Invasive Lionfish (Pterois volitans): A Potential Human Health Threat for Ciguatera Fish Poisoning in Tropical Waters
Abstract
:1. Introduction

2. Results and Discussion
3. Experimental Section
3.1. Reagents and Standards
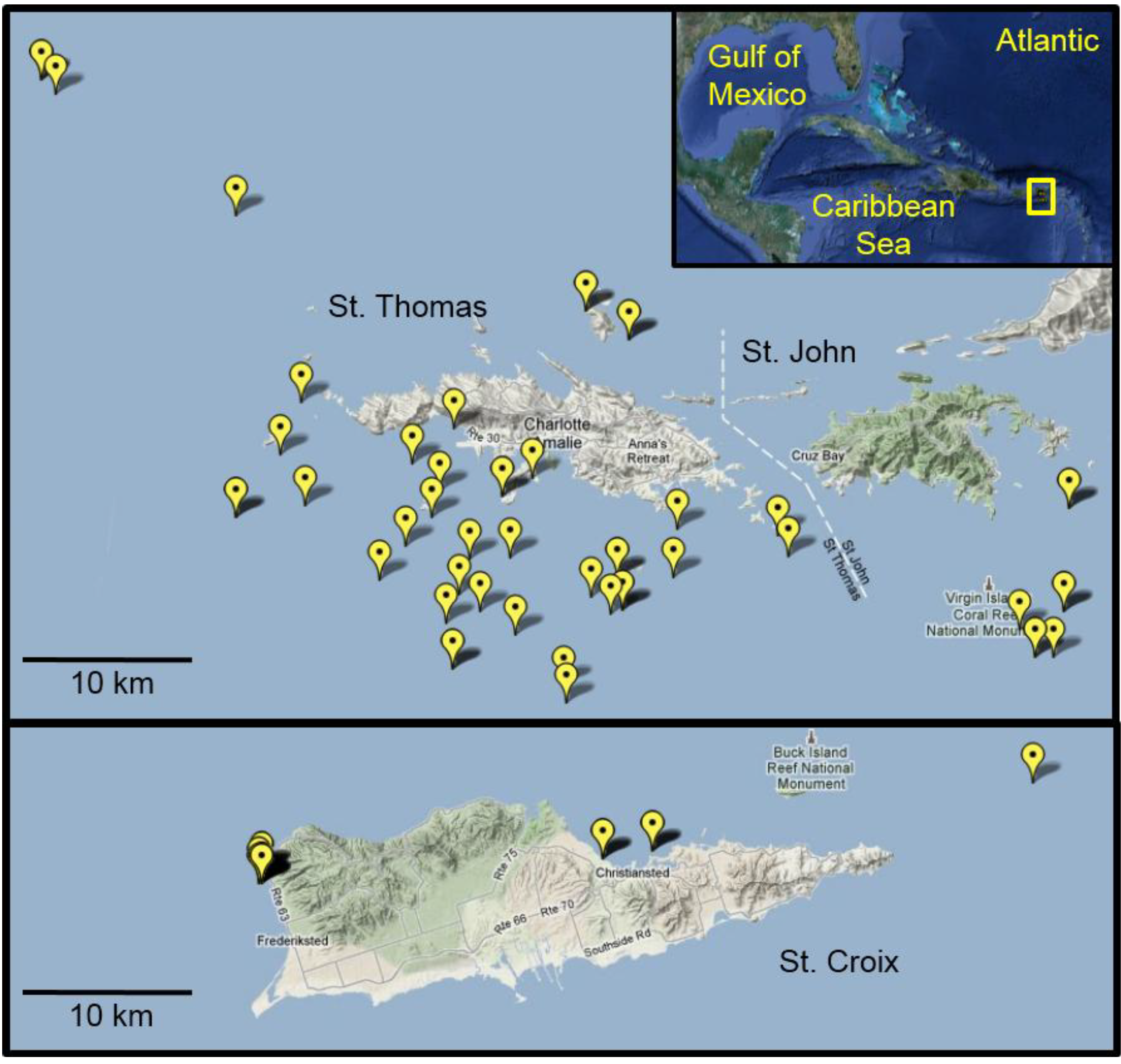
3.2. Lionfish Sampling
3.3. Sample Preparation and Toxin Extraction
3.4. In Vitro Neuroblastoma Cytotoxicity Assay
3.5. Confirmatory Liquid Chromatography-Mass Spectrometry:
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Schultz, E.T. Pterois volitans and Pterois miles: Two valid species. Copeia 1986, 1, 686–690. [Google Scholar] [CrossRef]
- U.S. Geological Survey: Non-indigenous Aquatic Species Database. Available online: http://nas.er.usgs.gov/ (accessed on 5 December 2012).
- Morris, J.A.; Akins, J.L. Feeding ecology of invasive lionfish (Pterois volitans) in the Bahamian archipelago. Environ. Biol. Fish. 2009, 86, 389–398. [Google Scholar]
- Albins, M.A.; Hixon, M.A. Invasive Indo-Pacific lionfish (Pterois volitans) reduce recruitment of Atlantic coral-reef fishes. Mar. Ecol. Prog. Ser. 2008, 367, 233–238. [Google Scholar] [CrossRef]
- Morris, J.A.; Whitfield, P.E. Biology, Ecology, Control and Management of the Invasive Indo-Pacific Lionfish: An Updated Integrated Assessment. In NOAA Technical Memorandum NOS NCCOS 99; National Oceanic and Atmospheric Administration: Washington, DC, USA, 2009. [Google Scholar]
- Whitfield, P.E.; Hare, J.A.; David, A.W.; Harter, S.L.; Muñoz, R.C.; Addison, C.M. Abundance estimates of the Indo-Pacific lionfish Pterois volitans/miles complex in the Western North Atlantic. Biol. Invasions 2007, 9, 53–64. [Google Scholar]
- Ragelis, E.P. Ciguatera seafood poisoning: An overview. In Seafood Toxins; Ragelis, E.P., Ed.; American Chemical Society: Washington, DC, USA, 1984; pp. 22–36. [Google Scholar]
- Fleming, L.E.; Baden, D.G.; Bean, J.A.; Weisman, R.; Blythe, D.G. Seafood toxin diseases: Issues in epidemiology and community outreach. In Harmful Algae; Reguera, B., Blanco, J., Fernandez, M.L., Wyatt, T., Eds.; Xunta de Galicia and Intergovernmental Oceanographic Commission of UNESCO: Galicia, Spain, 1998; pp. 245–248. [Google Scholar]
- Bagnis, R.; Legrand, A.M. Progress in Venom and Toxin Research. In Proceedings of the First Asia-Pacific Congress on Animal, Plant and Microbial Toxins, Singapore, 24–27 June 1987; Gopalakrishnakone, P., Tan, C.K., Eds.; National University of Singapore: Singapore, Singapore, 1987; pp. 372–384. [Google Scholar]
- Pearn, J. Neurology of ciguatera. J. Neurol. Neurosurg. Psychiatr. 2001, 70, 4–8. [Google Scholar] [CrossRef]
- Benoit, E.; Juzans, P.; Legrand, A.M.; Molgo, J. Nodal swelling produced by ciguatoxin-induced selective activation of sodium channels in myelinated nerve fibers. Neuroscience 1996, 71, 1121–1131. [Google Scholar] [CrossRef]
- Bidard, J.N.; Vijverberg, H.P.M.; Frelin, C.; Chungue, E.; Legrand, A.M.; Bagnis, R.; Lazdunski, M. Ciguatoxin is a novel type of Na+ channel toxin. J. Biol. Chem. 1984, 259, 8353–8357. [Google Scholar]
- Lombet, A.; Bidard, J.N.; Lazdunski, M. Ciguatoxins and brevetoxins share a common receptor site on the neuronal voltage-dependent Na+ channel. FEBS Lett. 1987, 219, 355–359. [Google Scholar] [CrossRef]
- Lewis, R.J.; Holmes, M.J. Origin and transfer of toxins involved in ciguatera. Comp. Biochem. Physiol. 1993, 106C, 615–628. [Google Scholar]
- Department of Health and Human Services. FDA Fish and Fishery Products Hazards and Control Guidance. Available online: http://www.fda.gov/ (accessed on 12 September 2012).
- Scheuer, P.J.; Takahashi, W.; Tsutsumi, J.; Yoshida, T. Ciguatoxin isolation and chemical nature. Science 1967, 155, 1267–1268. [Google Scholar] [CrossRef]
- Lewis, R.J.; Vernoux, J.P.; Brereton, I.M. Structure of Caribbean ciguatoxin isolated from Caranx latus. J. Am. Chem. Soc. 1998, 120, 5914–5920. [Google Scholar] [CrossRef]
- Robertson, A.; Loeffler, C.R.; Garcia, A.C.; Flores Quintana, H.A.; Liefer, J.D.; Olsen, D.A.; Smith, T.B. Prevalence and Distribution of Caribbean Ciguatoxins in Reef Fishes of the US Virgin Islands. 2013; to be submitted for publication. [Google Scholar]
- Fishelson, L. Experiments and observations on food consumption, growth and starvation in Dendrochirus brachypterus and Pterois volitans (Pteroinae, Scorpaenidae). Environ. Biol. Fish. 1997, 50, 391–403. [Google Scholar] [CrossRef]
- Morris, J.G.; Lewin, P.; Hargrett, N.T.; Smith, C.W. Ciguatera fish poisoning: Epidemiology of the disease on St. Thomas, U.S. Virgin Islands. Arch. Int. Med. 1982, 142, 1090–1092. [Google Scholar]
- Dickey, R.W. Ciguatera Toxins: Chemistry, Toxicity, and Detection. In Seafood and Freshwater Toxins: Pharmacology, Physiology, and Detection, 2nd ed.; Botana, L.M., Ed.; CRC Press, Taylor & Francis: Boca Raton, FL, USA, 2008; pp. 479–500. [Google Scholar]
- Manger, R.L.; Leja, L.S.; Lee, S.Y.; Hungerford, J.M.; Hokama, Y.; Dickey, R.W.; Granade, H.R.; Lewis, R.; Yasumoto, T.; Wekell, M.M. Detection of sodium channel effectors: Directed cytotoxicity assays of purified ciguatoxins, brevetoxins, saxitoxin, and seafood extracts. J. AOAC Int. 1995, 78, 521–527. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Robertson, A.; Garcia, A.C.; Quintana, H.A.F.; Smith, T.B.; II, B.F.C.; Reale-Munroe, K.; Gulli, J.A.; Olsen, D.A.; Hooe-Rollman, J.I.; Jester, E.L.E.; et al. Invasive Lionfish (Pterois volitans): A Potential Human Health Threat for Ciguatera Fish Poisoning in Tropical Waters. Mar. Drugs 2014, 12, 88-97. https://doi.org/10.3390/md12010088
Robertson A, Garcia AC, Quintana HAF, Smith TB, II BFC, Reale-Munroe K, Gulli JA, Olsen DA, Hooe-Rollman JI, Jester ELE, et al. Invasive Lionfish (Pterois volitans): A Potential Human Health Threat for Ciguatera Fish Poisoning in Tropical Waters. Marine Drugs. 2014; 12(1):88-97. https://doi.org/10.3390/md12010088
Chicago/Turabian StyleRobertson, Alison, Ana C. Garcia, Harold A. Flores Quintana, Tyler B. Smith, Bernard F. Castillo II, Kynoch Reale-Munroe, Joseph A. Gulli, David A. Olsen, Jennifer I. Hooe-Rollman, Edward L. E. Jester, and et al. 2014. "Invasive Lionfish (Pterois volitans): A Potential Human Health Threat for Ciguatera Fish Poisoning in Tropical Waters" Marine Drugs 12, no. 1: 88-97. https://doi.org/10.3390/md12010088
APA StyleRobertson, A., Garcia, A. C., Quintana, H. A. F., Smith, T. B., II, B. F. C., Reale-Munroe, K., Gulli, J. A., Olsen, D. A., Hooe-Rollman, J. I., Jester, E. L. E., Klimek, B. J., & Plakas, S. M. (2014). Invasive Lionfish (Pterois volitans): A Potential Human Health Threat for Ciguatera Fish Poisoning in Tropical Waters. Marine Drugs, 12(1), 88-97. https://doi.org/10.3390/md12010088




