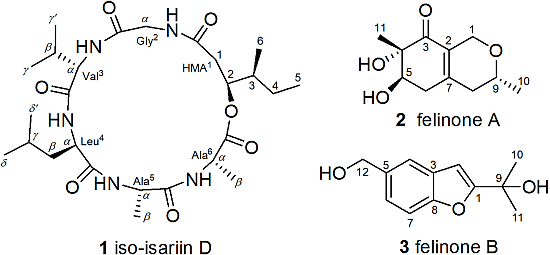
Cyclodepsipeptides and Other O-Containing Heterocyclic Metabolites from Beauveria felina EN-135, a Marine-Derived Entomopathogenic Fungus
Abstract
:1. Introduction
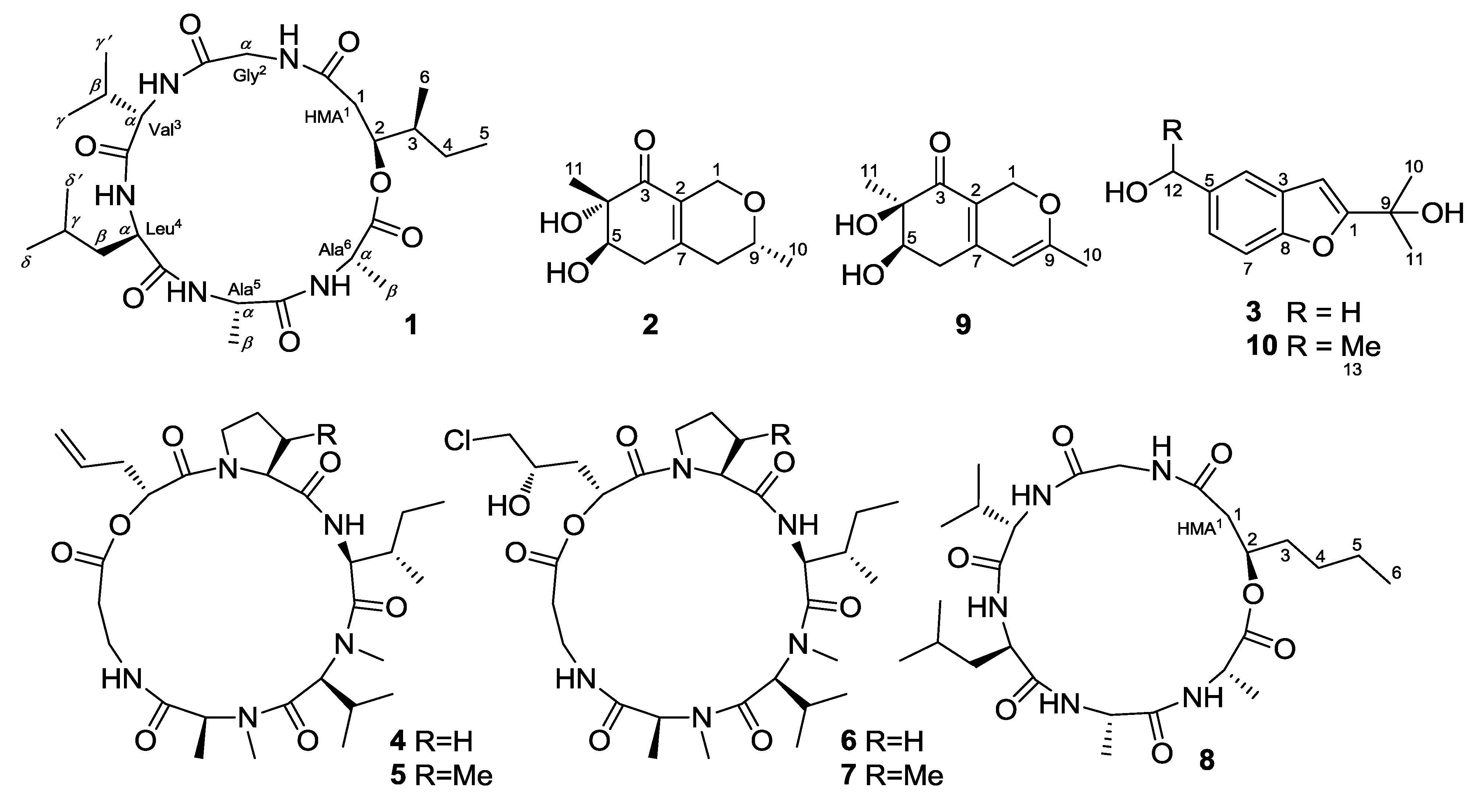
2. Results and Discussion
2.1. Structure Elucidation of the New Compounds
| Position | δH (J in Hz) | δC | Position | δH (J in Hz) | δC |
|---|---|---|---|---|---|
| HMA1 | - | - | Leu4 | - | - |
| CO | - | 169.7 C | CO | - | 171.1 C |
| 1 | 3.19, m; 2.92, d (13.6) | 37.6 CH2 | α | 4.68, m | 51.9 CH |
| 2 | 5.63, m | 74.3 CH | β | 2.13, m | 38.6 CH2 |
| 3 | 2.20, m | 38.0 CH | γ | 2.28, m | 24.1 CH |
| 4 | 2.03, m; 1.70, m | 24.8 CH2 | δ | 1.48, d (6.8) | 21.1 CH3 |
| 5 | 1.49, t (6.9) | 11.3 CH3 | δ’ | 1.53, d (6.8) | 18.8 CH3 |
| 6 | 1.46, d (6.8) | 14.1 CH3 | NH | 9.30, d (6.0) | - |
| Gly2 | - | - | Ala5 | - | - |
| CO | - | 168.9 C | CO | - | 171.6 C |
| α | 4.73, m; 4.64, dd (16.4, 3.4) | 42.4 CH2 | α | 4.84, m | 47.5 CH |
| NH | 8.54, br. s | - | β | 1.85, d (7.0) | 17.3 CH3 |
| Val3 | - | - | NH | 8.60, d (8.3) | - |
| CO | - | 171.7 C | Ala6 | - | - |
| α | 4.76, m | 58.1 CH | CO | - | 171.6 C |
| β | 2.51, m | 29.9 CH | α | 4.79, m | 48.3 CH |
| γ | 1.55, d (7.7) | 22.8 CH3 | β | 1.95, d (7.2) | 16.6 CH3 |
| γ’ | 1.51, d (7.7) | 18.6 CH3 | NH | 8.48, d (7.6) | - |
| NH | 8.48, d (7.6) | - | - | - | - |

| Compound 2 | Compound 3 | |||||
|---|---|---|---|---|---|---|
| Position | δH (J in Hz) a | δH (J in Hz) b | δC a | Position | δH (J in Hz) b | δC b |
| 1 | 4.29, d (15.5) 4.00, d (15.5) | 4.43, d (15.8) 4.04, d (15.8) | 62.9 CH2 | 1 | - | 164.1 C |
| 2 | - | - | 127.3 C | 2 | 6.61, s | 100.0 CH |
| 3 | - | - | 199.1 C | 3 | - | 128.5 C |
| 4 | - | - | 76.1 C | 4 | 7.52, s | 119.2 CH |
| 5 | 3.74, m | 3.91, dd (9.5, 5.3) | 71.4 CH | 5 | - | 135.8 C |
| 6α 6β | 2.52, m 2.14, m | 2.59, dd (18.1, 5.3) 2.35, dd (18.1, 9.5) | 36.7 CH2 | 6 | 7.24, d (8.4) | 123.1 CH |
| 7 | - | - | 151.6 C | 7 | 7.39, d (8.4) | 110.2 CH |
| 8α 8β | 2.07, m 2.20, m | 2.12, dd (18.3, 10.1) 2.24, br. s (18.3) | 36.9 CH2 | 8 | - | 154.2 C |
| 9 | 3.54, m | 3.60, ddd (10.1, 6.2, 3.5) | 68.6 CH | 9 | - | 68.4 C |
| 10 | 1.16, d (6.2) | 1.21, d (6.2) | 20.9 CH3 | 10 | 1.61, s | 27.6 CH3 |
| 11 | 1.08, s | 1.18, s | 18.0 CH3 | 11 | 1.61, s | 27.6 CH3 |
| 4-OH | 5.05, br. s | - | - | 12 | 4.65, s | 64.1 CH2 |
| 5-OH | 5.07, d (4.2) | - | - | - | - | - |
2.2. Biological Activities of the Isolated Compounds
3. Experimental Section
3.1. General
3.2. Fungal Material
3.3. Fermentation
3.4. Extraction and Isolation
 : −22.2 (c 0.36, MeOH); UV (MeOH) λmax (log ε) 206 (3.64) nm; 1H and 13C NMR data, see Table 1; ESIMS m/z 540 [M + H]+; HRESIMS m/z 540.3392 [M + H]+ (calcd for C26H46N5O7, 540.3392).
: −22.2 (c 0.36, MeOH); UV (MeOH) λmax (log ε) 206 (3.64) nm; 1H and 13C NMR data, see Table 1; ESIMS m/z 540 [M + H]+; HRESIMS m/z 540.3392 [M + H]+ (calcd for C26H46N5O7, 540.3392). : +104.8 (c 0.21, MeOH); UV (MeOH) λmax (log ε) 200 (3.45), 240 (3.77) nm; CD ëmax (Δε) 219 (−13.89), 248 (+27.06) nm; 1H and 13C NMR data, see Table 2; ESIMS m/z 213 [M + H]+; HRESIMS m/z 213.1123 [M + H]+ (calcd for C11H17O4, 213.1121).
: +104.8 (c 0.21, MeOH); UV (MeOH) λmax (log ε) 200 (3.45), 240 (3.77) nm; CD ëmax (Δε) 219 (−13.89), 248 (+27.06) nm; 1H and 13C NMR data, see Table 2; ESIMS m/z 213 [M + H]+; HRESIMS m/z 213.1123 [M + H]+ (calcd for C11H17O4, 213.1121). : −16.7 (c 0.12, MeOH); UV (MeOH) λmax (log ε) 209 (4.38), 247 (4.09), 279 (3.47), 286 (3.48) nm; 1H and 13C NMR data, see Table 2; ESIMS m/z 229 [M + Na]+; HRESIMS m/z 229.0828 [M + Na]+ (calcd for C12H14O3Na, 229.0835).
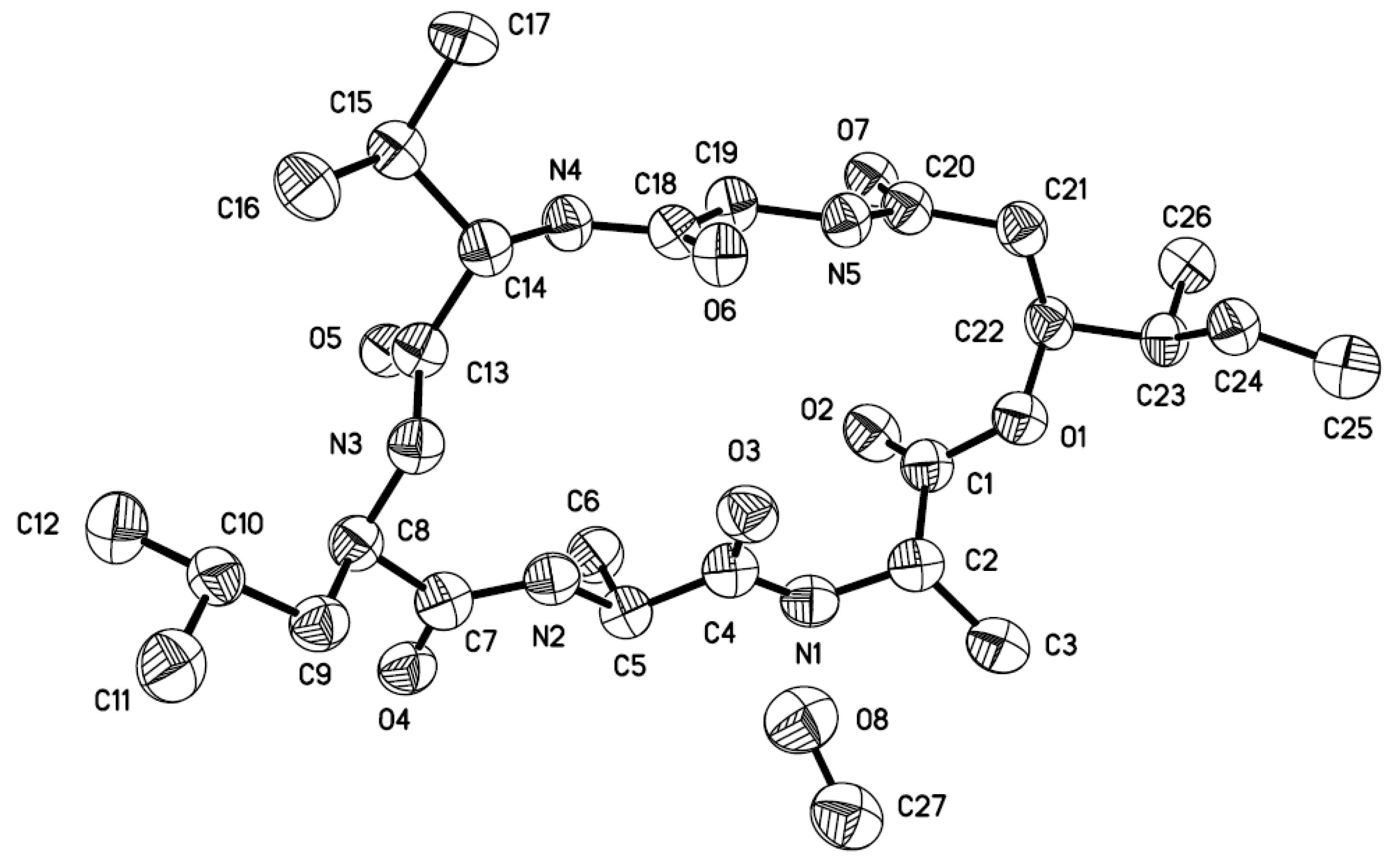
: −16.7 (c 0.12, MeOH); UV (MeOH) λmax (log ε) 209 (4.38), 247 (4.09), 279 (3.47), 286 (3.48) nm; 1H and 13C NMR data, see Table 2; ESIMS m/z 229 [M + Na]+; HRESIMS m/z 229.0828 [M + Na]+ (calcd for C12H14O3Na, 229.0835).3.5. X-ray Crystallographic Analysis of Compound 1
3.6. Brine-Shrimp Lethality
3.7. Antimicrobial Assay
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Molnar, I.; Gibson, D.M.; Krasnoff, S.B. Secondary metabolites from entomopathogenic hypocrealean fungi. Nat. Prod. Rep. 2010, 27, 1241–1275. [Google Scholar] [CrossRef]
- Zenitani, S.; Tashiro, S.; Shindo, K.; Nagai, K.; Suzuki, K.; Imoto, M. Gerfelin, a novel inhibitor of geranylgeranyl diphosphate synthase from Beauveria felina QN22047, I. Taxonomy, fermentation, isolation, and biological activities. J. Antibiot. 2003, 56, 617–621. [Google Scholar] [CrossRef]
- Lira, S.P.; Vita-Marques, A.M.; Seleghim, M.H.R.; Bugni, T.S.; Labarbera, D.V.; Sette, L.D.; Sponchiado, S.R.P.; Ireland, C.M. New destruxins from the marine-derived fungus Beauveria felina. J. Antibiot. 2006, 59, 553–563. [Google Scholar] [CrossRef]
- Langenfeld, A.; Blond, A.; Gueye, S.; Herson, P.; Nay, B.; Dupont, J.; Prado, S. Insecticidal cyclodepsipeptides from Beauveria felina. J. Nat. Prod. 2011, 74, 825–830. [Google Scholar] [CrossRef]
- Chung, Y.M.; EI-Shazly, M.; Chuang, D.W.; Hwang, T.L.; Asai, T.; Oshima, Y.; Ashour, M.L.; Wu, Y.C.; Chang, F.R. Suberoylanilide hydroxamic acid, a histone deacetylase inhibitor, induces the production of anti-inflammatory cyclodepsipeptides from Beauveria felina. J. Nat. Prod. 2013, 76, 1260–1266. [Google Scholar] [CrossRef]
- Gao, S.S.; Li, X.M.; Du, F.Y.; Li, C.S.; Proksch, P.; Wang, B.G. Secondary metabolites from a marine-derived endophytic fungus Penicillium chrysogenum QEN-24S. Mar. Drugs 2011, 9, 59–70. [Google Scholar]
- Wang, M.H.; Li, X.M.; Li, C.S.; Ji, N.Y.; Wang, B.G. Secondary metabolites from Penicillium pinophilum SD-272, a marine sediment-derived fungus. Mar. Drugs 2013, 11, 2230–2238. [Google Scholar] [CrossRef]
- An, C.Y.; Li, X.M.; Li, C.S.; Wang, M.H.; Xu, G.M.; Wang, B.G. Aniquinazolines A—D, four new quinazolinone alkaloids from marine-derived endophytic fungus Aspergillus nidulans. Mar. Drugs 2013, 11, 2682–2694. [Google Scholar] [CrossRef]
- Li, C.S.; Li, X.M.; Gao, S.S.; Lu, Y.H.; Wang, B.G. Cytotoxic anthranilic acid derivatives from deep sea sediment-derived fungus Penicillium paneum SD-44. Mar. Drugs 2013, 11, 3068–3076. [Google Scholar] [CrossRef]
- An, C.Y.; Li, X.M.; Luo, H.; Li, C.S.; Wang, M.H.; Xu, G.M.; Wang, B.G. 4-phenyl-3, 4-dihydroquinolone derivatives from Aspergillus nidulans MA-143, an endophytic fungus isolated from the mangrove plant Rhizophora stylosa. J. Nat. Prod. 2013, 76, 1896–1901. [Google Scholar] [CrossRef]
- Vining, L.C.; Taber, W.A. Isariin, a new depsipeptide from Isaria cretacea. Can. J. Chem. 1962, 40, 1579–1584. [Google Scholar] [CrossRef]
- Deffieux, G.; Merlet, D.; Baute, R.; Bourgeois, G.; Baute, M.A.; Neveu, A. New insecticidal cyclodepsispeptides from the fungus Isaria felina. J. Antibiot. 1981, 34, 1266–1270. [Google Scholar] [CrossRef]
- Sabareesh, V.; Ranganayaki, R.S.; Raghothama, S.; Bopanna, M.P.; Balaram, H.; Srinivasan, M.C.; Balaram, P. Identification and characterization of a library of microheterogeneous cyclohexadepsipeptides from the fungus Isaria. J. Nat. Prod. 2007, 70, 715–729. [Google Scholar] [CrossRef]
- Xu, Y.Q.; Rozco, R.; Wijeratne, E.M.K.; Espinosa-Artiles, P.; Gunatilaka, A.A.L.; Stock, S.P.; Molnar, I. Biosynthesis of the cyclooligomer depsipeptide bassianolide, an insecticidal virulence factor of Beauveria bassiana. Fungal Genet. Biol. 2009, 46, 353–364. [Google Scholar] [CrossRef]
- Xu, Y.Q.; Orozco, R.; Wijeratne, E.M.K.; Gunatilaka, A.A.L.; Stock, S.P.; Molnar, I. Biosynthesis of the cyclooligomer depsipeptide beauvericin, a virulence factor of the entomopathogenic fungus Beauveria bassiana. Chem. Biol. 2008, 15, 898–907. [Google Scholar] [CrossRef]
- Stark, L.M.; Pekari, K.; Sorensen, E.J. A nucleophile-catalyzed cycloisomerization permits a concise synthesis of (+)-harziphilone. Proc. Natl. Acad. Sci. USA 2004, 101, 12064–12066. [Google Scholar] [CrossRef]
- Ding, G.; Liu, S.C.; Guo, L.D.; Zhou, Y.G.; Che, Y.S. Antifungal metabolites from the plant endophytic fungus Pestalotiopsis foedan. J. Nat. Prod. 2008, 71, 615–618. [Google Scholar] [CrossRef]
- Zaitsev, V.G.; Mikhal’chuk, A.L. Enantioconvergent synthesis of (−)-(S)- and (+)-(R)-2-acetyl-3,6-dihydroxycyclohex-2-enone starting from rac-6-hydroxy-3-methoxycyclohex-2-enone. Chirality 2001, 13, 488–492. [Google Scholar] [CrossRef]
- Bohlmann, F.; Ziesche, J.; King, R.M.; Robinson, H. Neue melampolide aus Smallanthus Fruticosus. Phytochemistry 1980, 19, 973–974. [Google Scholar] [CrossRef]
- Gupta, S.; Roberts, D.W.; Renwick, J.A.A. Molecular conformation of destruxin A. Tetrahedron Lett. 1989, 30, 4189–4192. [Google Scholar] [CrossRef]
- Springer, J.P.; Cole, R.J.; Dorner, J.W.; Cox, R.H.; Richard, J.L.; Barnes, C.L.; Helm, D.V.D. Structure and conformation of roseotoxin B. J. Am. Chem. Soc. 1984, 106, 2388–2392. [Google Scholar] [CrossRef]
- Wang, S.; Li, X.M.; Teuscher, F.; Li, D.L.; Diesel, A.; Ebel, R.; Proksch, P.; Wang, B.G. Chaetopyranin, a benzaldehyde derivative, and other related metabolites from Chaetomium globosum, an endophytic fungus derived from the marine red alga Polysiphonia urceola. J. Nat. Prod. 2006, 69, 1622–1625. [Google Scholar] [CrossRef]
- Cambridge Crystallographic Data Centre as CCDC 983056. Crystallographic Data of Compound 1; Cambridge Crystallographic Data Centre: Cambridge, UK, 2014. Available online: http://www.ccdc.cam.ac.uk/data_request/cif (accessed on 23 January 2014).
- Sheldrick, G.M. SADABS, Software for Empirical Absorption Correction; University of Göttingen: Göttingen, Germany, 1996. [Google Scholar]
- Sheldrick, G.M. SHELXTL, Structure Determination Software Programs; Bruker Analytical X-ray System Inc.: Madison, WI, USA, 1997. [Google Scholar]
- Sheldrick, G.M. SHELXL-97 and SHELXS-97,Program for X-ray Crystal Structure Solution and Refinement; University of Göttingen: Göttingen, Germany, 1997. [Google Scholar]
- Gerwick, W.H.; Proteau, P.J.; Nagle, D.G.; Hamel, E.; Blokhin, A.; Slate, D.L. Structure of curacin A, a novel antimitotic, antiproliferative, and brine shrimp toxic natural product from the marine Cyanobacterium Lyngbya majuscula. J. Org. Chem. 1994, 59, 1243–1245. [Google Scholar] [CrossRef]
- Bergeron, R.J.; Cavanaugh, P.F., Jr.; Kline, S.J.; Hughes, R.G., Jr.; Elliott, G.T.; Porter, C.W. Antineoplastic and antiherpetic activity of spermidine catecholamide iron chelators. Biochem. Biophys. Res. Commun. 1984, 121, 848–854. [Google Scholar] [CrossRef]
- Al-Burtamani, S.K.S.; Fatope, M.O.; Marwah, R.G.; Onifade, A.K.; Al-Saidi, S.H. Chemical composition, antibacterial and antifungal activities of the essential oil of Haplophyllum tuberculatum from Oman. J. Ethnopharmacol. 2005, 96, 107–112. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Du, F.-Y.; Li, X.-M.; Zhang, P.; Li, C.-S.; Wang, B.-G. Cyclodepsipeptides and Other O-Containing Heterocyclic Metabolites from Beauveria felina EN-135, a Marine-Derived Entomopathogenic Fungus. Mar. Drugs 2014, 12, 2816-2826. https://doi.org/10.3390/md12052816
Du F-Y, Li X-M, Zhang P, Li C-S, Wang B-G. Cyclodepsipeptides and Other O-Containing Heterocyclic Metabolites from Beauveria felina EN-135, a Marine-Derived Entomopathogenic Fungus. Marine Drugs. 2014; 12(5):2816-2826. https://doi.org/10.3390/md12052816
Chicago/Turabian StyleDu, Feng-Yu, Xiao-Ming Li, Peng Zhang, Chun-Shun Li, and Bin-Gui Wang. 2014. "Cyclodepsipeptides and Other O-Containing Heterocyclic Metabolites from Beauveria felina EN-135, a Marine-Derived Entomopathogenic Fungus" Marine Drugs 12, no. 5: 2816-2826. https://doi.org/10.3390/md12052816