Structure of Fucoidan from Brown Seaweed Turbinaria ornata as Studied by Electrospray Ionization Mass Spectrometry (ESIMS) and Small Angle X-ray Scattering (SAXS) Techniques
Abstract
:1. Introduction
2. Results and Discussion
| Yield (% dried seaweed) | Sulfate content (% mass) | Uronic acid (% mass) | Neutral sugar composition (% mol) | ||||
|---|---|---|---|---|---|---|---|
| Fuc | Gal | Xyl | Man | Glu | |||
| 2.215 | 25.6 | 7.8 | 30.3 | 9.0 | trace | trace | trace |
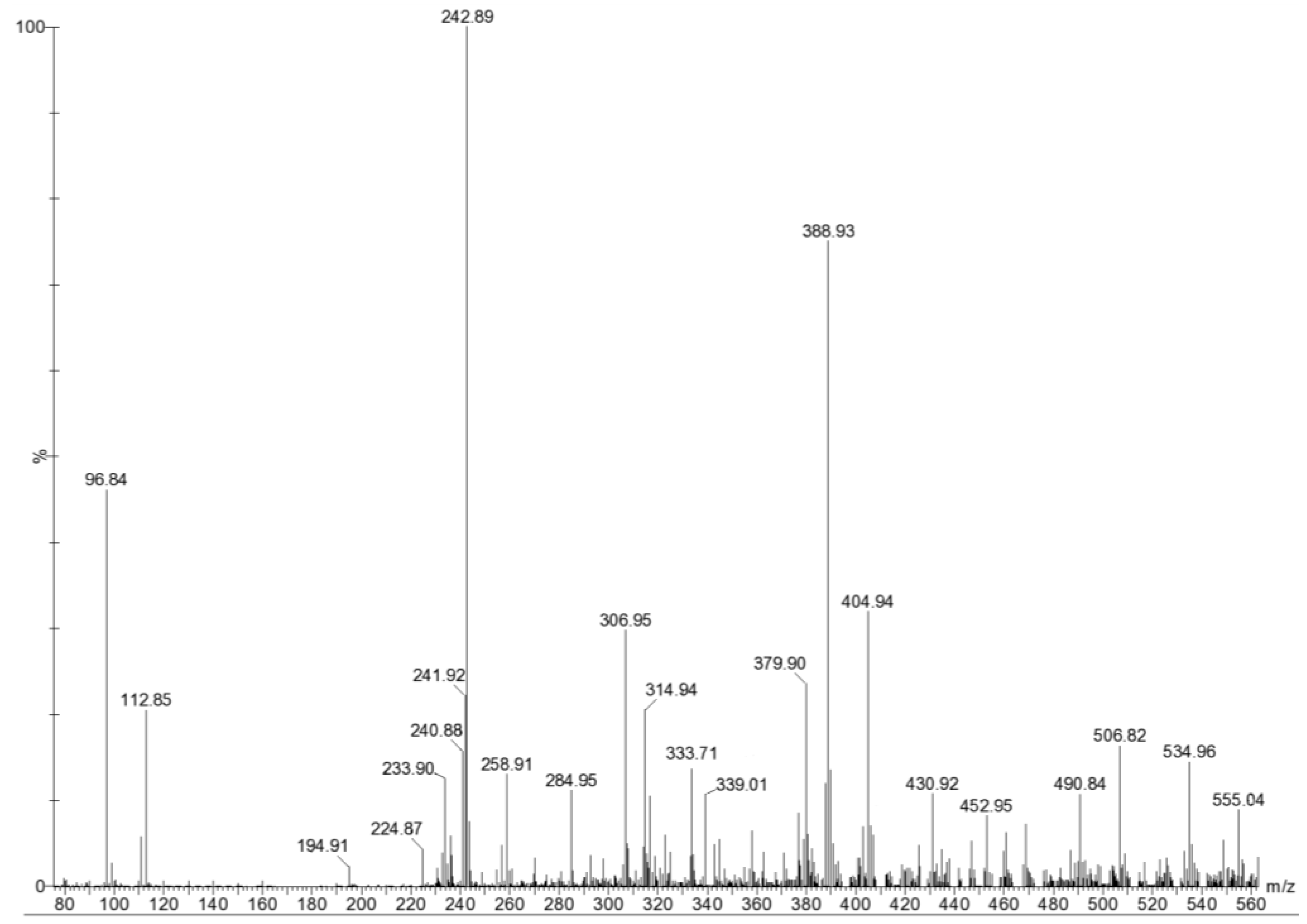
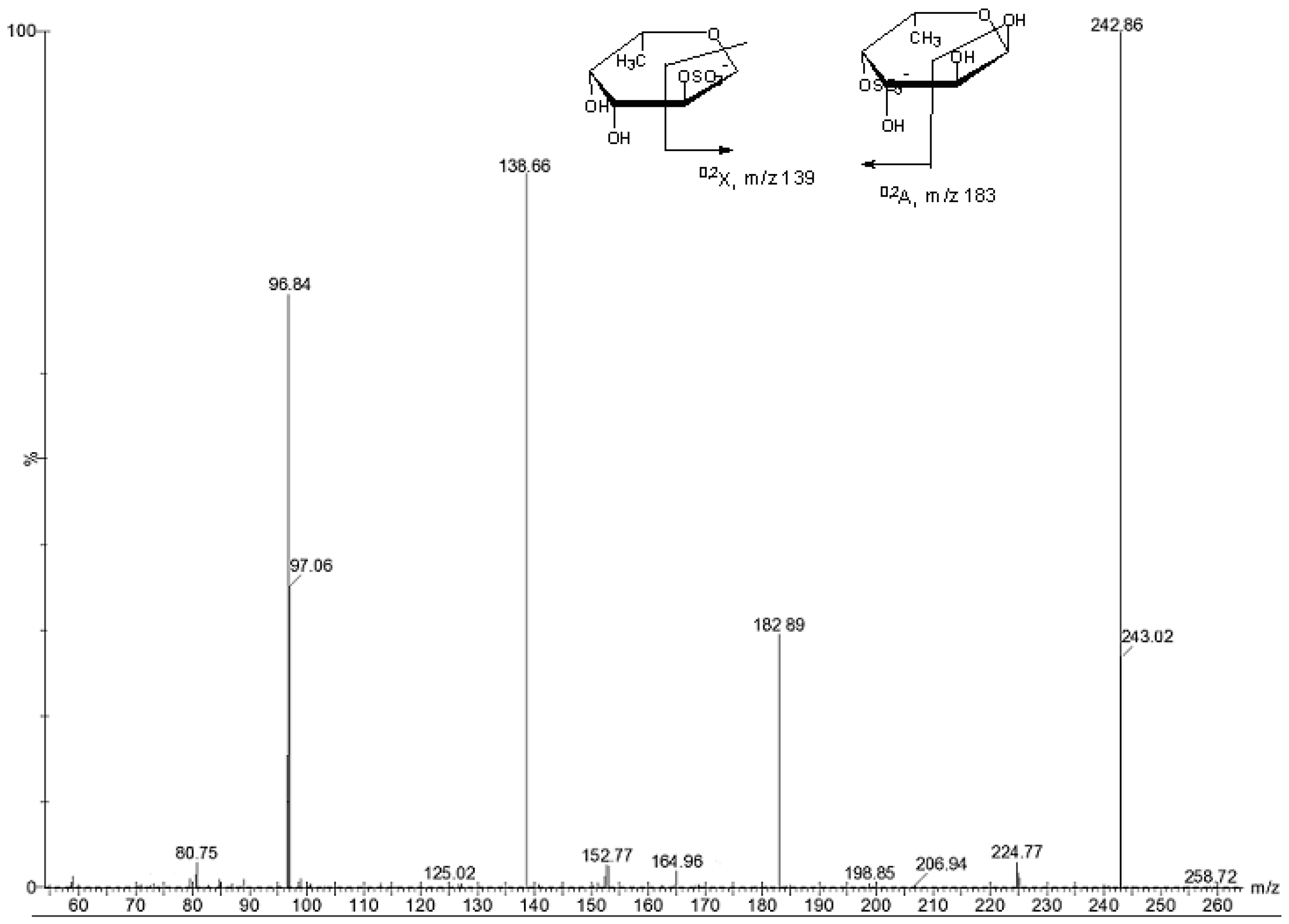
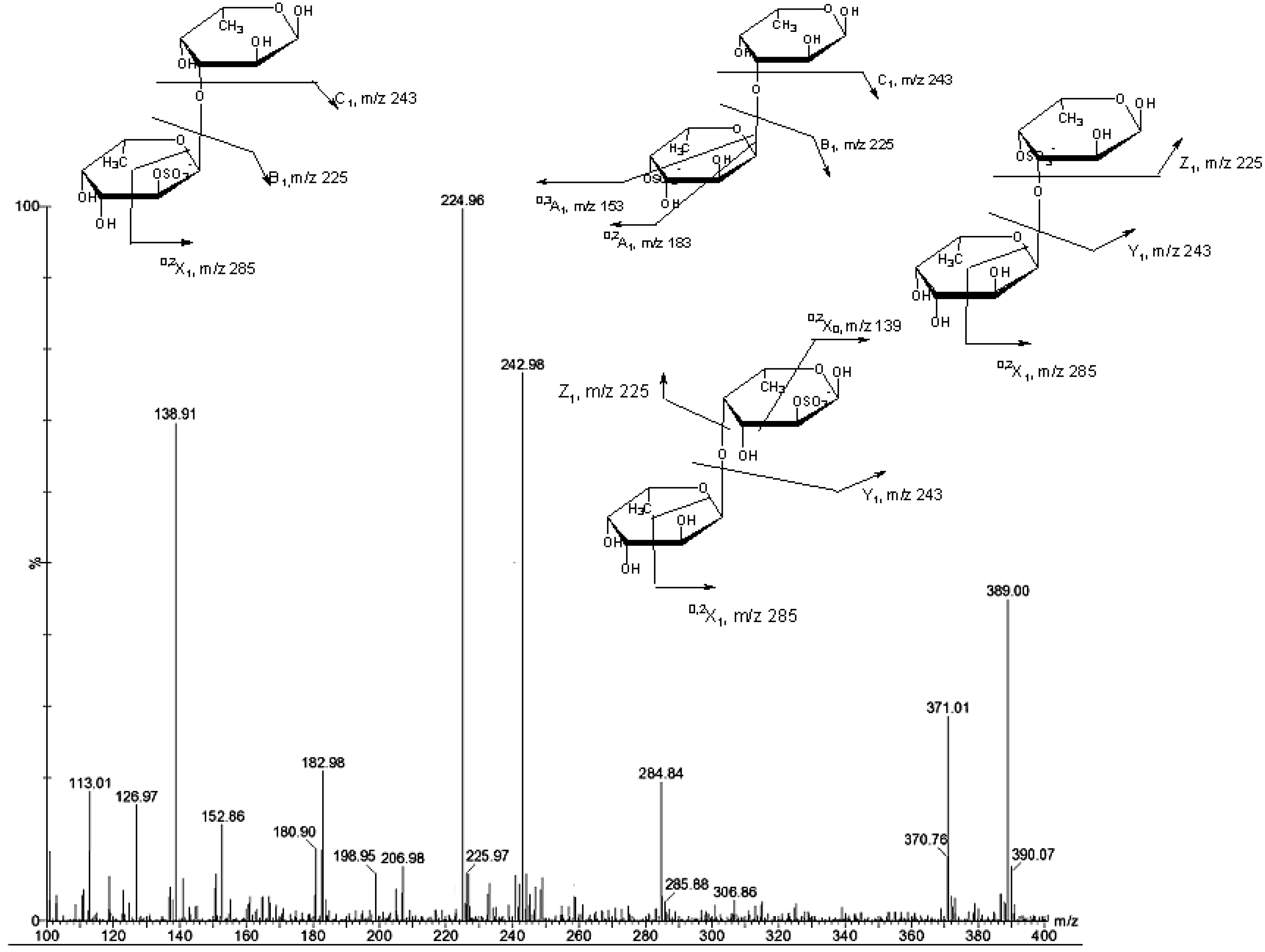
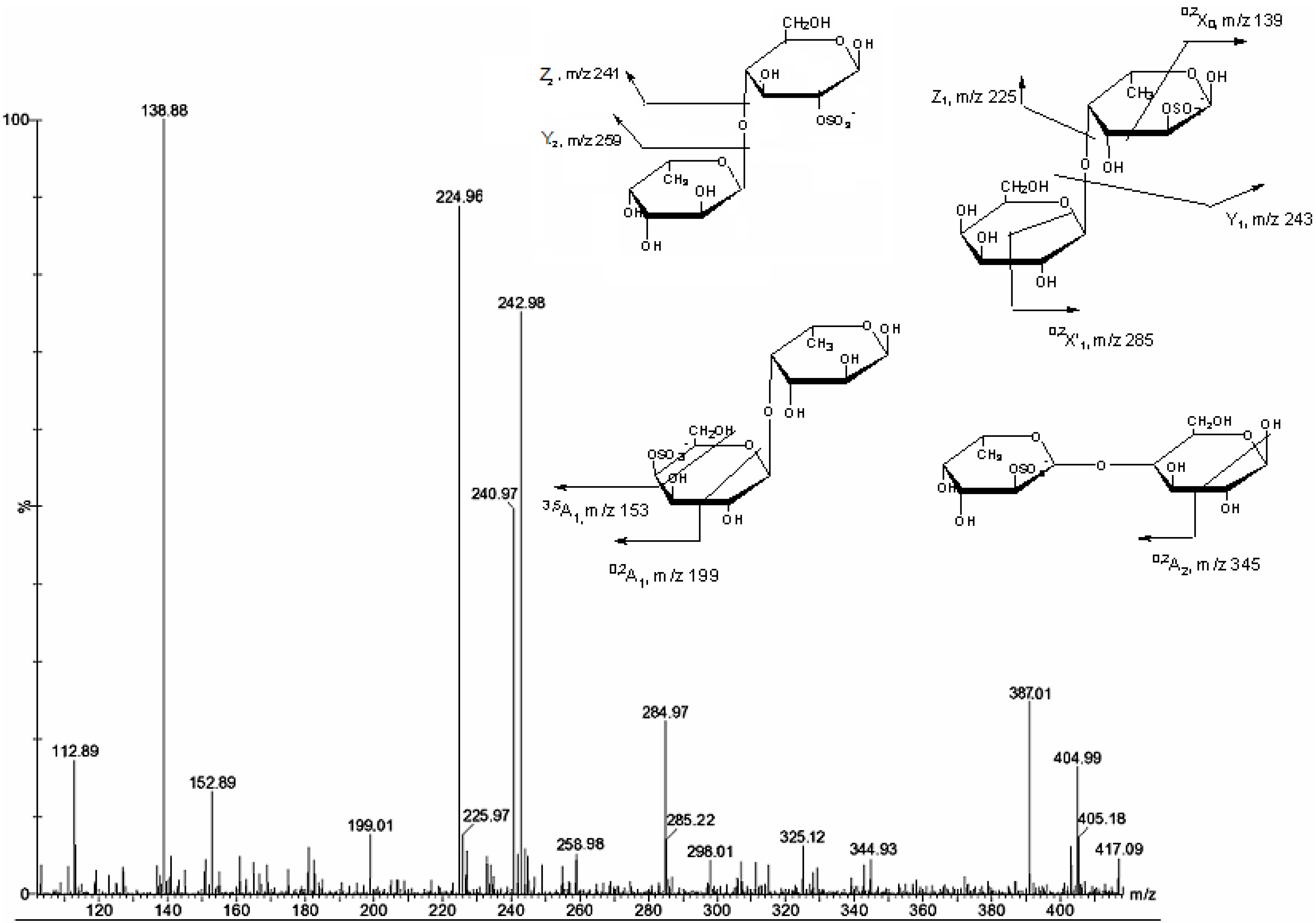
| m/z | composition | Proposed structure |
|---|---|---|
| 96 | HSO4− | |
| 225 | [FucSO3Na − Na − H2O]− | |
| 234 | [Fuc2(SO3Na)2 − 2Na]2− | |
| 243 | [FucSO3Na − Na]− | Fuc2S, Fuc4S |
| 259 | [GalSO3Na − Na]− | |
| 285 | [Fuc2(SO3Na)3 − 3Na + Na]2− | |
| 307 | [Fuc3(SO3Na)2 − 2Na]2− | |
| 371 | [Fuc2SO3Na − Na − H2O]− | |
| 333 | [Fuc4(SO3Na)5 − 3Na]3− | |
| 339 | [FucGlcA − Na]− | |
| 380 | [Fuc4(SO3Na)2 − 2Na]2− | |
| 389 | [Fuc2SO3Na − Na]− | Fuc2S-(1→3)Fuc Fuc4S-(1→3)Fuc Fuc(1→3)-Fuc4S Fuc(1→4)-Fuc2S |
| 405 | [FucGal(SO3Na) − Na]− | Gal(1→4)Fuc2S Fuc(1→4)Gal2S Fuc2S(1→4)Gal Gal4S(1→4)Fuc |
| 431 | [Fuc4(SO3Na)3 − 3Na + Na]2− | |
| 453 | [Fuc5(SO3Na)2 − 2Na]2− | |
| 491 | [Fuc2(SO3Na)2 − Na]− | Fuc2S(1→3)Fuc4S Fuc4S(1→3)Fuc2S |
| 507 | [FucGal(SO3Na)2 − Na]− | |
| 535 | [Fuc3SO3Na − Na]− |

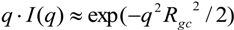
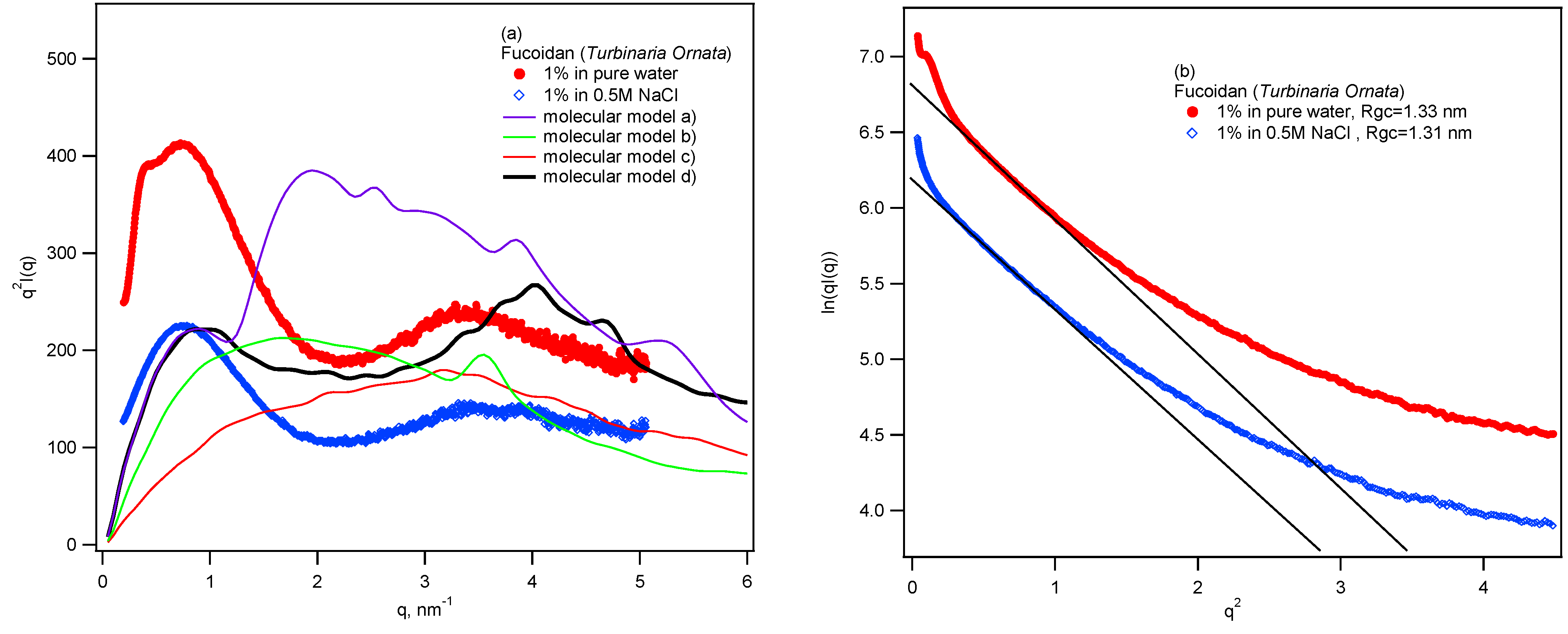
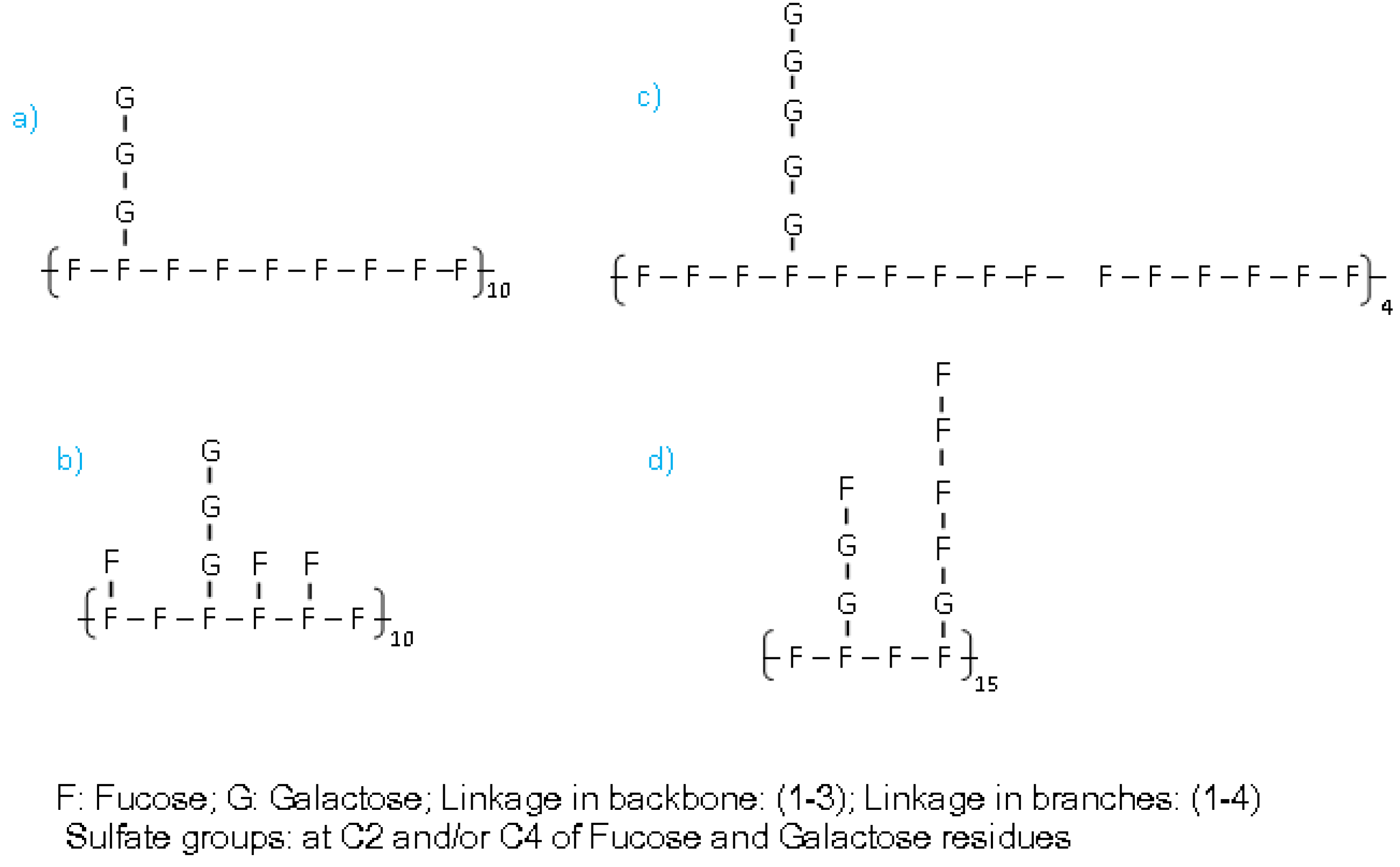

3. Experimental Section
3.1. Seaweed Collection
3.2. Extraction and Purification of Fucoidan
3.3. Chemical Analysis
3.4. Partial Acid Hydrolysis
3.5. ESI-MS
3.6. SAXS Measurement
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Li, B.; Lu, F.; Wei, F.; Zhao, R. Fucoidan: Structure and bioactivity. Molecules 2008, 13, 1671–1695. [Google Scholar] [CrossRef]
- Chevolot, L.; Mulloy, B.; Ratiskol, J.; Foucault, A.; Colliec-Jouault, S. A disaccharide repeat unit is the major structure in fucoidans from two species of brown algae. Carbohydr. Res. 2001, 330, 529–535. [Google Scholar] [CrossRef]
- Cumashi, A.; Ushakova, N.A.; Preobrazhenskaya, M.E.; D’Incecco, A.; Piccoli, A.; Totani, L.; Tinari, N.; Morozevich, G.E.; Berman, A.E.; Bilan, M.I.; et al. A comparative study of the anti-inflammatory, anticoagulant, antiangiogenic, and antiadhesive activities of nine different fucoidans from brown seaweeds. Glycobiology 2007, 17, 541–552. [Google Scholar]
- Kusaykin, M.; Bakunina, I.; Sova, V.; Ermakova, S.; Kuznetsova, T.; Besednova, N.; Zaporozhets, T.; Zvyagintseva, T. Structure, biological activity, and enzymatic transformation of fucoidans from the brown seaweeds. Biotechnol. J. 2008, 3, 904–915. [Google Scholar]
- Lee, J.B.; Hayashi, K.; Hashimoto, M.; Nakano, T.; Hayashi, T. Novel antiviral fucoidan from sporophyll of Undaria pinnatifida (Mekabu). Chem. Pharm. Bull. 2004, 52, 1091–1094. [Google Scholar] [CrossRef]
- Trinchero, J.; Ponce, N.M.A.; Cordoba, O.L.; Flores, M.L.; Pampuro, S.; Stortz, C.A.; Salomon, H.; Turk, G. Antiretroviral activity of fucoidans extracted from the brown seaweed Adenocystis utricularis. Phytother. Res. 2009, 23, 707–712. [Google Scholar] [CrossRef]
- Raghavendran, H.R.; Srinivasan, P.; Rekha, S. Immunomodulatory activity of fucoidan against aspirin-induced gastric mucosal damage in rats. Int. Immunopharmacol. 2011, 11, 157–163. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, Q.; Zhang, Z.; Song, H.; Li, P. Potential antioxidant and anticoagulant capacity oflow molecular weight fucoidan fractions extracted from Laminaria japonica. Int. J. Biol. Macromol. 2010, 46, 6–12. [Google Scholar] [CrossRef]
- Synytsya, A.; Kim, W.J.; Kim, S.M.; Pohl, R.; Synytsya, A.; Kvasnicka, F.; Copíkova, J.; Park, J.I. Structure and antitumour activity of fucoidan isolated from sporophyll of Korean brown seaweed Undaria pinnatifida. Carbohydr. Polym. 2010, 81, 41–48. [Google Scholar] [CrossRef]
- Saitô, H.; Yoshioka, Y.; Yokoi, M.; Yamada, J. Distinct gelation mechanism between linear and branched (1→3)-β-d-glucans as revealed by high-resolution solid-state 13C NMR. Biopolymers 1990, 29, 1689–1698. [Google Scholar] [CrossRef]
- Gao, Y.; Fukuda, A.; Katsuraya, K.; Kaneko, Y.; Mimura, T.; Nakashima, H.; Uryu, T. Synthesis of regioselective substituted curdlan sulfates with medium molecular weights and their specific anti-HIV-1 activities. Macromolecules 1997, 30, 3224–3228. [Google Scholar] [CrossRef]
- Daniel, R.; Chevolot, L.; Carrascal, M.; Tissot, B.; Mourao, P.A.S.; Abian, J. Electrospray ionization mass spectrometry of oligosaccharides derived from fucoidan of Ascophyllum nodosum. Carbohydr. Res. 2007, 342, 826–834. [Google Scholar] [CrossRef]
- Anastyuk, S.D.; Shevchenko, N.M.; Dmitrenok, P.S.; Zvyagintseva, T.N. ESIMS analysis of fucoidan preparations from Costaria costata, extracted from alga at different life-stages. Carbohydr. Polym. 2012, 90, 993–1002. [Google Scholar] [CrossRef]
- Anastyuk, S.D.; Shevchenko, N.M.; Dmitrenok, P.S.; Zvyagintseva, T.N. Anticancer activity in vitro of a fucoidan from the brown alga Fucus evanescens and its low-molecular fragments, structurally characterized by tandem mass-spectrometry. Carbohydr. Polym. 2012, 87, 186–194. [Google Scholar] [CrossRef]
- Glatter, O.; Kratky, O. Small Angle X-Ray Scattering; Academic Press: London, UK, 1982. [Google Scholar]
- Grant, T.D.; Luft, J.R.; Wolfley, J.R.; Tsuruta, H.; Martel, A.; Montelione, G.T.; Snell, E.H. Small angle X-ray scattering as a complementary tool for high-throughput structural studies. Biopolymers 2011, 95, 517–530. [Google Scholar] [CrossRef]
- Chattopadhyay, N.; Ghosh, T.; Sinha, S.; Chattopadhyay, K.; Karmakar, P.; Ray, B. Polysaccharides from Turbinaria conoides: Structural features and antioxidant capacity. Food Chem. 2010, 118, 823–829. [Google Scholar] [CrossRef]
- Huynh, Q.N.; Nguyen, H.D. The Seaweed Resources of Vietnam. In Seaweed Resources of the World,1998; Critchley, A.T., Ohno, M., Eds.; Japan International Cooperation Agency: Yokosuka, Japan, 2006; pp. 62–69. [Google Scholar]
- Tissot, B.; Salpin, J.Y.; Martinez, M.; Gaigeota, M.P.; Daniel, R. Differentiation of the fucoidan sulfated l-fucose isomers constituents by CE-ESIMS and molecular modeling. Carbohydr. Res. 2006, 341, 598–609. [Google Scholar] [CrossRef]
- Anastyuk, S.D.; Shevchenko, N.M.; Nazarenko, E.L.; Dmitrenok, P.S.; Zvyagintseva, T.N. Structural analysis of a fucoidan from the brown alga Fucus evanescens by MALDI-TOF and tandem ESI mass spectrometry. Carbohydr. Res. 2009, 344, 779–787. [Google Scholar] [CrossRef]
- Marais, M.F.; Joseleau, J.P. A fucoidan fraction from Ascophyllum nodosum. Carbohydr. Res. 2001, 336, 155–159. [Google Scholar] [CrossRef]
- Anastyuk, S.D.; Shevchenko, N.M.; Nazarenko, E.L.; Imbs, T.I.; Gorbach, V.I.; Dmitrenok, P.S.; Zvyagintseva, T.N. Structural analysis of a highly sulfated fucan from the brown alga Laminaria cichorioides by tandem MALDI and ESI mass spectrometry. Carbohydr. Res. 2010, 345, 2206–2212. [Google Scholar] [CrossRef]
- Saad, O.M.; Leary, J.A. Delineating mechanisms of dissociation for isomeric heparin disaccharides using isotope labeling and ion trap tandem massspectrometry. J. Am. Soc. Mass Spectrom. 2004, 15, 1274–1286. [Google Scholar] [CrossRef]
- Bilan, M.I.; Grachev, A.A.; Ustuzhamina, N.E.; Shashkov, A.S.; Nifantiev, N.E.; Usov, A.I. Structure of a fucoidan from the brown seaweed Fucus evanescens C. Ag. Carbohydr. Res. 2002, 337, 719–730. [Google Scholar] [CrossRef]
- Bitter, T.; Muir, H.M. A modified uronic acid carbazole reaction. Anal. Biochem. 1962, 4, 330–334. [Google Scholar] [CrossRef]
- Dodgson, K.S. Determination of inorganic sulphate in studies on the enzymic and non-enzymic hydrolysis of carbohydrate and other sulphate esters. Biochem. J. 1961, 78, 312–319. [Google Scholar]
- Shiroma, R.; KoniShi, T.; Uechi, S.; TaKo, M. Structural study of fucoidan from the brown seaweed Hizikia fusiformis. Food Sci. Technol. Res. 2008, 14, 176–182. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Thanh, T.T.T.; Tran, V.T.T.; Yuguchi, Y.; Bui, L.M.; Nguyen, T.T. Structure of Fucoidan from Brown Seaweed Turbinaria ornata as Studied by Electrospray Ionization Mass Spectrometry (ESIMS) and Small Angle X-ray Scattering (SAXS) Techniques. Mar. Drugs 2013, 11, 2431-2443. https://doi.org/10.3390/md11072431
Thanh TTT, Tran VTT, Yuguchi Y, Bui LM, Nguyen TT. Structure of Fucoidan from Brown Seaweed Turbinaria ornata as Studied by Electrospray Ionization Mass Spectrometry (ESIMS) and Small Angle X-ray Scattering (SAXS) Techniques. Marine Drugs. 2013; 11(7):2431-2443. https://doi.org/10.3390/md11072431
Chicago/Turabian StyleThanh, Thuy Thi Thu, Van Thi Thanh Tran, Yoshiaki Yuguchi, Ly Minh Bui, and Tai Tien Nguyen. 2013. "Structure of Fucoidan from Brown Seaweed Turbinaria ornata as Studied by Electrospray Ionization Mass Spectrometry (ESIMS) and Small Angle X-ray Scattering (SAXS) Techniques" Marine Drugs 11, no. 7: 2431-2443. https://doi.org/10.3390/md11072431
APA StyleThanh, T. T. T., Tran, V. T. T., Yuguchi, Y., Bui, L. M., & Nguyen, T. T. (2013). Structure of Fucoidan from Brown Seaweed Turbinaria ornata as Studied by Electrospray Ionization Mass Spectrometry (ESIMS) and Small Angle X-ray Scattering (SAXS) Techniques. Marine Drugs, 11(7), 2431-2443. https://doi.org/10.3390/md11072431




