Natural Products from Mangrove Actinomycetes
Abstract
:1. Introduction
2. Natural Products Produced by Mangrove Actinomycetes
2.1. Alkaloids
2.1.1. Cyclic Dipeptides

2.1.2. Indole Alkaloids

2.1.3. Naphthyridines
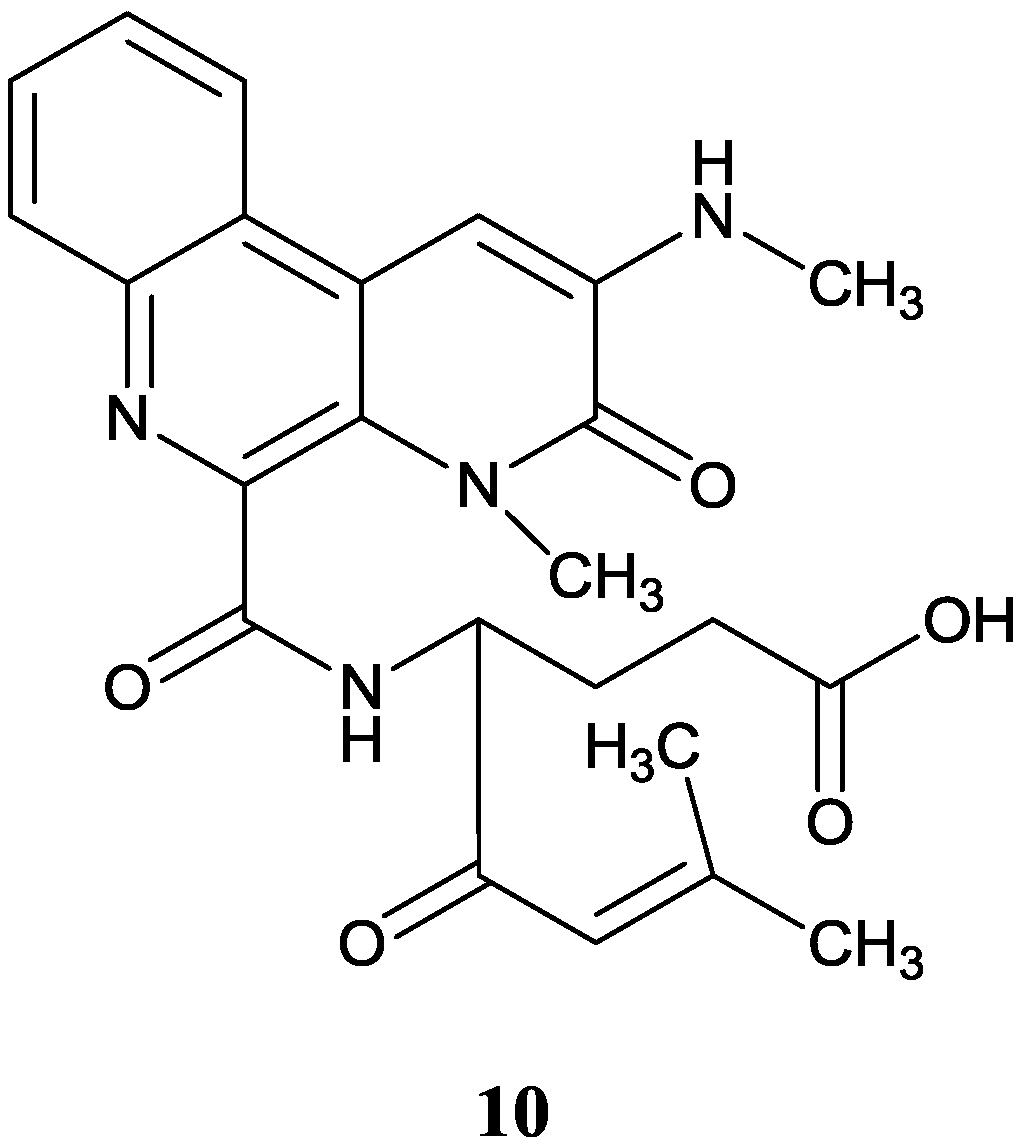
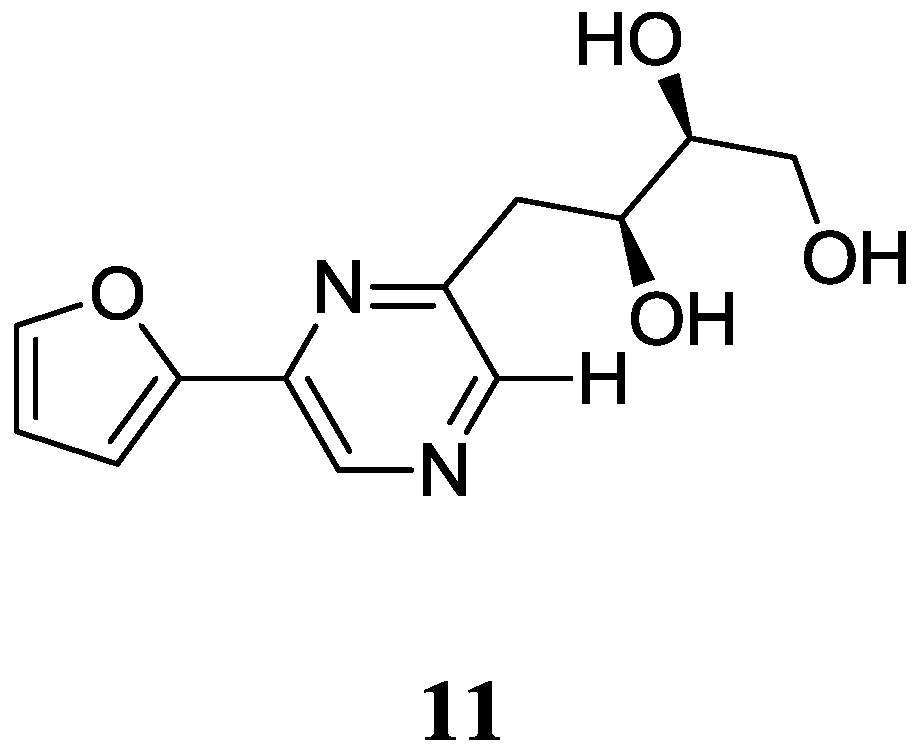
2.1.4. Pyrazine Derivatives
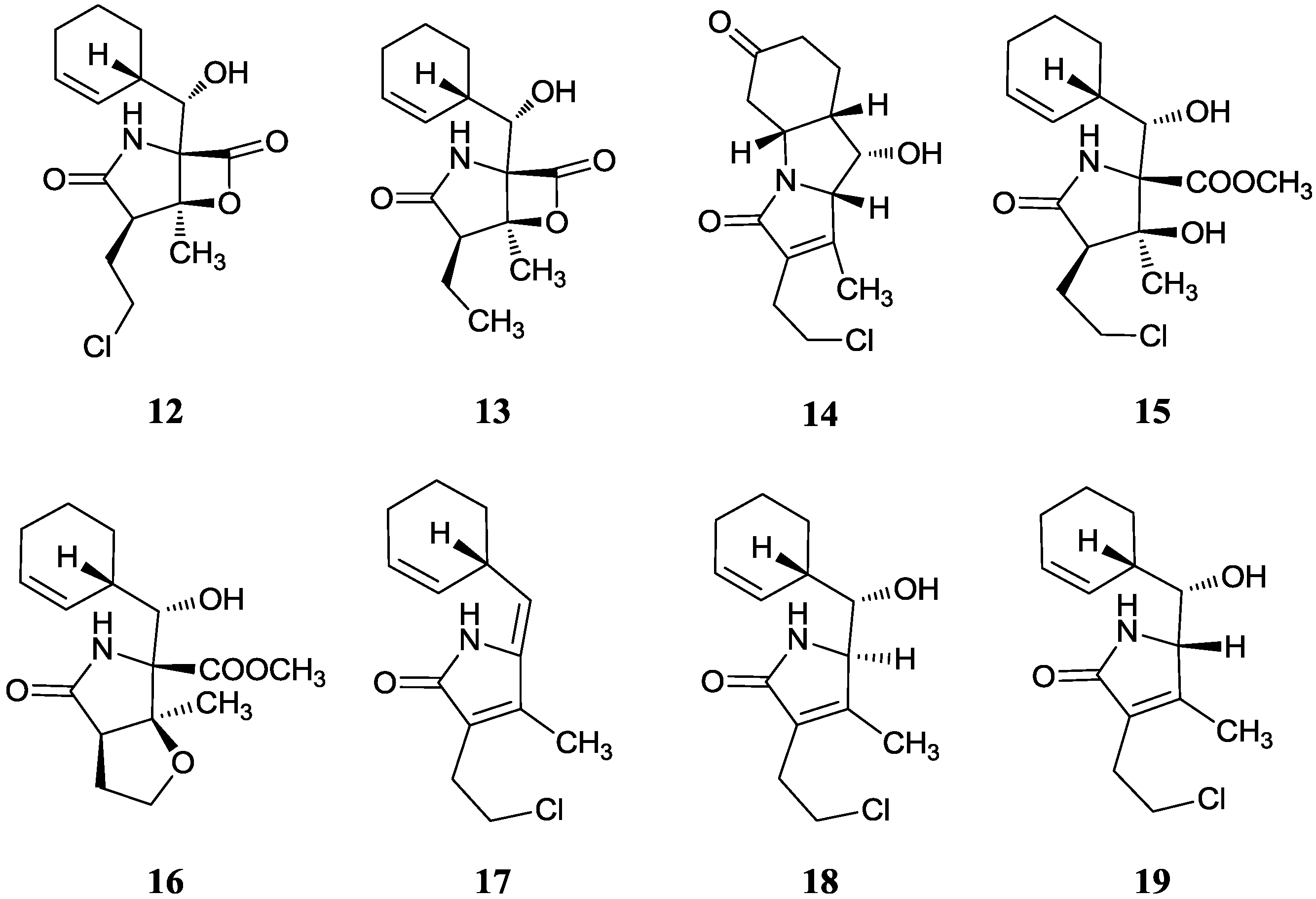
2.1.5. Salinosporamides
2.1.6. Other Alkaloids

2.2. Benzene Derivatives
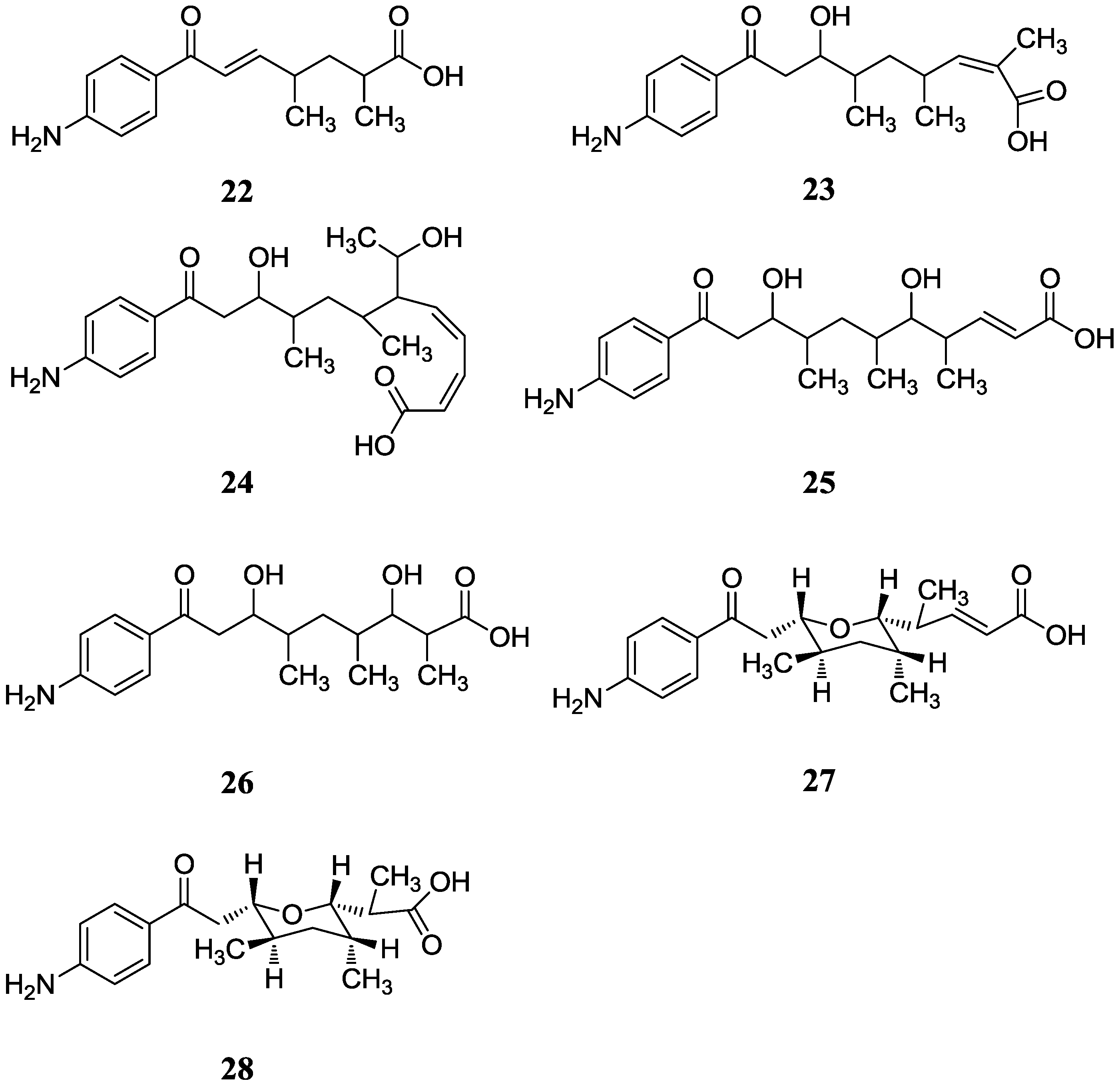
2.2.1. p-Aminoacetophenonic Acids
2.2.2. Benzamides

2.2.3. Benzopyran derivatives
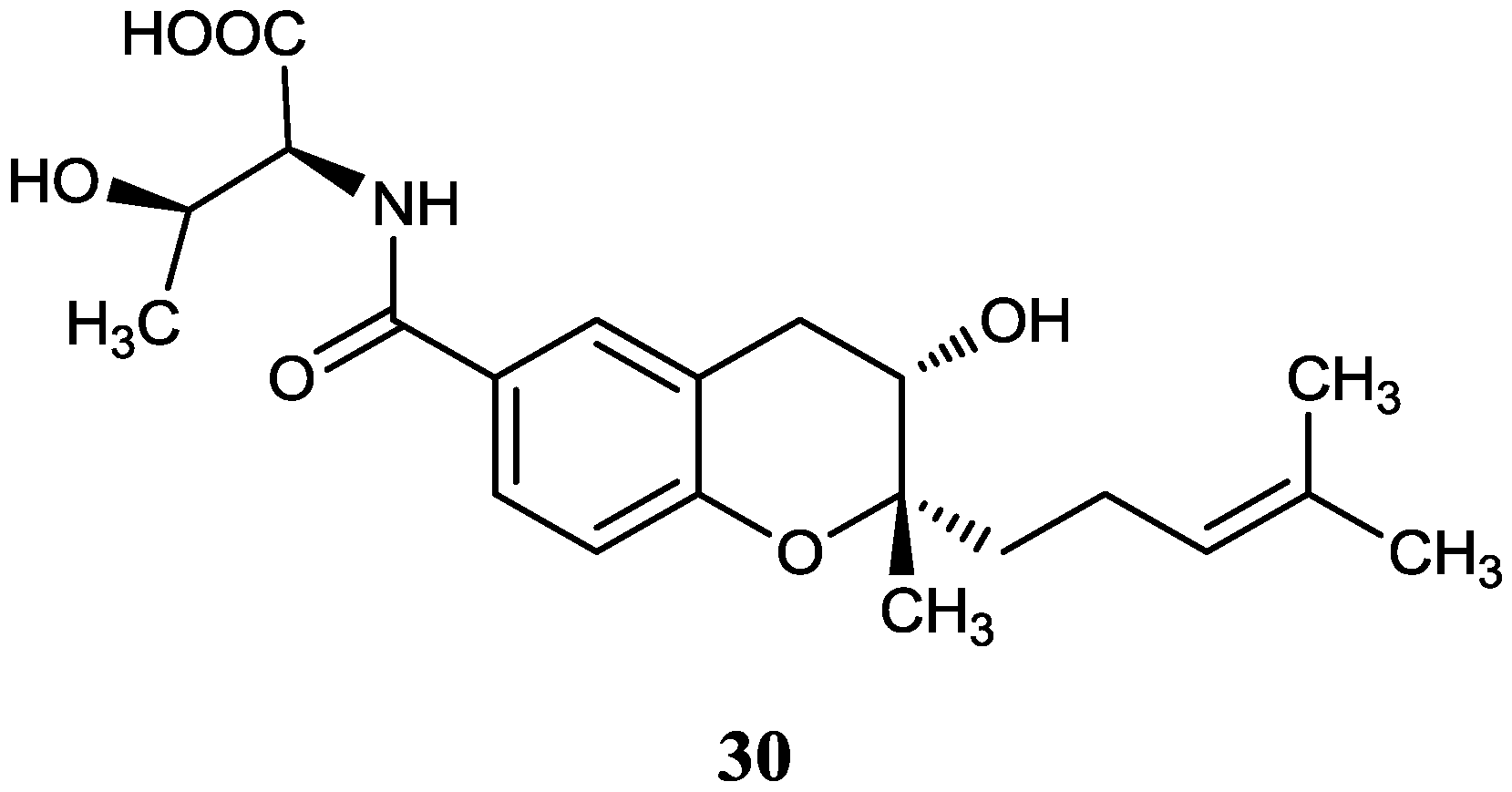
2.2.4. Phenols
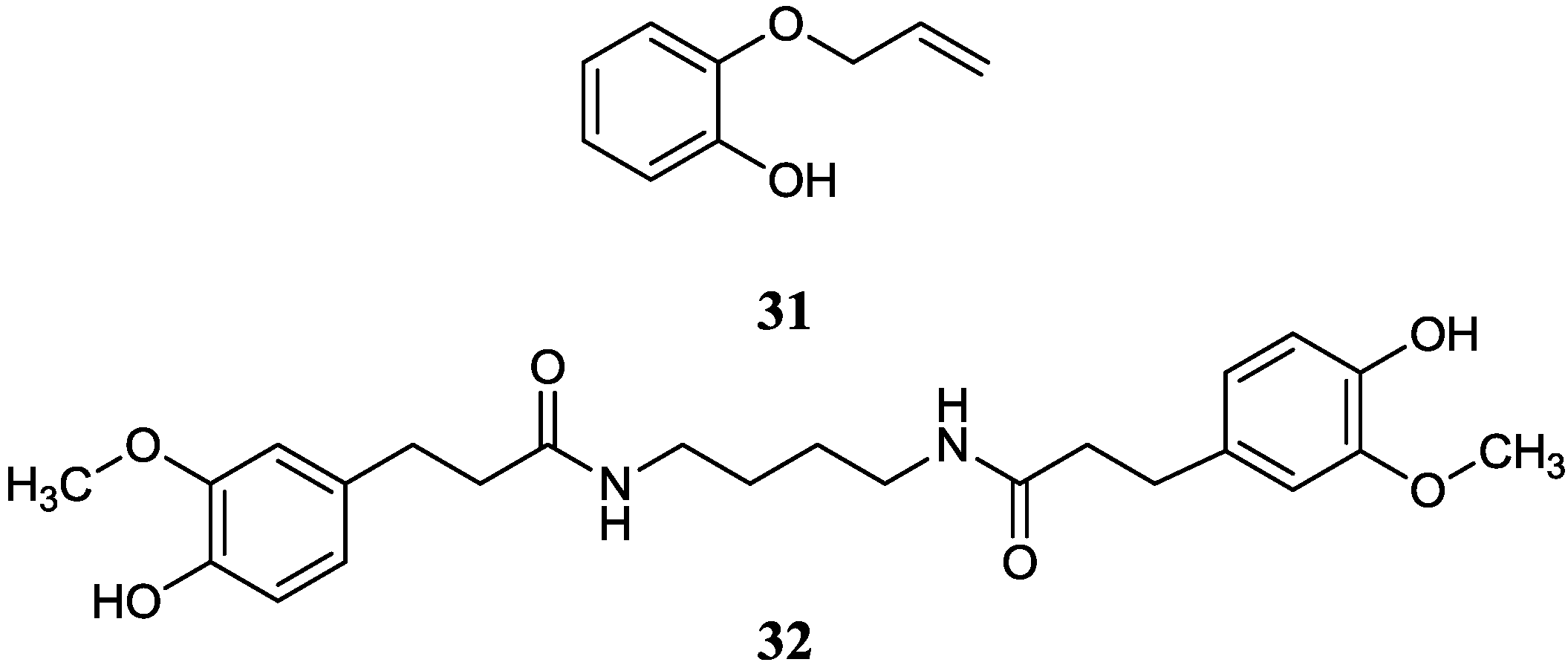
2.2.5. Other Benzene Derivative

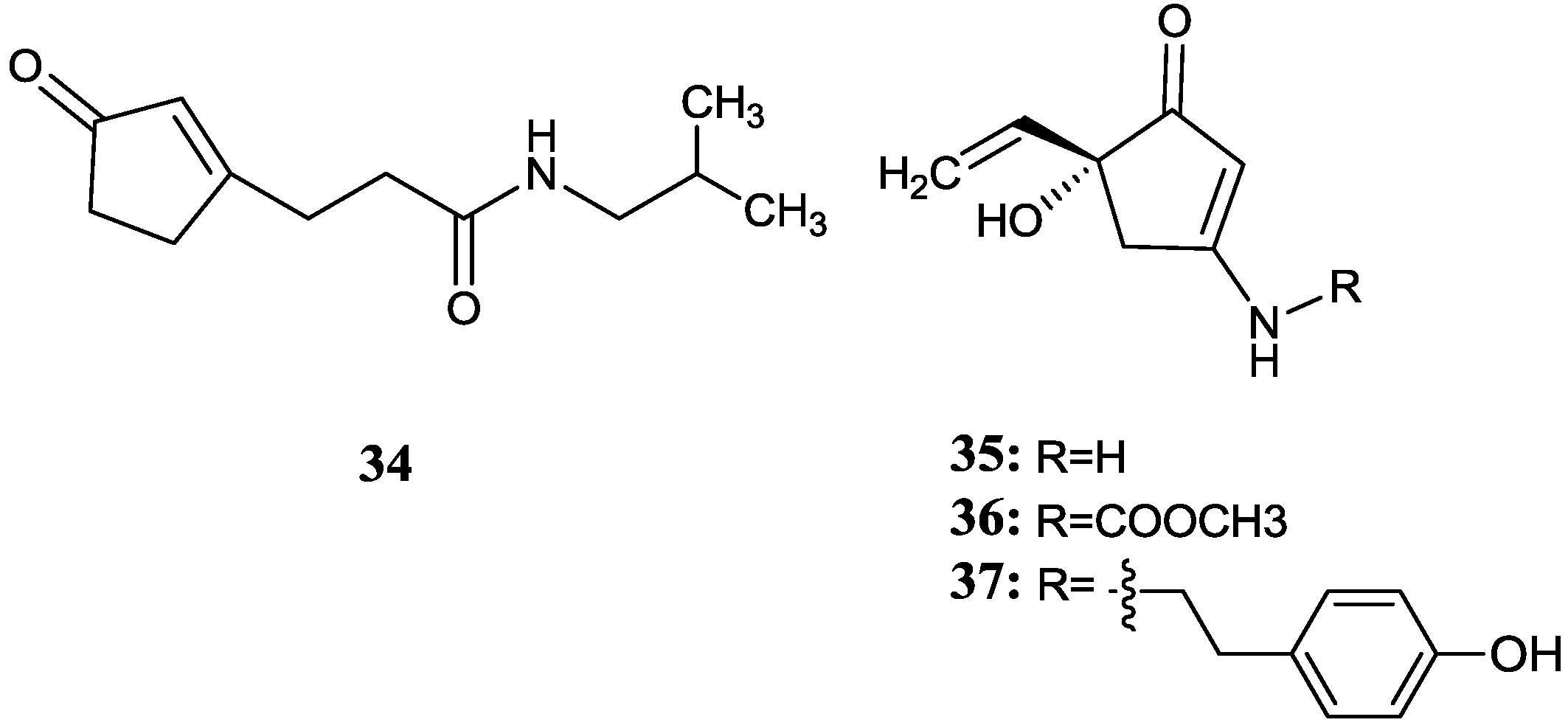
2.3. Cyclopentenone Derivatives
2.4. Dilactones
2.4.1. Antimycins
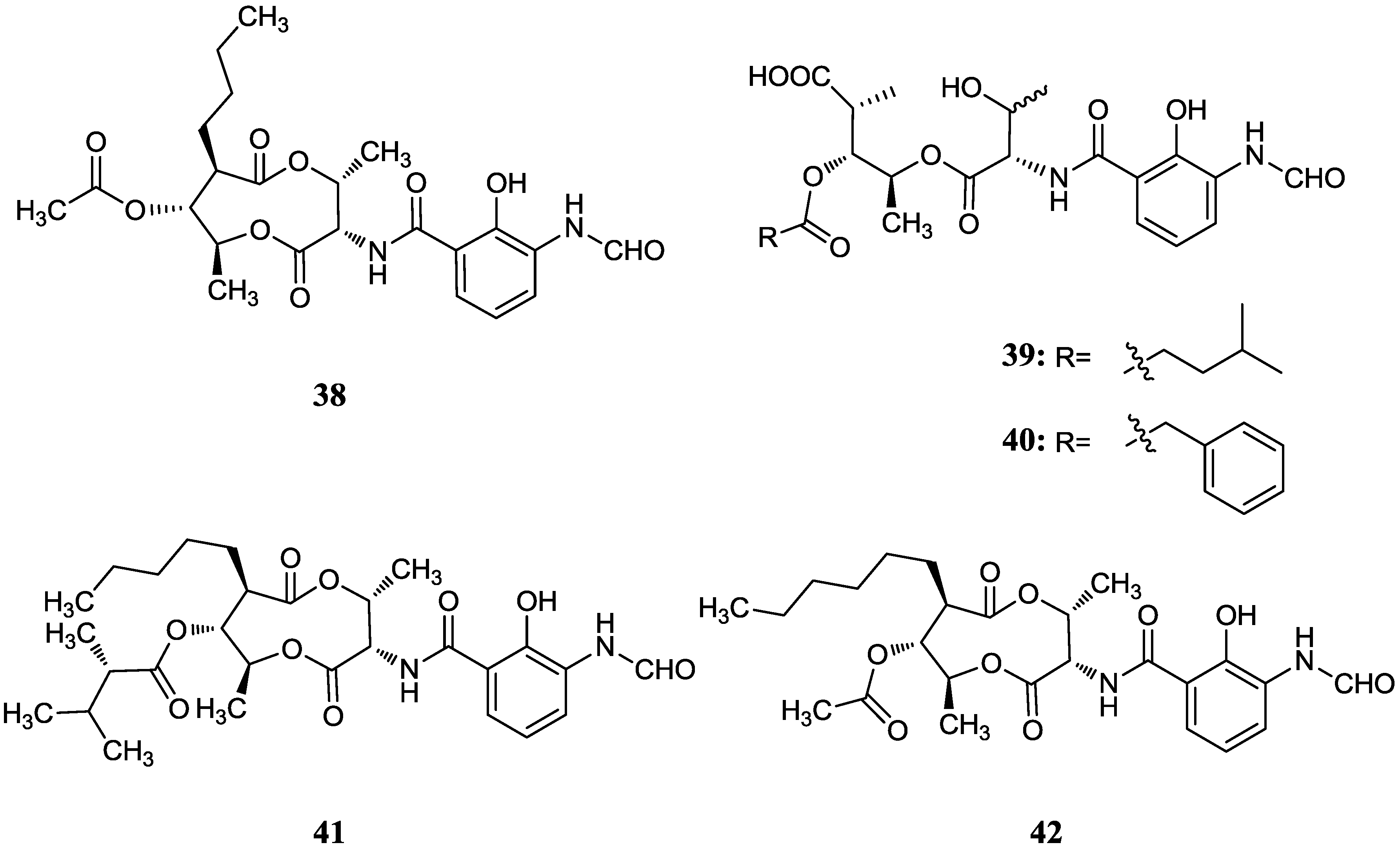
2.4.2. Other Dilactone
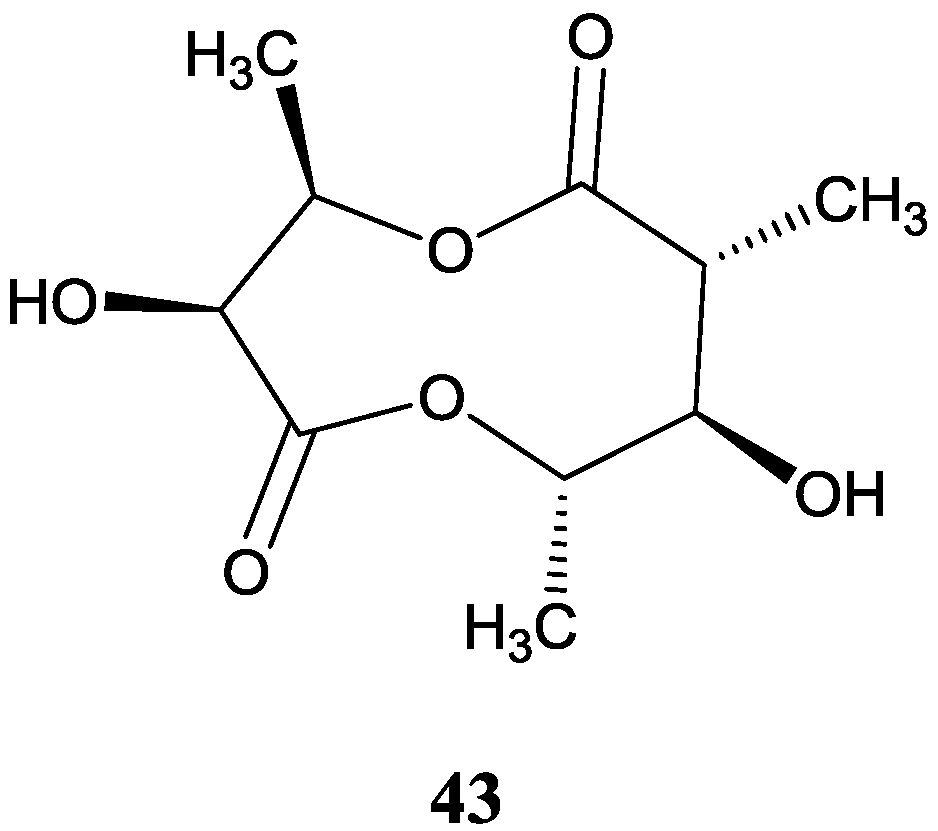
2.5. Macrolides
2.5.1. Ansa-macrolides
2.5.2. Macrocyclic Dilactones


2.5.3. Macrocyclic Lactones

2.6. 2-Pyranones Derivatives

2.7. Sesquiterpenes
2.7.1. Eudesmene-Type Sesquiterpenes
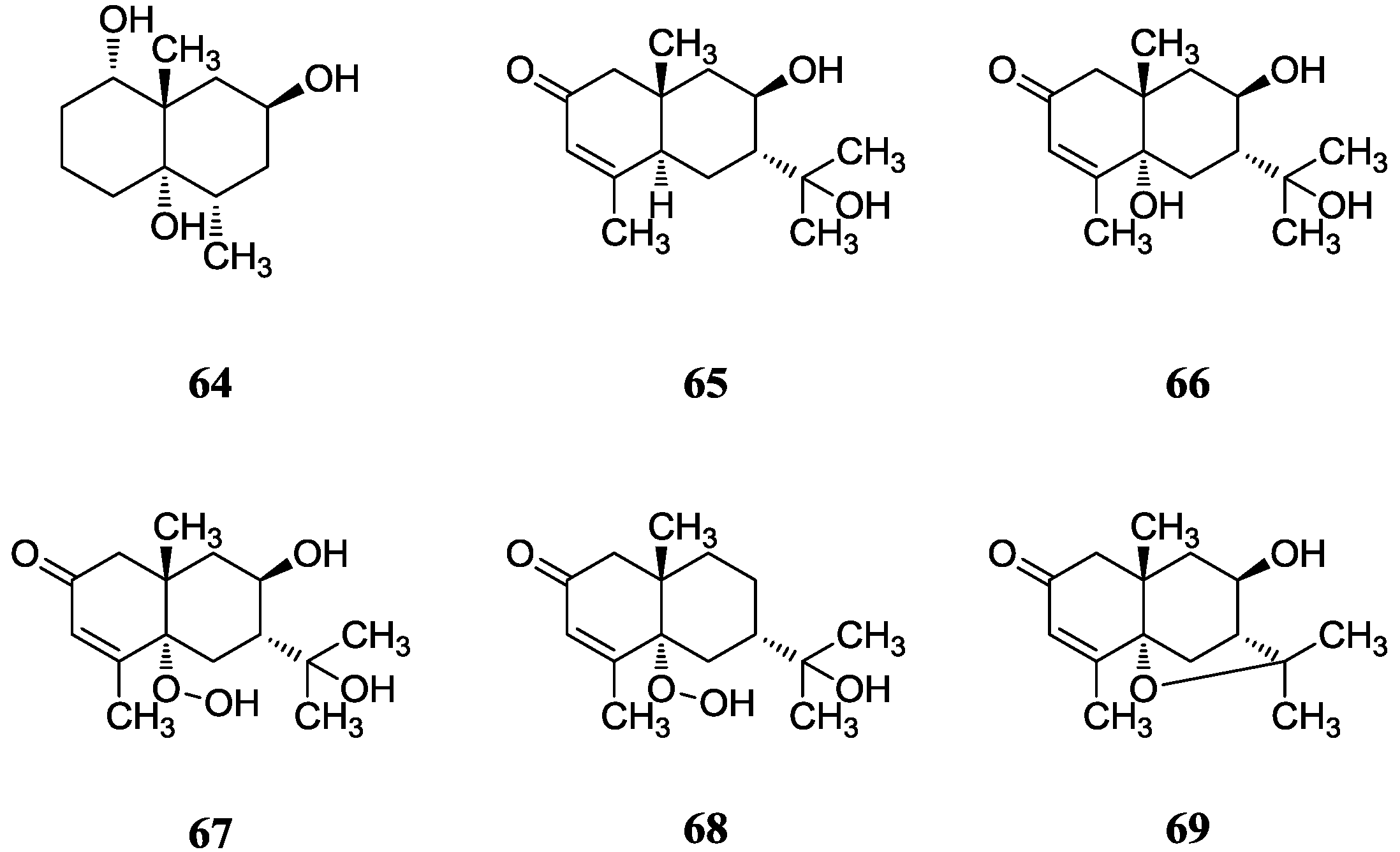
2.7.2. Germacrane-Type Sesquiterpenes

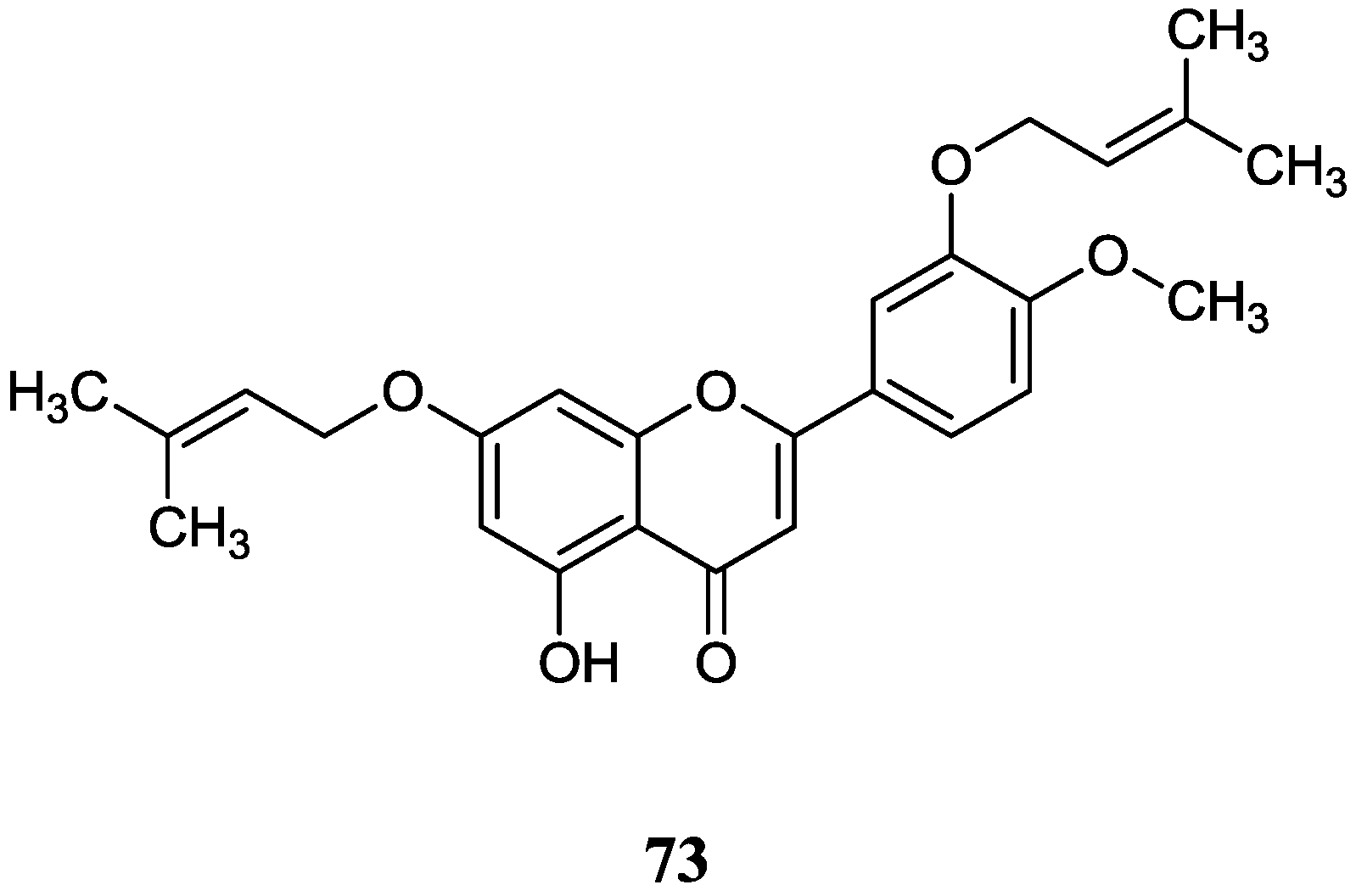
2.8. Miscellaneous
2.8.1. 2-Acylglycerol Derivatives

2.8.2. Flavones
| Compound | Isolate | Source | Bioactivity | Reference |
|---|---|---|---|---|
 | Streptomyces sp. 060524 | Mangrove seaweed (Haikou, China) | Antitumor | [76] |
 | Micromonospora sp. M2DG17 | Composite mangrove sediment (Haikou, China) | Antitumor | [77,78] |
 | Jishengella endophytica 161111 | Mangrove rhizosphere soil and root (Hainan, China) | Anti-H1N1 virus | [36,37] |
 | Streptomyces sp. 172614 | Mangrove soil (Fujian, China) | Antitumor | [33] |
 | Streptomyces sp. 061316 | Mangrove soil (Wenchang, China) | Caspase-3 inhibitory activity | [49] |
 | Marine actinomycete strain H83-3 | Mangrove bottom mud (South China Sea) | Antifungal | [79] |
 | Streptomycete sp. B7064 | Mangrove sediment (Pohoiki, Hawaii) | Antimicrobial | [67] |
 | Streptomyces antibioticus H74-18 | Mangrove bottom soil (South China Sea) | Antifungal | [59] |
 | Micromonospora rifamycinica AM105 | Mangrove sediment (South China Sea) | Antibacterial | [80] |
 | Streptomyces sp. 211726 | Mangrove rhizosphere soil (Wenchang, China) | Antimicrobial Antitumor | [68] |
 | Streptomyces griseus subsp. | Mangrove plant stems (Xiamen, China) | Unknown | [73] |
3. Concluding Remarks and Future Perspectives
Acknowledgments
Conflicts of Interest
References
- Debbab, A.; Aly, A.H.; Lin, W.H.; Proksch, P. Bioactive compounds from marine bacteria and fungi. Microb. Biotechnol. 2010, 3, 544–563. [Google Scholar] [CrossRef]
- Thornburg, C.C.; Zabriskie, T.M.; McPhail, K.L. Deep-sea hydrothermal vents: Potential hot spots for natural products discovery? J. Nat. Prod. 2010, 73, 489–499. [Google Scholar] [CrossRef]
- Abdel-Mageed, W.M.; Milne, B.F.; Wagner, M.; Schumacher, M.; Sandor, P.; Pathom-aree, W.; Goodfellow, M.; Bull, A.T.; Horikoshi, K.; Ebel, R.; et al. Dermacozines, a new phenazine family from deep-sea dermacocci isolated from a Mariana Trench sediment. Org. Biomol. Chem. 2010, 8, 2352–2362. [Google Scholar] [CrossRef]
- Bull, A.T.; Asenjo, J.A. Microbiology of hyper-arid environments: Recent insights from the Atacama Desert, Chile. Antonie Van Leeuwenhoek 2013, 103, 1173–1179. [Google Scholar] [CrossRef]
- Liu, J.T.; Lu, X.L.; Liu, X.Y.; Gao, Y.; Hu, B.; Jiao, B.H.; Zheng, H. Bioactive natural products from the antarctic and arctic organisms. Mini Rev. Med. Chem. 2013, 13, 617–626. [Google Scholar] [CrossRef]
- Amrita, K.; Nitin, J.; Devi, C.S. Novel bioactive compounds from mangrove derived actinomycetes. Int. Res. J. Pharm. 2012, 3, 25–29. [Google Scholar]
- Giri, C.; Ochieng, E.; Tieszen, L.L.; Zhu, Z.; Singh, A.; Loveland, T.; Masek, J.; Duke, N. Status and distribution of mangrove forests of the world using earth observation satellite data. Glob. Ecol. Biogeogr. 2011, 20, 154–159. [Google Scholar] [CrossRef]
- Holguin, G.; Vazquez, P.; Bashan, Y. The role of sediment microorganisms in the productivity, conservation, and rehabilitation of mangrove ecosystems: An overview. Biol. Fertil. Soils 2001, 33, 265–278. [Google Scholar] [CrossRef]
- Wu, S.L.; Jiang, L.M. Recent advances in mangrove actinomycetes. Curr. Biotechnol. 2012, 2, 335–340. [Google Scholar]
- Li, M.Y.; Xiao, Q.; Pan, J.Y.; Wu, J. Natural products from semi-mangrove flora: Source, chemistry and bioactivities. Nat. Prod. Rep. 2009, 26, 281–298. [Google Scholar] [CrossRef]
- Hong, K.; Gao, A.H.; Xie, Q.Y.; Gao, H.; Zhuang, L.; Lin, H.P.; Yu, H.P.; Li, J.; Yao, X.S.; Goodfellow, M.; et al. Actinomycetes for marine drug discovery isolated from mangrove soils and plants in China. Mar. Drugs 2009, 7, 24–44. [Google Scholar] [CrossRef]
- Calcul, L.; Waterman, C.; Ma, W.S.; Lebar, M.D.; Harter, C.; Mutka, T.; Morton, L.; Maignan, P.; van Olphen, A.; Kyle, D.E.; et al. Screening mangrove endophytic fungi for antimalarial natural products. Mar. Drugs 2013, 11, 5036–5050. [Google Scholar] [CrossRef]
- Liu, A.R.; Wu, X.P.; Xu, T. Research advances in endophytic fungi of mangrove. J. Appl. Ecol. 2007, 18, 912–918. [Google Scholar]
- Shilpi, J.A.; Islam, M.E.; Billah, M.; Islam, K.M.; Sabrin, F.; Uddin, S.J.; Nahar, L.; Sarker, S.D. Antinociceptive, anti-inflammatory, and antipyretic activity of mangrove plants: A mini review. Adv. Pharmacol. Sci. 2012, 2012, 576086:1–576086:7. [Google Scholar]
- Patra, J.K.; Mohanta, Y.K. Antimicrobial compounds from mangrove plants: A pharmaceutical prospective. Chin. J. Integr. Med. 2014, 20, 311–320. [Google Scholar] [CrossRef]
- Wu, J.; Xiao, Q.; Xu, J.; Li, M.Y.; Pan, J.Y.; Yang, M.H. Natural products from true mangrove flora: Source, chemistry and bioactivities. Nat. Prod. Rep. 2008, 25, 955–981. [Google Scholar] [CrossRef]
- Dharmaraj, S. Marine Streptomyces as a novel source of bioactive substances. World J. Microbiol. Biotechnol. 2010, 26, 2123–2139. [Google Scholar] [CrossRef]
- Berdy, J. Bioactive microbial metabolites. J. Antibiot. 2005, 58, 1–26. [Google Scholar] [CrossRef]
- Watve, M.G.; Tickoo, R.; Jog, M.M.; Bhole, B.D. How many antibiotics are produced by the genus Streptomyces? Arch. Microbiol. 2001, 176, 386–390. [Google Scholar] [CrossRef]
- He, J.; Zhang, D.; Xu, Y.; Zhang, X.; Tang, S.; Xu, L.; Li, W. Diversity and bioactivities of culturable marine actinobacteria isolated from mangrove sediment in Indian Ocean. Acta Microbiol. Sin. 2012, 52, 1195–1202. [Google Scholar]
- Usha, R.; Mala, K.K.; Venil, C.K.; Palaniswamy, M. Screening of actinomycetes from mangrove ecosystem for l-asparaginase activity and optimization by response surface methodology. Pol. Soc. Microbiol. 2011, 60, 213–221. [Google Scholar]
- Feling, R.H.; Buchanan, G.O.; Mincer, T.J.; Kauffman, C.A.; Jensen, P.R.; Fenical, W. Salinosporamide A: A highly cytotoxic proteasome inhibitor from a novel microbial source, a marine bacterium of the new genus Salinospora. Angew. Chem. Int. Ed. Engl. 2003, 42, 355–357. [Google Scholar] [CrossRef]
- Ding, L.; Munch, J.; Goerls, H.; Maier, A.; Fiebig, H.H.; Lin, W.H.; Hertweck, C. Xiamycin, a pentacyclic indolosesquiterpene with selective anti-HIV activity from a bacterial mangrove endophyte. Bioorg. Med. Chem. Lett. 2010, 20, 6685–6687. [Google Scholar] [CrossRef]
- Fu, P.; Yang, C.; Wang, Y.; Liu, P.; Ma, Y.; Xu, L.; Su, M.; Hong, K.; Zhu, W. Streptocarbazoles A and B, two novel indolocarbazoles from the marine-derived actinomycete strain Streptomyces sp. FMA. Org. Lett. 2012, 14, 2422–2425. [Google Scholar] [CrossRef]
- Hong, K. Actinomycetes from mangrove and their secondary metabolites—A review. Acta Microbiol. Sin. 2013, 53, 1131–1141. [Google Scholar]
- Jensen, P.R.; Mafnas, C. Biogeography of the marine actinomycete Salinispora. Environ. Microbiol. 2006, 8, 1881–1888. [Google Scholar] [CrossRef]
- Xie, X.C.; Mei, W.L.; Zeng, Y.B.; Lin, H.P.; Zhuang, L.; Dai, H.F.; Hong, K. Cytotoxic active components from marine Streptomyces sp. 124092. Chem. J. Chin. Univ. 2008, 29, 2183–2186. [Google Scholar]
- Ding, L.; Maier, A.; Fiebig, H.H.; Lin, W.H.; Hertweck, C. A family of multicyclic indolosesquiterpenes from a bacterial endophyte. Org. Biomol. Chem. 2011, 9, 4029–4031. [Google Scholar] [CrossRef]
- Xu, Z.; Baunach, M.; Ding, L.; Hertweck, C. Bacterial synthesis of diverse indole terpene alkaloids by an unparalleled cyclization sequence. Angew. Chem. Int. Ed. Engl. 2012, 51, 10293–10297. [Google Scholar] [CrossRef]
- Li, H.; Zhang, Q.; Li, S.; Zhu, Y.; Zhang, G.; Zhang, H.; Tian, X.; Zhang, S.; Ju, J.; Zhang, C. Identification and characterization of xiamycin A and oxiamycin gene cluster reveals an oxidative cyclization strategy tailoring indolosesquiterpene biosynthesis. J. Am. Chem. Soc. 2012, 134, 8996–9005. [Google Scholar] [CrossRef]
- Zhang, Q.; Li, H.; Li, S.; Zhu, Y.; Zhang, G.; Zhang, H.; Zhang, W.; Shi, R.; Zhang, C. Carboxyl formation from methyl via triple hydroxylations by XiaM in xiamycin A biosynthesis. Org. Lett. 2012, 14, 6142–6145. [Google Scholar] [CrossRef]
- Li, T.; Du, Y.; Cui, Q.; Zhang, J.; Zhu, W.; Hong, K.; Li, W. Cloning, characterization and heterologous expression of the indolocarbazole biosynthetic gene cluster from marine-derived Streptomyces sanyensis FMA. Mar. Drugs 2013, 11, 466–488. [Google Scholar] [CrossRef]
- Li, X.B.; Tang, J.S.; Gao, H.; Ding, R.; Li, J.; Hong, K.; Yao, X.S. A new staurosporine analog from actinomycetes Streptomyces sp. 172614. J. Asian Nat. Prod. Res. 2011, 13, 765–769. [Google Scholar] [CrossRef]
- Li, X.L.; Xu, M.J.; Zhao, Y.L.; Xu, J. A novel benzo[f][1,7]naphthyridine produced by Streptomyces albogriseolus from mangrove sediments. Molecules 2010, 15, 9298–9307. [Google Scholar]
- Tian, C.; Jiao, X.; Liu, X.; Li, R.; Dong, L.; Liu, X.; Zhang, Z.; Xu, J.; Xu, M.; Xie, P. First total synthesis and determination of the absolute configuration of 1-N-methyl-3-methylamino-[N-butanoicacid-3-(9-methyl-8-propen-7-one)-amide]-benzo[f][1,7]naphthyridine-2-one, a novel benzonaphthyridine alkaloid. Tetrahedron Lett. 2012, 53, 4892–4895. [Google Scholar]
- Wang, P.; Kong, F.; Wei, J.; Wang, Y.; Wang, W.; Hong, K.; Zhu, W. Alkaloids from the mangrove-derived actinomycete Jishengella endophytica 161111. Mar. Drugs 2014, 12, 477–490. [Google Scholar] [CrossRef]
- Xie, Q.Y.; Wang, C.; Wang, R.; Qu, Z.; Lin, H.P.; Goodfellow, M.; Hong, K. Jishengella endophytica gen. nov., sp. nov., a new member of the family micromonosporaceae. Int. J. Syst. Evol. Microbiol. 2011, 61, 1153–1159. [Google Scholar] [CrossRef]
- Williams, P.G.; Buchanan, G.O.; Feling, R.H.; Kauffman, C.A.; Jensen, P.R.; Fenical, W. New cytotoxic salinosporamides from the marine actinomycete Salinispora tropica. J. Org. Chem. 2005, 70, 6196–6203. [Google Scholar] [CrossRef]
- Liu, Y.; Hazzard, C.; Eustaquio, A.S.; Reynolds, K.A.; Moore, B.S. Biosynthesis of salinosporamides from α,β-unsaturated fatty acids: Implications for extending polyketide synthase diversity. J. Am. Chem. Soc. 2009, 131, 10376–10377. [Google Scholar]
- Endo, A.; Danishefsky, S.J. Total synthesis of salinosporamide A. J. Am. Chem. Soc. 2005, 127, 8298–8299. [Google Scholar] [CrossRef]
- Potts, B.C.; Lam, K.S. Generating a generation of proteasome inhibitors: From microbial fermentation to total synthesis of salinosporamide A (Marizomib) and other salinosporamides. Mar. Drugs 2010, 8, 835–880. [Google Scholar] [CrossRef]
- Potts, B.C.; Albitar, M.X.; Anderson, K.C.; Baritaki, S.; Berkers, C.; Bonavida, B.; Chandra, J.; Chauhan, D.; Cusack, J.C., Jr.; Fenical, W.; et al. Marizomib, a proteasome inhibitor for all seasons: Preclinical profile and a framework for clinical trials. Curr. Cancer Drug Targets 2011, 11, 254–284. [Google Scholar] [CrossRef]
- Millward, M.; Price, T.; Townsend, A.; Sweeney, C.; Spencer, A.; Sukumaran, S.; Longenecker, A.; Lee, L.; Lay, A.; Sharma, G.; et al. Phase 1 clinical trial of the novel proteasome inhibitor marizomib with the histone deacetylase inhibitor vorinostat in patients with melanoma, pancreatic and lung cancer based on in vitro assessments of the combination. Investig. New Drugs 2012, 30, 2303–2317. [Google Scholar] [CrossRef]
- Lawasut, P.; Chauhan, D.; Laubach, J.; Hayes, C.; Fabre, C.; Maglio, M.; Mitsiades, C.; Hideshima, T.; Anderson, K.C.; Richardson, P.G. New proteasome inhibitors in myeloma. Curr. Hematol. Malig. Rep. 2012, 7, 258–266. [Google Scholar] [CrossRef]
- Izumikawa, M.; Hosoya, T.; Takagi, M.; Shin-ya, K. A new cyclizidine analog-JBIR-102-from Saccharopolyspora sp. RL78 isolated from mangrove soil. J. Antibiot. 2012, 65, 41–43. [Google Scholar] [CrossRef]
- Kyeremeh, K.; Acquah, K.S.; Sazak, A.; Houssen, W.; Tabudravu, J.; Deng, H.; Jaspars, M. Butremycin, the 3-hydroxyl derivative of ikarugamycin and a protonated aromatic tautomer of 5'-methylthioinosine from a Ghanaian Micromonospora sp. K310. Mar. Drugs 2014, 12, 999–1012. [Google Scholar] [CrossRef] [Green Version]
- Guan, S.H.; Sattler, I.; Lin, W.H.; Guo, D.A.; Grabley, S. p-Aminoacetophenonic acids produced by a mangrove endophyte: Streptomyces griseus subsp. J. Nat. Prod. 2005, 68, 1198–1200. [Google Scholar] [CrossRef]
- Wang, F.; Xu, M.; Li, Q.; Sattler, I.; Lin, W. p-Aminoacetophenonic acids produced by a mangrove endophyte Streptomyces sp. (strain HK10552). Molecules 2010, 15, 2782–2790. [Google Scholar]
- Chen, G.; Gao, H.; Tang, J.; Huang, Y.; Chen, Y.; Wang, Y.; Zhao, H.; Lin, H.; Xie, Q.; Hong, K.; et al. Benzamides and quinazolines from a mangrove actinomycetes Streptomyces sp. (No. 061316) and their inhibiting caspase-3 catalytic activity in vitro. Chem. Pharm. Bull. 2011, 59, 447–451. [Google Scholar]
- Xu, M.J.; Liu, X.J.; Zhao, Y.L.; Liu, D.; Xu, Z.H.; Lang, X.M.; Ao, P.; Lin, W.H.; Yang, S.L.; Zhang, Z.G.; et al. Identification and characterization of an anti-fibrotic benzopyran compound isolated from mangrove-derived Streptomyces xiamenensis. Mar. Drugs 2012, 10, 639–654. [Google Scholar] [CrossRef]
- Arumugam, M.; Mitra, A.; Jaisankar, P.; Dasgupta, S.; Sen, T.; Gachhui, R.; Kumar Mukhopadhyay, U.; Mukherjee, J. Isolation of an unusual metabolite 2-allyloxyphenol from a marine actinobacterium, its biological activities and applications. Appl. Microbiol. Biotechnol. 2010, 86, 109–117. [Google Scholar]
- Arumugam, M.; Mitra, A.; Pramanik, A.; Saha, M.; Gachhui, R.; Mukherjee, J. Streptomyces sundarbansensis sp. nov., an actinomycete that produces 2-allyloxyphenol. Int. J. Syst. Evol. Microbiol. 2011, 61, 2664–2669. [Google Scholar]
- Takagi, M.; Shin-Ya, K. New species of actinomycetes do not always produce new compounds with high frequency. J. Antibiot. 2011, 64, 699–701. [Google Scholar] [CrossRef]
- Kawahara, T.; Izumikawa, M.; Otoguro, M.; Yamamura, H.; Hayakawa, M.; Takagi, M.; Shin-ya, K. JBIR-94 and JBIR-125, antioxidative phenolic compounds from Streptomyces sp. R56–07. J. Nat. Prod. 2012, 75, 107–110. [Google Scholar]
- Lin, W.; Li, L.; Fu, H.; Sattler, I.; Huang, X.; Grabley, S. New cyclopentenone derivatives from an endophytic Streptomyces sp. isolated from the mangrove plant Aegiceras comiculatum. J. Antibiot. 2005, 58, 594–598. [Google Scholar]
- Yan, L.L.; Han, N.N.; Zhang, Y.Q.; Yu, L.Y.; Chen, J.; Wei, Y.Z.; Li, Q.P.; Tao, L.; Zheng, G.H.; Yang, S.E.; et al. Antimycin A18 produced by an endophytic Streptomyces albidoflavus isolated from a mangrove plant. J. Antibiot. 2010, 63, 259–261. [Google Scholar]
- Yan, L.L.; Han, N.N.; He, Q.Y.; Chen, J.; Yang, S.E.; Liu, S.W.; Tao, L.; Guo, L.; Jin, J.; Yu, L.Y.; et al. Preliminary studies on cytotoxicity of antimycin A18 and the biological characteristics of its producing strain I07A-01824. Chin. J. Antibiot. 2011, 36, 269–279. [Google Scholar]
- Han, Z.; Xu, Y.; McConnell, O.; Liu, L.; Li, Y.; Qi, S.; Huang, X.; Qian, P. Two antimycin A analogues from marine-derived actinomycete Streptomyces lusitanus. Mar. Drugs 2012, 10, 668–676. [Google Scholar] [CrossRef]
- Xu, L.Y.; Quan, X.S.; Wang, C.; Sheng, H.F.; Zhou, G.X.; Lin, B.R.; Jiang, R.W.; Yao, X.S. Antimycins A(19) and A(20), two new antimycins produced by marine actinomycete Streptomyces antibioticus H74-18. J. Antibiot. 2011, 64, 661–665. [Google Scholar]
- Inai, M.; Nishii, T.; Tanaka, A.; Kaku, H.; Horikawa, M.; Tsunoda, T. Total synthesis of the (+)-antimycin A family. Eur. J. Org. Chem. 2011, 2011, 2719–2729. [Google Scholar]
- Zhou, Z.L.; Jin, B.; Yin, W.Q.; Fu, C.Y.; Feng, H.F. Components in antineoplastic actinomycete strain (N2010–37) of bottom mud in mangrove. Chin. Herb. Med. 2011, 3, 165–167. [Google Scholar]
- Ding, L.; Maier, A.; Fiebig, H.H.; Gorls, H.; Lin, W.H.; Peschel, G.; Hertweck, C. Divergolides A-D from a mangrove endophyte reveal an unparalleled plasticity in ansa-macrolide biosynthesis. Angew. Chem. Int. Ed. Engl. 2011, 50, 1630–1634. [Google Scholar]
- Dai, W.M.; Zhao, G.; Wu, J. Toward a total synthesis of divergolide A; Synthesis of the amido hydroquinone core and the C10-C15 fragment. Synlett 2012, 23, 2845–2849. [Google Scholar]
- Rasapalli, S.; Jarugumilli, G.; Yarrapothu, G.R.; Golen, J.A.; Rheingold, A.L. Studies toward total synthesis of divergolides C and D. Org. Lett. 2013, 15, 1736–1739. [Google Scholar]
- Izumikawa, M.; Takagi, M.; Shin-Ya, K. Isolation of a novel macrocyclic dilactone—JBIR-101—from Promicromonospora sp. RL26. J. Antibiot. 2011, 64, 689–691. [Google Scholar] [CrossRef]
- Zeng, Q.; Huang, H.; Zhu, J.; Fang, Z.; Sun, Q.; Bao, S. A new nematicidal compound produced by Streptomyces albogriseolus HA10002. Antonie Van Leeuwenhoek 2013, 103, 1107–1111. [Google Scholar] [CrossRef]
- Asolkar, R.N.; Maskey, R.P.; Helmke, E.; Laatsch, H. Chalcomycin B, a new macrolide antibiotic from the marine isolate Streptomyces sp. B7064. J. Antibiot. 2002, 55, 893–898. [Google Scholar]
- Yuan, G.; Lin, H.; Wang, C.; Hong, K.; Liu, Y.; Li, J. 1H and 13C assignments of two new macrocyclic lactones isolated from Streptomyces sp. 211726 and revised assignments of azalomycins F3a, F4a and F5a. Magn. Reson. Chem. MRC 2011, 49, 30–37. [Google Scholar] [CrossRef]
- Yuan, G.; Hong, K.; Lin, H.; She, Z.; Li, J. New azalomycin F analogs from mangrove Streptomyces sp. 211726 with activity against microbes and cancer cells. Mar. Drugs 2013, 11, 817–829. [Google Scholar] [CrossRef]
- Lin, C.; Lu, C.H.; Shen, Y.M. Three new 2-pyranone derivatives from mangrove endophytic actinomycete strain Nocardiopsis sp. A00203. Rec. Nat. Prod. 2010, 4, 176–179. [Google Scholar]
- Xie, X.C.; Mei, W.L.; Zhao, Y.X.; Hong, K.; Dai, H.F. A new degraded sesquiterpene from marine actinomycete Streptomyces sp. 0616208. Chin. Chem. Lett. 2006, 17, 1463–1465. [Google Scholar]
- Ding, L.; Maier, A.; Fiebig, H.H.; Lin, W.H.; Peschel, G.; Hertweck, C. Kandenols A–E, eudesmenes from an endophytic Streptomyces sp. of the mangrove tree Kandelia candel. J. Nat. Prod. 2012, 75, 2223–2227. [Google Scholar] [CrossRef]
- Guan, S.; Grabley, S.; Groth, I.; Lin, W.; Christner, A.; Guo, D.; Sattler, I. Structure determination of germacrane-type sesquiterpene alcohols from an endophyte Streptomyces griseus subsp. Magn. Reson. Chem. MRC 2005, 43, 1028–1031. [Google Scholar] [CrossRef]
- Yuan, G.J.; Lin, H.P.; Hong, K. Identification of a new 2-acylglycerol from the secondary metabolites produced by marine Streptomyces sp. 211726. Chin. J. Mar. Drugs 2011, 30, 1–4. [Google Scholar]
- Ding, W.J.; Zhang, S.Q.; Wang, J.H.; Lin, Y.X.; Liang, Q.X.; Zhao, W.J.; Li, C.Y. A new di-O-prenylated flavone from an actinomycete Streptomyces sp. MA-12. J. Asian Nat. Prod. Res. 2013, 15, 209–214. [Google Scholar] [CrossRef]
- Hu, S.C.; Tan, R.X.; Zhuang, L.; Yan, L.P.; Hong, K. Isolation of cytotoxic compounds from marine Streptomyces sp. 060524. Chin. J. Antibiot. 2009, 34, S01–S03. [Google Scholar]
- Huang, Z.Y.; Tang, J.S.; Gao, H.; Li, Y.J.; Hong, K.; Li, J.; Yao, X.S. Studies on the cytotoxic constituents from marine actinomycete Micromonospora sp. M2DG17. Chin. J. Mar. Drugs 2011, 30, 29–33. [Google Scholar]
- Xie, Q.Y.; Qu, Z.; Lin, H.P.; Li, L.; Hong, K. Micromonospora haikouensis sp. Nov., isolated from mangrove soil. Antonie Van Leeuwenhoek 2012, 101, 649–655. [Google Scholar] [CrossRef]
- Quan, X.S.; Pu, X.M.; Zhang, Y.; Shen, H.F.; Zhou, G.X.; Lin, B.R. Bioactive chemical constituents of an actinomycete strain (No. H83–3) from the bottom mud of the mangrove zone in the South China Sea. Chin. J. Mar. Drugs 2011, 30, 7–13. [Google Scholar]
- Huang, H.; Wu, X.; Yi, S.; Zhou, Z.; Zhu, J.; Fang, Z.; Yue, J.; Bao, S. Rifamycin S and its geometric isomer produced by a newly found actinomycete, Micromonospora rifamycinica. Antonie Van Leeuwenhoek 2009, 95, 143–148. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Xu, D.-B.; Ye, W.-W.; Han, Y.; Deng, Z.-X.; Hong, K. Natural Products from Mangrove Actinomycetes. Mar. Drugs 2014, 12, 2590-2613. https://doi.org/10.3390/md12052590
Xu D-B, Ye W-W, Han Y, Deng Z-X, Hong K. Natural Products from Mangrove Actinomycetes. Marine Drugs. 2014; 12(5):2590-2613. https://doi.org/10.3390/md12052590
Chicago/Turabian StyleXu, Dong-Bo, Wan-Wan Ye, Ying Han, Zi-Xin Deng, and Kui Hong. 2014. "Natural Products from Mangrove Actinomycetes" Marine Drugs 12, no. 5: 2590-2613. https://doi.org/10.3390/md12052590






