The Marine Fungal Metabolite, AD0157, Inhibits Angiogenesis by Targeting the Akt Signaling Pathway
Abstract
:1. Introduction
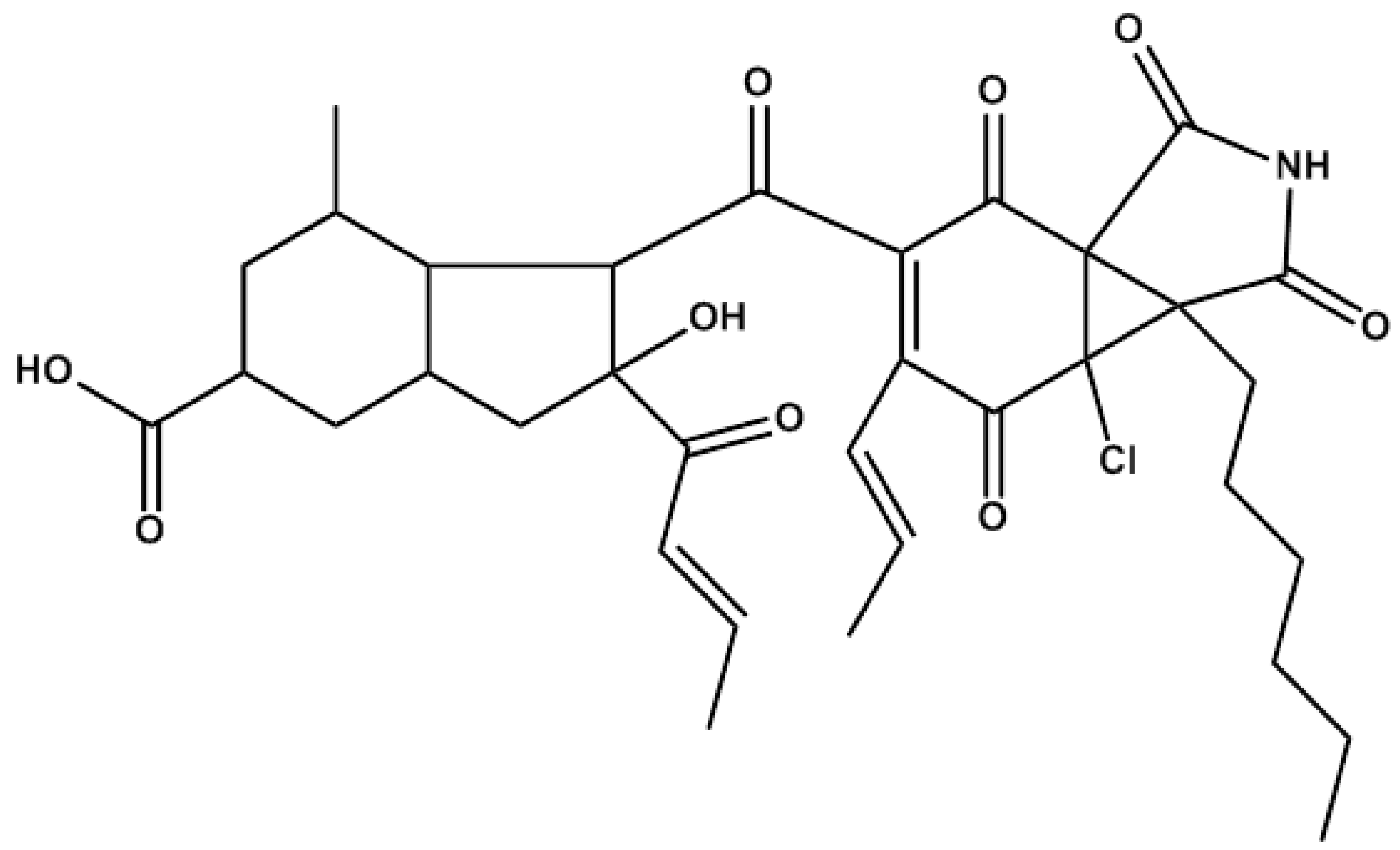
2. Results and Discussion
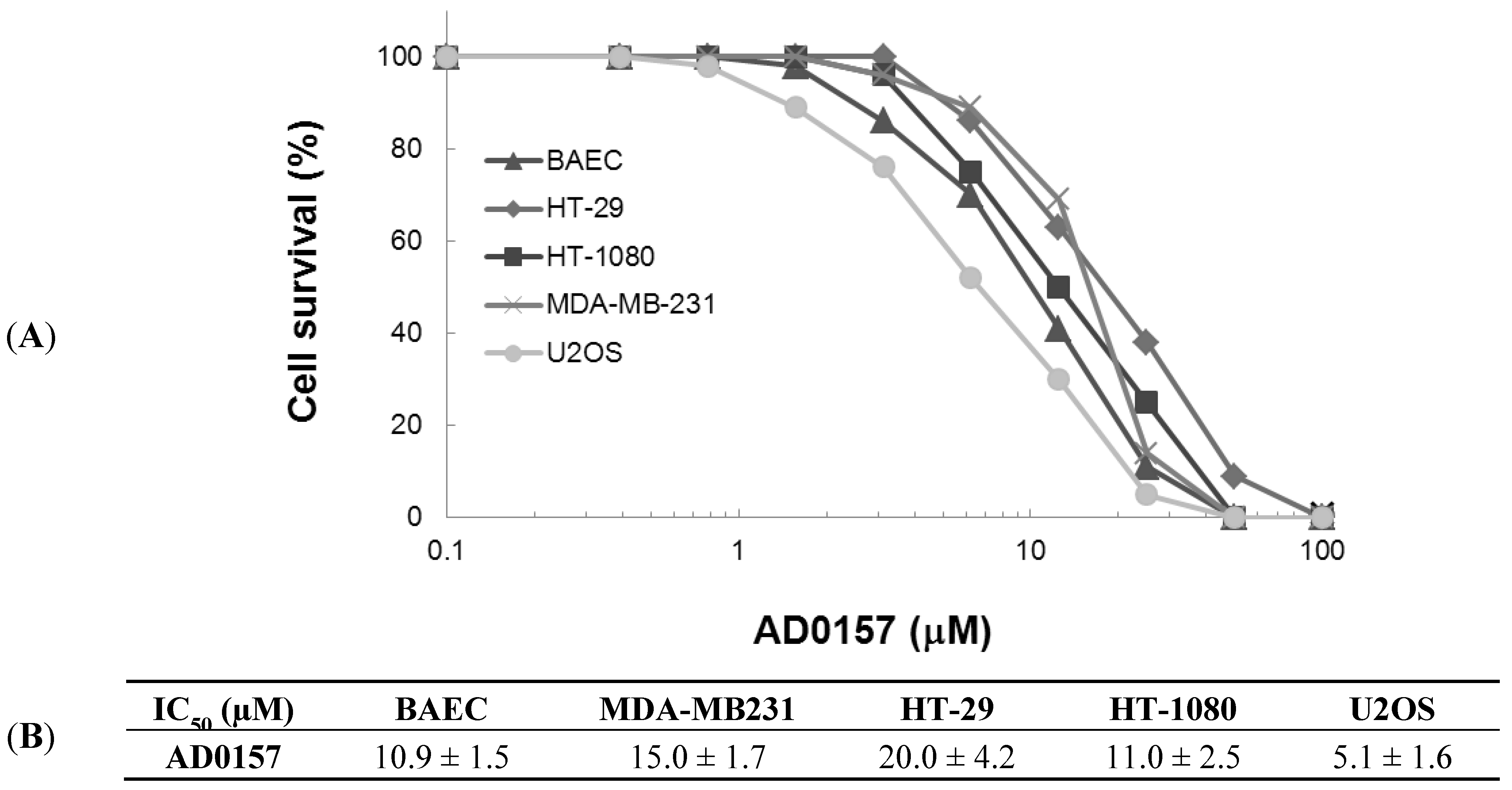
2.1. AD0157 Inhibits the Growth of Endothelial and Tumor Cells
2.2. AD0157 Inhibits Capillary Tube Formation by Endothelial Cells
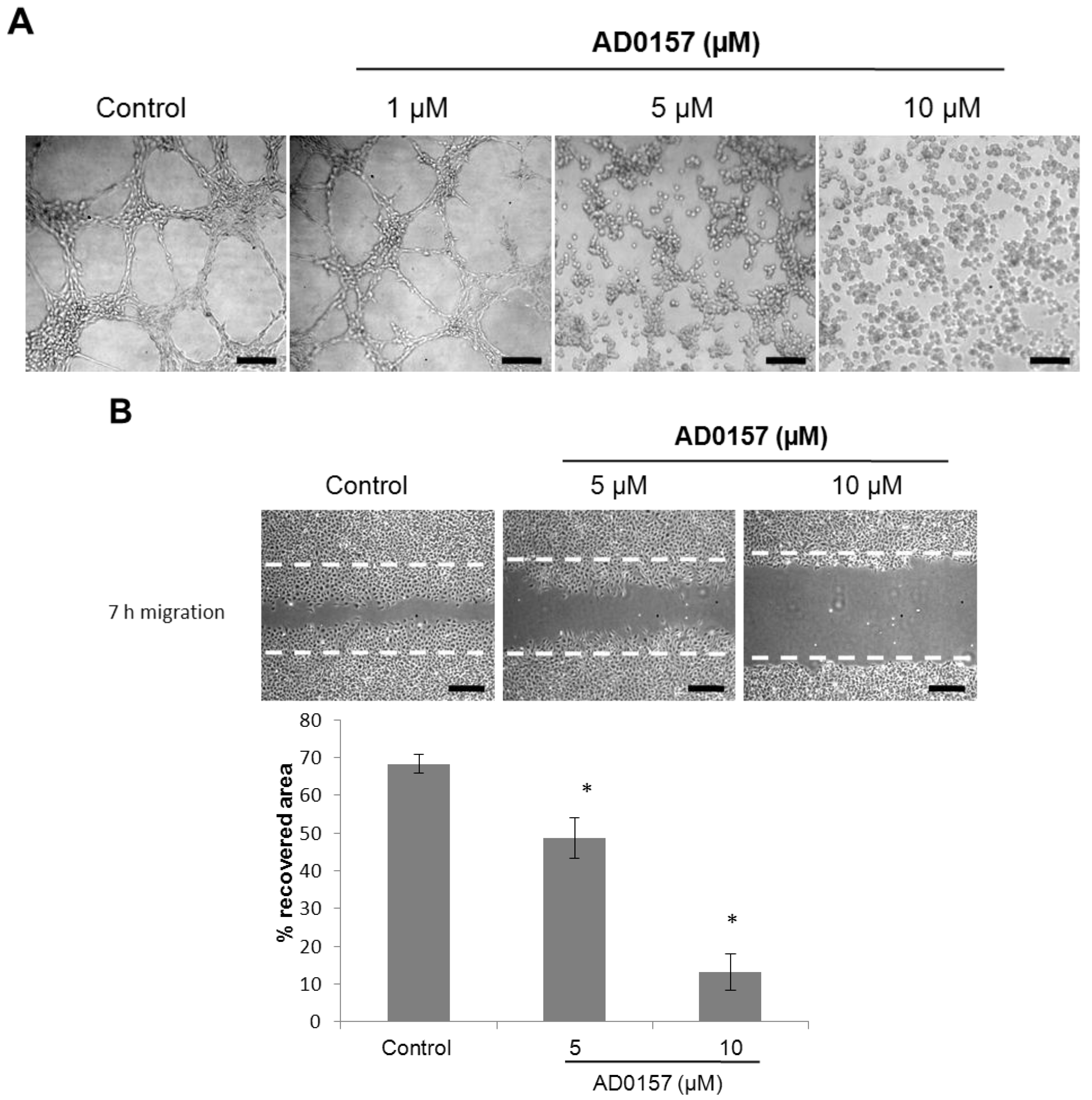
2.3. AD0157 Inhibits the Migratory Capacity of Endothelial Cells
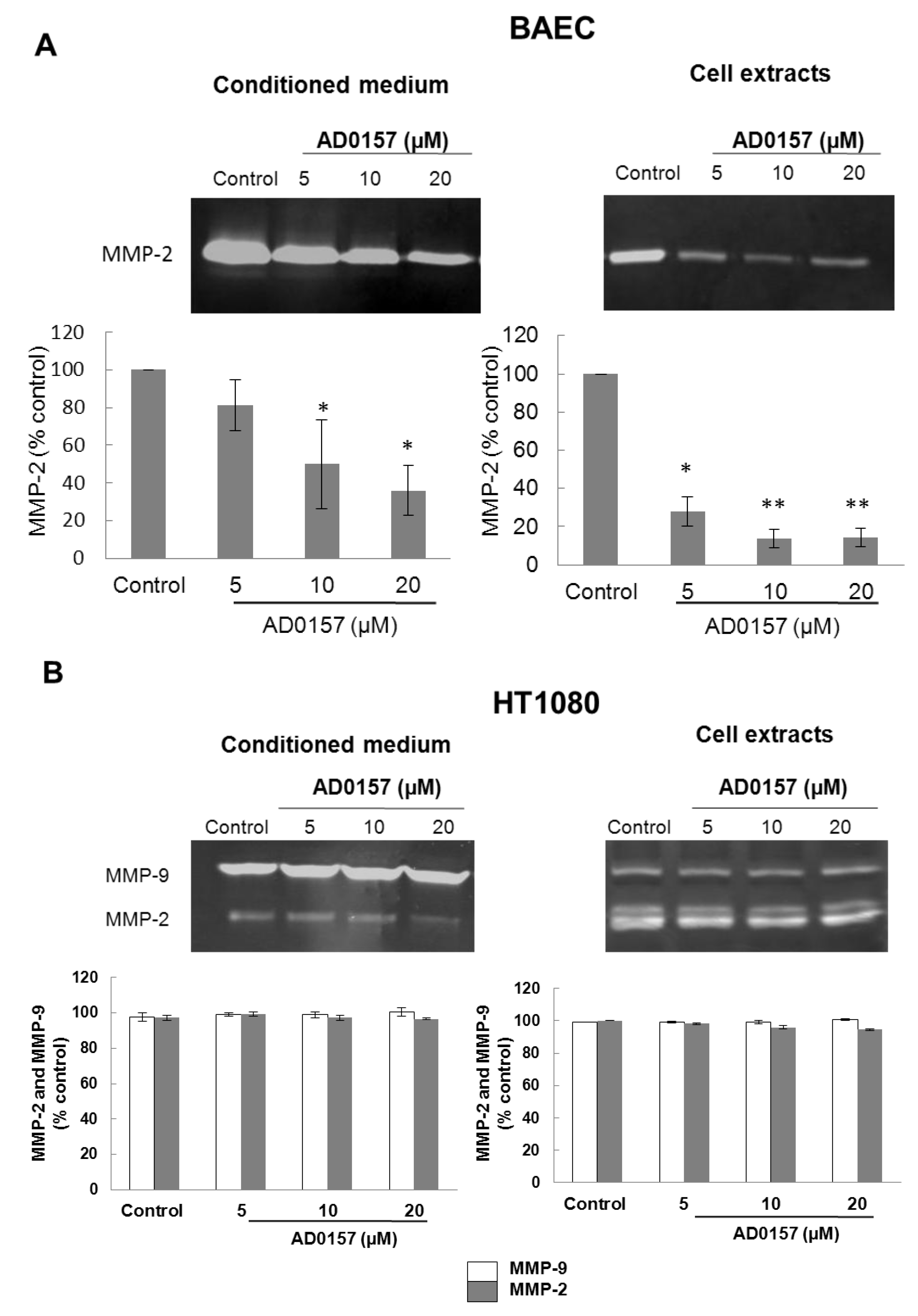
2.4. AD0157 Inhibits the Extracellular Matrix Degrading Potential of Endothelial Cells
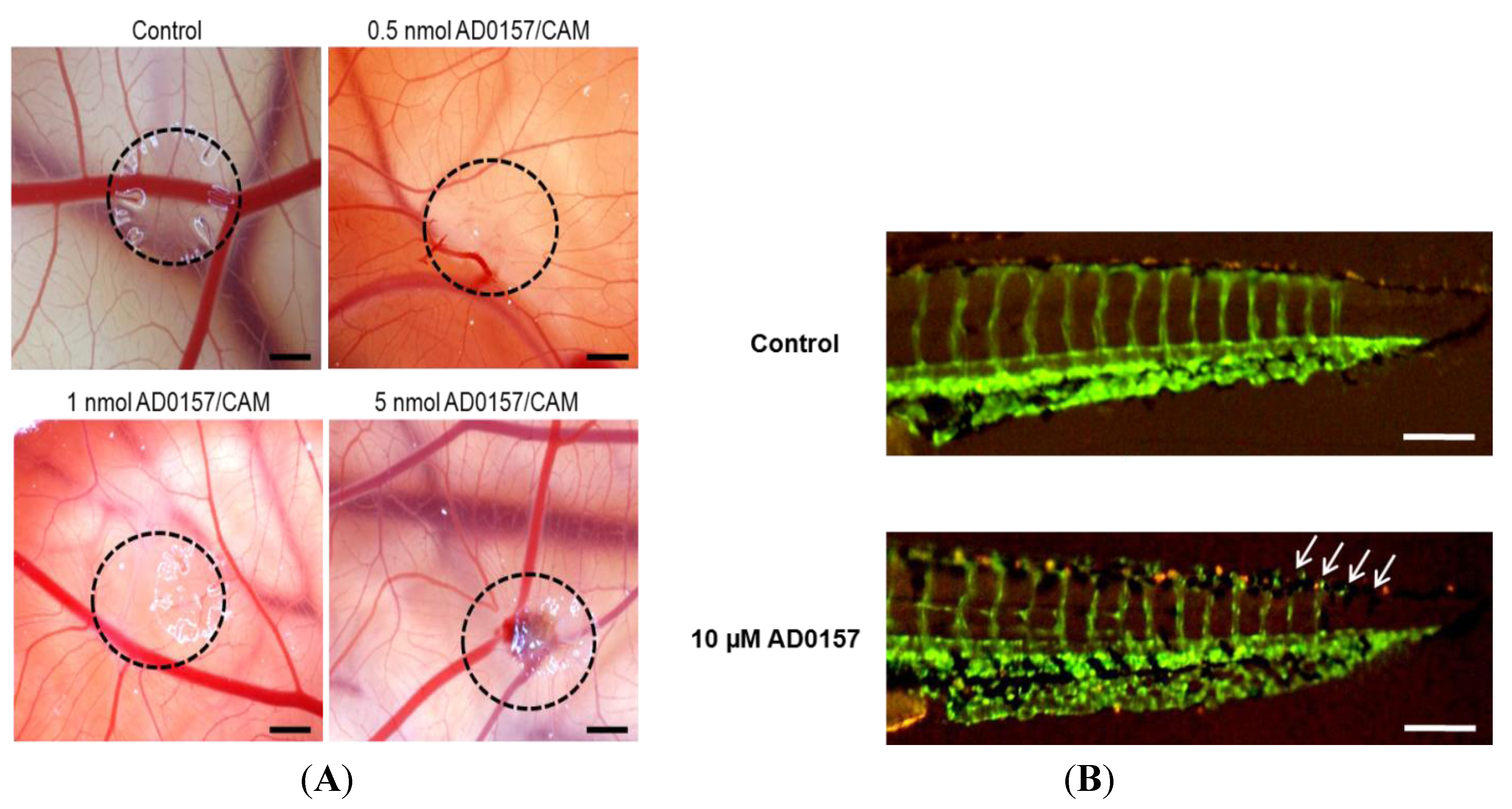
2.5. AD0157 Inhibits Angiogenesis in Vivo
| CAM assay | AD0157 (nmol/CAM) | Positive/Total | % inhibition |
| 0.1 | 1/5 | 20 | |
| 0.5 | 5/6 | 83 | |
| 1 | 6/6 | 100 | |
| 5 | 6/6 | 100 | |
| Zebrafish embryo assay | AD0157 (µM) | Positive/Total | % inhibition |
| 5 | 5/20 | 25 | |
| 10 | 12/25 | 48 | |
| 25 | 17/20 | 85 |
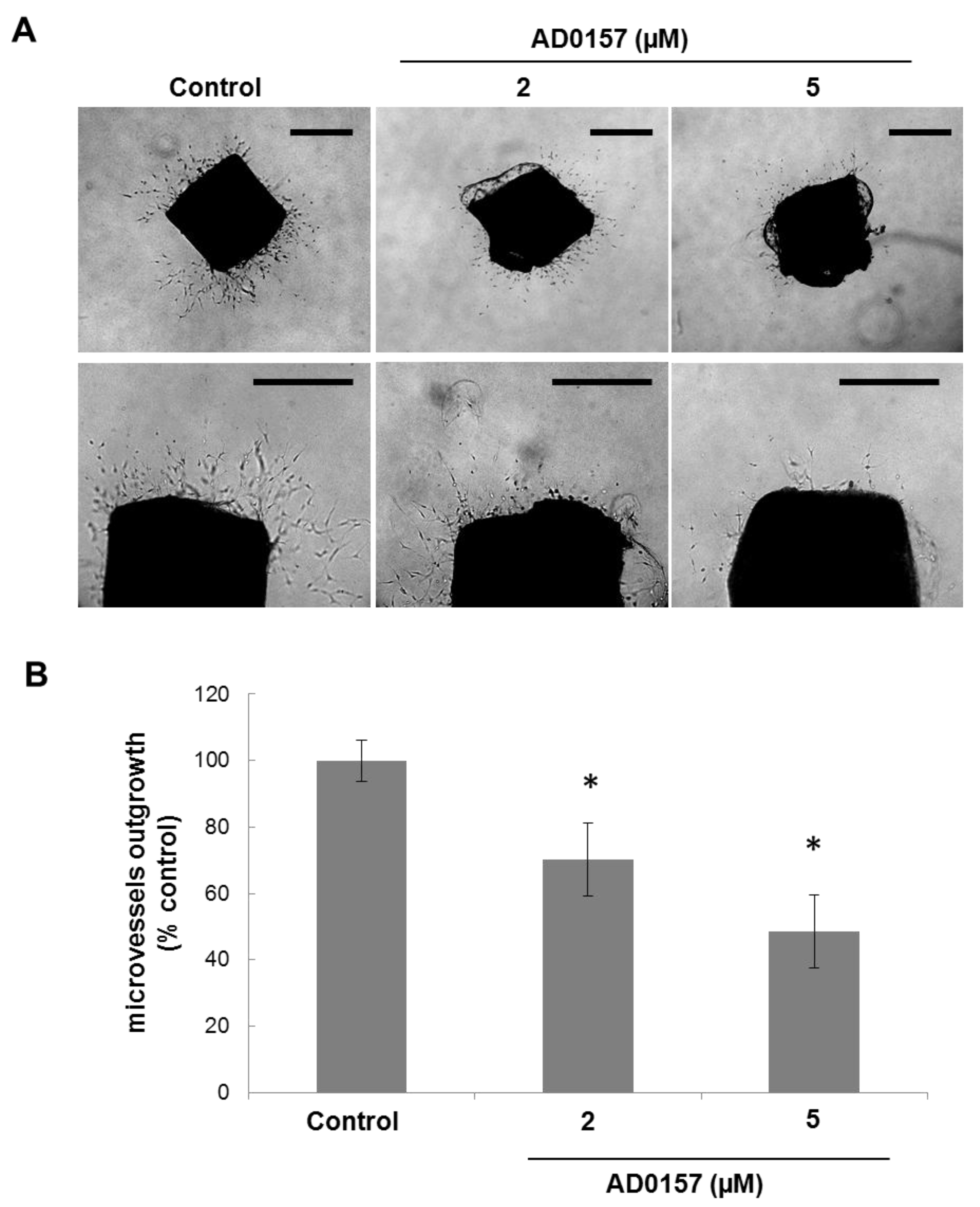
2.6. AD0157 Inhibits Microvessel Outgrowth in Mouse Aortic Rings
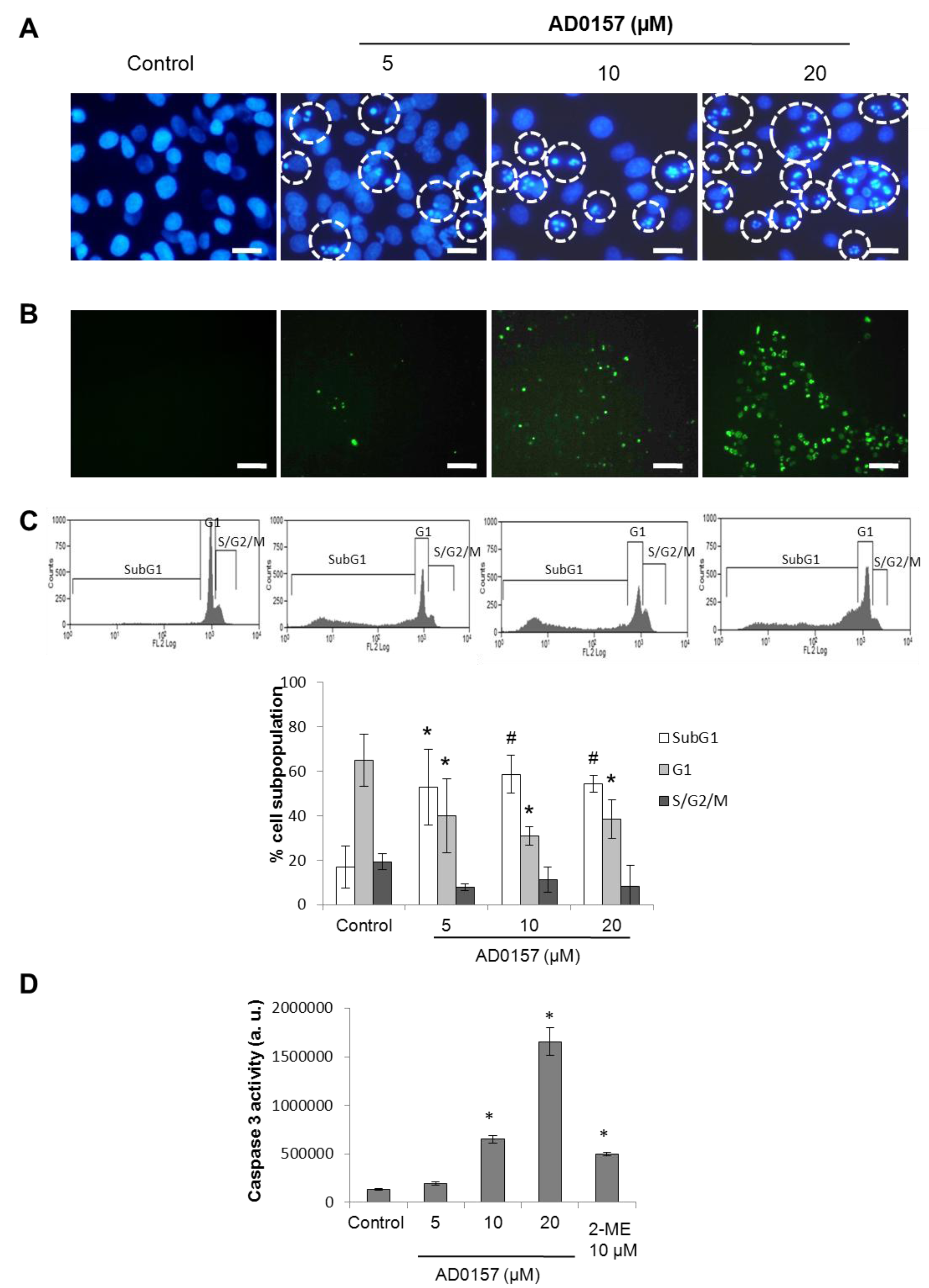
2.7. AD0157 Induces Apoptosis in Endothelial Cells
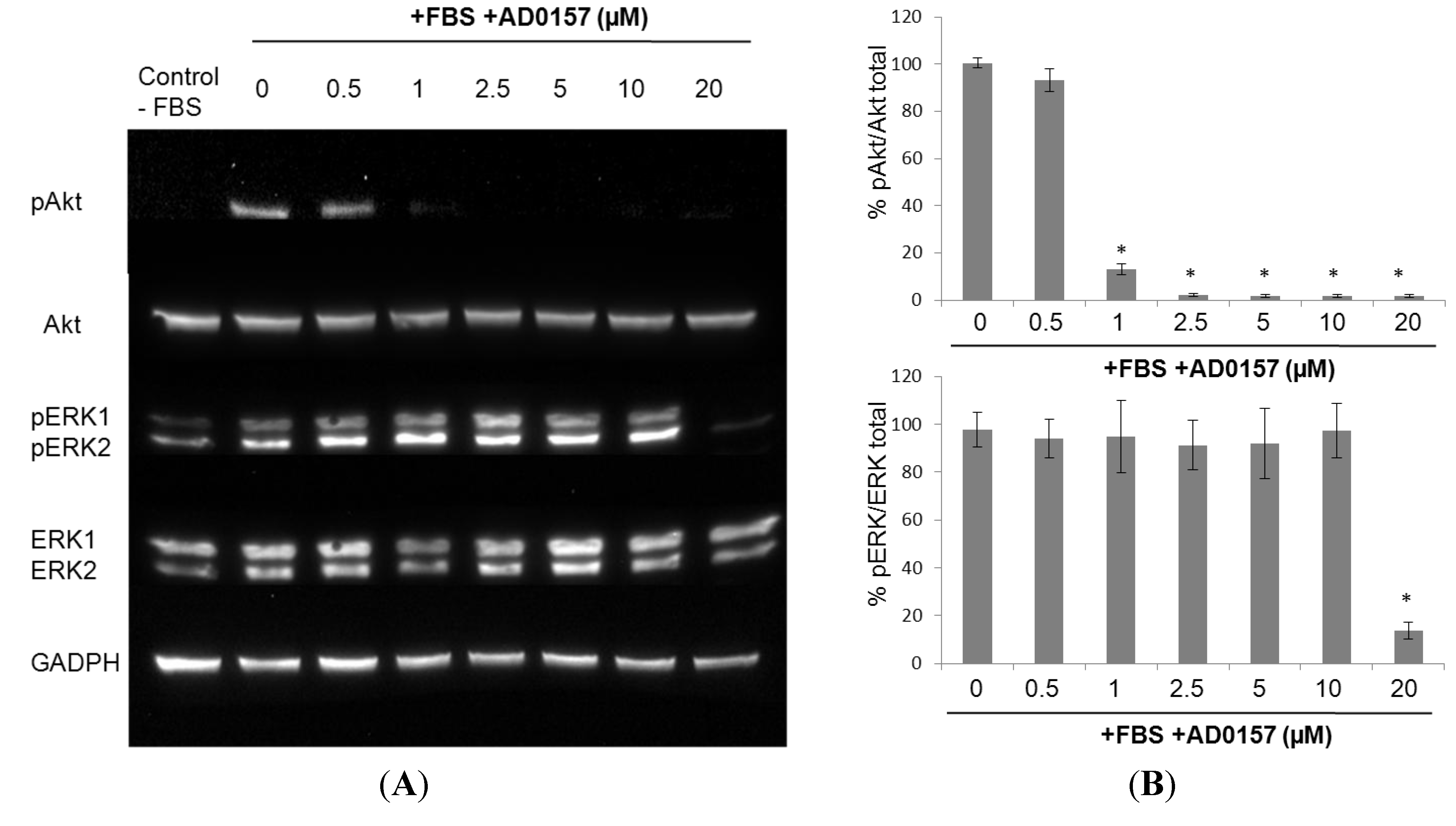
2.8. AD0157 Interference on the ERK1/2 and Akt Pathways
2.9. Global Discussion
3. Experimental Section
3.1. Materials
3.2. Cell Culture
3.3. Cell Growth Assay
3.4. Endothelial Cell Differentiation Assay: Tube Formation on Matrigel
3.5. Endothelial Cell Migration Assay
3.6. Gelatinolytic Activity
3.7. Chorioallantoic Membrane (CAM) Assay
3.8. Zebrafish Embryo Assay
3.9. Mouse Aortic Ring Assay
3.10. Apoptosis and Cell Cycle Assays
3.11. Western Blot Analysis
3.12. Statistical Analysis
3.13. Animal Testing Regulations
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef]
- Chung, A.S.; Ferrara, N. Developmental and pathological angiogenesis. Annu. Rev. Cell. Dev. Biol. 2011, 27, 563–584. [Google Scholar] [CrossRef]
- Carmeliet, P. Angiogenesis in life, disease and medicine. Nature 2005, 438, 932–936. [Google Scholar] [CrossRef]
- Quesada, A.R.; Medina, M.Á.; Muñoz-Chápuli, R.; Ponce, Á.L. Do not say ever never more: The ins and outs of antiangiogenic therapies. Curr. Pharm. Des. 2010, 1, 3932–3957. [Google Scholar]
- Quesada, A.R.; Muñoz-Chápuli, R.; Medina, M.A. Anti-angiogenic drugs: From bench to clinical trials. Med. Res. Rev. 2006, 26, 483–530. [Google Scholar] [CrossRef]
- Bhatnagar, I.; Kim, S.K. Immense essence of excellence: Marine microbial bioactive compounds. Mar. Drugs 2010, 8, 2673–2701. [Google Scholar] [CrossRef]
- Imhoff, J.F.; Labes, A.; Wiese, J. Bio-mining the microbial treasures of the ocean: New natural products. Biotechnol. Adv. 2011, 29, 468–482. [Google Scholar] [CrossRef]
- Rodríguez-Nieto, S.; González-Iriarte, M.; Carmona, R.; Muñoz-Chápuli, R.; Medina, M.A.; Quesada, A.R. Antiangiogenic activity of aeroplysinin-1, a brominated compound isolated from a marine sponge. FASEB J. 2002, 16, 261–263. [Google Scholar]
- Castro, M.E.; González-Iriarte, M.; Barrero, A.F.; Salvador-Tormo, N.; Muñoz-Chápuli, R.; Medina, M.A.; Quesada, A.R. Study of puupehenone and related compounds as inhibitors of angiogenesis. Int. J. Cancer 2004, 110, 31–38. [Google Scholar] [CrossRef]
- Martínez-Poveda, B.; Rodríguez-Nieto, S.; García-Caballero, M.; Medina, M.Á.; Quesada, A.R. The antiangiogenic compound aeroplysinin-1 induces apoptosis in endothelial cells by activating the mitochondrial pathway. Mar. Drugs 2012, 10, 2033–2046. [Google Scholar] [CrossRef]
- García-Caballero, M.; Marí-Beffa, M.; Cañedo, L.; Medina, M.Á.; Quesada, A.R. Toluquinol, a marine fungus metabolite, is a new angiosuppresor that interferes with the Akt pathway. Biochem. Pharmacol. 2013, 85, 1727–1740. [Google Scholar]
- Fernández-Medarde, A.; Cañedo, L.; Vinuesa, M.A.; Sánchez-López, J.M.; Calvo, B.; Martínez-Insua, M.; Quesada, A.R.; García-Caballero, M.; Medina, M.A. Compounds, Their Preparation And Use in Medicaments. European Patent Application 13382284, 9 July 2013. [Google Scholar]
- Staton, C.A.; Reed, M.W.; Brown, N.J. A critical analysis of current in vitro and in vivo angiogenesis assays. Int. J. Exp. Pathol. 2009, 90, 195–221. [Google Scholar] [CrossRef]
- Lawson, N.D.; Weinstein, B.M. In vivo imaging of embryonic vascular development using transgenic zebrafish. Dev. Biol. 2002, 248, 307–318. [Google Scholar] [CrossRef]
- Nicosia, R.F.; Zorzi, P.; Ligresti, G.; Morishita, A.; Aplin, A.C. Paracrine regulation of angiogenesis by different cell types in the aorta ring model. Int. J. Dev. Biol. 2011, 55, 447–453. [Google Scholar] [CrossRef]
- Muñoz-Chápuli, R.; Quesada, A.R.; Medina, M.Á. Angiogenesis and signal transduction in endothelial cells. Cell. Mol. Life Sci. 2004, 61, 2224–2243. [Google Scholar]
- Fan, T.P.; Yeh, J.C.; Leung, K.W.; Yue, P.Y.; Wong, R.N. Angiogenesis: From plants to blood vessels. Trends Pharmacol. Sci. 2006, 27, 297–309. [Google Scholar] [CrossRef]
- Bhatnagar, I.; Kim, S.K. Marine Antitumor drugs: Status, shortfalls and strategies. Mar. Drugs 2010, 8, 2702–2720. [Google Scholar] [CrossRef]
- Nastrucci, C.; Cesario, A.; Russo, P. Anticancer drug discovery from the marine environment. Recent Pat. Anticancer Drug Discov. 2012, 7, 218–232. [Google Scholar] [CrossRef]
- Blunt, J.W.; Copp, B.R.; Munro, M.H.; Northcote, P.T.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2011, 28, 196–268. [Google Scholar] [CrossRef]
- Wang, Y.Q.; Miao, Z.H. Marine-derived angiogenesis inhibitors for cancer therapy. Mar. Drugs 2013, 11, 903–933. [Google Scholar] [CrossRef]
- Kong, D.; Yamori, T.; Kobayashi, M.; Duan, H. Antiproliferative and antiangiogenic activities of smenospongine, a marine sponge sesquiterpene aminoquinone. Mar. Drugs 2011, 9, 154–161. [Google Scholar] [CrossRef]
- Kim, G.D.; Cheong, O.J.; Bae, S.Y.; Shin, S.J.; Lee, S.K. 6″-Debromohamacanthin A, a Bis (Indole) alkaloid, inhibits angiogenesis by targeting the VEGFR2-mediated PI3K/AKT/mTOR signaling pathways. Mar. Drugs 2013, 11, 1087–1103. [Google Scholar] [CrossRef]
- Martínez-Poveda, B.; Quesada, A.R.; Medina, M.A. Hypericin in the dark inhibits key steps of angiogenesis in vitro. Eur. J. Pharmacol. 2005, 516, 97–103. [Google Scholar] [CrossRef]
- López-Jiménez, A.; García-Caballero, M.; Medina, M.A.; Quesada, A.R. Anti-angiogenic properties of carnosol and carnosic acid, two major dietary compounds from rosemary. Eur. J. Nutr. 2013, 52, 85–95. [Google Scholar] [CrossRef]
- Kim, Y.H.; Shin, E.K.; Kim, D.H.; Lee, H.H.; Park, J.H.; Kim, J.K. Antiangiogenic effect of licochalcone A. Biochem. Pharmacol. 2010, 80, 1152–1159. [Google Scholar]
- García-Caballero, M.; Marí-Beffa, M.; Medina, M.A.; Quesada, A.R. Dimethylfumarate inhibits angiogenesis in vitro and in vivo: A possible role for its antipsoriatic effect? J. Investig. Dermatol. 2011, 131, 1347–1355. [Google Scholar] [CrossRef]
- Fortes, C.; García-Vilas, J.A.; Quesada, A.R.; Medina, M.A. Evaluation of the anti-angiogenic potential of hydroxytyrosol and tyrosol, two bio-active phenolic compounds of extra virgin olive oil, in endothelial cell cultures. Food Chem. 2012, 134, 134–140. [Google Scholar] [CrossRef]
- Igura, K.; Ohta, T.; Kuroda, Y.; Kaji, K. Resveratrol and quercetin inhibit angiogenesis in vitro. Cancer Lett. 2001, 171, 11–16. [Google Scholar] [CrossRef]
- Cárdenas, C.; Quesada, A.R.; Medina, M.A. Evaluation of the anti-angiogenic effect of aloe-emodin. Cell. Mol. Life Sci. 2006, 63, 3083–3089. [Google Scholar] [CrossRef]
- Rodriguez-Nieto, S.; Medina, M.A.; Quesada, A.R. A re-evaluation of fumagillin selectivity towards endothelial cells. Anticancer Res. 2001, 21, 3457–3460. [Google Scholar]
- Nguyen, M.; Arkell, J.; Jackson, C.J. Human endothelial gelatinases and angiogenesis. Int. J. Biochem. Cell. Biol. 2001, 33, 960–970. [Google Scholar] [CrossRef]
- Fang, J.; Shing, Y.; Wiederschain, D.; Yan, L.; Butterfield, C.; Jackson, G.; Harper, J.; Tamvakopoulos, G.; Mosess, M.A. Matrix metalloproteinase-2 is required for the switch to the angiogenic phenotype in a tumor model. Proc. Natl. Acad. Sci. USA 2000, 97, 3884–3889. [Google Scholar] [CrossRef]
- Itoh, T.; Tanioka, M.; Yoshida, H.; Yoshioka, T.; Nishimoto, H.; Itohara, S. Reduced angiogenesis and tumor progression in gelatinase A-deficient mice. Cancer Res. 1998, 58, 1048–1051. [Google Scholar]
- Masson, V.E.; Devy, L.; Grignet-Debrus, C.; Bernt, S.; Bajou, K.; Blacher, S.; Roland, G.; Chang, Y.; Fong, T.; Carmeliet, P.; et al. Mouse aortic ring assay: A new approach of the molecular genetics of angiogenesis. Biol. Proced. Online 2002, 4, 24–31. [Google Scholar] [CrossRef]
- Yi, C.H.; Yuan, J. The Jekyll and Hyde functions of caspases. Dev. Cell 2009, 16, 21–34. [Google Scholar] [CrossRef]
- Cheng, J.Q.; Lindsley, C.W.; Cheng, G.Z.; Yang, H.; Nicosia, S.V. The Akt/PKB pathway: Molecular target for cancer drug discovery. Oncogene 2005, 24, 7482–7492. [Google Scholar] [CrossRef]
- Jiang, B.H.; Liu, L.Z. AKT signaling in regulating angiogenesis. Curr. Cancer Drug Targets 2008, 8, 19–26. [Google Scholar] [CrossRef]
- Kim, E.K.; Choi, E.J. Pathological roles of MAPK signaling pathways in human diseases. Biochim. Biophys. Acta 2010, 1802, 396–405. [Google Scholar]
- Chakraborti, S.; Mandal, M.; Das, S.; Mandal, A.; Chakraborti, T. Regulation of matrix metalloproteinases: An overview. Mol. Cell. Biochem. 2003, 253, 269–285. [Google Scholar] [CrossRef]
- Gospodarowicz, D.; Moran, J.S. Mitogenic effect of fibroblast growth factor on early passage cultures of human and murine fibroblasts. J. Cell. Biol. 1975, 66, 451–457. [Google Scholar] [CrossRef]
Supplementary Files
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
García-Caballero, M.; Cañedo, L.; Fernández-Medarde, A.; Medina, M.Á.; Quesada, A.R. The Marine Fungal Metabolite, AD0157, Inhibits Angiogenesis by Targeting the Akt Signaling Pathway. Mar. Drugs 2014, 12, 279-299. https://doi.org/10.3390/md12010279
García-Caballero M, Cañedo L, Fernández-Medarde A, Medina MÁ, Quesada AR. The Marine Fungal Metabolite, AD0157, Inhibits Angiogenesis by Targeting the Akt Signaling Pathway. Marine Drugs. 2014; 12(1):279-299. https://doi.org/10.3390/md12010279
Chicago/Turabian StyleGarcía-Caballero, Melissa, Librada Cañedo, Antonio Fernández-Medarde, Miguel Ángel Medina, and Ana R. Quesada. 2014. "The Marine Fungal Metabolite, AD0157, Inhibits Angiogenesis by Targeting the Akt Signaling Pathway" Marine Drugs 12, no. 1: 279-299. https://doi.org/10.3390/md12010279
APA StyleGarcía-Caballero, M., Cañedo, L., Fernández-Medarde, A., Medina, M. Á., & Quesada, A. R. (2014). The Marine Fungal Metabolite, AD0157, Inhibits Angiogenesis by Targeting the Akt Signaling Pathway. Marine Drugs, 12(1), 279-299. https://doi.org/10.3390/md12010279








