Analysis of the Toxicity and Histopathology Induced by the Oral Administration of Pseudanabaena galeata and Geitlerinema splendidum (Cyanobacteria) Extracts to Mice
Abstract
:1. Introduction
2. Results
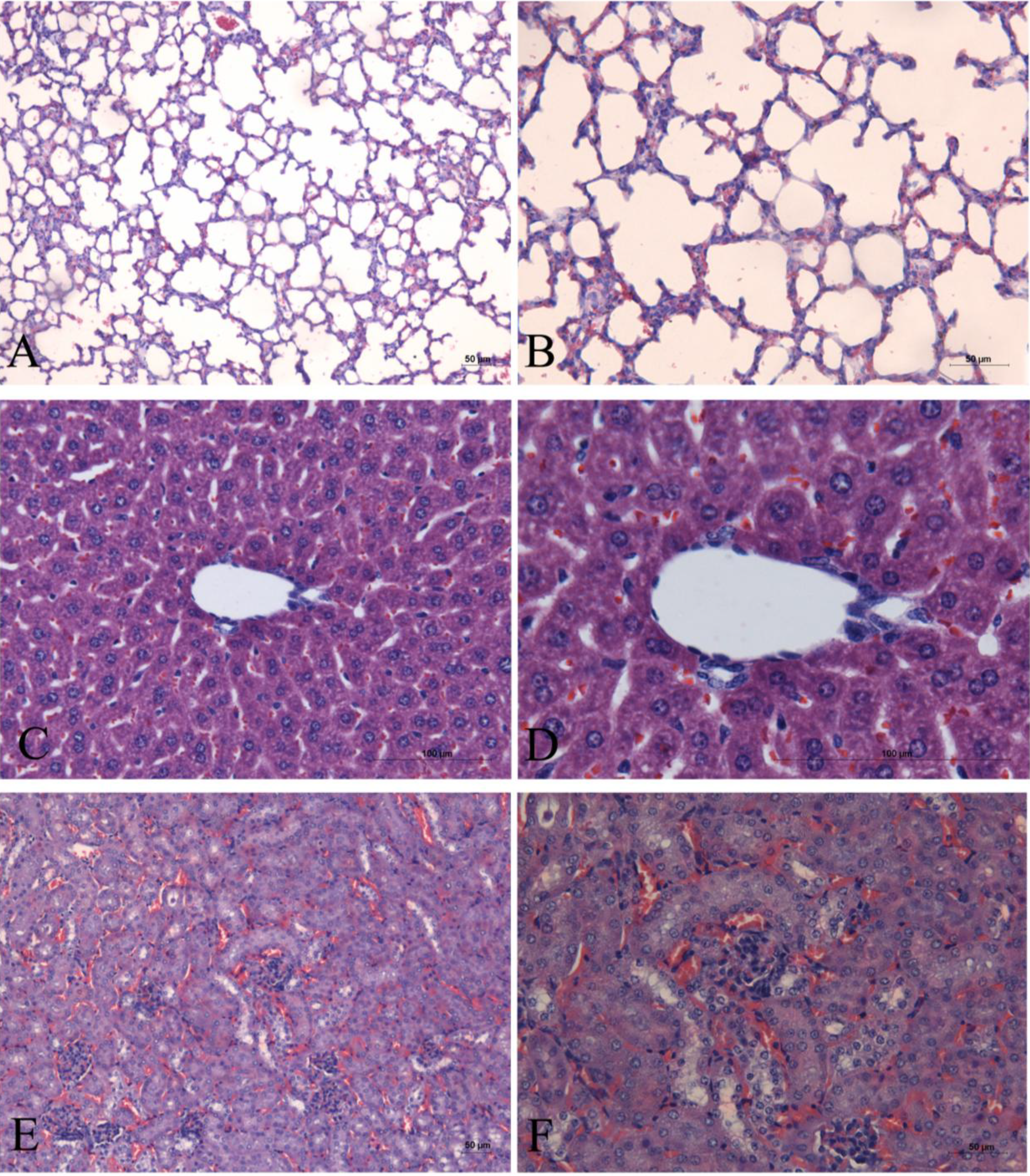
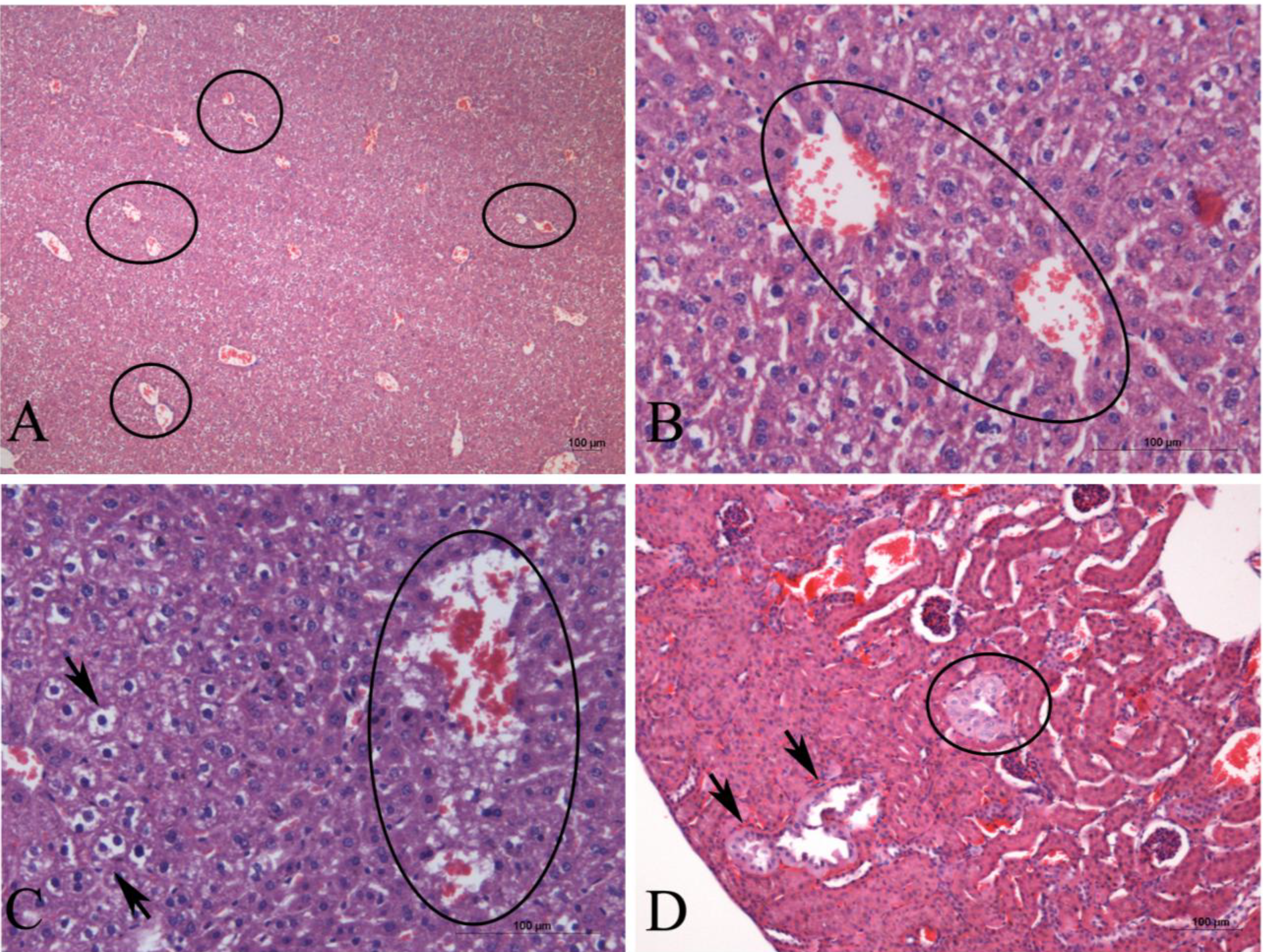


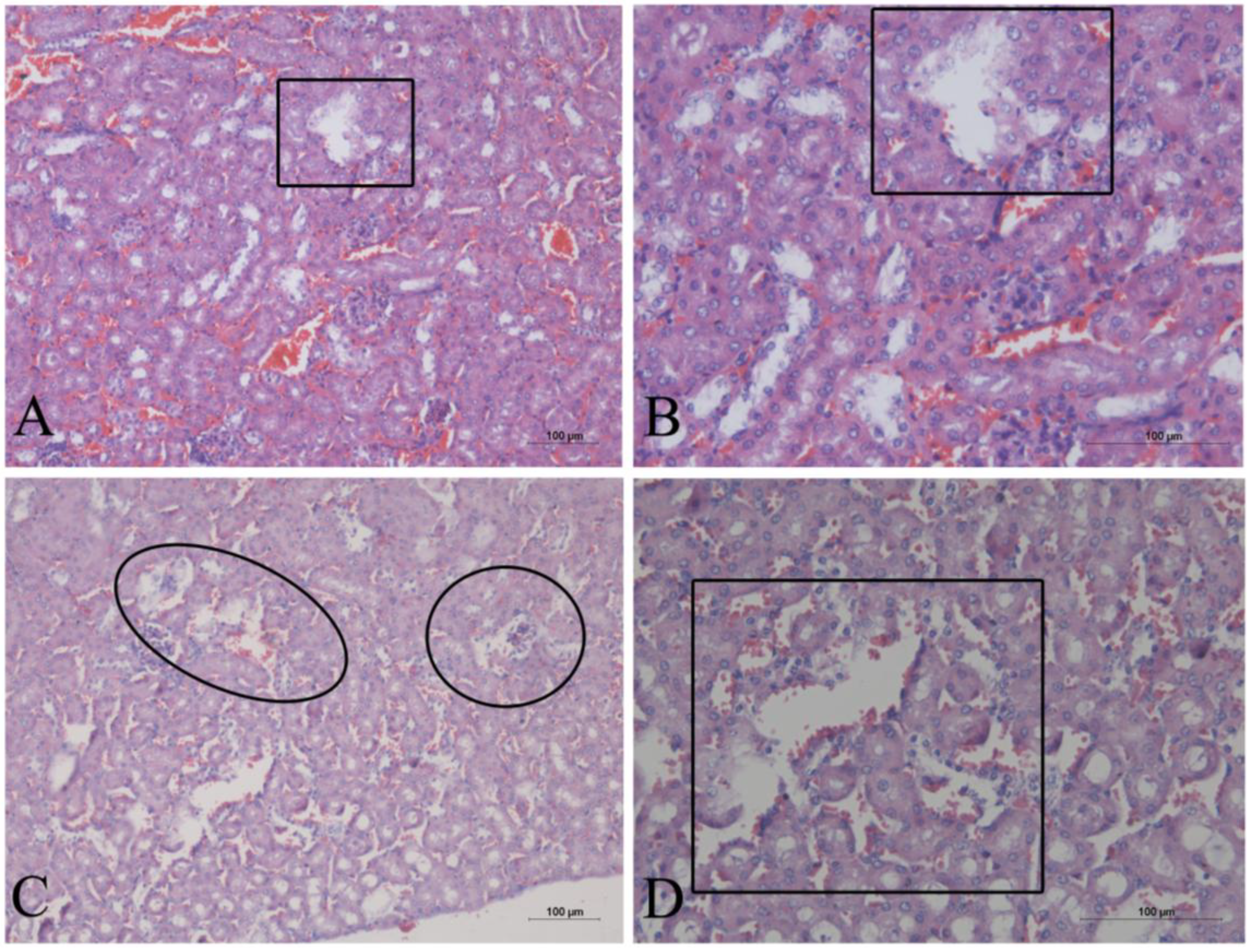
3. Discussion
4. Experimental Section
4.1. Biomass Production
4.2. Preparation of Cyanobacteria Extracts
4.3. Mouse Bioassay
4.4. Histological Analysis
4.5. Hemolytic Assay
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Blunt, J.W.; Copp, B.R.; Hu, W.P.; Munro, M.H.; Northcote, P.T.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2008, 25, 35–94. [Google Scholar] [CrossRef]
- Blunt, J.W.; Copp, B.R.; Hu, W.P.; Munro, M.H.; Northcote, P.T.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2009, 26, 170–244. [Google Scholar] [CrossRef]
- Blunt, J.W.; Copp, B.R.; Munro, M.H.; Northcote, P.T.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2010, 27, 165–237. [Google Scholar] [CrossRef]
- Blunt, J.W.; Copp, B.R.; Munro, M.H.; Northcote, P.T.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2011, 28, 196–268. [Google Scholar] [CrossRef]
- Blunt, J.W.; Copp, B.R.; Keyzers, R.A.; Munro, M.H.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2012, 29, 144–222. [Google Scholar] [CrossRef]
- Blunt, J.W.; Copp, B.R.; Keyzers, R.A.; Munro, M.H.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2013, 30, 237–323. [Google Scholar] [CrossRef]
- Hu, G.-P.; Yuan, J.; Sun, L.; She, Z.-G.; Wu, J.-H.; Lan, X.-J.; Zhu, X.; Lin, Y.-C.; Chen, S.P. Statistical research on marine natural products based on data obtained between 1985 and 2008. Mar. Drugs 2011, 9, 514–525. [Google Scholar] [CrossRef]
- Martins, R.; Pereira, P.; Welker, M.; Fastner, J.; Vasconcelos, V.M. Toxicity of culturable cyanobacteria strains isolated from the Portuguese coast. Toxicon 2005, 46, 454–464. [Google Scholar] [CrossRef]
- El-Sheekha, M.M.; Osmana, M.E.H.; Dyabb, M.A.; Amera, M.S. Production and characterization of antimicrobial active substance from the cyanobacterium Nostoc muscoru. Toxicol. Pharmacol. 2006, 21, 42–50. [Google Scholar]
- Falconer, I.R. Measurement of Toxins from Blue-Green Algae in Water and Foodstuffs. In Algal Toxins in Seafood and Drinking Water, 1st ed.; Falconer, I.R., Ed.; Academic Press: London, UK, 1993; Volume 1, pp. 165–175. [Google Scholar]
- Carmichael, W.W. The toxins of Cyanobacteria. Sci. Am. 1994, 170, 78–86. [Google Scholar] [CrossRef]
- Skulberg, O.M.; Carmichael, W.W.; Codd, G.A.; Skulberg, R. Taxonomy of Toxic Cyanophyceae (Cyanobacteria). In Algal Toxins in Seafood and Drinking Water, 1st ed.; Falconer, I.R., Ed.; Academic Press: London, UK, 1993; Volume 1, pp. 145–164. [Google Scholar]
- Komárek, J.; Anagnostidis, K. Cyanoprokaryota Part 2. Oscillatoriales. In Süsswasserflora von Mitteleuropa; 19/2; Büdel, B., Krienitz, L., Gärtner, G., Schagerl, M., Eds.; Elsevier Spektrum Akademischer Verlag: München, Germany, 2005; pp. 1–759. [Google Scholar]
- Osborne, N.J.T.; Webb, P.M.; Shaw, G.R. The toxins of Lyngbya majuscula and their human and ecological health effects. Environ. Int. 2001, 27, 381–392. [Google Scholar] [CrossRef]
- Fusetani, N.; Kem, W. Marine Toxins: An Overview. In Marine Toxins as Research Tools. Progress in Molecular and Subcellular Biology (Marine Molecular Biotechnology), 1st ed.; Fusetani, N., Kem, W., Eds.; Springer: Berlin, Germany, 2009; Volume 1, pp. 1–44. [Google Scholar]
- Pearson, L.; Mihali, T.; Moffitt, M.; Kellmann, R.; Neilan, B. On the chemistry, toxicology and genetics of the cyanobacterial toxins, microcystin, nodularin, saxitoxin and cylindrospermopsin. Mar. Drugs 2010, 8, 1650–1680. [Google Scholar] [CrossRef]
- Rangel, M.; Brunetti, R.L.; Garcia, A.N.; Cambui, C.C.N.; Conserva, G.A.A.; Neves, A.C.; Sant’Anna, C.L.; Carvalho, L.R. Acute effects of three Geitlerinema spp. (Cyanobacteria) extracts administrated in mice: Symptoms and histopathological aspects. Phytochem. Rev. 2012, 11, 1–11. [Google Scholar] [CrossRef]
- Sivonen, K.; Jones, G. Cyanobacterial Toxins. In Toxic Cyanobacteria in Water: A Guide to Their Public Health Consequences, Monitoring and Management, 1st ed.; Chorus, I., Bartram, J., Eds.; E & FN Spon: London, UK, 1999; Volume 1, pp. 43–112. [Google Scholar]
- Azevedo, S.M.F.O.; Carmichael, W.W.; Jochimsen, E.M.; Rinehart, K.L.; Lau, S.; Shaw, G.R.; Eaglesham, G.K. Human intoxication by microcystins during renal dialysed treatment in Caruaru-Brazil. Toxicology 2002, 181–182, 441–446. [Google Scholar] [CrossRef]
- Carvalho, L.R.; Pipole, F.; Werner, V.R.; Laughinghouse, H.D.; Camargo, A.C.M.; Rangel, M.; Konno, K.; Sant’Anna, C.L. A toxic cyanobacterial mixed bloom in an urban coastal lake, Rio Grande do Sul State, Southern Brazil. Braz. J. Microbiol. 2008, 39, 761–769. [Google Scholar] [CrossRef]
- Van Apeldoorn, M.E.; van Egmond, H.P.; Speijers, G.J.A.; Bakker, G.J.I. Review—Toxins of cyanobacteria. Mol. Nutr. Food Res. 2007, 51, 1–60. [Google Scholar]
- MacKintosh, C.; Beattie, K.A.; Klumpp, S.; Cohen, P.; Codd, G.A. Cyanobacterial microcystin-LR is a potent and specific inhibitor of protein phosphatases 1 and 2A from both mammals and higher plants. FEBS Lett. 1990, 264, 187–192. [Google Scholar] [CrossRef]
- Runnegar, M.; Berndt, N.; Kong, S.M.; Lee, E.Y.C.; Zhang, L.F. In vivo and in vitro binding of microcystin to protein phosphatase 1 and 2A. Biochem. Biophys. Res. Commun. 1995, 216, 162–169. [Google Scholar] [CrossRef]
- Carvalho, M.C.; Agujaro, L.F.; Pires, D.A.; Picoli, C. Planktonic Cyanobateria Manual: Legal and Environmental Aspects and Monitoring Guidelines; CETESB: São Paulo, Brazil, 2013; pp. 1–47. [Google Scholar]
- Harada, K.; Kondo, F.; Lawton, L. Laboratory Analysis of Cyanotoxins. In Toxic Cyanobacteria in Water: A Guide to Their Public Health Consequences, Monitoring and Management, 1st ed.; Chorus, I., Bartram, J., Eds.; E & FN Spon: New York, NY, USA, 1999; Volume 1, pp. 369–405. [Google Scholar]
- Dogo, C.R.; Bruni, F.M.; Elias, F.; Rangel, M.; Pantoja, P.A.; Sant’Anna, C.L.; Lima, C.; Lopes-Ferreira, M.; Carvalho, L.R. Inflammatory effects of the toxic cyanobacterium Geitlerinema amphibium. Toxicon 2011, 58, 464–470. [Google Scholar] [CrossRef]
- Dogo, C.R.; Rangel, M.; Cardoso-Lopes, E.M.; Sant’anna, C.L.; Bruni, F.M.; Lopes-Ferreira, M.; Carvalho, L.R. Evaluation of Anticholinesterasic Activity of Strain SPC 920 Geitlerinema Unigranulatum. In Poisoning by Plants, Micotoxins and Related Toxins, 1st ed.; Riet-Correa, F., Pfister, J., Schild, A.L., Wierenga, T., Eds.; CAB International: Wallingford, CT, USA, 2011; pp. 725–730. [Google Scholar]
- Carvalho, L.R.; Neves, A.C.; Conserva, G.A.A.; Brunetti, R.L.; Hentschke, G.S.; Malone, C.F.S.; Torres, L.M.B.; Sant’Anna, C.L.; Rangel, M. Biologically active compounds from cyanobacteria extracts: in vivo and in vitro aspects. Braz. J. Pharmacogn. 2013, 23, 471–480. [Google Scholar]
- Brunetti, R.L.; Campos-Junior, A.G.; Garcia, A.N.; Dogo, C.R.; Mattos, L.F.A.; Carvalho, L.R.; Rangel, M. Histopathological evaluation of injected mice with extracts of two toxic cyanobacteria strains. In Proceedings of the XI Reunião Cientifica Anual do Instituto Butantan, Sao Paulo, Brazil, 2–4 December 2009; Memórias do Instituto Butantan: Butantã, Brazil, 2009; Volume 66, pp. 267–267. [Google Scholar]
- Lawton, L.A.; Beattie, K.A.; Hawser, S.P.; Campbell, D.L.; Codd, G.A. Evaluation of Assay Methods for the Determination of Cyanobacterial Hepatotoxicity. In Detection Methods for Cyanobacterial Toxins; Codd, G.A., Jefferies, T.M., Keevil, C.W., Potter, E., Eds.; The Royal Society of Chemistry: Cambridge, UK, 1994; pp. 111–116. [Google Scholar]
- Pelander, A.; Ojanperä, I.; Lahti, K.; Niinivaara, K.; Vuori, E. Visual detection of cyanobacterial hepatotoxins by thin-layer chromatography and application to water analysis. Water Res. 2000, 34, 2643–2652. [Google Scholar] [CrossRef]
- Buckley, L.J.; Ikawa, M.; Sasner, J.J. Isolation of Gonyaulax tamarensis toxins from soft shell clams (Mya arenaria) and a thin-layer chromatographic-fluorometric method for their detection. J. Agric. Food Chem. 1976, 24, 107–110. [Google Scholar] [CrossRef]
- Ojanpera, I.; Vuori, E.; Himberg, K.; Waris, M.; Niinivaara, K. Facile detection of anatoxin-a in algal material by thin-layer chromatography with Fast Black K Salt. Analyst 1991, 116, 265–267. [Google Scholar] [CrossRef]
- Vega, A.; Bell, E.A. α-amino-β-methylaminopropionic acid, a new amino acid from seeds of Cycas circinalis. Phytochemistry 1967, 6, 759–762. [Google Scholar] [CrossRef]
- Conserva, G.A.A.; Sant’Anna, C.L.; Cambui, C.C.N.; Brunetti, R.L.; Rangel, M.; Torres, L.M.B.; Young, M.C.M.; Carvalho, L.R. Prospecção de atividades toxicológicas e farmacológicas em cepas de cianobactérias da Coleção de Culturas do Instituto de Botânica (“Screening of toxicological and pharmacological activities in cyanobacteria strains of the Culture Collection of the Institute of Botany”). In Proceedings of the 18ª Reunião Cientifica Anual do Instituto de Botânica, Sao Paulo, Brazil, 21–25 November 2011; pp. 1–4.
- Gupta, N.; Pant, S.C.; Vijayaraghavan, R.; Lakshmana Rao, P.V. Comparative toxicity evaluation of cyanobacterial cyclic peptide toxin microcystin variants (LR, RR, YR) in mice. Toxicology 2003, 188, 285–296. [Google Scholar] [CrossRef]
- Guzman, R.E.; Solter, P.F. Characterization of sublethal microcystin-LR exposure in mice. Vet. Pathol. 2002, 39, 17–26. [Google Scholar] [CrossRef]
- Hawkins, P.R.; Runnegar, M.T.C.; Jackson, A.R.B.; Falconer, I.R. Severe hepatotoxicity caused by the tropical Cyanobacterium (Blue-Green Alga) Cylindrospermopsis raciborskii (Woloszynska) Seenaya and Subba Raju isolated from a domestic water supply reservoir. Appl. Environ. Microbiol. 1985, 50, 1292–1295. [Google Scholar]
- Kuiper-Goodman, T.; Falconer, I.; Fitzgerald, J. Human Health Aspects. In Toxic Cyanobacteria in Water: A Guide to Their Public Health Consequences, Monitoring and Management, 1st ed.; Chorus, I., Bartram, J., Eds.; E & FN Spon: New York, NY, USA, 1999; Volume 1, pp. 125–160. [Google Scholar]
- Fastner, J.; Heinzeb, R.; Humpage, A.R.; Mischke, U.; Eaglesha, G.K.; Chorus, I. Cylindrospermopsin occurrence in two German lakes and preliminary assessment of toxicity and toxin production of Cylindrospermopsis raciborskii (Cyanobacteria) isolates. Toxicon 2003, 42, 313–321. [Google Scholar]
- Bazin, E.; Huet, S.; Jarry, G.; Le He´garat, L.; Munday, J.S.; Humpage, A.R.; Fessard, V. Cytotoxic and genotoxic effects of cylindrospermopsin in mice treated by gavage or intraperitoneal injection. Environ. Toxicol. 2012, 27, 277–284. [Google Scholar]
- Stewart, I.; Schluter, P.J.; Shaw, G.R. Cyanobacterial lipopolysaccharides and human health—A review. Environ. Health 2006, 5, 7. [Google Scholar] [CrossRef]
- Teneva, I.; Mladenov, R.; Dzhambazov, B. Toxic effects of extracts from Pseudanabaena galeata (Cyanoprokaryota) in mice and cell cultures in vitro. Nat. Sci. Hum. 2009, 12, 237–243. [Google Scholar]
- Humpage, A.; Falconer, I.; Bernard, C.; Froscio, S.; Fabbro, L. Toxicity of the cyanobacterium Limnothrix AC0243 to male Balb/c mice. Water Res. 2012, 46, 1576–1583. [Google Scholar]
- Rangel, M.; Konno, K.; Brunaldi, K.; Procopio, J.; Freitas, J.C. Neurotoxic activity induced by a haemolytic substance in the extract of the marine sponge Geodia corticostylifera. Comp. Biochem. Physiol. (Part C) 2005, 141, 207–215. [Google Scholar]
- Grasso, A.; Alema, S.; Rufini, S.; Senni, M.I. Black widow spider toxin-induced calcium fluxes and transmitter release in neurosecretory cell line. Nature 1980, 283, 714–716. [Google Scholar]
- Robello, M.; Fresia, M.; Maga, L.; Grasso, A.; Ciani, S. Permeation of divalent cations through α-latrotoxin channels in lipid bilayers: steady-state current-voltage relationships. J. Membr. Biol. 1987, 95, 55–62. [Google Scholar]
- Lazarovici, P.; Primor, N.; Caratsch, C.G.; Munz, K.; Lelkes, P.; Loew, L.M.; Shai, Y.; Louini, K.; Contreras, M.L.; Fox, J.; et al. Action on Artificial and Neuronal Membranes of Pardaxin, a New Presynaptic Excitatory Polypeptide Neurotoxin with Ionophore Activity. In Neurotoxins in Neurochemistry, 1st ed.; Dolly, J.O., Ed.; Camelot: Southampton, UK, 1988; Volume 1, pp. 219–240. [Google Scholar]
- Shai, Y.; Fox, J.; Caratsch, C.; Shi, Y.L.; Edwards, C.; Lazarovici, P. Sequencing and synthesis of pardaxin, a polypeptide from the Red Sea Moses sole with ionophore activity. FEBS Lett. 1988, 242, 161–166. [Google Scholar]
- Shi, Y.L.; Edwards, C.; Lazarovici, P. Ion selectivity of the channels formed by pardaxin, an ionophore, in bilayer membranes. Nat. Toxins 1995, 3, 151–155. [Google Scholar]
- Berlinck, R.G.S.; Ogawa, C.A.; Almeida, A.M.P; Sanchez, M.A.A.; Malpezzi, E.L.A.; Costa, L.V.; Hajdu, E.; Freitas, J.C. Chemical and pharmacological characterization of halitoxin from Amphimedon viridis (Porifera) from the southeastern Brazilian coast. Comp. Biochem. Physiol. (Part C) 1996, 115, 155–163. [Google Scholar]
- Umbach, J.A.; Grasso, A.; Zurcher, S.D.; Kornblum, H.I.; Mastrogiacomo, A.; Gundersen, C.B. Electrical and optical monitoring of alatrotoxin action at Drosophila neuromuscular junctions. Neuroscience 1998, 87, 913–924. [Google Scholar]
- Yan, L.; Adams, M.E. Lycotoxins, antimicrobial peptides from venom of the wolf spider Lycosa carolinensis. J. Biol. Chem. 1998, 273, 2059–2066. [Google Scholar]
- Scott, R.H.; Whyment, A.D.; Gordon, K.H.; Milne, B.F.; Jaspars, M. Analysis of the structure and electrophysiological actions of halitoxins: 1,3 alkyl-pyridinium salts from Callyspongia ridleyi. J. Membr. Biol. 2000, 176, 119–131. [Google Scholar]
- Azevedo, M.T.P.; Sant’Anna, C.L. Sphaerocavum brasiliense, a new planktic genus and species of Cyanobacteria from reservoirs of Sao Paulo State, Brazil. Algol. Stud. 2003, 109, 79–92. [Google Scholar]
- Pyo, D.; Shin, H. Extraction and analysis of microcystins RR and LR in cyanobacteria using a cyano cartridge. J. Biochem. Biophys. Methods 2002, 51, 103–109. [Google Scholar]
- Rangel, M.; Cabrera, M.P.S.; Kazuma, K.; Ando, K.; Wang, X.; Kato, M.; Nihei, K.; Hirata, I.Y.; Cross, T.J.; Garcia, A.N.; et al. Chemical and biological characterization of four new linear cationic α-helical peptides from the venoms of two solitary eumenine wasps. Toxicon 2011, 57, 1081–1092. [Google Scholar]
- Rangel, M.; Malpezzi, E.L.A.; Susini, S.M.M.; Freitas, J.C. Hemolytic activity in extracts of the diatom Nitzschia. Toxicon 1997, 35, 305–309. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Rangel, M.; Martins, J.C.G.; Garcia, A.N.; Conserva, G.A.A.; Costa-Neves, A.; Sant'Anna, C.L.; De Carvalho, L.R. Analysis of the Toxicity and Histopathology Induced by the Oral Administration of Pseudanabaena galeata and Geitlerinema splendidum (Cyanobacteria) Extracts to Mice. Mar. Drugs 2014, 12, 508-524. https://doi.org/10.3390/md12010508
Rangel M, Martins JCG, Garcia AN, Conserva GAA, Costa-Neves A, Sant'Anna CL, De Carvalho LR. Analysis of the Toxicity and Histopathology Induced by the Oral Administration of Pseudanabaena galeata and Geitlerinema splendidum (Cyanobacteria) Extracts to Mice. Marine Drugs. 2014; 12(1):508-524. https://doi.org/10.3390/md12010508
Chicago/Turabian StyleRangel, Marisa, Joyce C. G. Martins, Angélica Nunes Garcia, Geanne A. A. Conserva, Adriana Costa-Neves, Célia Leite Sant'Anna, and Luciana Retz De Carvalho. 2014. "Analysis of the Toxicity and Histopathology Induced by the Oral Administration of Pseudanabaena galeata and Geitlerinema splendidum (Cyanobacteria) Extracts to Mice" Marine Drugs 12, no. 1: 508-524. https://doi.org/10.3390/md12010508



