, M.; Smith, D.G.; McLachlan, J.; McInnes, A.G. Studies on chitin (b-(1–4)-linked 2-acetamido-2-deoxy-d-glucan) fibers of the diatom Thalassiosira fluviatilis Hustedt. II.Proton magnetic resonance, infrared and X-ray studies. Can. J. Chem. 1966, 44, 2269–2281. [Google Scholar] [CrossRef]
- Galat, A.; Koput, J.; Popowicz, J. Analyses of infrared amide bands of chitin. Acta Biochim. Pol. 1979, 26, 303–308. [Google Scholar] [PubMed]
- Iwamoto, R.; Miya, M.; Mima, S. Vibrational polarization spectra of α-type chitin. In Chitin and Chitosan, Proceedings of the Second International Conference on Chitin and Chitosan, Sapporo, Japan, 12–14 July 1982; Hirano, S., Tokura, S., Eds.; The Japanese Society of Chitin and Chitosan: Tottori, Japan, 1982; pp. 82–86. [Google Scholar]
- Focher, B.; Naggi, A.; Torri, G.; Cosani, A.; Terbojevich, M. Structural differences between chitin polymorphs and their precipitates from solutions-evidence from CP-MAS 13CNMR, FT-IR and FT-Raman spectroscopy. Carbohydr. Polym. 1992, 17, 97–102. [Google Scholar] [CrossRef]
- Brugnerotto, J.; Lizardi, J.; Goycoolea, F.M.; Arguelles-Monal, W.; Desbrieres, J.; Rinaudo, M. An infrared investigation in relation with chitin and chitosan characterization. Polymer 2001, 42, 3569–3580. [Google Scholar] [CrossRef]
- Saito, H.; Tabeta, R.; Hirano, S. Conformation of chitin and N-acyl chitosans in solid state as revealed by 13C cross polarization/magic angle spinning (CP/MAS) NMR spectroscopy. Chem. Lett. 1981, 10, 1479–1482. [Google Scholar] [CrossRef]
- Tanner, S.F.; Chanzy, H.; Vincendon, M.; Roux, J.C.; Gaill, F. High resolution solid-state carbon-13 nuclear magnetic resonance study of chitin. Macromolecules 1990, 23, 3576–3583. [Google Scholar] [CrossRef]
- Kono, H. Two-dimensional magic angle spinning NMR investigation of naturally occurring chitins: Precise 1H and 13C resonance assignment of α- and β-chitin. Biopolymers 2004, 75, 255–263. [Google Scholar] [CrossRef] [PubMed]
- Heux, L.; Brugnerotto, J.; Desbrieres, J.; Versali, M.F.; Rinaudo, M. Solid state NMR for determination of degree of acetylation of chitin and chitosan. Biomacromolecules 2000, 1, 746–751. [Google Scholar] [CrossRef] [PubMed]
- Poirier, M.; Charlet, G. Chitin fractionation and characterization in N, N-dimethylacetamide/lithium chloride solvent system. Carbohydr. Polym. 2002, 50, 363–370. [Google Scholar] [CrossRef]
- Terbojevich, M.; Carraro, C.; Cosani, A. Solution studies of the chitin-lithium chloride-N, N-dimethylacetamide system. Carbohydr. Res. 1988, 180, 73–86. [Google Scholar] [CrossRef]
- Pillai, C.K.S.; Paul, W.; Sharma, C.P. Chitin and chitosan polymers: Chemistry, solubility and fiber formation. Prog. Polym. Sci. 2009, 34, 641–678. [Google Scholar] [CrossRef]
- Roberts, G.A.F. Structure of chitin and chitosan. In Chitin Chemistry; Roberts, G.A.E., Ed.; Palgrave Macmillan: London, UK, 1992; pp. 85–91. [Google Scholar]
- Kafetzopoulos, D.; Martinou, A.; Bouriotis, V. Bioconversion of chitin to chitosan: Purification and characterization of chitin deacetylase from Mucor rouxii. Proc. Natl. Acad. Sci. USA 1993, 90, 2564–2568. [Google Scholar] [CrossRef] [PubMed]
- Aiba, S.I. Preparation of N-acetylchitooligosaccharides by hydrolysis of chitosan with chitinase followed by N-acetylation. Carbohyd. Res. 1994, 265, 323–328. [Google Scholar] [CrossRef]
- Ilyina, A.V.; Tatarinova, N.Y.; Varlamov, V.P. The preparation of low-molecular-weight chitosan using chitinolytic complex from Streptomyces kurssanovii. Process Biochem. 1999, 34, 875–878. [Google Scholar] [CrossRef]
- Tokuyasu, K.; Mitsutomi, M.; Yamaguchi, I.; Hayashi, K.; Mori, Y. Recognition of chitooligosaccharides and their N-acetyl groups by putative subsites of chitin deacetylase from a deuteromycete, Colletotrichum lindemuthianum. Biochemistry 2000, 39, 8837–8843. [Google Scholar] [CrossRef] [PubMed]
- Kurita, K.; Sannan, T.; Iwakura, Y. Studies on chitin, 4: Evidence for formation of block and random copolymers of N-acetyl-d-glucosamine and d-glucosamine by hetero- and homogeneous hydrolyses. Makromol. Chem. 1977, 178, 3197–3202. [Google Scholar] [CrossRef]
- No, H.K.; Meyers, S.P. Preparation and characterization of chitin and chitosan—A review. J. Aquat. Food Prod. Technol. 1995, 2, 27–52. [Google Scholar] [CrossRef]
- Hajji, S.; Younes, I.; Ghorbel-Bellaaj, O.; Hajji, R.; Rinaudo, M.; Nasri, M.; Jellouli, K. Structural differences between chitin and chitosan extracted from three different marine sources. Int. J. Biol. Macromol. 2014, 65, 298–306. [Google Scholar] [CrossRef] [PubMed]
- Chang, K.L.B.; Tsai, G.; Lee, J.; Fu, W.R. Heterogeneous N-deacetylation of chitin in alkaline solution. Carbohyd. Res. 1997, 303, 327–332. [Google Scholar] [CrossRef]
- Sannan, T.; Kurita, K.; Iwakura, Y. Studies on chitin, 2. Effect of deacetylation on solubility. Makromol. Chem. 1976, 177, 3589–3600. [Google Scholar] [CrossRef]
- Rinaudo, M.; Domard, A. Solution properties of chitosan. In Chitin and Chitosan; Skjak-Bræk, G., Anthonsen, T., Stanford, P., Eds.; Kluwer Academic Publisher: Dordrecht, The Netherlands, 1989; pp. 71–86. [Google Scholar]
- Aiba, S.I. Studies on chitosan: 3. evidence for the presence of random and block copolymer structures in partially N-acetylated chitosans. Int. J. Biol. Macromol. 1991, 13, 40–44. [Google Scholar] [CrossRef] [PubMed]
- Berger, J.; Reist, M.; Chenite, A.; Felt-Baeyens, O.; Mayer, J.M.; Gurny, R. Erratum to Pseudo-thermosetting chitosan hydrogels for biomedical application. Int. J. Pharm. 2005, 28, 197–206. [Google Scholar] [CrossRef]
- Rong, H.C.; Hwa, H.D. Effect of molecular weight of chitosan with the same degree of deacetylation on the thermal, mechanical, and permeability properties of the prepared membrane. Carbohydr. Polym. 1996, 29, 353–358. [Google Scholar] [CrossRef]
- Li, Q.; Dunn, E.T.; Grandmaison, E.W.; Goosen, M.F.A. Applications and properties of chitosan. J. Bioact. Compat. Pol. 1992, 7, 370–397. [Google Scholar] [CrossRef]
- Rege, P.R.; Block, L.H. Chitosan processing: Influence of process parameters during acidic and alkaline hydrolysis and effect of the processing sequence on the resultant chitosan’s properties. Carbohydr. Res. 1999, 321, 235–245. [Google Scholar] [CrossRef]
- Tolaimate, A.; Desbrieres, J.; Rhazi, M.; Alagui, A.; Vincendon, M.; Vottero, P. On the influence of deacetylation process on the physicochemical characteristics of chitosan from squid chitin. Polymer 2000, 41, 2463–2469. [Google Scholar] [CrossRef]
- Tsaih, M.L.; Chen, R.H. The effect of reaction time and temperature during heterogenous alkali deacetylation on degree of deacetylation and molecular weight of resulting chitosan. J. Appl. Polym. Sci. 2003, 88, 2917–2923. [Google Scholar] [CrossRef]
- Sannan, T.; Kurita, K.; Iwakura, Y. Studies on chitin. V. Kinetics of deacetylation reaction. Polym. J. 1977, 9, 649–651. [Google Scholar] [CrossRef]
- Weska, R.F.; Moura, J.M.; Batista, L.M.; Rizzi, J.; Pinto, L.A.A. Optimization of deacetylation in the production of chitosan from shrimp wastes: Use of response surface methodology. J. Food Eng. 2007, 80, 749–753. [Google Scholar] [CrossRef]
- Hwang, K.T.; Jung, S.T.; Lee, G.D.; Chinnan, M.S.; Park, Y.S.; Park, H.J. Controlling molecular weight and degree of deacetylation of chitosan by response surface methodology. J. Agr. Food Chem. 2002, 50, 1876–1882. [Google Scholar] [CrossRef]
- Younes, I.; Ghorbel-Bellaaj, O.; Chaabouni, M.; Rinaudo, M.; Souard, F.; Vanhaverbeke, C.; Nasri, M. Use of a fractional factorial design to study the effects of experimental factors on the chitin deacetylation. Int. J. Biol. Macromol. 2014, 70, 385–390. [Google Scholar] [CrossRef] [PubMed]
- Le Dung, P.; Milas, M.; Rinaudo, M.; Desbrières, J. Water soluble derivatives obtained by controlled chemical modifications of chitosan. Carbohydr. Polym. 1994, 24, 209–214. [Google Scholar] [CrossRef]
- Tsigos, I.; Martinou, A.; Kafetzopoulos, D.; Bouriotis, V. Chitin deacetylases: New, versatile tools in biotechnology. Trends Biotechnol. 2000, 18, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Araki, Y.; Ito, E. A pathway of chitosan formation in Mucor rouxii: Enzymatic deacetylation of chitin. Eur. J. Biochem. 1975, 189, 249–253. [Google Scholar]
- Martinou, A.; Kafetzopoulos, D.; Bouriotis, V. Isolation of chitin deacetylase from Mucor rouxii by immunoaffinity chromatography. J. Chromatogr. 1993, 644, 35–41. [Google Scholar] [CrossRef]
- Gao, X.D.; Katsumoto, T.; Onodera, K. Purification and characterization of chitin deacetylase from Absidia coerulea. J. Biochem. 1995, 117, 257–263. [Google Scholar] [CrossRef] [PubMed]
- Alfonso, C.; Nuero, O.M.; Santamaría, F.; Reyes, F. Purification of a heat stable chitin deacetylase from Aspergillus nidulans and its role in cell wall degradation. Curr. Microbiol. 1995, 30, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Tsigos, I.; Bouriotis, V. Purification and characterization of chitin deacetylase from Colletotrichum lindemuthianum. J. Biol. Chem. 1995, 270, 26286–26291. [Google Scholar] [CrossRef] [PubMed]
- Tokuyasu, K.; Kameyama, M.O.; Hiyashi, K. Purification and characterization of extracellular chitin deacetylase from Colletotrichum lindemuthianum. Biosci. Biotechnol. Biochem. 1996, 60, 1598–1603. [Google Scholar] [CrossRef]
- Sundara, R.G.; Aruchami, M.; Gowri, N. Natural deacetylation of chitin to chitosan in the abdominal cuticle of the physogastric queen of Macrotermes estherae. In Proceeding Second International Conference Chitin/Chitosan, Sapporo, Japan, 12–14 July 1982; Tokura, S., Hirano, S., Eds.; Japanese Soc. Chitin: Tottori, Japan, 1982. [Google Scholar]
- Martinou, A.; Kafetzopoulos, D.; Bouriotis, V. Chitin deacetylation by enzymatic means: Monitoring of deacetylation processes. Carbohydr. Res. 1995, 273, 235–242. [Google Scholar] [CrossRef]
- Martinou, A.; Bouriotis, V.; Stokke, B.T.; Vårum, K.M. Mode of action of chitin deacetylase from M. rouxii on partially N-acetylated chitosans. Carbohydr. Res. 1998, 311, 71–78. [Google Scholar] [CrossRef]
- Philippova, O.E.; Volkov, E.V.; Sitnikova, N.L.; Khokhlov, A.; Desbrières, J.; Rinaudo, M. Two types of hydrophobic aggregates in aqueous solutions of chitosan and its hydrophobic derivative. Biomacromolecules 2001, 2, 483–490. [Google Scholar] [CrossRef] [PubMed]
- Philippova, O.E.; Korchagina, E.V.; Volkov, E.V.; Smirnov, V.A.; Khokhlov, A.R.; Rinaudo, M. Aggregation of some water-soluble derivatives of chitin in aqueous solutions: Role of the degree of acetylation and effect of hydrogen bond breaker. Carbohydr. Polym. 2012, 87, 687–694. [Google Scholar] [CrossRef]
- Maghami, G.G.; Roberts, G.A.F. Evaluation of the viscometric constants for chitosan. Makromol. Chem. 1988, 189, 195–200. [Google Scholar] [CrossRef]
- Auzely, R.; Rinaudo, M. Controlled chemical modifications of chitosan characterization and investigation of original properties. Macromol. Biosci. 2003, 3, 562–565. [Google Scholar] [CrossRef]
- Rinaudo, M.; le Dung, P.; Milas, M. A new and simple method of synthesis of carboxymethyl chitosans. In Advances in Chitin and Chitosan; Brine, C.J., Sanford, P.A., Zitakis, J.P., Eds.; Elsevier: London, UK, 1992; pp. 516–525. [Google Scholar]
- Desbrieres, J.; Martinez, C.; Rinaudo, M. Hydrophobic derivatives of chitosan: Characterization and rheological behaviour. Int. J. Biol. Macromol. 1996, 19, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Rinaudo, M.; Auzely, R.; Vallin, C.; Mullagaliev, I. Specific interactions in modified chitosan systems. Biomacromolecules 2005, 6, 2396–2407. [Google Scholar] [CrossRef] [PubMed]
- Auzely-Velty, R.; Rinaudo, M. New supramolecular assemblies of a cyclodextrin grafted chitosan through specific complexation. Macromolecules 2002, 35, 7955–7962. [Google Scholar] [CrossRef]
- Recillas, M.; Silva, L.L.; Peniche, C.; Goycoolea, F.M.; Rinaudo, M.; Argelles-Monal, W.M. Thermoresponsive behavior of chitosan-g-N-isopropylacrylamide copolymer solutions. Biomacromolecules 2009, 10, 1633–1641. [Google Scholar] [CrossRef] [PubMed]
- Rinaudo, M. New way to crosslink chitosan in aqueous solution. Eur. Polym. J. 2010, 46, 1537–1544. [Google Scholar] [CrossRef]
- Saito, H.; Tabeta, R.; Ogawa, K. High-resolution solid-state 13C-NMR study of chitosan and its salts with acids: Conformational characterization of polymorphs and helical structures as viewed from the conformation-dependent 13C chemical shifts. Macromolecules 1987, 20, 2424–2430. [Google Scholar] [CrossRef]
- Raymond, L.; Morin, F.G.; Marchessault, R.H. Degree of deacetylation of chitosan using conductometric titration and solid-state NMR. Carbohydr. Res. 1993, 246, 331–336. [Google Scholar] [CrossRef]
- Varum, K.M.; Anthonsen, M.W.; Grasdalen, H.; Smisrød, O. Determination of the degree of N-acetylation and the distribution of N-acetyl groups in partially N-deacetylated chitins (chitosans) by high-field n.m.r. spectroscopy. Carbohydr. Res. 1991, 211, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Varum, K.M.; Anthonsen, M.W.; Grasdalen, H.; Smisrød, O. 13 C-NMR studies of the acetylation sequences in partially N-deacetylated chitins (chitosans). Carbohydr. Res. 1991, 217, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Rinaudo, M.; Milas, M.; le Dung, P. Characterization of chitosan. Influence of ionic strength and degree of acetylation on chain expansion. Int. J. Biol. Macromol. 1993, 15, 281–285. [Google Scholar] [CrossRef] [PubMed]
- Brugnerotto, J.; Desbrieres, J.; Roberts, G.; Rinaudo, M. Characterization of chitosan by steric exclusion chromatography. Polymer 2001, 42, 9921–9927. [Google Scholar] [CrossRef]
- Odijk, T. On the ionic-strength dependence of the intrinsic viscosity of DNA. Biopolymers 1979, 18, 3111–3113. [Google Scholar] [CrossRef]
- Mazeau, K.; Perez, S.; Rinaudo, M. Predicted influence of N-acetyl group content on the conformational extension of chitin and chitosan chains. J. Carbohydr. Chem. 2000, 19, 1269–1284. [Google Scholar] [CrossRef]
- Wei, Y.C.; Hudson, S.M.; Mayer, J.M.; Kaplan, D.L. The crosslinking of chitosan fibers. J. Polym. Sci. Part A Polym. Chem. 1992, 30, 2187–2193. [Google Scholar] [CrossRef]
- Welsh, E.R.; Price, R.R. Chitosan cross-linking with a water-soluble, blocked diisocyanate. 2. Solvates and hydrogels. Biomacromolecules 2003, 4, 1357–1361. [Google Scholar] [CrossRef] [PubMed]
- Arguelles-Monal, W.; Goycoolea, F.M.; Peniche, C.; Higuera-Ciapara, I. Rheological study of the chitosan/glutaraldehyde chemical gel system. Polym. Gels Netw. 1998, 6, 429–440. [Google Scholar] [CrossRef]
- Hirano, S.; Yamaguchi, R.; Fukui, N.; Iwata, M. A chitosan oxalate gel: Its conversion to an N-acetylchitosan gel via a chitosan gel. Carbohydr. Res. 1990, 201, 145–149. [Google Scholar] [CrossRef]
- Yamaguchi, R.; Hirano, S.; Arai, Y.; Ito, T. Chitosan salt gels thermally reversible gelation of chitosan. Agric. Biol. Chem. 1978, 42, 1981–1982. [Google Scholar] [CrossRef]
- Yokoyama, A.; Yamamoto, S.; Kawasaki, T.; Kohgo, T.; Nakasu, M. Development of calcium phosphate cement using chitosan and citric acid for bone substitute materials. Biomaterials 2002, 23, 1091–1101. [Google Scholar] [CrossRef] [PubMed]
- Shen, X.; Tong, H.; Jiang, T.; Zhu, Z.; Wan, P.; Hu, J. Homogeneous chitosan/carbonate apatite/citric acid nanocomposites prepared through a novel in situ precipitation. Compos. Sci. Technol. 2007, 67, 2238–2245. [Google Scholar] [CrossRef]
- Hsieh, S.H.; Chen, W.H.; Wei, L.L. A spectroscopic analysis of the reaction mechanism of polycarboxylic acid crosslinking with chitosan and cotton fabric. Cellul. Chem. Technol. 2003, 37, 359–369. [Google Scholar]
- Desai, K.G.H.; Park, H.J. Encapsulation of vitamine C in tripolyphosphate cross-linked chitosan microspheres by spray drying. J. Microencapsul. 2005, 22, 179–192. [Google Scholar] [CrossRef]
- Quemeneur, F.; Rinaudo, M.; Maret, G.; Pepin-Donat, B. Decoration of lipid vesicles by polyelectrolytes: Mechanism and structure. Soft Matter 2010, 6, 4471–4481. [Google Scholar] [CrossRef]
- Rinaudo, M.; Quemeneur, F.; Pepin-Donat, B. Stabilization of liposomes against stress using polyelectrolytes: Interaction mechanisms, influence of pH, molecular weight, and polyelectrolyte structure. Int. J. Polym. Anal. Charact. 2009, 14, 667–677. [Google Scholar] [CrossRef]
- Bordi, F.; Sennato, S.; Truzzolillo, D. Polyelectrolyte-induced aggregation of liposomes: A new cluster phase with interesting applications. J. Phys. Condens. Matter 2009, 21, 203102:1–203102:26. [Google Scholar] [CrossRef]
- Haidar, Z.S.; Hamdy, R.C.; Tabrizian, M. Protein release kinetics for core-shell hybrid nanoparticles based on the layer-by-layer assembly of alginate and chitosan on liposomes. Biomaterials 2008, 29, 1207–1215. [Google Scholar] [CrossRef] [PubMed]
- Boddohi, S.; Killingsworth, C.E.; Kipper, M.J. Polyelectrolyte multilayer assembly as a function of pH and ionic strength using the polysaccharides chitosan and heparin. Biomacromolecules 2008, 9, 2021–2028. [Google Scholar] [CrossRef] [PubMed]
- Hillberg, A.L.; Tabrizian, M. Biorecognition through layer-by-layer polyelectrolyte assembly: In situ hybridation on living cells. Biomacromolecules 2006, 7, 2742–2750. [Google Scholar] [CrossRef] [PubMed]
- Majima, T.; Funakosi, T.; Iwasaki, N.; Yamane, S.T.; Harada, K.; Nonaka, S.; Minami, A.; Nishimura, S.I. Alginate and chitosan polyion complex hybrid fibers for scaffolds in ligament and tendon tissue engineering. J. Orthopaedic. Sci. 2005, 10, 302–307. [Google Scholar] [CrossRef]
- Iwasaki, N.; Yamane, S.T.; Majima, T.; Kasahara, Y.; Minami, A.; Harada, K.; Nanaka, S.; Maekawa, N.; Tamura, H.; Tokura, S.; et al. Feasibility of polysaccharide hybrid materials for scaffolds in cartilage tissue engineering: Evaluation of chondrocyte adhesion to polyion complex fibers prepared from alginate and chitosan. Biomacromolecules 2004, 5, 828–823. [Google Scholar] [CrossRef] [PubMed]
- Chung, T.W.; Yang, J.; Akaike, T.; Cho, K.Y.; Nah, J.W.; Kim, S.I.; Cho, C.S. Preparation of alginate/galactosylated chitosan scaffold for hepatocyte attachment. Biomaterials 2002, 23, 282–283. [Google Scholar]
- Lavertu, M.; Méthot, S.; Tran-Khanh, N.; Buschmann, M.D. High efficiency gene transfer using chitosan/DNA nanoparticles with specific combinations of molecular weight and degree of deacetylation. Biomaterials 2006, 27, 4815–4824. [Google Scholar] [CrossRef] [PubMed]
- Jean, M.; Smaoui, F.; Lavertu, M.; Méthot, S.; Bouhdoud, L.; Buschmann, M.D.; Merzouki, A. Chitosan-plasmid nanoparticle formulations for IM and SC delivery of recombinant FGF-2 and PDGF-BB or generation of antibodies. Gene Ther. 2009, 16, 1097–1110. [Google Scholar] [CrossRef] [PubMed]
- Alameh, M.Z.; Jean, M.; Dejesus, D.; Buschmann, M.D.; Merzouki, A. Chitosanase-based method for RNA isolation from cells transfected with chitosan/siRNA nanocomplexes for real-time RT-PCR in gene delivery. Int. J. Nanomedecine 2010, 5, 473–481. [Google Scholar]
- Thibault, M.; Nimesh, S.; Lavertu, M.; Buschmann, M. Intracellular trafficking and decondensation kinetics of chitosan-pDNA polyplexes. Mol. Ther. 2010, 18, 1787–1795. [Google Scholar] [CrossRef] [PubMed]
- Strand, S.P.; Danielsen, S.; Christensen, B.E.; Varum, K.M. Influence of chitosan structure on the formation and stability of DNA-Chitosan polyelectrolyte complexes. Biomacromolecules 2005, 6, 3357–3366. [Google Scholar] [CrossRef] [PubMed]
- Farkas, V. Fungal cell walls: Their structure, biosynthesis and biotechnological aspects. Acta Biotechnol. 1990, 10, 225–238. [Google Scholar] [CrossRef]
- Fleet, G.H.; Phaff, H.J. Fungal glucans-structure and metabolism. Encycl. Plant Physiol. N S 1981, 13B, 416–440. [Google Scholar]
- Friedman, M.; Juneja, V.K. Review of Antimicrobial and Antioxidative Activities of Chitosans in Food. J. Food Protect. 2010, 73, 1737–1761. [Google Scholar]
- Kardas, I.; Struszczyk, M.H.; Kucharska, M.; van den Broek, L.A.M.; van Dam, J.E.G.; Ciechańska, D. Chitin and chitosan as functional biopolymers for industrial applications. In The European Polysaccharide Network of Excellence (EPNOE). Research Initiatives and Results; Narvard, P., Ed.; Springer-Verlag: Wien, Austria, 2012; pp. 329–374. [Google Scholar]
- Alishahi, A.; Aïder, M. Applications of chitosan in the seafood industry and aquaculture: A review. Food Bioprocess Technol. 2012, 5, 817–830. [Google Scholar] [CrossRef]
- Bordenave, N.; Grelier, S.; Cama, V. Water and moisture susceptibility of chitosan and paper-based materials: Structure-property relationships. J. Agric. Food Chem. 2007, 55, 9479–9488. [Google Scholar] [CrossRef]
- Franklin, T.J.; Snow, G.A. Biochemistry of Antimicrobial Action, 3rd ed.; Chapman and Hall: London, UK, 1981; p. 217. [Google Scholar]
- Synowiecki, J.; Al-khatteb, N.A.A. Production, properties and some new applications of chitin and its derivatives. Crit. Rev. Food Sci. Nut. 2003, 43, 144–171. [Google Scholar] [CrossRef]
- Sudarshan, N.R.; Hoover, D.G.; Knorr, D. Antibacterial action of chitosan. Food Biotechnol. 1992, 6, 257–272. [Google Scholar] [CrossRef]
- Leuba, S.; Stossel, P. Chitosan and other polyamines: Antifungal activity and interaction with biological membranes. In Chitin in Nature and Technology; Muzzarelli, R.A.A., Jeuniaux, C., Gooday, C., Eds.; Plenum Press: New York, NY, USA, 1985; p. 217. [Google Scholar]
- Choi, B.K.; Kim, K.Y.; Yoo, Y.J.; Oh, S.J.; Choi, J.H.; Kim, C.Y. In vitro antimicrobial activity of a chitooligosaccharide mixture against Actinobacillus actinomycetemcomitans and Streptococcus mutans. Int. J. Antimicrob. Agent 2001, 18, 553–557. [Google Scholar]
- Eaton, P.; Fernandes, J.C.; Pereira, E.; Pintado, M.E.; Malcata, F.X. Atomic force microscopy study of the antibacterial effects of chitosans on Escherichia coli and Staphylococcus aureus. Ultramicroscopy 2008, 108, 1128–1134. [Google Scholar]
- Chung, Y.C.; Su, Y.P.; Chen, C.C.; Jia, G.; Wang, H.L.; Wu, J.G.; Lin, J.G. Relationship between antibacterial activity of chitosan and surface characteristics of cell wall. Acta Pharm. Sin. 2004, 25, 932–936. [Google Scholar]
- Jeon, Y.J.; Park, P.J.; Kim, S.K. Antimicrobial effect of chitooligosaccharides produced by bioreactor. Carbohydr. Polym. 2001, 44, 71–76. [Google Scholar] [CrossRef]
- Muzzarelli, R.; Tarsi, R.; Filippini, O.; Giovanetti, E.; Biagini, G.; Varaldo, P.E. Antimicrob. Agents Chemother. 1990, 34, 2019–2023. [CrossRef]
- Rhoades, J.; Roller, S. Antimicrobial actions of degraded and native chitosan against spoilage organisms in laboratory media and foods. Appl. Environ. Microbiol. 2000, 66, 80–86. [Google Scholar] [CrossRef] [PubMed]
- Helander, I.M.; Nurmiaho-Lassila, E.L.; Ahvenainen, R.; Rhoades, J.; Roller, S. Chitosan disrupts the barrier properties of the outer membrane of Gram-negative bacteria. Int. J. Food Microbiol. 2001, 71, 235–244. [Google Scholar] [CrossRef] [PubMed]
- No, H.K.; Young Park, N.; ho Lee, S.; Meyers, S.P. Antibacterial activity of chitosans and chitosan oligomers with different molecular weights. Int. J. Food Microbiol. 2002, 74, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Younes, I.; Sellimi, S.; Rinaudo, M.; Jellouli, K.; Nasri, M. Influence of acetylation degree and molecular weight of homogeneous chitosans on antibacterial and antifungal activities. Int. J. Food Microbiol. 2014, 185, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Zheng, L.Y.; Zhu, J.F. Study on antimicrobial activity of chitosan with different molecular weights. Carbohydr. Polym. 2003, 54, 527–530. [Google Scholar] [CrossRef]
- Benhabiles, M.S.; Salah, R.; Lounici, H.; Drouiche, N.; Goosen, M.F.A.; Mameri, N. Antibacterial activity of chitin, chitosan and its oligomers prepared from shrimp shell waste. Food Hydrocolloid. 2012, 29, 48–56. [Google Scholar] [CrossRef]
- Bell, A.A.; Hubbard, J.C.; Liu, L.; Davis, R.M.; Subbarao, K.V. Effects of chitin and chitosan on the incidence and severity of Fusarium yellows of celery. Plant Dis. 1998, 82, 322–328. [Google Scholar] [CrossRef]
- Ben-Shalom, N.; Ardi, R.; Pinto, R.; Aki, C.; Fallik, E. Controlling gray mould caused by Botrytis cinerea in cucumber plants by means of chitosan. Crop. Prot. 2003, 22, 285–290. [Google Scholar] [CrossRef]
- Wojdyła, A.T. Chitosan (Biochikol 020 PC) in the control of some ornamental foliage diseases. Commun. Agric. Appl. Biol. Sci. 2004, 69, 705–715. [Google Scholar]
- Atia, M.M.M.; Buchenauer, H.; Aly, A.Z.; Abou-Zaid, M.I. Antifungal activity of chitosan against Phytophthora infestans and activation of defence mechanisms in tomato to late blight. Biol. Agric. Hortic. 2005, 23, 175–197. [Google Scholar] [CrossRef]
- Photchanachai, S.; Singkaew, J.; Thamthong, J. Effects of chitosan seed treatment on colletotrichum sp. and seedling growth of chili cv. “jinda”. In ISHS Acta Horticulturae 712, Proceedings of the IV International Conference on Managing Quality in Chains-The Integrated View on Fruits and Vegetables Quality, Bangkok, Thailand, 30 June 2006; Purvis, A.C., McGlasson, W.B., Kanlayanarat, S., Eds.; International Society for Horticultural Science: Leuven, Belgium, 2006; pp. 585–590. [Google Scholar]
- Bai, R.K.; Huang, M.Y.; Jiang, Y.Y. Selective permeabilities of chitosan-acetic acid complex membrane and chitosan-polymer complex membranes for oxygen and carbon dioxide. Polym. Bull. 1988, 20, 83–88. [Google Scholar] [CrossRef]
- El-Ghaouth, A.; Arul, J.; Asselin, A.; Benhamou, N. Antifungal activity of chitosan on two postharvest pathogens of strawberry fruits. Phytopathology 1992, 82, 398–402. [Google Scholar] [CrossRef]
- Ames, B.N.; Gold, L.S.; Willet, W.C. Causes and prevention of cancer. Proc. Nat. Acad. Sci. USA 1995, 92, 5258–5265. [Google Scholar] [CrossRef] [PubMed]
- Pincernail, J. Free radicals and antioxidants in human disease. In Analysis of Free Radicals in Biological Systems; Favier, A.E., Cadet, J., Kalyanaraman, B., Fontecave, M., Pierre, J.L., Eds.; Birkhauser: Basel, Switzerland, 1995; pp. 83–98. [Google Scholar]
- Stadtman, E.R. Protein oxidation and aging. Science 1992, 257, 1220–1224. [Google Scholar] [CrossRef] [PubMed]
- Witztum, J.L. The oxidation hypothesis of atherosclerosis. Lancet 1994, 344, 793–795. [Google Scholar] [CrossRef] [PubMed]
- Ames, B.N.; Shigenaga, M.K.; Hagan, T.M. Oxidants, antioxidants and the degenerative diseases of aging. Proc. Natl. Acad. Sci. USA 1993, 90, 7915–7922. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B.; Murcia, M.A.; Chirico, S.; Aruoma, O.I. Free radicals and antioxidants in food and in vivo: What they do and how they work. Crit. Rev. Food Sci. Nutr. 1995, 35, 7–20. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B. Free radicals, antioxidants and human disease: Curiosity, cause or consequence. Lancet 1994, 344, 721–724. [Google Scholar] [CrossRef] [PubMed]
- Chiang, M.T.; Yao, H.T.; Chen, H.C. Effect of dietary chitosans with different viscosity on plasma lipids and lipid peroxidation in rats fed on a diet enriched with cholesterol. Biosci. Biotech. Bioch. 2000, 5, 965–971. [Google Scholar] [CrossRef]
- Park, P.J.; Je, J.Y.; Kim, S.K. Free radical scavenging activity of chitooligosaccharides by electron spin resonance spectrometry. J. Agric. Food Chem. 2003, 51, 4624–4627. [Google Scholar]
- Sun, T.; Xie, W.; Xu, P. Antioxidant activity of graft chitosan derivatives. Macromol. Biosci. 2003, 3, 320–323. [Google Scholar] [CrossRef]
- Yin, X.Q.; Lin, Q.; Zhang, Q.; Yang, L.C. O2− scavenging activity of chitosan and its metal complexes. Chin. J. Appl. Chem. 2002, 19, 325–328. [Google Scholar]
- Kim, K.W.; Thomas, R.L. Antioxidative activity of chitosans with varying molecular weights. Food Chem. 2007, 101, 308–313. [Google Scholar] [CrossRef]
- Je, J.Y.; Park, P.J.; Kim, S.K. Free radical scavenging properties of hetero-chitooligosaccharides using an ESR spectroscopy. Food Chem. Toxicol. 2004, 42, 381–387. [Google Scholar] [CrossRef] [PubMed]
- Qi, L.; Xu, Z. In vivo antitumor activity of chitosan nanoparticles. Bioorg. Med. Chem. Lett. 2006, 16, 4243–4245. [Google Scholar] [CrossRef] [PubMed]
- Dass, C.R.; Choong, P.F. The use of chitosan formulations in cancer therapy. J. Microencapsul. 2008, 25, 275–279. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.R.; Adams, R.L.; Carubelli, R.; Nordquist, R.E. Laser-photosensitizer assisted immunotherapy: A novel modality for cancer treatment. Cancer Lett. 1997, 115, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, K.; Nishimura, S.; Nishi, N.; Saiki, I.; Tokura, S.; Azuma, I. Immunological activity of chitin and its derivatives. Vaccine 1984, 2, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Tokoro, A.; Tatewaki, N.; Suzuki, K.; Mikami, T.; Suzuki, S.; Suzuki, M. Growth inhibitory effect of hexa-N-acetylchitohexaose and chitohexaos and Meth-A solid tumor. Chem. Pharm. Bull. (Tokyo) 1998, 36, 784–790. [Google Scholar] [CrossRef]
- Murata, J.; Saiki, I.; Nishimura, S.; Nishi, N.; Tokura, S.; Azuma, I. Inhibitory effect of chitin heparinoids on the lung metastasis of B16-BL6 melanoma. Jpn. J. Cancer Res. 1989, 80, 866–872. [Google Scholar] [CrossRef] [PubMed]
- Hasegawa, M.; Yagi, K.; Iwakawa, S.; Hirai, M. Chitosan induces apoptosis via caspase-3 activation in bladder tumor cells. Jpn. J. Cancer Res. 2001, 92, 459–466. [Google Scholar] [CrossRef] [PubMed]
- Qi, L.; Xu, Z.; Chen, M. In vitro and in vivo suppression of hepatocellular carcinoma growth by chitosan nanoparticles. Eur. J. Cancer 2007, 43, 184–193. [Google Scholar] [CrossRef] [PubMed]
- Guminska, M.; Ignacak, J.; Wojcik, E. In vitro inhibitory effect of chitosan and its degradation products on energy metabolism in Ehrlich ascites tumour cells (EAT). Polish J. Pharmacol. 1996, 48, 495–501. [Google Scholar]
- Lin, S.Y.; Chan, H.Y.; Shen, F.H.; Chen, M.H.; Wang, Y.J.; Yu, C.K. Chitosan prevents the development of AOM-induced aberrant crypt foci in mice and suppressed the proliferation of AGS cells by inhibiting DNA synthesis. J. Cell Biochem. 2007, 100, 1573–1580. [Google Scholar] [CrossRef] [PubMed]
- Jeon, Y.J.; Kim, S.K. Antitumor activity of chitosan oligosaccharides produced in an ultra filtration membrane reactor system. J. Microbiol. Biotechn. 2002, 12, 503–507. [Google Scholar]
- Suzuki, K.; Mikami, T.; Okawa, Y.; Tokoro, A.; Suzuki, S.; Suzuki, M. Antitumor effect of hexa-N-acetylchitohexaose and chitohexaose. Carbohydr. Res. 1986, 151, 403–408. [Google Scholar] [CrossRef] [PubMed]
- Huang, M.; Khor, E.; Lim, L.Y. Uptake and cytotoxicity of chitosan molecules and nanoparticles: Effects of molecular weight and degree of deacetylation. Pharm. Res. 2004, 21, 344–353. [Google Scholar] [CrossRef] [PubMed]
- Younes, I.; Frachet, V.; Rinaudo, M.; Jellouli, K.; Nasri, M. Sfax University, Sfax, Tunisia. Cytotoxicity of homogeneous chitosans with different acetylation degrees and molecular weight on bladder carcinoma cells. 2015; Unpublished work. [Google Scholar]
- Ravi Kumar, M.N.V.; Muzzarelli, R.A.A.; Muzzarelli, C.; Sashiwa, H.; Domb, A.J. Chitosan Chemistry and Pharmaceutical Perspectives. React. Funct. Polym. 2000, 46, 1–27. [Google Scholar] [CrossRef]
- Patil, R.S.; Ghormade, V.; Deshpande, M.V. Chitinolytic enzymes: An exploration. Enzym. Microb. Technol. 2000, 26, 473–483. [Google Scholar] [CrossRef]
- Venter, J.P.; Kotze, A.F.; Auzely-Velty, R.; Rinaudo, M. Synthesis and evaluation of the mucoadhesivity of a CD-chitosan derivative. Int. J. Pharm. 2006, 313, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Khor, E. Chitin: A biomaterial in waiting. Curr. Opin. Solid State Mater. Sci. 2002, 6, 313–317. [Google Scholar] [CrossRef]
- Maeda, Y.; Jayakumar, R.; Nagahama, H.; Furuike, T.; Tamura, H. Synthesis, characterization and bioactivity studies of novel b-chitin scaffolds for tissue-engineering applications. Int. J. Biol. Macromol. 2008, 42, 463–467. [Google Scholar] [CrossRef]
- Nagahama, H.; Nwe, N.; Jayakumar, R.; Koiwa, S.; Furuike, T.; Tamura, H. Novel biodegradable chitin membranes for tissue engineering applications. Carbohydr. Polym. 2008, 73, 295–302. [Google Scholar] [CrossRef]
- Yang, T.L. Chitin-based materials in tissue engineering: Applications in soft tissue and epithelial organ. Int. J. Mol. Sci. 2011, 12, 1936–1963. [Google Scholar] [CrossRef] [PubMed]
- Mi, F.L.; Lin, Y.M.; Wu, Y.B.; Shyu, S.S.; Tsai, Y.H. Chitin/PLGA blend microspheres as a biodegradable drug-delivery system: Phase-separation, degradation and release behavior. Biomaterials 2002, 23, 3257–3267. [Google Scholar] [CrossRef] [PubMed]
- Illum, L.; Davis, S. Chitosan as a delivery system for the transmucosal administration of drugs. In Polysaccharides. Structural Diversity and Functional Versatility, 2nd ed.; Dumitriu, S., Ed.; Marcel Dekker Publisher: New York, NY, USA, 2005; pp. 643–660. [Google Scholar]
- Kumirska, J.; Weinhold, M.X.; Thöming, J.; Stepnowski, P. Biomedical Activity of Chitin/Chitosan Based Materials—Influence of Physicochemical Properties Apart from Molecular Weight and Degree of N-Acetylation. Polymers 2011, 3, 1875–1901. [Google Scholar] [CrossRef]
- Jayakumar, R.; Menon, D.; Manzoor, K.; Nair, S.V.; Tamura, H. Biomedical applications of chitin and chitosan based nanomaterials-A short review. Carbohydr. Polym. 2011, 82, 227–232. [Google Scholar] [CrossRef]
- Aranaz, I.; Mengíbar, M.; Harris, R.; Paños, I.; Miralles, B.; Acosta, N.; Galed, G.; Heras, A. Functional Characterization of Chitin and Chitosan. Curr. Chem. Biol. 2009, 3, 203–230. [Google Scholar]
- Dutta, P.K.; Dutta, J.; Tripathi, V.S. Chitin and chitosan: Chemistry, properties and applications. J. Sci. Ind. Res. India 2004, 63, 20–31. [Google Scholar]
- Park, B.K.; Kim, M-M. Applications of chitin and its derivatives in biological medicine. Int. J. Mol. Sci. 2010, 11, 5152–5164. [Google Scholar] [CrossRef] [PubMed]
- Cheba, B.A. Chitin and Chitosan: Marine Biopolymers with Unique Properties and Versatile Applications. Glob. J. Biotechnol. Biochem. 2011, 6, 149–153. [Google Scholar]
- De Alvarenga, E.S. Characterization and properties of chitosan. In Biotechnology of Biopolymers; Elnashar, M., Ed.; In Tech: Rijeka, Croatia, 2011; pp. 91–108. [Google Scholar]
- Kim, S.K. Chitin and Chitosan Derivatives: Advances in Drug Discovery and Developments; Kim, S.K., Ed.; CRC Press: Boca Raton, FL, USA, 2013. [Google Scholar]
- Sarmento, B.; Das Neves, J. Chitosan-Based Systems for Biopharmaceuticals: Delivery, Targeting and Polymer Therapeutics; Wiley & Sons: Hoboken, NJ, USA, 2012. [Google Scholar]
- Park, B.K.; Kim, M.M. Applications of Chitin and Its Derivatives in Biological Medicine. Int. J. Mol. Sci. 2010, 11, 5152–5164. [Google Scholar] [CrossRef] [PubMed]
- Dash, M.; Chiellini, F.; Ottenbrite, R.M.; Chiellini, E. Chitosan—A versatile semi-synthetic polymer in biomedical applications. Prog. Polym. Sci. 2011, 36, 981–1014. [Google Scholar] [CrossRef]
- Kanke, M.; Katayama, H.; Tsuzuki, S.; Kuramoto, H. Application of chitin and chitosan to pharmaceutical preparations. Chem. Pharm. Bull. 1989, 37, 523–525. [Google Scholar] [CrossRef]
- Kato, Y.; Onishi, H.; Machida, Y. Application of chitin and chitosan derivatives in the pharmaceutical field. Curr. Pharm. Biotechnol. 2003, 4, 303–309. [Google Scholar] [CrossRef] [PubMed]
- Felse, P.A.; Panda, T. Studies on applications of chitin and its derivatives. Bioprocess Eng. 1999, 20, 505–512. [Google Scholar] [CrossRef]
- Yusof, N.L.; Wee, A.; Lim, L.Y.; Khor, E. Flexible chitin films as potential wound-dressing materials: Wound model studies. J. Biomed. Mater. Res. Part A 2003, 66A, 224–232. [Google Scholar] [CrossRef]
- Liu, J.; Zhu, L. Method for preparing chitin composite artificial skin that can be used as woundplast. Faming Zhuanli Shenqing CN 101411897 A 20090422 2009. [Google Scholar]
- Wongpanit, P.; Sanchavanakit, N.; Pavasant, P.; Supaphol, P.; Tokura, S.; Rujiravanit, R. Preparation and characterization of microwave-treated carboxymethylchitin and carboxymethylchitosan films for potential use in wound care application. Macromol. Biosci. 2005, 5, 1001–1012. [Google Scholar] [CrossRef] [PubMed]
- Muzzarelli, R.A.A.; Morganti, G.; Palombo, P.; Biagini, G.; Mattioli Belmonte, M.; Giantomassi, F.; Orlandi, F.; Muzzarelli, C. Chitin nanofibrils/chitosan glycolate composites as wound medicaments. Carbohydr. Polym. 2007, 70, 274–284. [Google Scholar] [CrossRef]
- Bernkop-Schnurch, A. Mucoadhesive polymers. In Polymeric Biomaterials; Dumitriu, S., Ed.; Marcel Dekker: New York, NY, USA, 2002; pp. 147–165. [Google Scholar]
- Ito, M. In vitro properties of a chitosan-bonded hydroxyapatite bone-filling paste. Biomaterials 1991, 12, 41–45. [Google Scholar] [CrossRef] [PubMed]
- Swetha, M.; Sahithi, K.; Moorthi, A.; Srinivasan, N.; Ramasamy, K.; Selvamurugan, N. Biocomposites containing natural polymers and hydroxyapatite for bone tissue engineering. Int. J. Biol. Macromol. 2010, 47, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Khor, E.; Lim, L.Y. Implantable applications of chitin and chitosan. Biomaterials 2003, 24, 2339–2349. [Google Scholar] [CrossRef] [PubMed]
- Vankatesan, J.; Kim, S.K. Chitosan composites for bone tissue engineering—an overview. Mar. Drugs 2010, 8, 2252–2266. [Google Scholar] [CrossRef] [PubMed]
- El Zein, A.R.; Dabbarh, F.; Chaput, C. Injectable self-setting calcium phosphate cement. In Chitosan in Pharmacy and Chemistry; Muzzarelli, R.A.A., Muzzarelli, C., Eds.; ATEC: Grottammare, Italy, 2002; pp. 365–370. [Google Scholar]
- Yi, H.; Wu, L.Q.; Bentley, W.E.; Ghadssi, R.; Rubloff, G.W.; Culver, J.N.; Payne, G.F. Biofabrication with chitosan. Biomacromolecules 2005, 6, 2881–2894. [Google Scholar] [CrossRef] [PubMed]
- Saravanan, S.; Nethala, S.; Pattnaik, S.; Tripathi, A.; Moorthi, A.; Selvamurugan, N. Preparation, characterization and antimicrobial activity of a bio-composite scaffold containing chitosan/nano-hydroxyapatite/nano-silver for bone tissue engineering. Int. J. Biol. Macromol. 2011, 49, 188–193. [Google Scholar] [CrossRef] [PubMed]
- Venkatesan, J.; Kim, S.K. Chitosan Composites for Bone Tissue Engineering—An Overview. Mar. Drugs 2010, 8, 2252–2266. [Google Scholar] [CrossRef] [PubMed]
- Teng, S.; Lee, E.; Yoon, B.; Shin, D.; Kim, H.; Oh, J. Chitosan/nanohydroxyapatite composite membranes via dynamic filtration for guided bone regeneration. J. Biomed. Mater. Res. Part A 2009, 88, 569–580. [Google Scholar] [CrossRef]
- Bin, J.; Feng, Y.; Zhi-kun, L. Basic fibroblast growth factor combined with nano-hydroxyapatite/chitosan composites for repair of radial bone defects in rabbits. Chin. J. Tissue Eng. Res. 2012, 16, 6343–6348. [Google Scholar]
- Costa-Pinto, A.R.; Reis, R.L.; Neves, N.M. Scaffolds based bone tissue engineering: The role of chitosan. Tissue Eng. Part B Rev. 2011, 17, 331–347. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Krajewska, B. Application of chitin- and chitosan-based materials for enzyme immobilizations: A review. Enzym. Microb. Technol. 2004, 35, 126–139. [Google Scholar] [CrossRef]
- Freier, T.; Montenegro, R.; Koh, S.; Shoichet, M.S. Chitin tubes for tissue engineering in the nervous system. Biomaterials 2005, 26, 4624–4632. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.L. Chitin-based Materials in Tissue Engineering: Applications in Soft Tissue and Epithelial Organ. Int. J. Mol. Sci. 2011, 12, 1936–1963. [Google Scholar] [CrossRef] [PubMed]
- Kuo, S.M.; Niu, G.C.; Chang, S.J.; Kuo, C.H.; Bair, M.S. A one‐step method for fabricating chitosan microspheres. J. Appl. Polym. Sci. 2004, 94, 2150–2157. [Google Scholar] [CrossRef]
- Honarkar, H.; Barikani, M. Applications of biopolymers I: Chitosan. Monatsh. Chem. 2009, 140, 1403–1420. [Google Scholar] [CrossRef]
- Özbas-Turan, S.; Aral, C.; Kabasakal, L.; Keyer-Uysal, M.; Akbuga, J. Co-encapsulation of two plasmids in chitosan microspheres as a non-viral gene delivery vehicle. J. Pharm. Pharmaceut. Sci. 2003, 6, 27–32. [Google Scholar]
- Ohkawa, K.; Minato, K.I.; Kumagai, G.; Hayashi, S.; Yamamoto, H. Chitosan nanofiber. Biomacromolecules 2006, 7, 3291–3294. [Google Scholar] [CrossRef] [PubMed]
- Kucharska, M.; Niekraszewicz, A.; Lebioda, J.; Malczewska-Brzoza, K.; Wesołowska, E. Bioactive Composite Materials In Progress on Chemistry and Application of Chitin and Its Derivatives; Jaworska, M.M., Ed.; Polish Chitin Society: Lodz, Poland, 2007; Volume 12, pp. 131–138. [Google Scholar]
- Dhanikula, A.B.; Panchagnula, R. Development and characterization of biodegradable chitosan films for local delivery of paclitaxel. AAPS J. 2004, 6, 88–89. [Google Scholar] [CrossRef]
- Kucharska, M.; Struszczyk, M.H.; Cichecka, M.; Brzoza, K. Preliminary studies on the usable properties of innovative wound dressings. In Progress on Chemistry and Application of Chitin and Its Derivatives; Jaworska, M.M., Ed.; Polish chitin Society: Lotz, Poland, 2011; Volume 16, pp. 131–137. [Google Scholar]
- Pereira, A.O.; Cartucho, D.J.; Duarte, A.S.; Gil, M.H.; Cabrita, A.; Patricio, J.A.; Barros, M.M. Immobilisation of cardosin A in chitosan sponges as a novel implant for drug delivery. Curr. Drug Discov. Technol. 2005, 2, 231–238. [Google Scholar] [CrossRef] [PubMed]
- Illum, L. Chitosan and its use as a pharmaceutical excipient. Pharm. Res. 1998, 15, 1326–1331. [Google Scholar] [CrossRef] [PubMed]
- Dai, T.; Tanaka, M.; Huang, Y.Y.; Hamblin, M.R. Chitosan preparations for wounds and burns: Antimicrobial and wound-healing effects. Expert Rev. Anti. Infect. Ther. 2011, 9, 857–879. [Google Scholar] [CrossRef] [PubMed]
- Jayakumar, R.; Prabaharan, M.; Sudheesh Kumar, P.T.; Nair, S.V.; Tamura, H. Biomaterials based on chitin and chitosan in wound dressing applications. Biotechnol. Adv. 2011, 29, 322–337. [Google Scholar] [CrossRef] [PubMed]
- Loke, W.K.; Lau, S.K.; Yong, L.L.; Khor, E.; Sum, C.K. Wound dressing with sustained anti-microbial capability. J. Biomed. Mater. Res. 2000, 53, 8–17. [Google Scholar] [CrossRef] [PubMed]
- Kumari, R.; Dutta, P.K. Physicochemical and biological activity study of genipin-crosslinked chitosan scaffolds prepared by using supercritical carbon dioxide for tissue engineering applications. Int. J. Biol. Macromol. 2010, 46, 261–266. [Google Scholar] [CrossRef] [PubMed]
- Luna-barcenas, G.; Prokhorov, E.; Elizalde-pena, E.; Nuno-licona, A.; Sanchez, I.C. Chitosan-Based Hydrogels for Tissue Engineering Applications, Biotechnology in Agriculture, Industry and Medicine Series; Nova Science Publisher: New York, NY, USA, 2011. [Google Scholar]
- Croisier, F.; Jérôme, C. Chitosan-based biomaterials for tissue engineering. Eur. Polym. J. 2013, 49, 780–792. [Google Scholar] [CrossRef]
- Riva, R.; Ragelle, H.; des Rieux, A.; Duhem, N.; Jérôme, C.; Préat, V. Chitosan and chitosan derivatives in drug delivery and tissue engineering. In Chitosan for Biomaterials II; Jayakumar, R., Prabaharan, M., Muzzarelli, R.A.A., Eds.; Springer: Berlin-Heidelberg, Germany, 2011; pp. 19–44. [Google Scholar]
- Suh, J.K.F.; Matthew, H.W.T. Application of chitosan-based polysaccharide biomaterials in cartilage tissue engineering: A review. Biomaterials 2000, 21, 2589–2598. [Google Scholar] [CrossRef] [PubMed]
- Shi, C.; Zhu, Y.; Ran, X.; Wang, M.; Su, Y.; Cheng, T. Therapeutic potential of chitosan and its derivatives in regenerative medicine. J. Surg. Res. 2006, 133, 185–192. [Google Scholar] [CrossRef] [PubMed]
- Farzaneh, N.H.; Soheila, S.K.; Faramarz, A.T.; Zahra, A. Novel Topical Biocompatible Tissue Adhesive Based on Chitosan-modified Urethane Pre- S polymer. Iran Polym. J. 2011, 20, 671–680. [Google Scholar]
- Kotzé, A.F.; Hamman, J.H.; Snyman, D.; Jonker, C.; Stander, M. Mucoadhesive and absorption enhancing properties of N-trimethyl chitosan chloride. In Chitosan in Pharmacy and Chemistry; Muzzarelli, R.A.A., Muzzarelli, C., Eds.; ATEC: Grottammare, Italy, 2002; pp. 31–40. [Google Scholar]
- Rickett, T.A.; Amoozgar, Z.; Tuchek, C.A.; Park, J.; Yeo, Y.; Shi, R. Rapidly photo-cross-linkable chitosan hydrogel for peripheral neurosurgeries. Biomacromolecules 2011, 12, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Hamman, J.H.; Kotzé, A.F. Paracellular absorption enhancement across intestinal epithelia by N-trimethyl chitosan chloride. In Chitosan in Pharmacy and Chemistry; Muzzarelli, R.A.A., Muzzarelli, C., Eds.; ATEC: Grottammare, Italy, 2002; pp. 41–50. [Google Scholar]
- Miwa, A.; Ishibe, A.; Nakano, M.; Yamahira, T.; Itai, S.; Jinno, S.; Kawahara, H. Development of novel chitosan derivatives as micellar carriers of taxol. Pharm. Res. 1998, 15, 1844–1850. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Sun, S.J.; Zhang, X.; de Yao, K. Self-aggregation behaviour of alkylated chitosan and its effect on the release of a hydrophobic drug. J. Biomater. Sci. Polym. Edn. 2003, 14, 851–859. [Google Scholar] [CrossRef]
- Liu, W.; Zang, X.; Sun, S.J.; Sun, G.J.; Yao, K.D.; Liang, D.C.; Guo, G.; Zhang, J.Y. N-alkylated chitosan as a potential nonviral vector for gene transfection. Bioconjug. Chem. 2003, 14, 782–789. [Google Scholar] [CrossRef] [PubMed]
- Ouchi, T.; Murata, J.I.; Ohya, Y. Gene delivery by quaternary chitosan with antennary galactose residues. In Polysaccharide Applications: Cosmetics and Pharmaceuticals; El-Nokaly, M.A., Soini, H.A., Eds.; American Chemical Society: Washington, DC, USA, 1999; pp. 15–23. [Google Scholar]
- Mi, F.L.; Shyu, S.S.; Chen, C.T.; Schoung, J.Y. Porous chitosan microspheres suitable for controlling the antigen release of Newcastle disease vaccine: Preparation of antigen-adsorbed microsphere and in vitro release. Biomaterials 1999, 20, 1603–1612. [Google Scholar] [CrossRef] [PubMed]
- Park, I.K.; Jiang, H.L.; Yun, C.H.; Choi, Y.J.; Kim, S.J.; Akaike, T.; Kim, S.I.; Cho, C.S. Release of Newcastle disease virus vaccine from chitosan microspheres in vitro and in vivo. Asian-Aust. J. Anim. Sci. 2004, 17, 543–547. [Google Scholar] [CrossRef]
- Foda, N.H.; El-Iaithy, M.; Tadros, I. Implantable biodegradable sponges: Effect of interpolymer complex formation of chitosan with gelatin on the release behavior of tramadol hydrochloride. Drug Dev. Ind. Pharm. 2007, 33, 7–17. [Google Scholar] [CrossRef] [PubMed]
- Dhanaraj, S.A.; Selvadurai, M.; Santhi, K.; Hui, A.L.S.; Wen, C.J.; Teng, H.C. Targeted drug delivery system:-formulation and evaluation of chitosan nanospheres containing doxorubicin hydrochloride. Int. J. Drug Deliv. 2014, 6, 186–193. [Google Scholar]
- Makhlof, A.; Tozuka, Y.; Takeuchi, H. Design and evaluation of novel pH-sensitive chitosan nanoparticles for oral insulin delivery. Eur. J. Pharm. Sci. 2011, 42, 445–451. [Google Scholar] [CrossRef] [PubMed]
- Cheng, S.Y.; Yuen, M.C.; Lam, P.L.; Gambari, R.; Wong, R.S.; Cheng, G.Y.; Lai, P.B.; Tong, S.W.; Chan, K.W.; Lau, F.Y.; et al. Synthesis, characterization and preliminary analysis of in vivo biological activity of chitosan/celecoxib microcapsules. Bioorg. Med. Chem. Lett. 2010, 20, 4147–4151. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Younes, I.; Rinaudo, M. Chitin and Chitosan Preparation from Marine Sources. Structure, Properties and Applications. Mar. Drugs 2015, 13, 1133-1174. https://doi.org/10.3390/md13031133
Younes I, Rinaudo M. Chitin and Chitosan Preparation from Marine Sources. Structure, Properties and Applications. Marine Drugs. 2015; 13(3):1133-1174. https://doi.org/10.3390/md13031133
Chicago/Turabian StyleYounes, Islem, and Marguerite Rinaudo. 2015. "Chitin and Chitosan Preparation from Marine Sources. Structure, Properties and Applications" Marine Drugs 13, no. 3: 1133-1174. https://doi.org/10.3390/md13031133






