Chitosan-Alginate Biocomposite Containing Fucoidan for Bone Tissue Engineering
Abstract
:1. Introduction
2. Results and Discussion
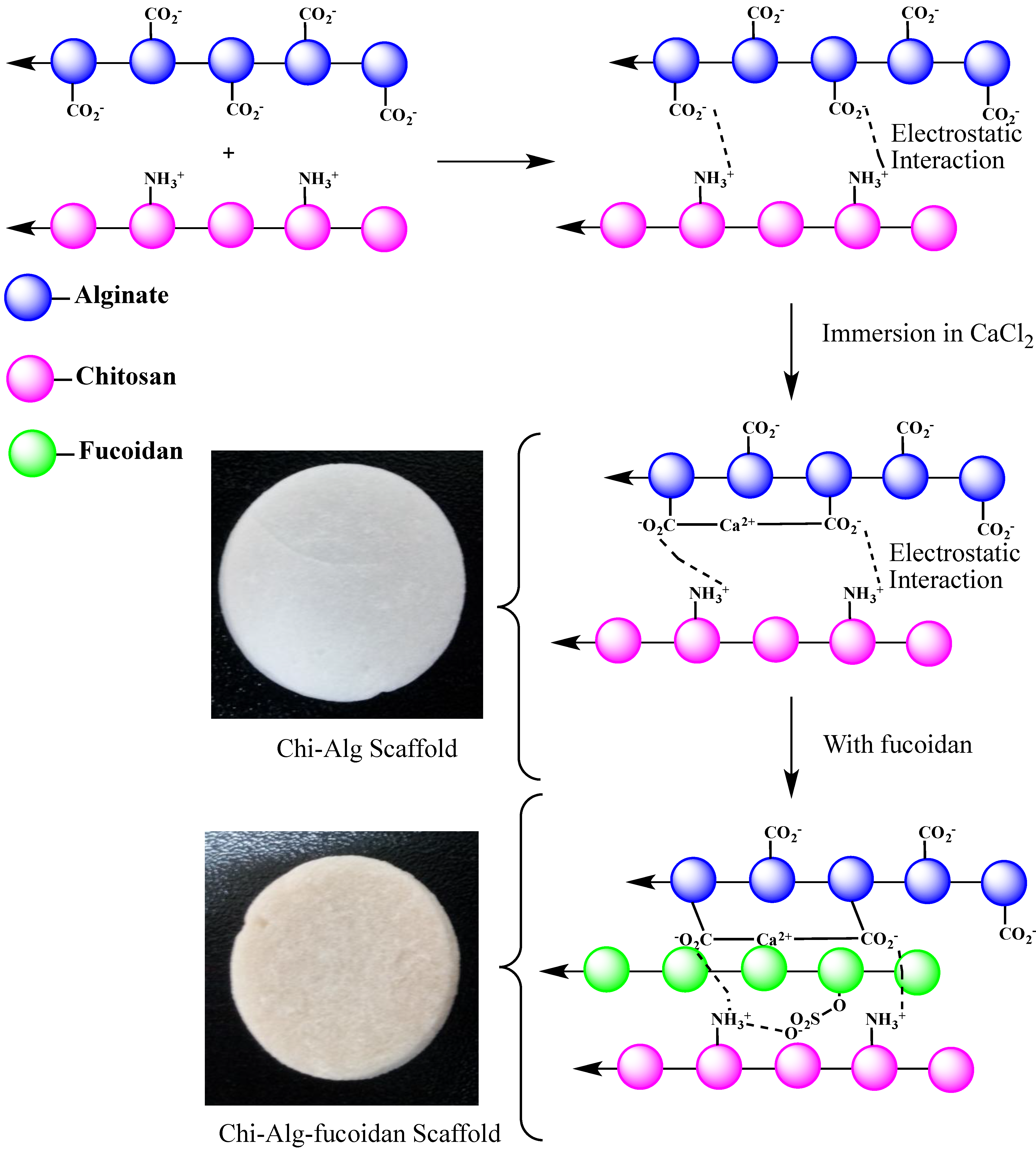
2.1. General Observation
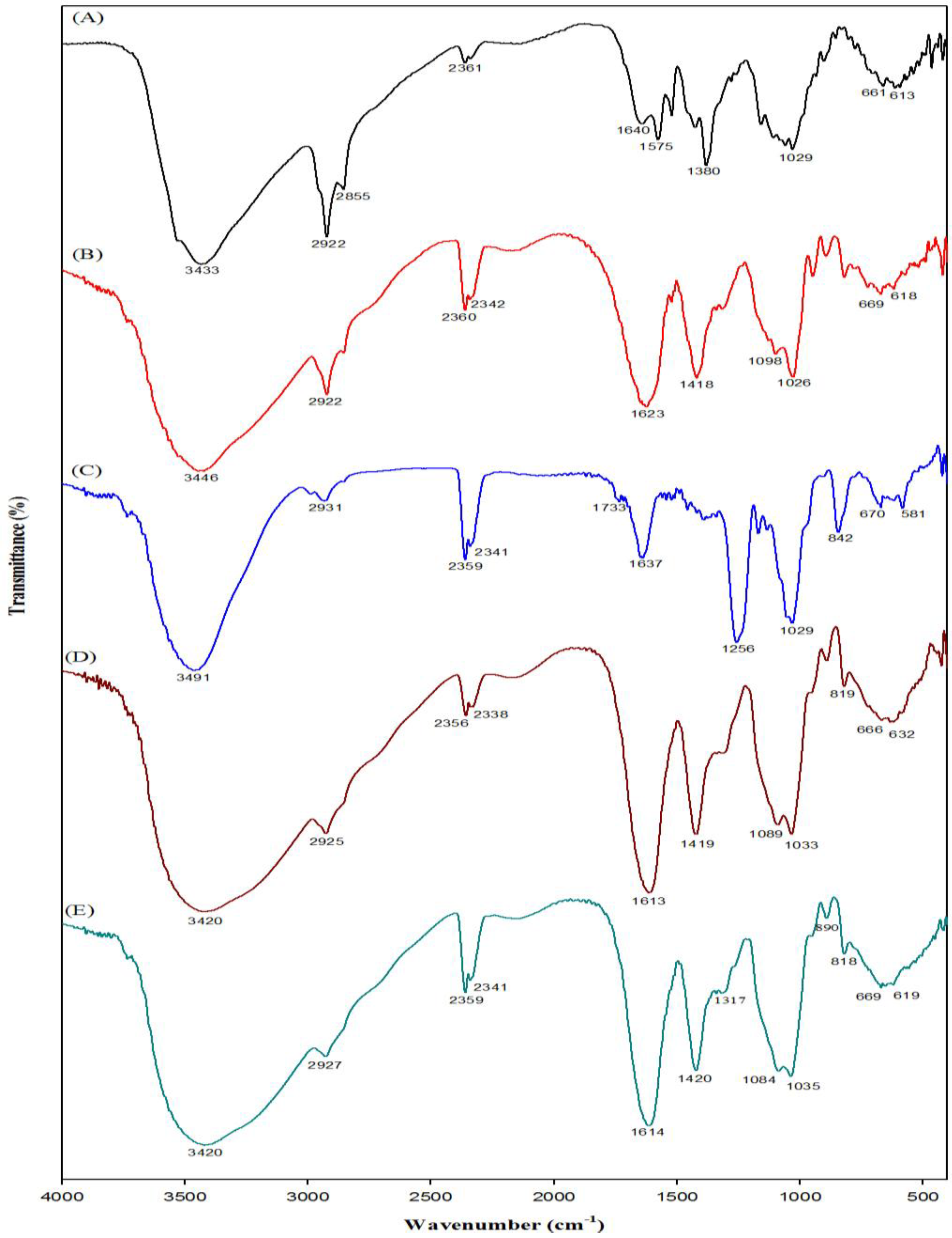
2.2. Fourier Transform-Infrared Spectroscopy
2.3. Porosity of the Scaffolds
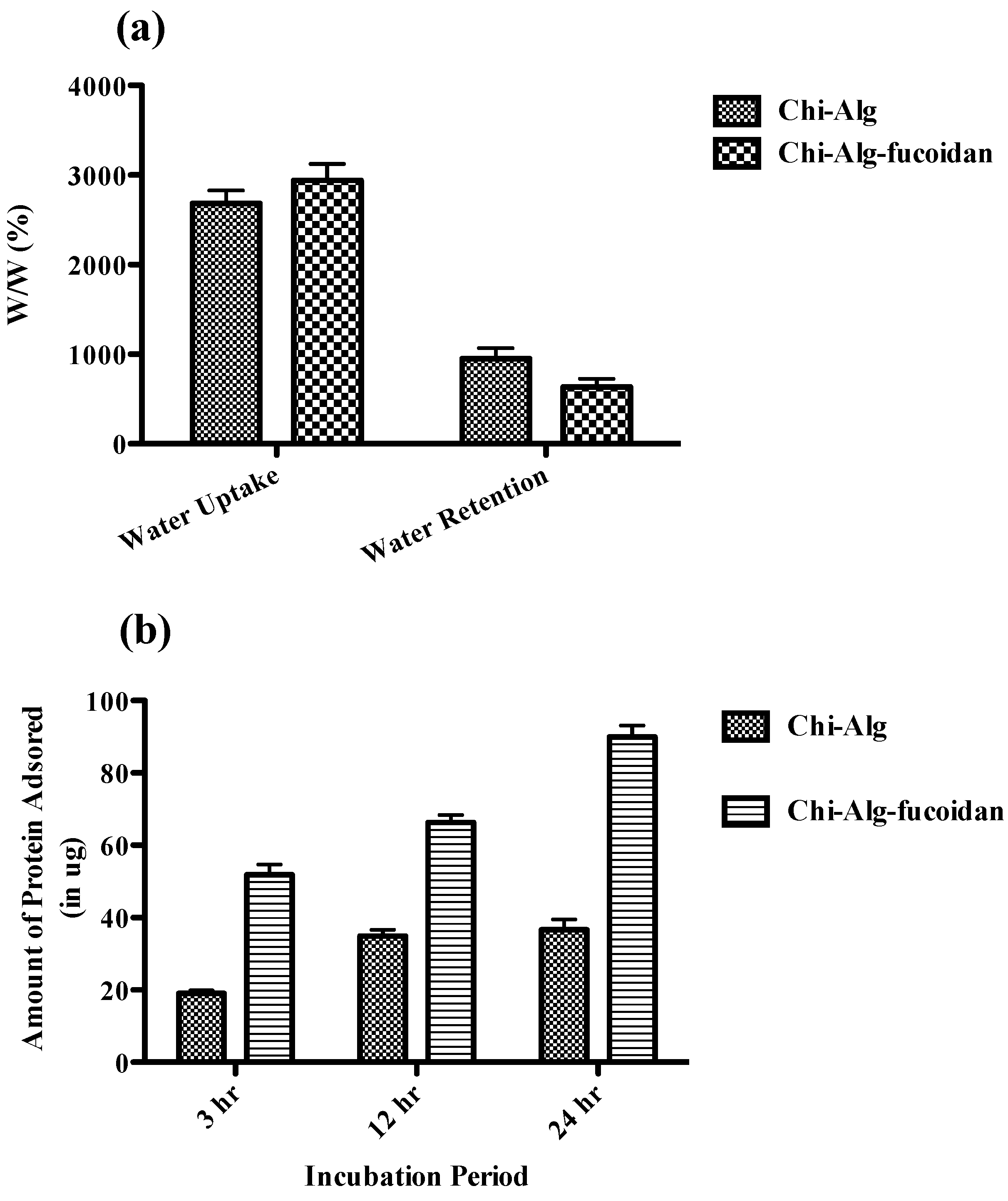
2.4. Water Uptake and Retention Ability of the Scaffolds
2.5. Protein Adsorption Efficiency
2.6. In vitro Biodegradation Behavior
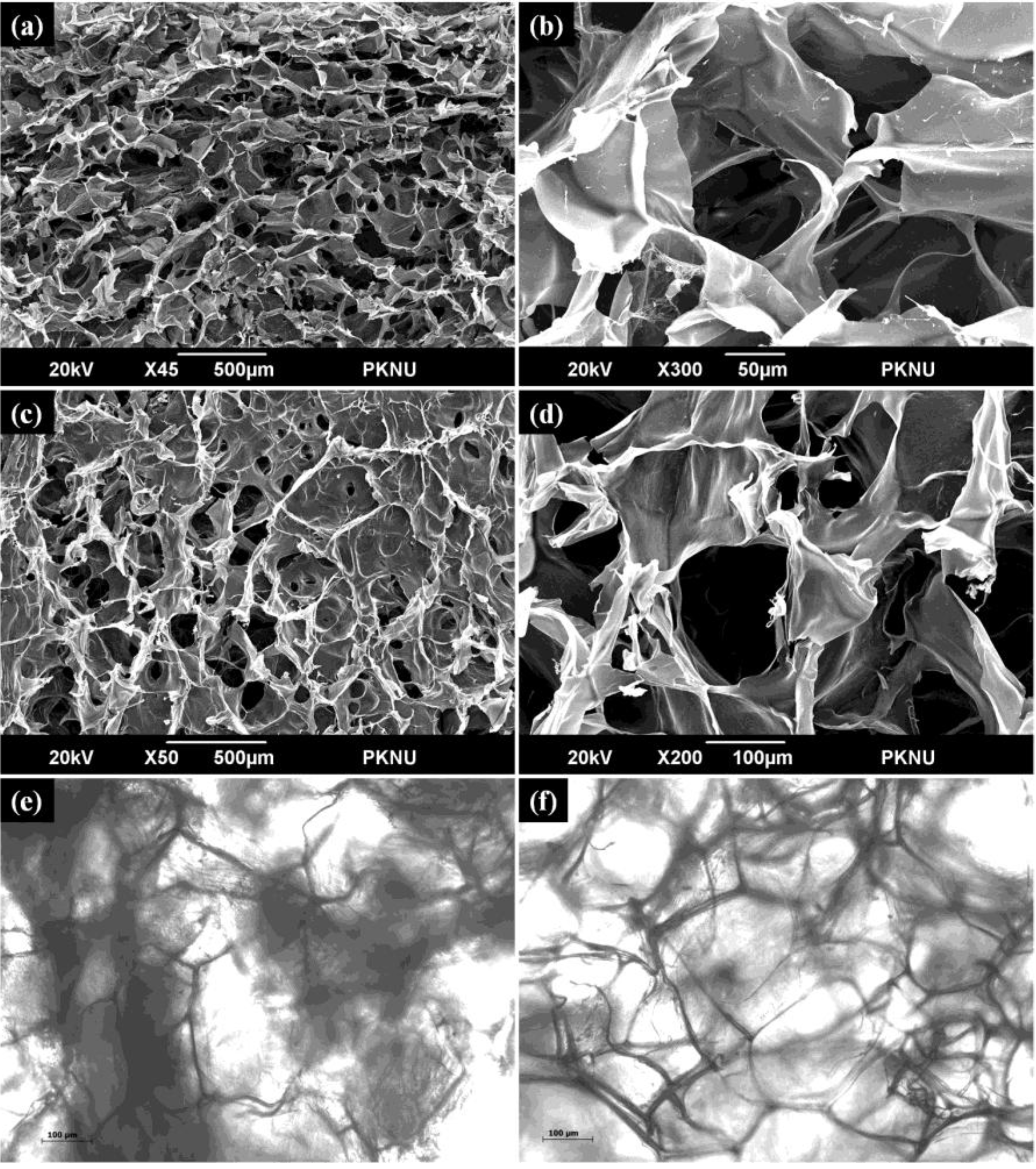
2.7. Scanning Electron Microscopy and Optical Microscopy Analysis
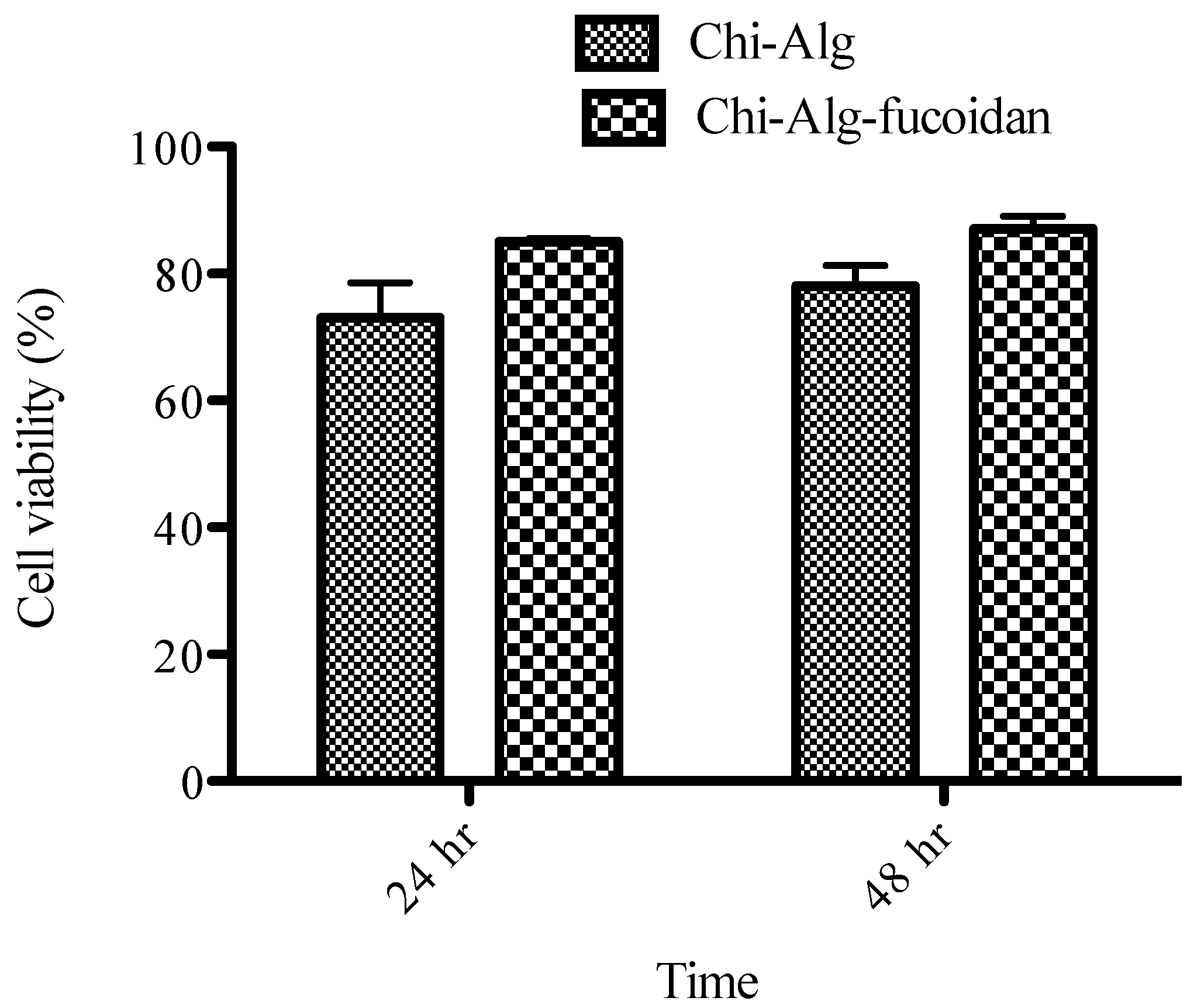
2.8. Biocompatibility of the Scaffolds
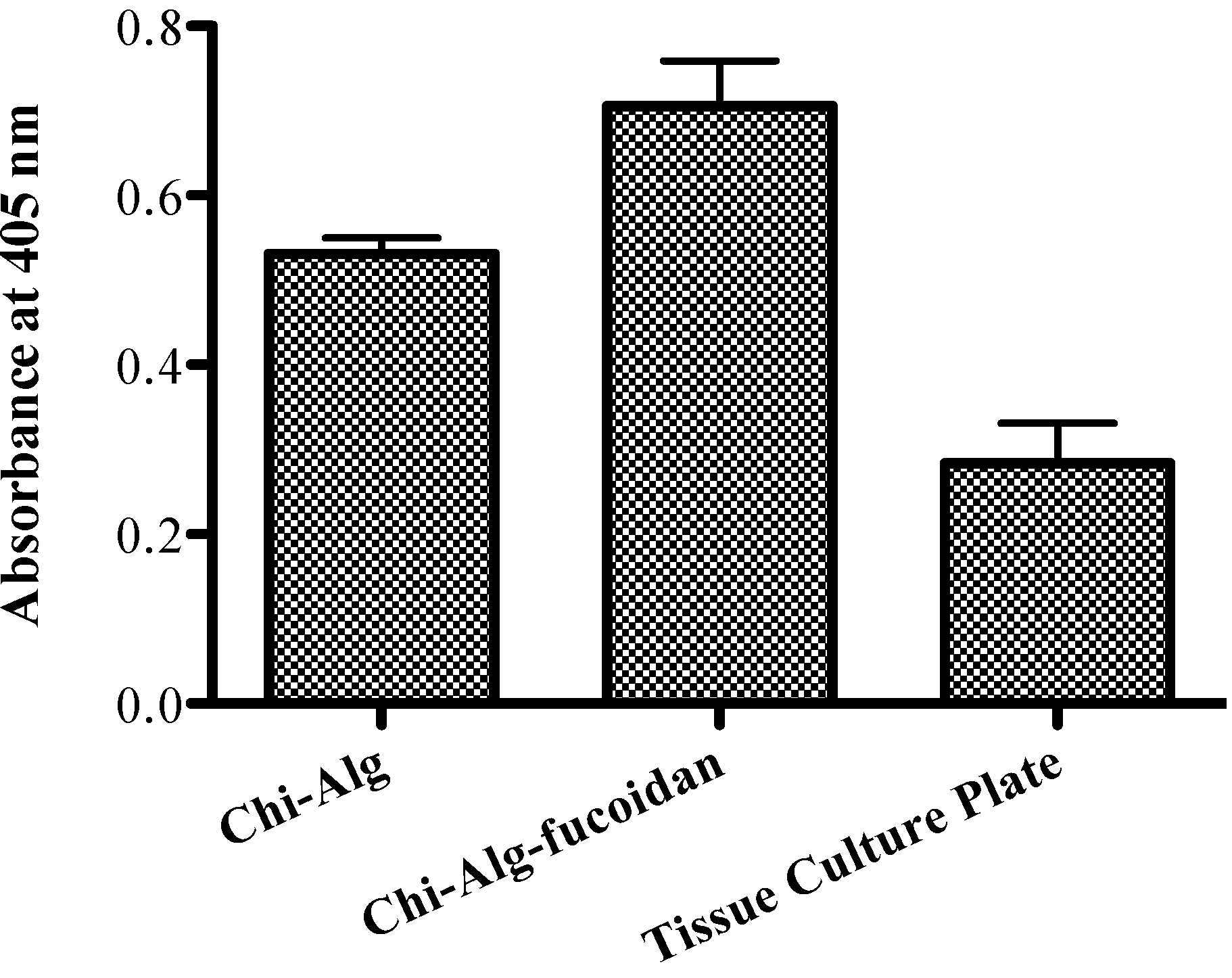
2.9. Alkaline Phosphatase Activity
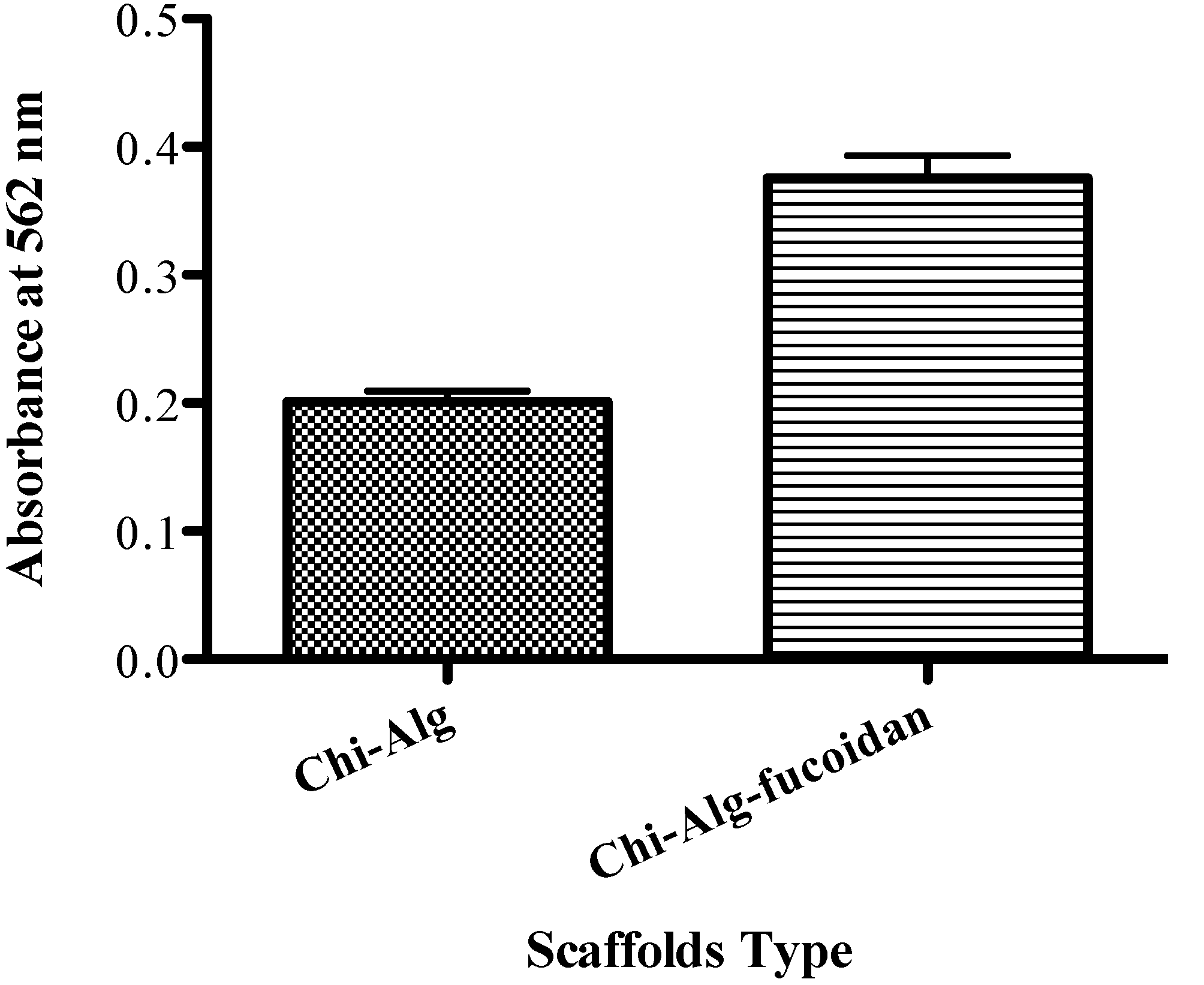
2.10. Mineralization Results
3. Experimental Section
3.1. Preparation of the Chitosan-Alginate (Chi-Alg) Scaffold
3.2. Chitosan-Alginate-Fucoidan (Chi-Alg-Fucoidan) Scaffold
3.3. Physicochemical Characterization
3.3.1. Fourier Transform-Infrared (FT-IR) Spectroscopy
3.3.2. Porosity Measurement
3.3.3. Water Uptake and Retention Abilities
3.3.4. Protein Adsorption Study
3.3.5. In Vitro Biodegradation Behavior
3.3.6. Scanning Electron Microscopy (SEM) and Optical Microscopy
3.4. Cell Culture Studies
3.4.1. Cytotoxic Studies
3.4.2. Alkaline Phosphatase Activity
3.4.3. Mineralization Assay
3.5. Statistical Analysis
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- American Academy of Orthopaedic Surgeons Home Page. Available online: http://www.aaos.org/news/aaosnow/jan08/reimbursement2.asp (accessed on 21 December 2013).
- Giannoudis, P.V.; Dinopoulos, H.; Tsiridis, E. Bone substitutes: An update. Injury 2005, 36, S20–S27. [Google Scholar] [CrossRef]
- Sowjanya, J.; Singh, J.; Mohita, T.; Sarvanan, S.; Moorthi, A.; Srinivasan, N.; Selvamurugan, N. Biocomposite scaffolds containing chitosan/alginate/nano-silica for bone tissue engineering. Colloids Surf. B Biointerfaces 2013, 109, 294–300. [Google Scholar] [CrossRef]
- Jayakumar, R.; Menon, D.; Manzoor, K.; Nair, S.; Tamura, H. Biomedical applications of chitin and chitosan based nanomaterials—A short review. Carbohydr. Polym. 2010, 82, 227–232. [Google Scholar] [CrossRef]
- Muzzarelli, R.A. Chitins and chitosans for the repair of wounded skin, nerve, cartilage and bone. Carbohydr. Polym. 2009, 76, 167–182. [Google Scholar] [CrossRef]
- Di Martino, A.; Sittinger, M.; Risbud, M.V. Chitosan: A versatile biopolymer for orthopaedic tissue-engineering. Biomaterials 2005, 26, 5983–5990. [Google Scholar] [CrossRef]
- Pallela, R.; Venkatesan, J.; Janapala, V.R.; Kim, S.K. Biophysicochemical evaluation of chitosan-hydroxyapatite-marine sponge collagen composite for bone tissue engineering. J. Biomed. Mater. Res. A 2012, 100, 486–495. [Google Scholar]
- Thein-Han, W.W.; Misra, R.D.K. Biomimetic chitosan–nanohydroxyapatite composite scaffolds for bone tissue engineering. Acta Biomater. 2009, 5, 1182–1197. [Google Scholar] [CrossRef]
- Venkatesan, J.; Pallela, R.; Bhatnagar, I.; Kim, S.-K. Chitosan–amylopectin/hydroxyapatite and chitosan–chondroitin sulphate/hydroxyapatite composite scaffolds for bone tissue engineering. Int. J. Biol. Macromol. 2012, 51, 1033–1042. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, M. Synthesis and characterization of macroporous chitosan/calcium phosphate composite scaffolds for tissue engineering. J. Biomed. Mater. Res. 2001, 55, 304–312. [Google Scholar] [CrossRef]
- Croisier, F.; Jérôme, C. Chitosan-based biomaterials for tissue engineering. Eur. Polym. J. 2013, 49, 780–792. [Google Scholar] [CrossRef]
- Dash, M.; Chiellini, F.; Ottenbrite, R.; Chiellini, E. Chitosan—A versatile semi-synthetic polymer in biomedical applications. Prog. Polym. Sci. 2011, 36, 981–1014. [Google Scholar] [CrossRef]
- Venkatesan, J.; Kim, S.-K. Chitosan composites for bone tissue engineering—An overview. Mar. Drugs 2010, 8, 2252–2266. [Google Scholar] [CrossRef]
- Sun, J.; Tan, H. Alginate-based biomaterials for regenerative medicine applications. Materials 2013, 6, 1285–1309. [Google Scholar] [CrossRef]
- Lee, K.Y.; Mooney, D.J. Alginate: properties and biomedical applications. Prog. Polym. Sci. 2012, 37, 106–126. [Google Scholar] [CrossRef]
- Anal, A.K.; Stevens, W.F. Chitosan–alginate multilayer beads for controlled release of ampicillin. Int. J. Pharm. 2005, 290, 45–54. [Google Scholar]
- Hamman, J.H. Chitosan based polyelectrolyte complexes as potential carrier materials in drug delivery systems. Mar. Drugs 2010, 8, 1305–1322. [Google Scholar] [CrossRef]
- Lai, H.L.; Abu'Khalil, A.; Craig, D.Q. The preparation and characterisation of drug-loaded alginate and chitosan sponges. Int. J. Pharm. 2003, 251, 175–181. [Google Scholar] [CrossRef]
- Lee, M.; Li, W.; Siu, R.K.; Whang, J.; Zhang, X.; Soo, C.; Ting, K.; Wu, B.M. Biomimetic apatite-coated alginate/chitosan microparticles as osteogenic protein carriers. Biomaterials 2009, 30, 6094–6101. [Google Scholar] [CrossRef]
- Mi, F.-L.; Sung, H.-W.; Shyu, S.-S. Drug release from chitosan–alginate complex beads reinforced by a naturally occurring cross-linking agent. Carbohydr. Polym. 2002, 48, 61–72. [Google Scholar] [CrossRef]
- Ribeiro, A.J.; Silva, C.; Ferreira, D.; Veiga, F. Chitosan-reinforced alginate microspheres obtained through the emulsification/internal gelation technique. Eur. J. Pharm. Sci. 2005, 25, 31–40. [Google Scholar] [CrossRef]
- Xu, Y.; Zhan, C.; Fan, L.; Wang, L.; Zheng, H. Preparation of dual crosslinked alginate–chitosan blend gel beads and in vitro controlled release in oral site-specific drug delivery system. Int. J. Pharm. 2007, 336, 329–337. [Google Scholar] [CrossRef]
- Hong, H.-J.; Jin, S.-E.; Park, J.-S.; Ahn, W.S.; Kim, C.-K. Accelerated wound healing by smad3 antisense oligonucleotides-impregnated chitosan/alginate polyelectrolyte complex. Biomaterials 2008, 29, 4831–4837. [Google Scholar] [CrossRef]
- Murakami, K.; Aoki, H.; Nakamura, S.; Nakamura, S.-I.; Takikawa, M.; Hanzawa, M.; Kishimoto, S.; Hattori, H.; Tanaka, Y.; Kiyosawa, T.; et al. Hydrogel blends of chitin/chitosan, fucoidan and alginate as healing-impaired wound dressings. Biomaterials 2010, 31, 83–90. [Google Scholar] [CrossRef]
- Wang, L.; Khor, E.; Wee, A.; Lim, L.Y. Chitosan-alginate PEC membrane as a wound dressing: Assessment of incisional wound healing. J. Biomed. Mater. Res. 2002, 63, 610–618. [Google Scholar] [CrossRef]
- Majima, T.; Funakosi, T.; Iwasaki, N.; Yamane, S.-T.; Harada, K.; Nonaka, S.; Minami, A.; Nishimura, S.-I. Alginate and chitosan polyion complex hybrid fibers for scaffolds in ligament and tendon tissue engineering. J. Orthop. Sci. 2005, 10, 302–307. [Google Scholar] [CrossRef]
- Shao, X.; Hunter, C.J. Developing an alginate/chitosan hybrid fiber scaffold for annulus fibrosus cells. J. Biomed. Mater. Res. A 2007, 82, 701–710. [Google Scholar] [CrossRef]
- Gomez d’Ayala, G.; De Rosa, A.; Laurienzo, P.; Malinconico, M. Development of a new calcium sulphate-based composite using alginate and chemically modified chitosan for bone regeneration. J. Biomed. Mater. Res. A 2007, 81, 811–820. [Google Scholar]
- Park, D.J.; Choi, B.H.; Zhu, S.J.; Huh, J.Y.; Kim, B.Y.; Lee, S.H. Injectable bone using chitosan-alginate gel/mesenchymal stem cells/BMP-2 composites. J. Cranio-Maxillofac. Surg. 2005, 33, 50–54. [Google Scholar] [CrossRef]
- Cho, Y.-S.; Jung, W.-K.; Kim, J.; Choi, I.-W.; Kim, S.-K. Beneficial effects of fucoidan on osteoblastic MG-63 cell differentiation. Food Chem. 2009, 116, 990–994. [Google Scholar] [CrossRef]
- Changotade, S.; Korb, G.; Bassil, J.; Barroukh, B.; Willig, C.; Colliec-Jouault, S.; Durand, P.; Godeau, G.; Senni, K. Potential effects of a low-molecular-weight fucoidan extracted from brown algae on bone biomaterial osteoconductive properties. J. Biomed. Mater. Res. A 2008, 87, 666–675. [Google Scholar]
- Park, S.-J.; Lee, K.W.; Lim, D.-S.; Lee, S. The sulfated polysaccharide fucoidan stimulates osteogenic differentiation of human adipose-derived stem cells. Stem Cells Dev. 2012, 21, 2204–2211. [Google Scholar] [CrossRef]
- Jin, G.; Kim, G.H. Rapid-prototyped PCL/fucoidan composite scaffolds for bone tissue regeneration: design, fabrication, and physical/biological properties. J. Mater. Chem. 2011, 21, 17710–17718. [Google Scholar] [CrossRef]
- Lee, J.S.; Jin, G.H.; Yeo, M.G.; Jang, C.H.; Lee, H.; Kim, G.H. Fabrication of electrospun biocomposites comprising polycaprolactone/fucoidan for tissue regeneration. Carbohydr. Polym. 2012, 90, 181–188. [Google Scholar] [CrossRef]
- Sezer, A.; Hatipoglu, F.; Cevher, E.; Oğurtan, Z.; Bas, A.; Akbuğa, J. Chitosan film containing fucoidan as a wound dressing for dermal burn healing: Preparation and in vitro/in vivo evaluation. AAPS PharmSciTech 2007, 8, E94–E101. [Google Scholar]
- Zhang, L.; Guo, J.; Zhou, J.; Yang, G.; Du, Y. Blend membranes from carboxymethylated chitosan/alginate in aqueous solution. J. Appl. Polym. Sci. 2000, 77, 610–616. [Google Scholar] [CrossRef]
- Ho, Y.-C.; Mi, F.-L.; Sung, H.-W.; Kuo, P.-L. Heparin-functionalized chitosan–alginate scaffolds for controlled release of growth factor. Int. J. Pharm. 2009, 376, 69–75. [Google Scholar] [CrossRef]
- Rodriguez-Jasso, R.M.; Mussatto, S.I.; Pastrana, L.; Aguilar, C.N.; Teixeira, J.A. Microwave-assisted extraction of sulfated polysaccharides (fucoidan) from brown seaweed. Carbohydr. Polym. 2011, 86, 1137–1144. [Google Scholar] [CrossRef]
- Levene, H.B.; Lhommeau, C.M.; Kohn, J.B. Porous polymer scaffolds for tissue engineering. U.S. Patent 6103255 A, 15 August 2000. [Google Scholar]
- Kim, S.; Pyo, H.-B.; Ko, S.H.; Ah, C.S.; Kim, A.; Kim, W.-J. Fabrication of anionic sulfate-functionalized nanoparticles as an immunosensor by protein immobilization. Langmuir 2010, 26, 7355–7364. [Google Scholar] [CrossRef]
- Fisher, J.; Reddi, A. Functional tissue engineering of bone: Signals and scaffolds. In Topics inTissue Engineering; Ashammakhi, N., Ferretti, P., Eds.; University of Oulu: Oulu, Finland, 2003. [Google Scholar]
- Venkatesan, J.; Qian, Z.-J.; Ryu, B.; Ashok Kumar, N.; Kim, S.-K. Preparation and characterization of carbon nanotube-grafted-chitosan—Natural hydroxyapatite composite for bone tissue engineering. Carbohydr. Polym. 2011, 83, 569–577. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Venkatesan, J.; Bhatnagar, I.; Kim, S.-K. Chitosan-Alginate Biocomposite Containing Fucoidan for Bone Tissue Engineering. Mar. Drugs 2014, 12, 300-316. https://doi.org/10.3390/md12010300
Venkatesan J, Bhatnagar I, Kim S-K. Chitosan-Alginate Biocomposite Containing Fucoidan for Bone Tissue Engineering. Marine Drugs. 2014; 12(1):300-316. https://doi.org/10.3390/md12010300
Chicago/Turabian StyleVenkatesan, Jayachandran, Ira Bhatnagar, and Se-Kwon Kim. 2014. "Chitosan-Alginate Biocomposite Containing Fucoidan for Bone Tissue Engineering" Marine Drugs 12, no. 1: 300-316. https://doi.org/10.3390/md12010300
APA StyleVenkatesan, J., Bhatnagar, I., & Kim, S.-K. (2014). Chitosan-Alginate Biocomposite Containing Fucoidan for Bone Tissue Engineering. Marine Drugs, 12(1), 300-316. https://doi.org/10.3390/md12010300




