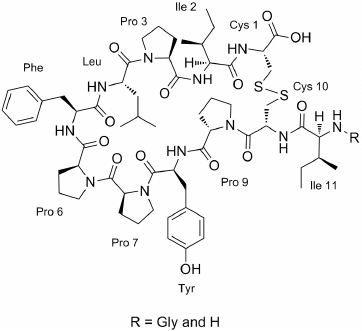
New Cyclic Cystine Bridged Peptides from the Sponge Suberites waedoensis
Abstract
:1. Introduction

2. Results and Discussion
| A. A. unit | Position | chujamide A | chujamide B | ||
|---|---|---|---|---|---|
| δC | δH (J in Hz) | δC | δH (J in Hz) | ||
| Cys 1 | 1 | 176.7, C | 176.9, C | ||
| 2 | 60.1, CH | 4.71, dd (11.4, 2.4) | 60.3, CH | 4.71, dd (11.4, 2.4) | |
| 3 | 43.0, CH2 | 3.45, m | 43.0, CH2 | 3.47, m | |
| 2.55, m | 2.58, m | ||||
| 2-NH a | 7.81, d (9.6) | 8.50, bs | |||
| Ile 2 | 4 | 172.4, C | 173.6, C | ||
| 5 | 59.1, CH | 4.82, d (3.6) | 59.2, CH | 4.82, m | |
| 6 | 37.7, CH | 2.34, m | 38.0, CH | 2.32, m | |
| 7 | 16.1, CH3 | 0.84, d (6.6) | 16.1, CH3 | 0.83, d (6.6) | |
| 8 | 25.2, CH2 | 1.54, m | 25.4, CH2 | 1.55, m | |
| 0.94, m | 0.95, m | ||||
| 9 | 12.2 CH3 | 0.90, t (6.6) | 12.2, CH3 | 0.90, t (6.6) | |
| 5-NH a | 7.52, d (10.2) | 7.67, bs | |||
| Pro 3 | 10 | 174.6, C | 173.4, C | ||
| 11 | 61.9, CH | 5.24, d (7.8) | 61.5, CH | 5.25, m | |
| 12 | 26.8, CH2 | 2.53, m | 27.0, CH2 | 2.48, m | |
| 1.72, m | 1.73, m | ||||
| 13 | 26.2, CH2 | 2.15, m | 26.3, CH2 | 2.13, m | |
| 1.97, m | 1.93, m | ||||
| 14 | 48.3, CH2 | 3.81, m | 48.3, CH2 | 3.80, m | |
| 3.63, dd (8.4, 8.4) | 3.64, m | ||||
| Leu | 15 | 176.4, C | 176.5, C | ||
| 16 | 51.7, CH | 4.50, dd (12.0, 1.8) | 51.6, CH | 4.51, dd (12.0, 1.8) | |
| 17 | 41.4, CH2 | 1.73, m | 41.6, CH2 | 1.72, m | |
| 1.40, m | 1.34, m | ||||
| 18 | 25.9, CH | 1.71, m | 26.0, CH | 1.70, m | |
| 19 | 20.5, CH3 | 0.97, d (6.6) | 20.5, CH3 | 0.97, d (6.6) | |
| 20 | 24.0, CH3 | 0.99, d (6.6) | 24.0, CH3 | 0.98, d (6.6) | |
| 16-NH a | 7.78, d (4.8) | 7.77, d (4.8) | |||
| Phe | 21 | 173.7, C | 173.7, C | ||
| 22 | 56.3, CH | 4.46, dd (12.0, 4.8) | 56.4, CH | 4.46, dd (12.0, 4.8) | |
| 23 | 33.8, CH2 | 3.41, m | 33.9, CH2 | 3.41, m | |
| 3.01, m | 3.00, m | ||||
| 24 | 140.2, C | 140.3, C | |||
| 25/29 | 131.1, CH | 7.27, dd (7.2, 1.8) | 131.2, CH | 7.27, dd (7.2, 1.8) | |
| 26/28 | 129.1, CH | 7.14, m | 129.1, CH | 7.14, m | |
| 27 | 127.3, CH | 7.14, m | 127.3, CH | 7.14, m | |
| 22-NH a | 8.79, d (7.2) | 8.80, d (7.2) | |||
| Pro 6 | 30 | 171.9, C | 171.9, C | ||
| 31 | 62.5, CH | 4.17, d (7.8) | 62.5, CH | 4.15, d (7.8) | |
| 32 | 30.3, CH2 | 2.55, m | 30.3, CH2 | 2.54, m | |
| 1.97, m | 1.99, m | ||||
| 33 | 23.1, CH2 | 1.97, m | 23.1,CH2 | 1.90, m | |
| 1.60, m | 1.62, m | ||||
| 34 | 47.8, CH2 | 3.50, m | 47.8, CH2 | 3.50, m | |
| 3.44, m | 3.44, m | ||||
| Pro 7 | 35 | 172.0, C | 172.1, C | ||
| 36 | 59.9, CH | 2.99, m | 59.9, CH | 2.99, m | |
| 37 | 29.4, CH2 | 2.00, m | 29.4, CH2 | 2.01, m | |
| 1.61, m | 1.62, m | ||||
| 38 | 26.0, CH2 | 2.00, m | 25.9, CH2 | 2.01, m | |
| 1.94, m | 1.93, m | ||||
| 39 | 48.8, CH2 | 3.61, m | 48.7, CH2 | 3.61, m | |
| 3.53, m | 3.53, m | ||||
| Tyr | 40 | 171.2, C | 171.3, C | ||
| 41 | 52.7, CH | 4.85, t (6.0) | 52.6, CH | 4.84, t (6.0) | |
| 42 | 36.9, CH2 | 3.00, m | 37.0, CH2 | 3.01, m | |
| 2.74, dd (14.4, 6.0) | 2.75, m | ||||
| 43 | 127.2, C | 127.4, C | |||
| 44/48 | 132.1, CH | 6.98, d (8.4) | 132.1, CH | 6.98, d (8.4) | |
| 45/47 | 116.1, CH | 6.71, d (8.4) | 116.3, CH | 6.70, d (8.4) | |
| 46 | 157.7, C | 157.7, C | |||
| 41-NH a | 7.05, d (7.2) | 7.11, m | |||
| Pro 9 | 49 | 172.5, C | 172.4, C | ||
| 50 | 62.2, CH | 4.68, d (8.4) | 62.2, CH | 4.69, d (8.4) | |
| 51 | 28.7, CH2 | 2.32, m | 28.6, CH2 | 2.32, m | |
| 2.00, m | 2.00, m | ||||
| 52 | 25.7, CH2 | 2.09, m | 25.7, CH2 | 2.09, m | |
| 1.80, m | 1.80, m | ||||
| 53 | 48.6, CH2 | 3.87, m | 48.6, CH2 | 3.87, m | |
| 3.73, m | 3.73, m | ||||
| Cys 10 | 54 | 172.7, C | 172.7, C | ||
| 55 | 51.2, CH | 4.77, dd (12.0, 2.4) | 51.5, CH | 4.76, ddd (12.0, 2.4, 2.4) | |
| 56 | 40.3, CH2 | 3.09, m | 40.5, CH2 | 3.08, m | |
| 2.77, dd (14.4, 3.0) | 2.73, m | ||||
| 55-NH a | 8.58, d (4.8) | 8.27, bs | |||
| Ile 11 | 57 | 174.3, C | 174.3, C | ||
| 58 | 59.7, CH | 4.01, d (10.2) | 60.0, CH | 3.95, m | |
| 59 | 37.2, CH | 1.72, m | 37.2, CH | 1.72, m | |
| 60 | 15.8, CH3 | 0.93, d (6.6) | 15.8, CH3 | 0.93, d (6.6) | |
| 61 | 26.7, CH2 | 1.55, m | 26.8, CH2 | 1.55, m | |
| 1.18, m | 1.17, m | ||||
| 62 | 10.7, CH3 | 0.86, t (6.6) | 10.8, CH3 | 0.86, t (6.6) | |
| 58-NH a | 8.73, d (6.6) | ||||
| Gly | 63 | 169.4, C | |||
| 64 | 41.8, CH2 | 3.57, m | |||
| 3.55, m | |||||
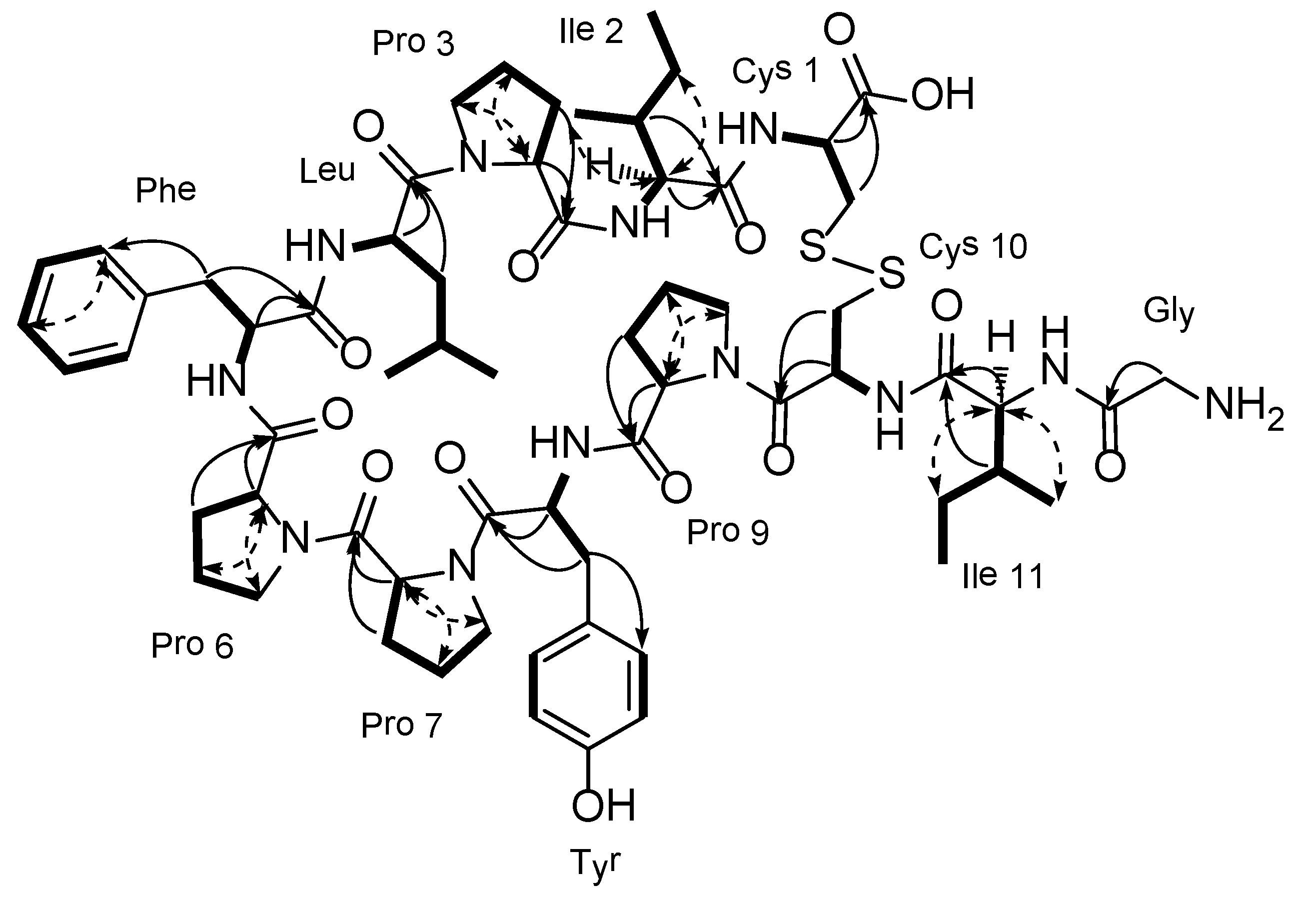
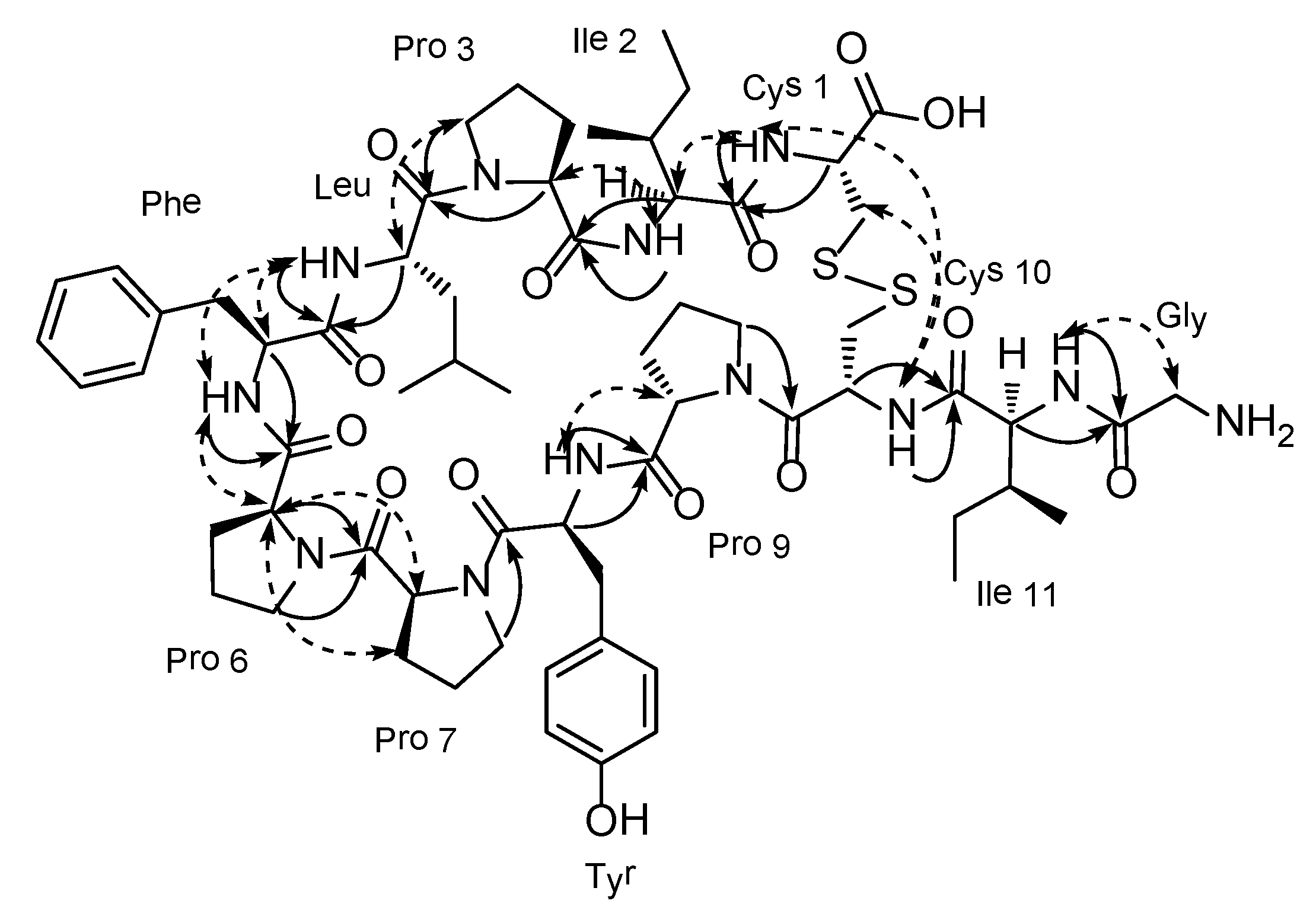
3. Experimental Section
3.1. General Experimental Procedures
3.2. Animal Materials
3.3. Extraction and Isolation
 −15 (c 0.50, MeOH); UV (MeOH) λmax (log ε) 210 (4.40), 227 (4.14), 277 (3.16) nm; IR (ZnSe) vmax 3300, 2958, 1655 cm−1; 1H and 13C NMR data, see Table 1; HRFABMS m/z 1317.6371 [M + H]+ (calcd for C64H93N12O14S2, 1317.6367).
−15 (c 0.50, MeOH); UV (MeOH) λmax (log ε) 210 (4.40), 227 (4.14), 277 (3.16) nm; IR (ZnSe) vmax 3300, 2958, 1655 cm−1; 1H and 13C NMR data, see Table 1; HRFABMS m/z 1317.6371 [M + H]+ (calcd for C64H93N12O14S2, 1317.6367). −52 (c 0.45, MeOH); UV (MeOH) λmax (log ε) 210 (4.41), 227 (4.10), 276 (3.16) nm; IR (ZnSe) vmax 3308, 2956, 1637 cm−1; 1H and 13C NMR data, see Table 1; MALDITOF-MS m/z 1260 [M]+ (calcd for C62H90N11O13S2, 1260).
−52 (c 0.45, MeOH); UV (MeOH) λmax (log ε) 210 (4.41), 227 (4.10), 276 (3.16) nm; IR (ZnSe) vmax 3308, 2956, 1637 cm−1; 1H and 13C NMR data, see Table 1; MALDITOF-MS m/z 1260 [M]+ (calcd for C62H90N11O13S2, 1260).3.4. Advanced Marfey’s Analysis of Compound 1
3.5. Analysis of the Configuration of the l-Ile Residue in Compound 1
3.6. Biological Assays
4. Conclusions
Supplementary Files
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Blunt, J.W.; Copp, B.R.; Keyzers, R.A.; Munro, M.H.G.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2013, 30, 237–323. [Google Scholar] [CrossRef]
- Yeung, B.K.S.; Nakao, Y.; Kinnel, R.B.; Carney, J.R.; Yoshida, W.Y.; Scheuer, P.J.; Kelly-Borges, M. The kapakahines, cyclic peptides from the marine sponge Cribrochalina olemda. J. Org. Chem. 1996, 61, 7168–7173. [Google Scholar]
- Nakao, Y.; Kuo, J.; Yoshida, W.Y.; Kelly, M.; Scheuer, P.J. More kapakahines from the marine sponge Cribrochalina olemda. Org. Lett. 2003, 5, 1387–1390. [Google Scholar] [CrossRef]
- Williams, D.E.; Austin, P.; Diaz-Marrero, A.R.; van Soest, R.; Matainaho, T.; Roskelley, C.D.; Roberge, M.; Andersen, R.J. Neopetrosiamides, peptides from the marine sponge Neopetrosia sp. that inhibit ameoboid invasion by human tumor cells. Org. Lett. 2005, 7, 4173–4176. [Google Scholar] [CrossRef]
- Araki, T.; Matsunaga, S.; Nakao, Y.; Furihata, K.; West, L.; Faulkner, D.J.; Fusetani, N. Koshikamide B, a cytotoxic peptide lactone from a marine sponge Theonella sp. J. Org. Chem. 2008, 73, 7889–7894. [Google Scholar] [CrossRef]
- Plaza, A.; Bifulco, G.; Masullo, M.; Lloyd, J.R.; Keffer, J.L.; Colin, P.L.; Hooper, J.N.A.; Bell, L.J.; Bewley, C.A. Mutremdamide A and koshikamides C-H, peptide inhibitors of HIV-1 entry from different Theonella species. J. Org. Chem. 2010, 75, 4344–4355. [Google Scholar] [CrossRef]
- Yeoka, R.; Ise, Y.; Ohtsuka, S.; Okada, S.; Yamori, T.; Matsunaga, S. Yaku’amide A and B, cytotoxic linear peptides rich in dehydroamino acids from the marine sponge Ceratopsion sp. J. Am. Chem. Soc. 2010, 132, 17692–17694. [Google Scholar] [CrossRef]
- Festa, C.; de Marino, S.; Sepe, V.; D’Auria, M.V.; Bifulco, G.; Débitus, C.; Bucci, M.; Vellecco, V.; Zampella, A. Solomonamides A and B, new anti-inflammatory peptides from Theonella swinhoei. Org. Lett. 2011, 13, 1532–1535. [Google Scholar] [CrossRef]
- Woo, J.-K.; Jeon, J.-E.; Kim, C.-K.; Sim, C.J.; Oh, D.-C.; Oh, K.-B.; Shin, J. Gombamide A, a cyclic thiopeptide from the sponge Clathra gombawuiensis. J. Nat. Prod. 2013, 76, 1380–1383. [Google Scholar] [CrossRef]
- Fujii, K.; Ikai, Y.; Mayumi, T.; Oka, H.; Suzuki, M.; Harada, K. A nonempirical method using LC/MS for determination of the absolute configuration of constituent amino acids in a peptide: Elucidation of limitations of Marfey’s method and of its separation mechanism. Anal. Chem. 1997, 69, 3346–3352. [Google Scholar] [CrossRef]
- Fujii, K.; Ikai, Y.; Oka, H.; Suzuki, M.; Harada, K. A nonempirical method using LC/MS for determination of the absolute configuration of constituent amino acids in a peptide: Combination of Marfey’s method with mass spectrometry and its practical application. Anal. Chem. 1997, 69, 5146–5151. [Google Scholar] [CrossRef]
- Davis, R.A.; Mangalindan, G.C.; Bojo, Z.P.; Antemano, R.R.; Rodriguez, N.O.; Concepcion, G.P.; Samson, S.C.; Guzman, D.; Cruz, L.J.; Tasdemir, D.; et al. Microcionamides A and B, bioactive peptides from the Philippine sponge Clathria (Thalysias) abietina. J. Org. Chem. 2004, 69, 4170–4176. [Google Scholar] [CrossRef]
- Katritzky, A.R.; Tala, S.R.; Abo-Dya, N.E.; Ibrahim, T.S.; El-Feky, S.A.; Gyanda, K.; Pandya, K.M. Chemical ligation of S-scylated cysteine peptides to form native peptides via 5-, 11-, and 14-membered cyclic transition states. J. Org. Chem. 2011, 76, 85–96. [Google Scholar] [CrossRef]
- Laird, D.W.; LaBarbera, D.V.; Feng, X.; Bugni, T.S.; Harper, M.K.; Ireland, C.M. Halogenated cyclic peptides isolated from the sponge Corticium sp. J. Nat. Prod. 2007, 70, 741–746. [Google Scholar] [CrossRef]
- Li, H.; Bowling, J.J.; Fronczek, F.R.; Hong, J.; Jabba, S.V.; Murray, T.F.; Ha, N.C.; Hamman, M.T.; Jung, J.H. Asteropsin A: An unusual cystine-crosslinked peptide from porifera enhances neuronal Ca2+ influx. Biochim. Biophys. Acta 2013, 1830, 2591–2599. [Google Scholar] [CrossRef]
- Tanaka, C.; Tanaka, J.; Bolland, R.; Marriott, G.; Higa, T. Seragamides A-F, new actin-targeting depsipeptides from the sponge Suberites japonicus Thiele. Tetrahedron 2006, 62, 3536–3542. [Google Scholar] [CrossRef]
- Crews, P.; Manes, L.V.; Boehler, M. Jasplakinolide, a cyclodepsipeptide from the marine sponge, Jaspis sp. Tetrahedron Lett. 1986, 27, 2797–2800. [Google Scholar] [CrossRef]
- Zabriskie, T.M.; Klocke, J.A.; Ireland, C.M.; Marcus, A.H.; Molinski, T.F.; Faulkner, D.J.; Xu, C.; Clardy, J.C. Jaspamide, a modified peptide from a Jaspis Sponge, with insecticidal and antifungal activity. J. Am. Chem. Soc. 1986, 108, 3123–3124. [Google Scholar] [CrossRef]
- Oh, K.-B.; Lee, J.H.; Chung, S.-C.; Shin, J.; Shin, H.J.; Kim, H.-K.; Lee., H.-S. Antimicrobial activities of the bromophenols from the red alga Odonthalia corymbifera and some synthetic derivatives. Bioorg. Med. Chem. Lett. 2008, 18, 104–108. [Google Scholar] [CrossRef]
- Shim, E.J.; Sim, C.J. Two new species of genus Suberites (Hadromerida: Suberitidae) from Korea. Korean J. Syst. Zool. 2008, 24, 215–218. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid colometric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Ulukaya, E.; Ozdikicioglu, F.; Oral, A.Y.; Demirci, M. The MTT assay yields a relatively lower result of growth inhibition than the ATP assay depending on the chemotherapeutic drugs tested. Toxicol. In Vitro 2008, 22, 232–239. [Google Scholar] [CrossRef]
- Johansson, M.; Karlsson, L.; Wennergren, M.; Jansson, T.; Powell, T.L. Activity and protein expression of Na+/K+ ATPase are reduced in microvillous syncytiotrophoblast plasma membranes isolated from pregnancies complicated by intrauterine growth restriction. J. Clin. Endocrinol. MeTab. 2003, 88, 2831–2837. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Song, J.; Jeon, J.-e.; Won, T.H.; Sim, C.J.; Oh, D.-C.; Oh, K.-B.; Shin, J. New Cyclic Cystine Bridged Peptides from the Sponge Suberites waedoensis. Mar. Drugs 2014, 12, 2760-2770. https://doi.org/10.3390/md12052760
Song J, Jeon J-e, Won TH, Sim CJ, Oh D-C, Oh K-B, Shin J. New Cyclic Cystine Bridged Peptides from the Sponge Suberites waedoensis. Marine Drugs. 2014; 12(5):2760-2770. https://doi.org/10.3390/md12052760
Chicago/Turabian StyleSong, Jinhaeng, Ju-eun Jeon, Tae Hyung Won, Chung J. Sim, Dong-Chan Oh, Ki-Bong Oh, and Jongheon Shin. 2014. "New Cyclic Cystine Bridged Peptides from the Sponge Suberites waedoensis" Marine Drugs 12, no. 5: 2760-2770. https://doi.org/10.3390/md12052760
APA StyleSong, J., Jeon, J.-e., Won, T. H., Sim, C. J., Oh, D.-C., Oh, K.-B., & Shin, J. (2014). New Cyclic Cystine Bridged Peptides from the Sponge Suberites waedoensis. Marine Drugs, 12(5), 2760-2770. https://doi.org/10.3390/md12052760