Optimization of Medium Using Response Surface Methodology for Lipid Production by Scenedesmus sp.
Abstract
:1. Introduction
2. Results and Discussion
2.1. Evaluating the Significant Nutrient Factors Using Plackett–Burman Design
| Run | X1 | X2 | X3 | X4 | X5 | X6 | X7 | X8 | X9 | Lipid production (mg·L−1) |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 4 | 200 | 25 | 1000 | 40 | 100 | 0.5 | 0.5 | 0.5 | 230.38 |
| 2 | 1 | 200 | 100 | 250 | 40 | 100 | 2 | 0.5 | 0.5 | 174.27 |
| 3 | 4 | 50 | 100 | 1000 | 10 | 100 | 2 | 2 | 0.5 | 188.56 |
| 4 | 1 | 200 | 25 | 1000 | 40 | 20 | 2 | 2 | 2 | 208.85 |
| 5 | 1 | 50 | 100 | 250 | 40 | 100 | 0.5 | 2 | 2 | 152.85 |
| 6 | 1 | 50 | 25 | 1000 | 10 | 100 | 2 | 0.5 | 2 | 199.58 |
| 7 | 4 | 50 | 25 | 250 | 40 | 20 | 2 | 2 | 0.5 | 215.37 |
| 8 | 4 | 200 | 25 | 250 | 10 | 100 | 0.5 | 2 | 2 | 214.41 |
| 9 | 4 | 200 | 100 | 250 | 10 | 20 | 2 | 0.5 | 2 | 173.23 |
| 10 | 1 | 200 | 100 | 1000 | 10 | 20 | 0.5 | 2 | 0.5 | 166.16 |
| 11 | 4 | 50 | 100 | 1000 | 40 | 20 | 0.5 | 0.5 | 2 | 188.82 |
| 12 | 1 | 50 | 25 | 250 | 10 | 20 | 0.5 | 0.5 | 0.5 | 196.58 |
| Factor | Level | Effect | Sum of Squares | Contribution % | Coefficient Estimate | t-value | p-value | Effect |
|---|---|---|---|---|---|---|---|---|
| −1 +1 | ||||||||
| NaHCO3 | 1 | 4 | 18.7476 | 1,054.3100 | 18.2703 | 9.37 | 0.0034 | 0.0015 a |
| KCl | 50 | 200 | 4.2567 | 54.3576 | 0.9420 | 1.9608 | 0.1537 | — |
| NaH2PO4·2H2O | 25 | 100 | −36.8800 | 4080.4000 | 70.7096 | −18.6075 | 0.0006 | <0.0001 a |
| NaNO3 | 250 | 1000 | 9.2733 | 257.9840 | 4.4706 | 4.4692 | 0.0582 | 0.0477 a |
| CaCl2 | 10 | 40 | 5.3367 | 85.4400 | 1.4806 | 2.8358 | 0.1306 | — |
| MgSO4·7H2O | 20 | 100 | 1.8400 | 10.1568 | 0.1760 | 1.0875 | 0.3203 | — |
| EDTA-Fe3+ | 0.5 | 2 | 1.7767 | 9.4696 | 0.1641 | 1.0558 | 0.2877 | — |
| A5 | 0.5 | 2 | −2.7767 | 23.1296 | 0.4008 | −1.5558 | 0.1907 | — |
| Soil extract | 0.5 | 2 | −5.5967 | 93.9680 | 1.6284 | −2.6308 | 0.1046 | — |
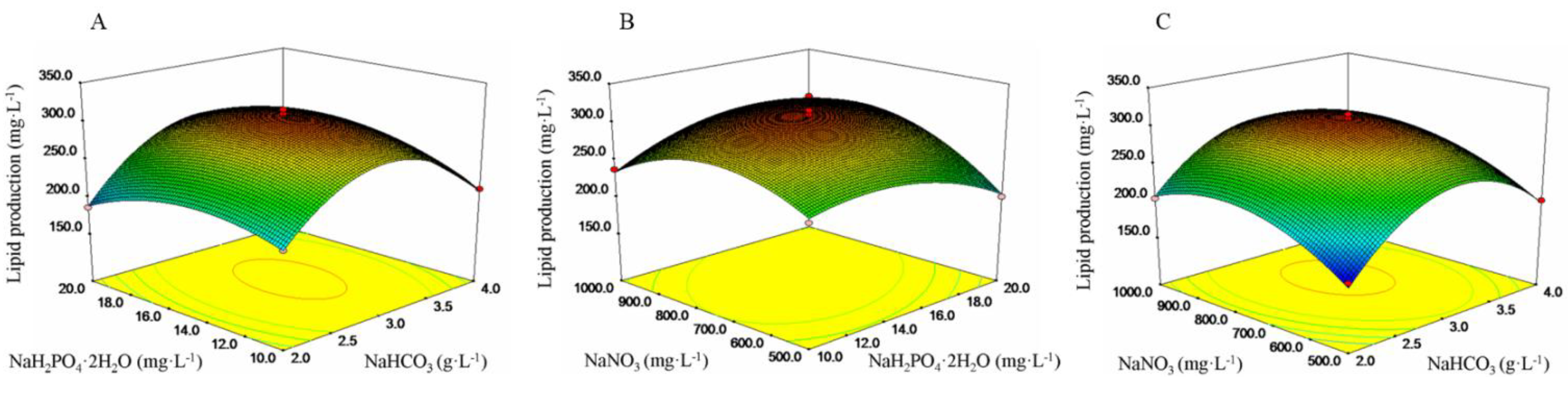
2.2. Identifying the Best Culture Conditions for Lipid Production Using Box–Behnken Design
| Run | NaHCO3 (g L−1) | NaH2PO4·2H2O (mg L−1) | NaNO3 (mg L−1) | Lipid production (mg L−1) | |
|---|---|---|---|---|---|
| Experimental | Predicted | ||||
| 1 | 2 | 10 | 750 | 207.53 | 207.77 |
| 2 | 4 | 10 | 750 | 211.29 | 209.08 |
| 3 | 2 | 20 | 750 | 186.23 | 188.44 |
| 4 | 4 | 20 | 750 | 229.44 | 229.20 |
| 5 | 2 | 15 | 500 | 171.96 | 166.17 |
| 6 | 4 | 15 | 500 | 201.35 | 198.01 |
| 7 | 2 | 15 | 1000 | 203.78 | 207.12 |
| 8 | 4 | 15 | 1000 | 211.58 | 217.37 |
| 9 | 3 | 10 | 500 | 240.55 | 246.1 |
| 10 | 3 | 20 | 500 | 200.97 | 204.55 |
| 11 | 3 | 10 | 1000 | 237.89 | 234.31 |
| 12 | 3 | 20 | 1000 | 282.19 | 276.64 |
| 13 | 3 | 15 | 750 | 304.86 | 307.51 |
| 14 | 3 | 15 | 750 | 305.22 | 307.51 |
| 15 | 3 | 15 | 750 | 310.39 | 307.51 |
| 16 | 3 | 15 | 750 | 301.35 | 307.51 |
| 17 | 3 | 15 | 750 | 315.74 | 307.51 |
| Factor | Sum of squares | Degree of Freedom | Mean square | Coefficient Estimate | F-value | p-value |
|---|---|---|---|---|---|---|
| Model | 38,929.9101 | 9 | 4,325.5456 | 307.5120 | 96.8736 | <0.0001 |
| NaHCO3 | 885.3632 | 1 | 885.3632 | 10.5200 | 19.8283 | 0.0030 |
| NaH2PO4·2H2O | 0.3081 | 1 | 0.3081 | 0.1963 | 0.0069 | 0.9361 |
| NaNO3 | 1,818.3465 | 1 | 1,818.3465 | 15.0763 | 40.7231 | 0.0004 |
| NaHCO3*NaH2PO4·2H2O | 389.0756 | 1 | 389.0756 | 9.8625 | 8.7136 | 0.0213 |
| NaHCO3*NaNO3 | 116.5320 | 1 | 116.5320 | −5.3975 | 2.6098 | 0.1502 |
| NaH2PO4·2H2O*NaNO3 | 1,758.9636 | 1 | 1,758.9396 | 20.9700 | 39.3932 | 0.0004 |
| NaHCO32 | 21,261.7504 | 1 | 21,261.7504 | −71.0610 | 476.1715 | <0.0001 |
| NaH2PO4·2H2O 2 | 3,260.7386 | 1 | 3,260.7386 | −27.8285 | 73.0265 | <0.0001 |
| NaNO32 | 6,497.6563 | 1 | 6,497.6563 | −39.2835 | 145.5195 | <0.0001 |
| Residual | 312.5602 | 7 | 44.6515 | |||
| Lack of fit | 186.3207 | 3 | 62.1069 | 1.9679 | 0.2609 | |
| Pure error | 126.2395 | 4 | 31.5599 | |||
| Corr. total | 39,242.4703 | 16 | ||||
| Model | 38,929.9101 | 9 | 4,325.5456 | 307.5120 | 96.8736 | <0.0001 |
2.3. Lipid Analysis and Fatty Acid Composition
| Fatty acid (%) | C16:0 | C16:1 | C16:2 | C18:0 | C18:1 | C18:2 | C18:3 |
|---|---|---|---|---|---|---|---|
| a | 29.43 ± 1.75 | 1.99 ± 0.69 | 2.42 ± 0.24 | 7.33 ± 0.13 | 30.04 ± 1.02 | 13.60 ± 0.45 | 12.76 ± 0.26 |
| b | 30.77 ± 0.76 | 1.96 ± 0.54 | 1.37 ± 0.02 | 3.41 ± 0.09 | 36.27 ± 0.78 | 11.55 ± 0.12 | 12.52 ± 0.39 |
| Lipid class | Composition (wt% of total lipids) | |
|---|---|---|
| a | b | |
| Neutral lipid | 81.29 ± 0.65 | 82.32 ± 0.89 |
| Glycolipid | 12.56 ± 0.53 | 10.93 ± 0.47 |
| Phospholipid | 6.14 ± 0.35 | 6.74 ± 0.76 |
3. Experimental Section
3.1. Strain and Culture Conditions
3.2. RSM Experimental Design
3.2.1. Plackett–Burman Design
3.2.2. Box–Behnken Design
3.3. Biomass Determination and Lipid Extraction
3.4. Lipid Analysis and Fatty Acid Composition
4. Conclusions
Supplementary Files
Acknowledgments
Conflicts of Interest
References
- Rodolfi, L.; Zittelli, G.C.; Bassi, N.; Padovan, G.; Biondi, N.; Bonini, G.; Tredici, M.R. Microalgae for oil: Strain selection, induction of lipid synthesis and outdoor mass cultivation in a low-cost photobioreactor. Biotechnol. Bioeng. 2009, 102, 100–112. [Google Scholar] [CrossRef]
- Mata, T.M.; Martins, A.A.; Caetano, N.S. Microalgae for biodiesel production and other applications: A review. Renew. Sustain. Energy Rev. 2010, 14, 217–232. [Google Scholar]
- Rawat, I.; Kumar, R.R.; Mutanda, T.; Bux, F. Biodiesel from microalgae: A critical evaluation from laboratory to large scale production. Appl. Energy 2013, 103, 444–467. [Google Scholar] [CrossRef]
- Chisti, Y. Biodiesel from microalgae. Biotechnol. Adv. 2007, 25, 294–306. [Google Scholar] [CrossRef]
- Helena, M.A.; Catarina, G.A.; Xavier, M.F. Advances and perspectives in using microalgae to produce biodiesel. Appl. Energy 2011, 88, 3401–3410. [Google Scholar]
- Huang, G.; Chen, F.; Wei, D.; Zhang, X.; Chen, G. Biodiesel production by microalgal biotechnology. Appl. Energy 2010, 87, 38–46. [Google Scholar] [CrossRef]
- Hu, Q.; Sommerfeld, M.; Jarvis, E.; Ghirardi, M.; Posewitz, M.; Seibert, M.; Darzins, A. Microalgal triacylglycerols as feedstocks for biofuel production: Perspectives and advances. Plant J. 2008, 54, 621–639. [Google Scholar] [CrossRef]
- Feng, D.A.; Chen, Z.A.; Xue, S.; Zhang, W. Increased lipid production of the marine oleaginous microalgae Isochrysis zhangjiangensis (Chrysophyta) by nitrogen supplement. Bioresour. Technol. 2011, 102, 6710–6716. [Google Scholar] [CrossRef]
- Roessler, P.G. Environmental control of glycerol lipid metabolism in microalgae: Commercial implications and future research directions. J. Phycol. 1990, 26, 3933–3939. [Google Scholar]
- Guschina, I.A.; Harwood, J.L. Lipids and lipid metabolism in eukaryotic algae. Prog. Lipid Res. 2006, 45, 160–186. [Google Scholar] [CrossRef]
- Sharma, K.K.; Schuhmann, H.; Schenk, P.M. High lipid induction in microalgae for biodiesel production. Energies 2012, 5, 1532–1553. [Google Scholar] [CrossRef]
- Khozin-Goldberg, I.; Cohen, Z. The effect of phosphate starvation on the lipid and fatty acid composition of the fresh water eustigmatophyte Monodus subterraneus. Phytochemistry 2006, 7, 696–701. [Google Scholar] [CrossRef]
- Li, Y.Q.; Horsman, M.; Wang, B.; Wu, N.; Lan, C.Q. Effects of nitrogen sources on cell growth and lipid accumulation of green alga Neochloris oleoabundans. Appl. Microbiol. Biotechnol. 2008, 81, 629–636. [Google Scholar] [CrossRef]
- Liu, Z.Y.; Wang, G.C.; Zhou, B.C. Effect of iron on growth and lipid accumulation in Chlorella vulgaris. Bioresour. Technol. 2008, 11, 4717–4722. [Google Scholar]
- Yeesang, C.; Cheirsilp, B. Effect of nitrogen, salt, and iron content in the growth medium and light intensity on lipid production by microalgae isolated from freshwater sources in Thailand. Bioresour. Technol. 2011, 3, 3034–3040. [Google Scholar] [CrossRef]
- White, D.A.; Pagarette, A.; Rooks, P.; Ali, S.T. The effect of sodium bicarbonate supplementation on growth and biochemical composition of marine microalgae cultures. J. Appl. Phycol. 2013, 25, 153–165. [Google Scholar] [CrossRef]
- Reitan, K.I.; Rainuzzo, J.R.; Olsen, Y. Effect of nutrient limitation on fatty acid and lipid content of marine microalgae. J. Phycol. 1994, 30, 972–979. [Google Scholar]
- Dean, A.P.; Sigee, D.C.; Estrada, B.; Pittman, J.K. Using FTIR spectroscopy for rapid determination of lipid accumulation in response to nitrogen limitation in freshwater microalgae. Bioresour. Technol. 2010, 101, 4499–4507. [Google Scholar] [CrossRef]
- Takagi, M.; Watanabe, K.; Yamaberi, K.; Yoshida, T. Limited feeding of potassium nitrate for intracellular lipid and triglyceride accumulation of Nannochloris sp. UTEX LB1999. Appl. Microbiol. Biotechnol. 2002, 54, 112–117. [Google Scholar]
- Griffiths, M.J.; Harrison, S.T.L. Lipid productivity as a key characteristic for choosing algal species for biodiesel production. J. Appl. Phycol. 2009, 21, 493–507. [Google Scholar] [CrossRef]
- Zhang, J.; Fu, D.; Xu, Y.; Liu, C. Optimization of parameters on photocatalytic degradation of chloramphenicol using TiO2 as photocatalyist by response surface methodology. J. Environ. Sci. 2012, 22, 1281–1289. [Google Scholar] [CrossRef]
- Qin, J.Z.; Song, F.F.; Qiu, Y.F.; Li, X.X.; Guan, X. Optimization of the medium composition of a biphasic production system for mycelial growth and spore production of Aschersonia placenta using response surface methodology. J. Invertebr. Pathol. 2013, 112, 108–115. [Google Scholar] [CrossRef]
- Aybastier, O.; Demir, C. Optimization of immobilization conditions of Thermomyces lanuginosus lipase on styrene-divinylbenzene copolymer using response surface methodology. J. Mol. Catal. B Enzym. 2010, 63, 170–178. [Google Scholar] [CrossRef]
- Pandian, M.; Sivapirakasam, S.P.; Udayakumar, M. Investigation on the effect of injection system parameters on performance and emission characteristics of a twin cylinder compression ignition direct injection engine fuelled with pongamia biodiesel–diesel blend using response surface methodology. Appl. Energy 2011, 88, 2663–2676. [Google Scholar] [CrossRef]
- Yücel, Y.S. Optimization of biocatalytic biodiesel production from pomace oil using response surface methodology. Fuel Process. Technol. 2012, 99, 97–102. [Google Scholar] [CrossRef]
- Cheng, K.C.; Ren, M.; Ogden, K.L. Statistical optimization of culture media for growth and lipid production of Chlorella protothecoides UTEX 250. Bioresour. Technol. 2013, 128, 44–48. [Google Scholar] [CrossRef]
- Karemore, A.; Pal, R.; Sen, R. Strategic enhancement of algal biomass and lipid in Chlorococcum infusionum as bioenergy feedstock. Algal Res. 2013, 2, 113–121. [Google Scholar] [CrossRef]
- Jiang, P.; Zhang, Y.; Shan, Z.X.; Zheng, Q.H. Optimizing the extraction yield of polyprenols from needles of Cunninghamia Ianceolata (Lamb.) hook using response surface methodology and its antioxidative activities. BioResources 2013, 1, 545–556. [Google Scholar]
- Mandal, S.; Mallick, N. Microalga Scenedesmus obliquus as a potential source for biodiesel production. Appl. Microbiol. Biotechnol. 2009, 84, 281–291. [Google Scholar] [CrossRef]
- Welter, C.; Schwenk, J.; Kanani, B.; Blargan, J.V.; Belovicha, J.M. Minimal Medium for Optimal Growth and Lipid Production of the Microalgae Scenedesmus dimorphus. Environ. Prog. Sustain. Energy 2013, 4, 937–945. [Google Scholar]
- Yeh, K.L.; Chang, J.S.; Chen, W.M. Effect of light supply and carbon source on cell growth and cellular composition of a newly isolated microalga Chlorella vulgaris ESP-31. Eng. Life Sci. 2010, 10, 201–208. [Google Scholar] [CrossRef]
- Gardner, R.D.; Cooksey, K.E.; Mus, F.; Macur, R.; Moll, K.; Eustance, E.; Carlson, R.P.; Gerlach, R.; Fields, M.W.; Peyton, B.M. Use of sodium bicarbonate to stimulate triacylglycerol accumulation in the chlorophyte Scenedesmus sp. and the diatom Phaeodactylum tricornutum. J. Appl. Phycol. 2012, 24, 1311–1320. [Google Scholar] [CrossRef]
- Demirbas, A.; Demirbas, M.F. Importance of algal oil as a source of biodiesel. Energy Convers Manag. 2011, 52, 163–170. [Google Scholar] [CrossRef]
- Chen, Z.; Gong, Y.M.; Fang, X.T.; Hu, H.H. Scenedesmus sp. NJ-1 isolated from Antarctica: A suitable renewable lipid source for biodiesel production. World J. Microbiol. Biotechnol. 2012, 28, 3219–3225. [Google Scholar] [CrossRef]
- Miao, X.; Wu, Q. Biodiesel production from heterotrophic microalgal oil. Bioresour. Technol. 2006, 6, 841–846. [Google Scholar] [CrossRef]
- Hugo, P.; Luisa, B.; Filipe, F.; Luisa, C.; Catarina, V.D.; Cristina, P.; Eva, R.; Aschwin, E.; Joao, V. Polyunsaturated fatty acids of marine macroalgae: Potential for nutritional and pharmaceutical applications. Mar. Drugs 2012, 10, 1920–1935. [Google Scholar] [CrossRef]
- Guckert, J.B.; Cooksey, K.E. Triglyceride accumulation and fatty acid profile changes in Chlorella (Chlorophyta) during high pH-induced cell cycle inhibition. J. Phycol. 1990, 26, 72–79. [Google Scholar]
- Plackett, R.L.; Burman, J.P. The design of optimum multifactorial experiments. Biometrika 1946, 33, 305–325. [Google Scholar] [CrossRef]
- Box, G.E.P.; Behnken, D.W. Some new three level designs for the study of quantitative variables. Technometrices 1960, 2, 455–475. [Google Scholar] [CrossRef]
- Khozin-Goldberg, I.; Shrestha, P.; Cohen, Z. Mobilization of arachidonyl moieties from triacylglycerols into chloroplastic lipids following recovery from nitrogen starvation of the microalga parietochloris incisa. BBA-Mol. Cell Biol. Lipids 2005, 1738, 63–71. [Google Scholar] [CrossRef]
- Christie, W.W. Lipid Analysis: Isolation, Separation, Identification and Structural Analysis of Lipids, 3rd ed.; The Oily Press: Bridgewater, UK, 2003; pp. 373–387. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yang, F.; Long, L.; Sun, X.; Wu, H.; Li, T.; Xiang, W. Optimization of Medium Using Response Surface Methodology for Lipid Production by Scenedesmus sp. Mar. Drugs 2014, 12, 1245-1257. https://doi.org/10.3390/md12031245
Yang F, Long L, Sun X, Wu H, Li T, Xiang W. Optimization of Medium Using Response Surface Methodology for Lipid Production by Scenedesmus sp. Marine Drugs. 2014; 12(3):1245-1257. https://doi.org/10.3390/md12031245
Chicago/Turabian StyleYang, Fangfang, Lijuan Long, Xiumei Sun, Hualian Wu, Tao Li, and Wenzhou Xiang. 2014. "Optimization of Medium Using Response Surface Methodology for Lipid Production by Scenedesmus sp." Marine Drugs 12, no. 3: 1245-1257. https://doi.org/10.3390/md12031245
APA StyleYang, F., Long, L., Sun, X., Wu, H., Li, T., & Xiang, W. (2014). Optimization of Medium Using Response Surface Methodology for Lipid Production by Scenedesmus sp. Marine Drugs, 12(3), 1245-1257. https://doi.org/10.3390/md12031245






