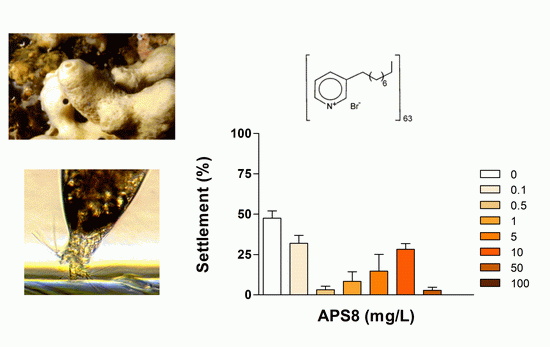
Antifouling Activity of Synthetic Alkylpyridinium Polymers Using the Barnacle Model
Abstract
:1. Introduction

2. Results and Discussion
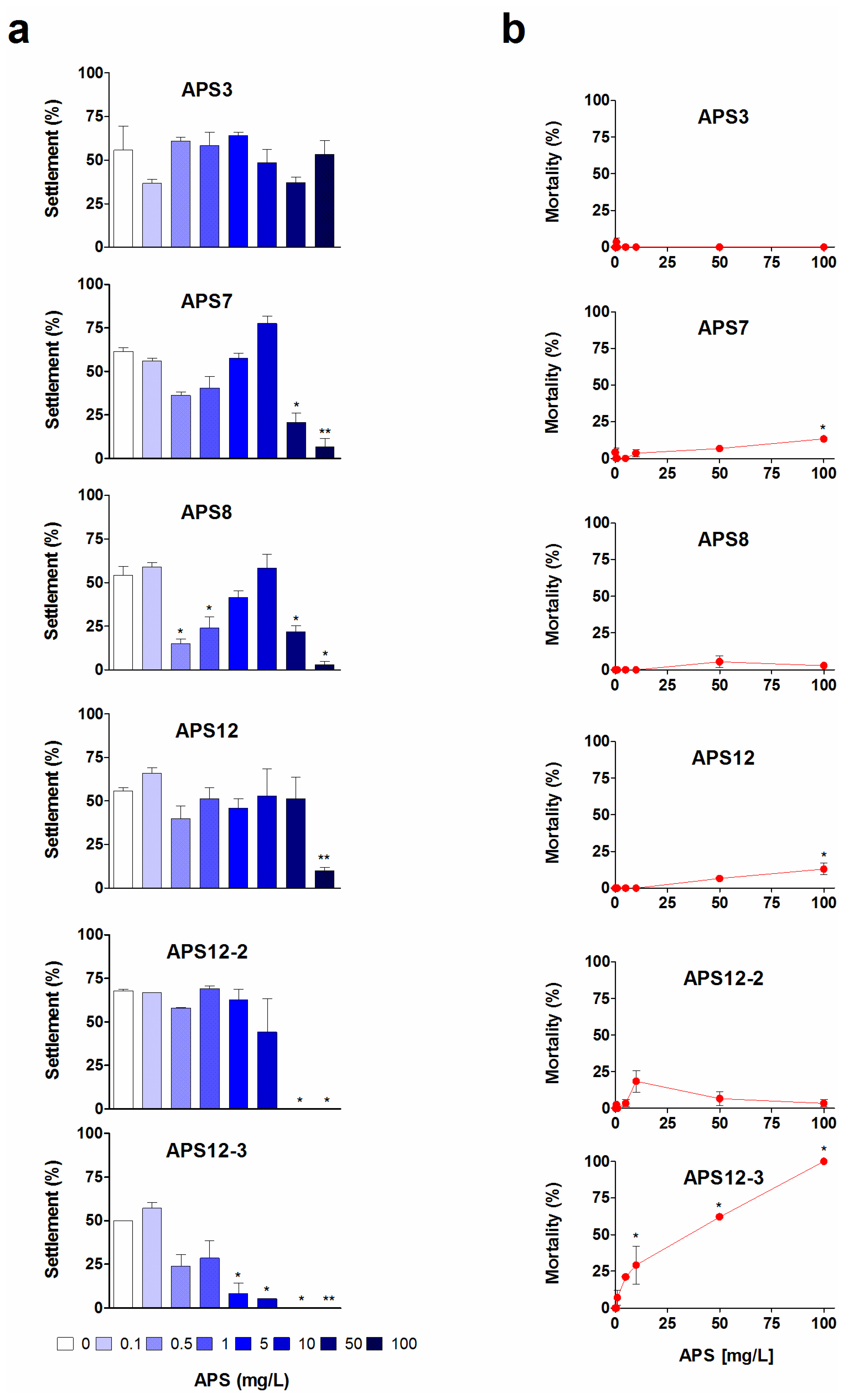
2.1. Anti-Settlement Assay
| Compound | Treatment (h) | EC50 (mg/L) | LC50 (mg/L) | EC50 (μM) | LC50 (μM) |
|---|---|---|---|---|---|
| Poly-APS 1 | 24 | 0.27 (0.15–0.47) | 0.049 | ||
| Zn pyrithione 1 | 24 | 0.02 | 0.063 | ||
| Cu pyrithione 1 | 24 | <0.01 | <0.032 | ||
| APS3 | 24 | 5.72 (4.24–7.72) | >100 | 3.9 | |
| 48 | >100 | >100 | |||
| 72 | >100 | >100 | |||
| APS7 | 24 | 10.50 (8.47–13.01) | >100 | 4.5 | |
| 48 | 25.86 (23.29–28.71) | >100 | 11.1 | ||
| 72 | 29.38 (26.17–32.99) | >100 | 12.6 | ||
| APS8 2 | 24 | 0.32 (0.26–0.39) | >100 | 0.026 | |
| 48 | 0.50 (0.36–0.70) | >100 | 0.042 | ||
| 72 | 2.33 (1.78–3.04) | >100 | 0.195 | ||
| APS12 | 24 | nc | >100 | ||
| 48 | nc | >100 | |||
| 72 | 49.82 (37.18–66.76) | >100 | 4.0 | ||
| APS12-2 | 24 | 8.78 (8.37-9.20) | >100 | 0.597 | |
| 48 | 9.38 (8.76–10.05) | >100 | 0.638 | ||
| 72 | 11.13 (10.38–11.94) | >100 | 0.757 | ||
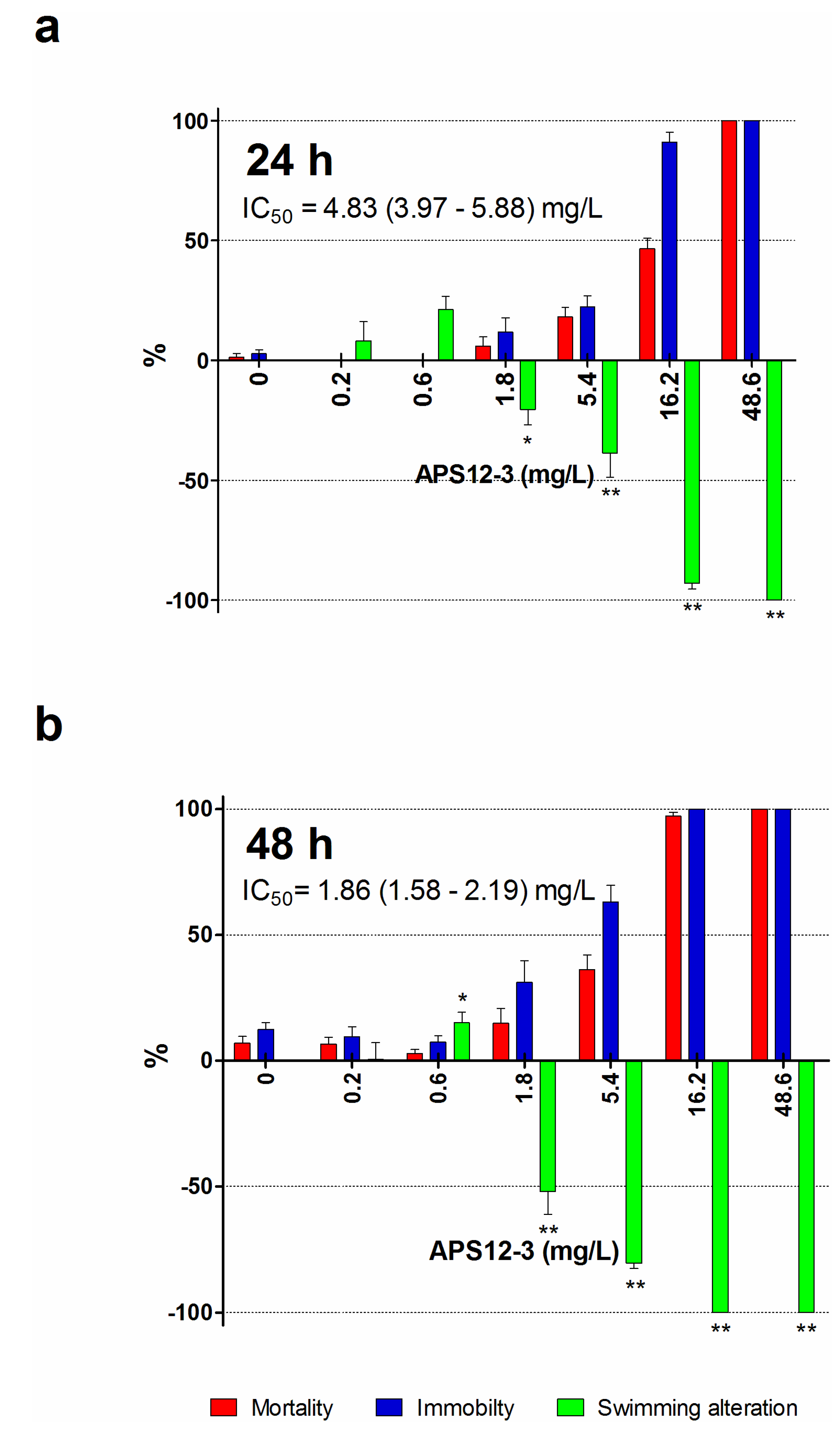
| APS12-3 2 | 24 | 0.89 (0.48–1.65) | 61.13 (51.65–72.36) | 0.146 | 10.0 |
| 48 | 4.03 (3.49–4.65) | 24.24 (20.09–29.24) | 0.661 | 4.0 | |
| 72 | 4.76 (4.44–5.11) | 17.97 (14.88–21.70) | 0.781 | 2.9 |

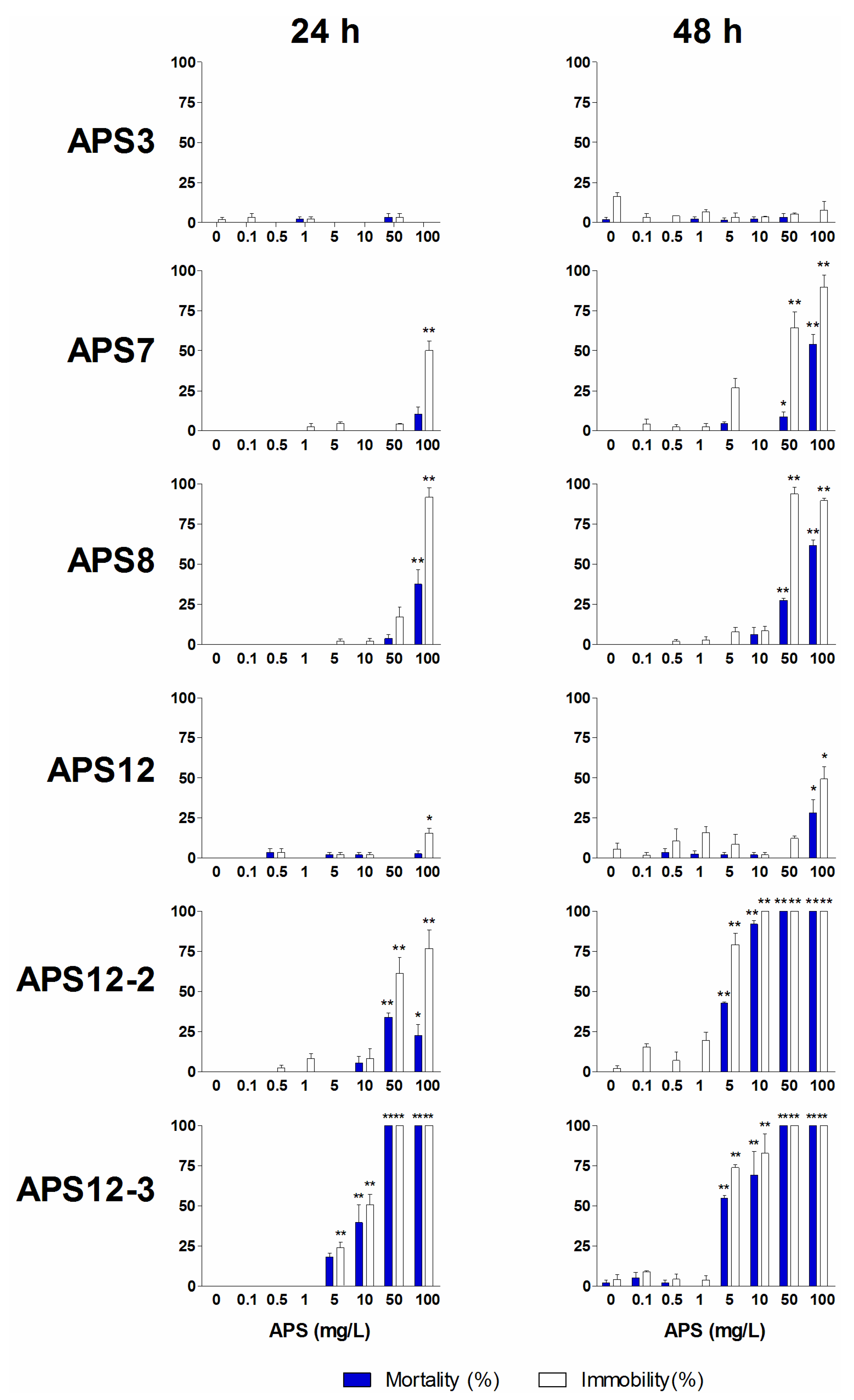
2.2. Naupliar Mortality, Immobility and Swimming Speed Alteration Assays
| Compound | Treatment (h) | EC50 (mg/L) | LC50 (mg/L) | EC50 (μM) | LC50 (μM) |
|---|---|---|---|---|---|
| Poly-APS 1 | 24 | >10 | 30.01 (21.71–41.49) | >1.81 | 5.43 |
| Zn pyrithione 1 | 24 | 0.23 (0.16–0.33) | 0.19 (0.13–0.30) | 0.725 | 0.6 |
| Cu pyrithione 1 | 24 | 0.03 (0.03–0.04) | <0.01 | 0.095 | <0.032 |
| APS3 | 24 | >100 | >100 | ||
| 48 | >100 | >100 | |||
| APS7 | 24 | >100 | >100 | ||
| 48 | 30.64 (25.43–36.9) | 94.02 (81.73–108.17) | 13.1 | 40.3 | |
| APS8 | 24 | >100 | >100 | ||
| 48 | >100 | 79.37 (68.29–92.25) | 6.7 | ||
| APS12 | 24 | >100 | >100 | ||
| 48 | >100 | >100 | |||
| APS12-2 | 24 | 36.92 (29.33–46.46) | >100 | 2.5 | |
| 48 | 2.28 (1.95–2.67) | 4.80 (4.21–5.46) | 0.15 | 0.32 | |
| APS12-3 | 24 | 9.43 (8.10–10.97) | 11.60 (10.06–13.38) | 1.5 | 1.9 |
| 48 | 3.61 (3.14–4.16) | 5.44 (4.64–6.37) | 0.59 | 0.89 |
| Compounds | Treatment (h) | EC50 (mg/L) | LC50(N) (mg/L) | LC50(C) (mg/L) | TRC | TRN |
|---|---|---|---|---|---|---|
| Poly-APS 1 | 24 | 0.27 | 30.01 | / | / | 111.15 |
| Zn pyrithione 1 | 24 | 0.02 | 0.19 | / | / | 9.50 |
| Cu pyrithione 1 | 24 | <0.01 | <0.01 | / | / | / |
| APS3 | 24 | 5.72 | >100 | >100 | nc | nc |
| 48 | nc | >100 | >100 | nc | nc | |
| 72 | nc | / | >100 | nc | / | |
| APS7 | 24 | 10.50 | >100 | >100 | nc | nc |
| 48 | 25.86 | 94.02 | >100 | nc | 3.64 | |
| 72 | 29.38 | / | >100 | nc | / | |
| APS8 | 24 | 0.32 | >100 | >100 | nc | |
| 48 | 0.50 | 79.37 | >100 | nc | 158.74 | |
| 72 | 2.33 | / | >100 | nc | / | |
| APS12 | 24 | nc | >100 | >100 | nc | nc |
| 48 | nc | >100 | >100 | nc | nc | |
| 72 | 49.82 | / | >100 | nc | / | |
| APS12-2 | 24 | 8.78 | >100 | >100 | nc | 11.39 |
| 48 | 9.38 | 4.8 | >100 | nc | 0.51 | |
| 72 | 11.13 | / | >100 | nc | / | |
| APS12-3 | 24 | 0.89 | 11.6 | 61.13 | 68.68 | 13.3 |
| 48 | 4.03 | 5.44 | 24.24 | 6.01 | 1.35 | |
| 72 | 4.76 | / | 17.97 | 3.77 | / |
3. Experimental Section
3.1. Chemicals
3.2. Rearing of Amphibalanus amphitrite Larvae
3.3. Settlement Assay
3.4. Toxicity Assay
3.5. Naupliar Swimming Speed Test
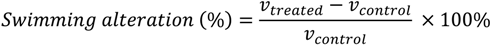
3.6. Statistical Analyses
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kirschner, C.M.; Brennan, A.B. Bio-Inspired Antifouling Strategies. Annu. Rev. Mater. Res. 2012, 42, 211–229. [Google Scholar] [CrossRef]
- Briand, J.-F. Marine antifouling laboratory bioassays: An overview of their diversity. Biofouling 2009, 25, 297–311. [Google Scholar]
- Jain, A.; Bhosle, N.B. Biochemical composition of the marine conditioning film: Implications for bacterial adhesion. Biofouling 2009, 25, 13–19. [Google Scholar] [CrossRef]
- Dobretsov, S. Marine Biofilms. In Biofouling; Dürr, S., Thomason, J.C., Eds.; Wiley-Blackwell: Oxford, UK, 2009; pp. 123–136. [Google Scholar]
- Railkin, A.I.; Ganf, T.A.; Manylov, O.G. Biofouling as a Process. In Marine Biofouling: Colonization Processes and Defenses; CRC Press: Boca Raton, FL, USA, 2003; pp. 25–39. [Google Scholar]
- Joint, I.; Tait, K.; Callow, M.E.; Callow, J.A.; Milton, D.; Williams, P.; Cámara, M. Cell-to-cell communication across the prokaryote-eukaryote boundary. Science 2002, 298, 1207. [Google Scholar] [CrossRef]
- Hadfield, M.G.; Paul, V.J. Natural chemical cues for settlement and metamorphosis of marine-invertebrate larvae. In Marine Chemical Ecology; McClintock, J.B., Baker, B.J., Eds.; CRC Press: Boca Raton, FL, USA, 2001; pp. 431–461. [Google Scholar]
- Hadfield, M.G. Biofilms and marine invertebrate larvae: What bacteria produce that larvae use to choose settlement sites. Ann. Rev. Mar. Sci. 2011, 3, 453–470. [Google Scholar] [CrossRef]
- Schultz, M.P. Effects of coating roughness and biofouling on ship resistance and powering. Biofouling 2007, 23, 331–341. [Google Scholar] [CrossRef]
- Schultz, M.P.; Bendick, J.A.; Holm, E.R.; Hertel, W.M. Economic impact of biofouling on a naval surface ship. Biofouling 2011, 27, 87–98. [Google Scholar] [CrossRef]
- Elimelech, M.; Phillip, W.A. The future of seawater desalination: Energy, technology, and the environment. Science 2011, 333, 712–717. [Google Scholar] [CrossRef]
- Page, H.M.; Dugan, J.E.; Piltz, F. Fouling and antifouling in oil and other offshore industries. In Biofouling; Dürr, S., Thomason, J.C., Eds.; Wiley-Blackwell: Oxford, UK, 2009; pp. 252–266. [Google Scholar]
- Fitridge, I.; Dempster, T.; Guenther, J.; de Nys, R. The impact and control of biofouling in marine aquaculture: A review. Biofouling 2012, 28, 649–669. [Google Scholar] [CrossRef]
- Dafforn, K.A.; Lewis, J.A.; Johnston, E.L. Antifouling strategies: History and regulation, ecological impacts and mitigation. Mar. Pollut. Bull. 2011, 62, 453–465. [Google Scholar] [CrossRef]
- Rosenhahn, A.; Schilp, S.; Kreuzer, H.J.; Grunze, M. The role of “inert” surface chemistry in marine biofouling prevention. Phys. Chem. Chem. Phys. 2010, 12, 4275–4286. [Google Scholar] [CrossRef]
- Sonak, S.; Pangam, P.; Giriyan, A.; Hawaldar, K. Implications of the ban on organotins for protection of global coastal and marine ecology. J. Environ. Manag. 2009, 90, S96–S108. [Google Scholar] [CrossRef]
- Ralston, E.; Swain, G. Bioinspiration—The solution for biofouling control? Bioinspir. Biomim. 2009, 4, 015007. [Google Scholar] [CrossRef]
- Salta, M.; Wharton, J.A.; Stoodley, P.; Dennington, S.P.; Goodes, L.R.; Werwinski, S.; Mart, U.; Wood, R.J.K.; Stokes, K.R. Designing biomimetic antifouling surfaces. Philos. Trans. A Math. Phys. Eng. Sci. 2010, 368, 4729–4754. [Google Scholar] [CrossRef]
- Scardino, A.J.; de Nys, R. Mini review: Biomimetic models and bioinspired surfaces for fouling control. Biofouling 2011, 27, 73–86. [Google Scholar] [CrossRef]
- Banerjee, I.; Pangule, R.C.; Kane, R.S. Antifouling coatings: Recent developments in the design of surfaces that prevent fouling by proteins, bacteria, and marine organisms. Adv. Mater. 2011, 23, 690–718. [Google Scholar] [CrossRef]
- Rittschof, D. Natural product antifoulants: One perspective on the challenges related to coatings development. Biofouling 2000, 15, 119–127. [Google Scholar] [CrossRef]
- Fusetani, N. Biofouling and antifouling. Nat. Prod. Rep. 2004, 21, 94–104. [Google Scholar] [CrossRef]
- Turk, T.; Frangež, R.; Sepčić, K. Mechanisms of toxicity of 3-alkylpyridinium polymers from marine sponge Reneira sarai. Mar. Drugs 2007, 5, 157–167. [Google Scholar] [CrossRef]
- Raveendran, T.V.; Limna Mol, V.P. Natural product antifoulants. Curr. Sci. 2009, 97, 508–520. [Google Scholar]
- Qian, P.-Y.; Xu, Y.; Fusetani, N. Natural products as antifouling compounds: Recent progress and future perspectives. Biofouling 2010, 26, 223–234. [Google Scholar] [CrossRef]
- Fusetani, N. Antifouling marine natural products. Nat. Prod. Rep. 2011, 28, 400–410. [Google Scholar] [CrossRef]
- Turk, T.; Sepčić, K.; Mancini, I.; Guella, G. 3-Akylpyridinium and 3-alkylpyridine compounds from marine sponges, their synthesis, biological activities and potential use. In Studies in Natural Products Chemistry; Elsevier: Amsterdam, The Netherlands, 2008; Volume 35, pp. 355–397. [Google Scholar]
- Sepčić, K.; Turk, T. 3-Alkylpyridinium compounds as potential non-toxic antifouling agents. In Antifouling Compounds SE-4; Fusetani, N., Clare, A., Eds.; Springer: Berlin/Heidelberg, Germany, 2006; Volume 42, pp. 105–124. [Google Scholar]
- Sepčić, K.; Guella, G.; Mancini, I.; Pietra, F.; Serra, M.D.; Menestrina, G.; Tubbs, K.; Maček, P.; Turk, T. Characterization of anticholinesterase-active 3-alkylpyridinium polymers from the marine sponge Reniera sarai in aqueous solutions. J. Nat. Prod. 1997, 60, 991–996. [Google Scholar] [CrossRef]
- Grandič, M.; Sepčić, K.; Turk, T.; Juntes, P.; Frangež, R. In vivo toxic and lethal cardiovascular effects of a synthetic polymeric 1,3-dodecylpyridinium salt in rodents. Toxicol. Appl. Pharmacol. 2011, 255, 86–93. [Google Scholar] [CrossRef]
- Malovrh, P.; Sepčić, K.; Turk, T.; Maček, P. Characterization of hemolytic activity of 3-alkylpyridinium polymers from the marine sponge Reniera sarai. Comp. Biochem. Physiol. Part C Pharmacol. Toxicol. Endocrinol. 1999, 124, 221–226. [Google Scholar] [CrossRef]
- Faimali, M.; Sepčić, K.; Turk, T.; Geraci, S. Non-toxic antifouling activity of polymeric 3-alkylpyridinium salts from the Mediterranean sponge Reniera sarai (Pulitzer-Finali). Biofouling 2003, 19, 47–56. [Google Scholar] [CrossRef]
- Garaventa, F.; Faimali, M.; Sepčić, K.; Geraci, S. Laboratory analysis of antimicrofouling activity of Poly-APS extracted from Reniera sarai (Porifera: Demospongiae). Biol. Mar. Mediterr. 2003, 10, 565–567. [Google Scholar]
- Qian, P.-Y.; Chen, L.; Xu, Y. Mini-review: Molecular mechanisms of antifouling compounds. Biofouling 2013, 29, 381–400. [Google Scholar] [CrossRef]
- Mancini, I.; Defant, A.; Guella, G. Recent synthesis of marine natural products with antibacterial activities. Anti-Infect. Agents Med. Chem. 2007, 6, 17–48. [Google Scholar] [CrossRef]
- Mancini, I.; Sicurelli, A.; Guella, G.; Turk, T.; Maček, P.; Sepčić, K. Synthesis and bioactivity of linear oligomers related to polymeric alkylpyridinium metabolites from the Mediterranean sponge Reniera sarai. Org. Biomol. Chem. 2004, 2, 1368–1375. [Google Scholar] [CrossRef]
- Faimali, M.; Garaventa, F.; Mancini, I.; Sicurelli, A.; Guella, G.; Piazza, V.; Greco, G. Antisettlement activity of synthetic analogues of polymeric 3-alkylpyridinium salts isolated from the sponge Reniera sarai. Biofouling 2005, 21, 49–57. [Google Scholar] [CrossRef]
- Chelossi, E.; Mancini, I.; Sepčić, K.; Turk, T.; Faimali, M. Comparative antibacterial activity of polymeric 3-alkylpyridinium salts isolated from the Mediterranean sponge Reniera sarai and their synthetic analogues. Biomol. Eng. 2006, 23, 317–323. [Google Scholar] [CrossRef]
- Houssen, W.E.; Lu, Z.; Edrada-Ebel, R.; Chatzi, C.; Tucker, S.J.; Sepčić, K.; Turk, T.; Zovko, A.; Shen, S.; Mancini, I.; et al. Chemical synthesis and biological activities of 3-alkyl pyridinium polymeric analogues of marine toxins. J. Chem. Biol. 2010, 3, 113–125. [Google Scholar] [CrossRef]
- Zovko, A.; Vaukner Gabrič, M.; Sepčić, K.; Pohleven, F.; Jaklič, D.; Gunde-Cimerman, N.; Lu, Z.; Edrada-Ebel, R.; Houssen, W.E.; Mancini, I.; et al. Antifungal and antibacterial activity of 3-alkylpyridinium polymeric analogs of marine toxins. Int. Biodeterior. Biodegrad. 2012, 68, 71–77. [Google Scholar] [CrossRef]
- Grandič, M.; Aráoz, R.; Molgó, J.; Turk, T.; Sepčić, K.; Benoit, E.; Frangež, R. Toxicity of the synthetic polymeric 3-alkylpyridinium salt (APS3) is due to specific block of nicotinic acetylcholine receptors. Toxicology 2013, 303, 25–33. [Google Scholar] [CrossRef]
- Eleršek, T.; Kosi, G.; Turk, T.; Pohleven, F.; Sepčić, K. Influence of polymeric 3-alkylpyridinium salts from the marine sponge Reniera sarai on the growth of algae and wood decay fungi. Biofouling 2008, 24, 137–143. [Google Scholar] [CrossRef]
- Holm, E.R. Barnacles and biofouling. Integr. Comp. Biol. 2012, 52, 348–355. [Google Scholar] [CrossRef]
- Calabrese, E.J. Biphasic dose responses in biology, toxicology and medicine: Accounting for their generalizability and quantitative features. Environ. Pollut. 2013, 182, 452–460. [Google Scholar] [CrossRef]
- Calabrese, E.J.; Mattson, M.P. Hormesis provides a generalized quantitative estimate of biological plasticity. J. Cell Commun. Signal. 2011, 5, 25–38. [Google Scholar] [CrossRef]
- Calabrese, E.J. Hormetic mechanisms. Crit. Rev. Toxicol. 2013, 43, 580–606. [Google Scholar] [CrossRef]
- Wiegant, F.A.C.; de Poot, S.A.H.; Boers-Trilles, V.E.; Schreij, A.M.A. Hormesis and cellular quality control: A possible explanation for the molecular mechanisms that underlie the benefits of mild stress. Dose-Response 2012, 11, 413–430. [Google Scholar]
- Rittschof, D.; Clare, A.S.; Gerhart, D.J.; Mary, S.A.; Bonaventura, J. Barnacle in vitro assays for biologically active substances: Toxicity and settlement inhibition assays using mass cultured Balanus amphitrite amphitrite Darwin. Biofouling 1992, 6, 115–122. [Google Scholar] [CrossRef]
- Piazza, V.; Roussis, V.; Garaventa, F.; Greco, G.; Smyrniotopoulos, V.; Vagias, C.; Faimali, M. Terpenes from the red alga Sphaerococcus coronopifolius inhibit the settlement of barnacles. Mar. Biotechnol. 2011, 13, 764–772. [Google Scholar] [CrossRef]
- Faimali, M.; Falugi, C.; Gallus, L.; Piazza, V.; Tagliafierro, G. Involvement of acetyl choline in settlement of Balanus amphitrite. Biofouling 2003, S19, 213–220. [Google Scholar]
- Faimali, M.; Garaventa, F.; Piazza, V.; Greco, G.; Corrà, C.; Magillo, F.; Pittore, M.; Giacco, E.; Gallus, L.; Falugi, C.; et al. Swimming speed alteration of larvae of Balanus amphitrite as a behavioural end-point for laboratory toxicological bioassays. Mar. Biol. 2006, 149, 87–96. [Google Scholar] [CrossRef]
- Garaventa, F.; Gambardella, C.; di Fino, A.; Pittore, M.; Faimali, M. Swimming speed alteration of Artemia sp. and Brachionus plicatilis as a sub-lethal behavioural end-point for ecotoxicological surveys. Ecotoxicology 2010, 19, 512–519. [Google Scholar] [CrossRef]
- Hamilton, M.A.; Russo, R.C.; Thurston, R.V. Trimmed Spearman-Karber method for estimating median lethal concentrations in toxicity bioassays. Environ. Sci. Technol. 1977, 11, 714–719. [Google Scholar] [CrossRef]
- De Muth, J.E. Basic Statistics and Pharmaceutical Statistical Applications, 2nd ed; Chapman and Hall/CRC: Boca Raton, FL, USA, 2006; p. 744. [Google Scholar]
- Kleszczyńska, H.; Bielecki, K.; Sarapuk, J.; Bonarska-Kujawa, D.; Pruchnik, H.; Trela, Z.; Łuczyński, J. Biological activity of new N-oxides of tertiary amines. Z. Naturforsch. C J. Biosci. 2009, 61, 715–720. [Google Scholar]
- Zarif, L.; Riess, J.G.; Pucci, B.; Pavia, A.A. Biocompatibility of alkyl and perfluoroalkyl telomeric surfactants derived from THAM. Biomater. Artif. Cells Immobil. Biotechnol. 1993, 21, 597–608. [Google Scholar]
- Kuroda, K.; DeGrado, W.F. Amphiphilic polymethacrylate derivatives as antimicrobial agents. J. Am. Chem. Soc. 2005, 127, 4128–4129. [Google Scholar] [CrossRef]
- Sarapuk, J.; Kleszczyńska, H.; Pernak, J.; Kalewska, J.; Rózycka-Roszak, B. Influence of counterions on the interaction of pyridinium salts with model membranes. Z. Naturforsch. C 1999, 54, 952–955. [Google Scholar]
- Kleszczynska, H.; Sarapuk, J.; Rozycka-Roszak, B. The role of counterions in the interaction of some cationic surfactants with model membranes. Pol. J. Environ. Stud. 1998, 7, 327–329. [Google Scholar]
- Zovko, A.; Viktorsson, K.; Lewensohn, R.; Kološa, K.; Filipič, M.; Xing, H.; Kem, W.R.; Paleari, L.; Turk, T. APS8, a polymeric alkylpyridinium salt blocks α7 nAChR and induces apoptosis in non-small cell lung carcinoma. Mar. Drugs 2013, 11, 2574–2594. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Piazza, V.; Dragić, I.; Sepčić, K.; Faimali, M.; Garaventa, F.; Turk, T.; Berne, S. Antifouling Activity of Synthetic Alkylpyridinium Polymers Using the Barnacle Model. Mar. Drugs 2014, 12, 1959-1976. https://doi.org/10.3390/md12041959
Piazza V, Dragić I, Sepčić K, Faimali M, Garaventa F, Turk T, Berne S. Antifouling Activity of Synthetic Alkylpyridinium Polymers Using the Barnacle Model. Marine Drugs. 2014; 12(4):1959-1976. https://doi.org/10.3390/md12041959
Chicago/Turabian StylePiazza, Veronica, Ivanka Dragić, Kristina Sepčić, Marco Faimali, Francesca Garaventa, Tom Turk, and Sabina Berne. 2014. "Antifouling Activity of Synthetic Alkylpyridinium Polymers Using the Barnacle Model" Marine Drugs 12, no. 4: 1959-1976. https://doi.org/10.3390/md12041959
APA StylePiazza, V., Dragić, I., Sepčić, K., Faimali, M., Garaventa, F., Turk, T., & Berne, S. (2014). Antifouling Activity of Synthetic Alkylpyridinium Polymers Using the Barnacle Model. Marine Drugs, 12(4), 1959-1976. https://doi.org/10.3390/md12041959