Neuritogenic and Neuroprotective Effects of Polar Steroids from the Far East Starfishes Patiria pectinifera and Distolasterias nipon
Abstract
:1. Introduction
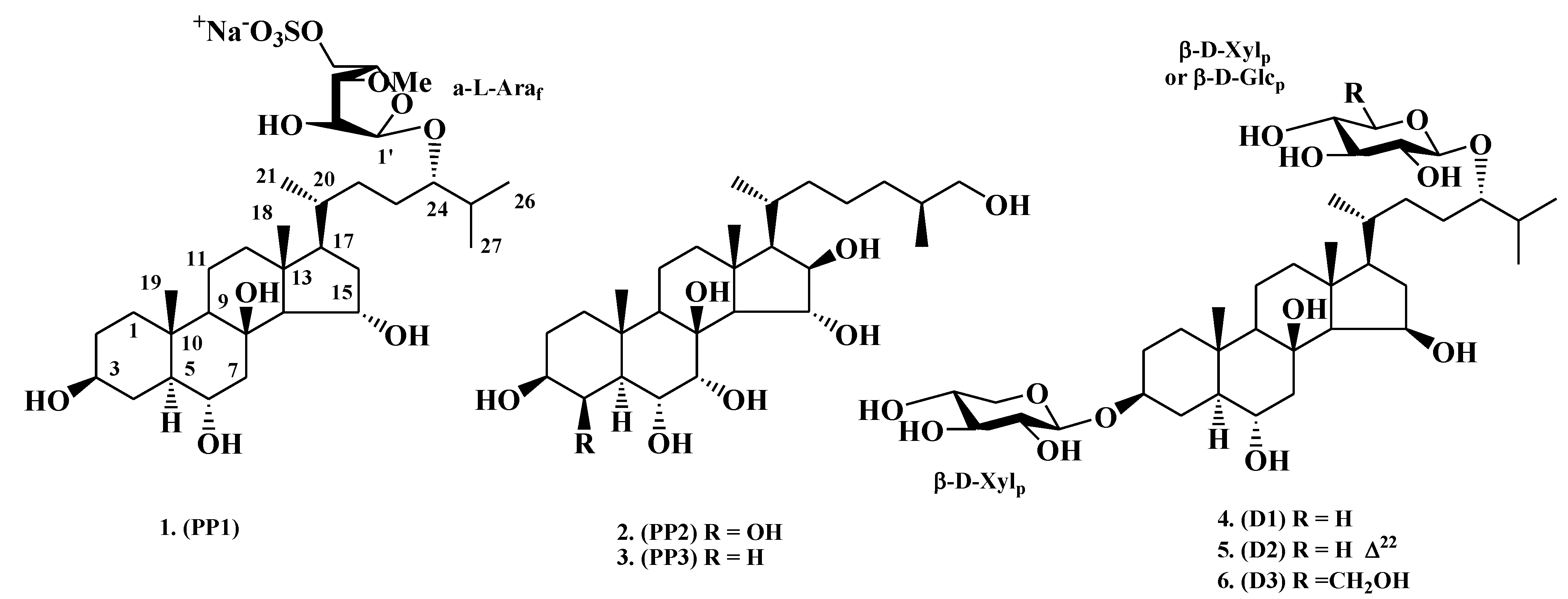
2. Results and Discussion
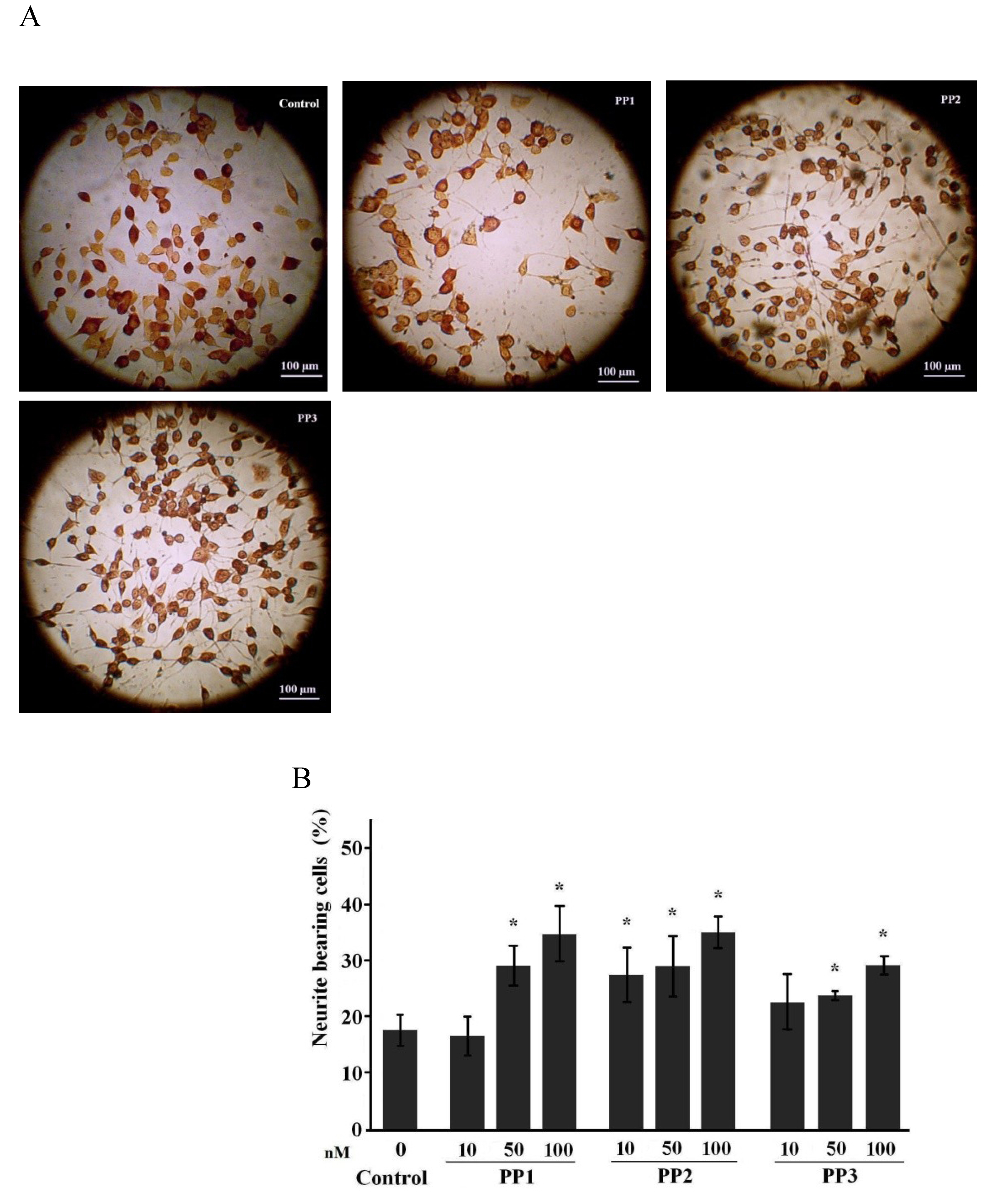
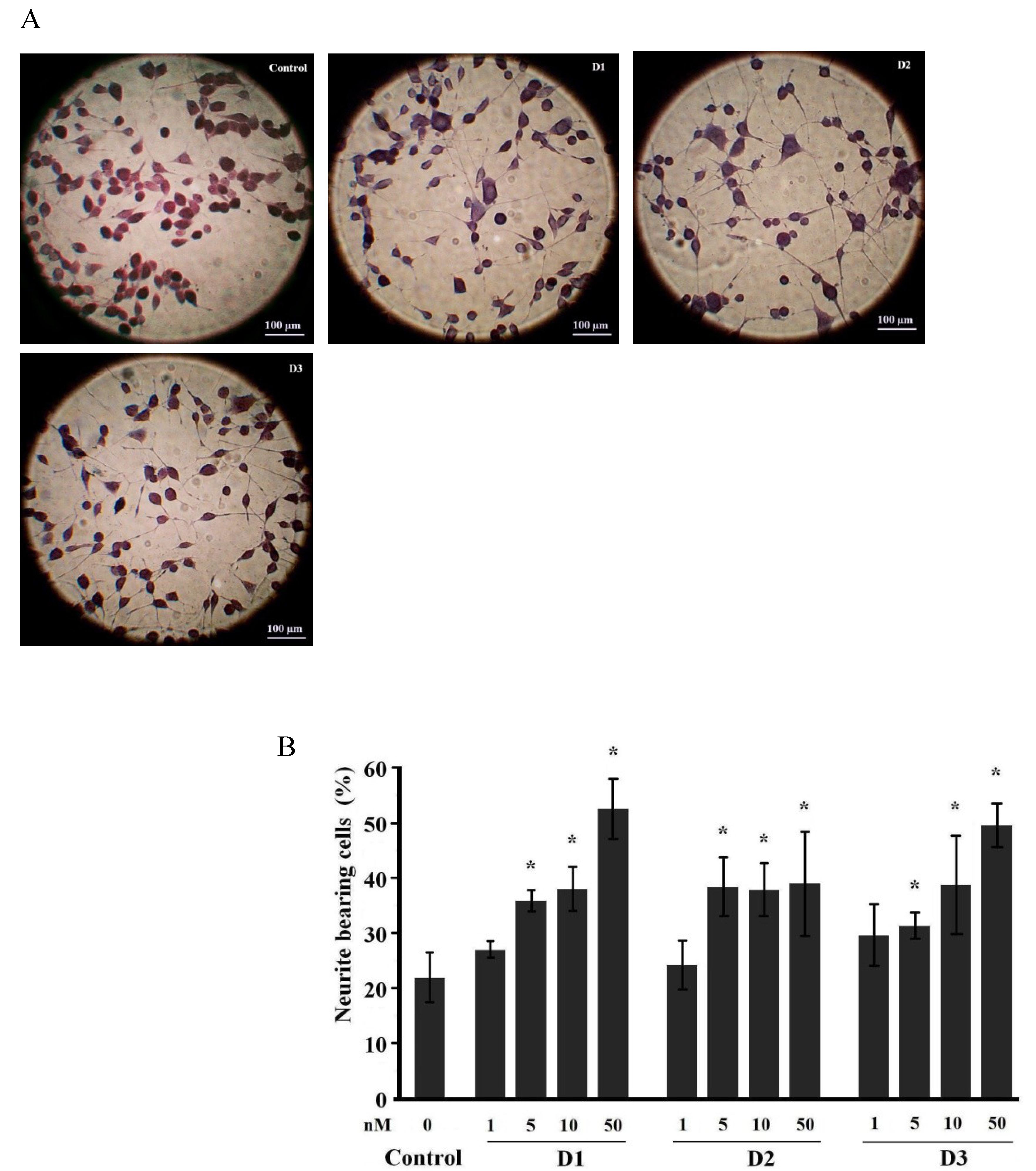
2.1. Neuritogenic Activity of Starfish Polar Steroids in Cultured NB Cells
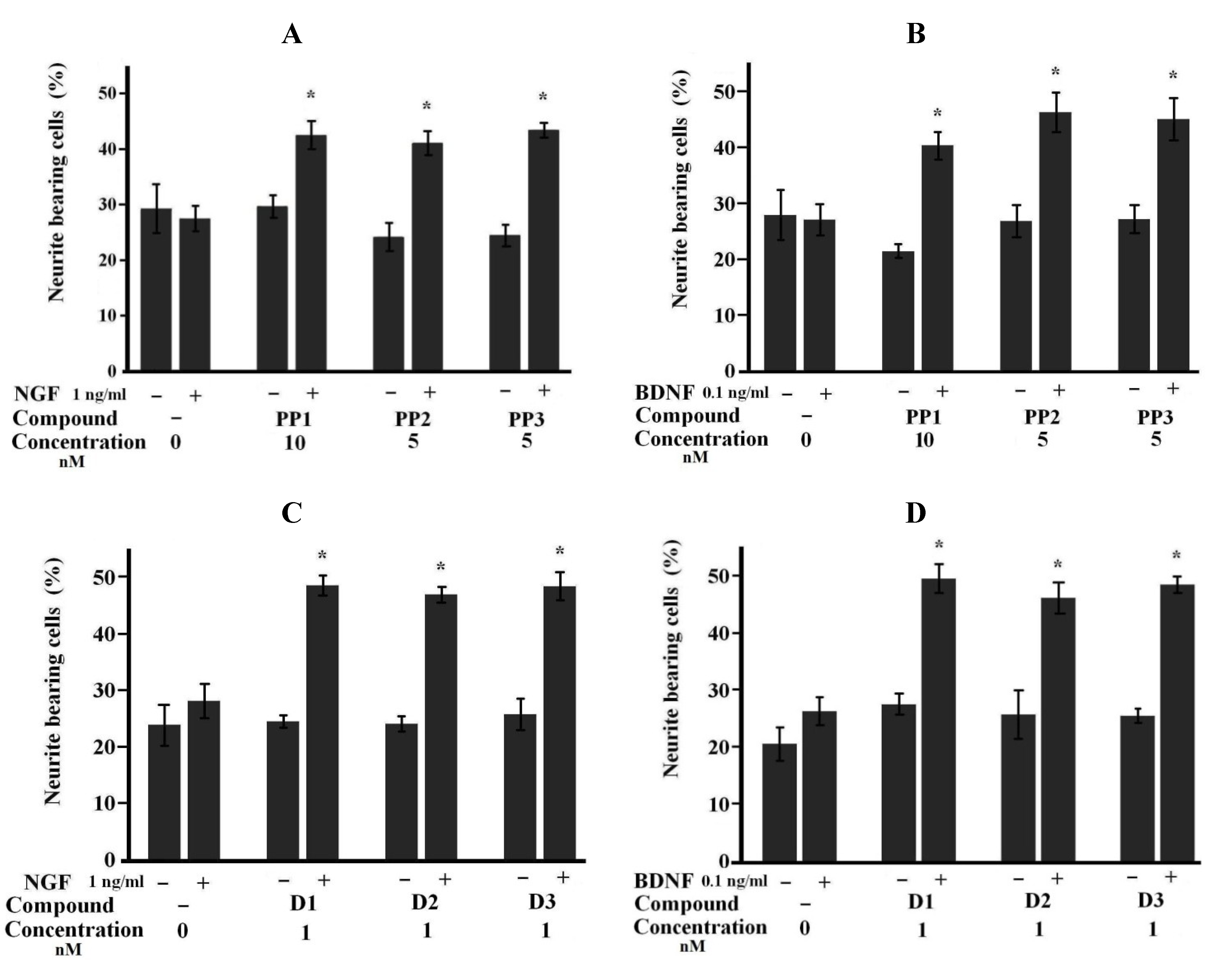
2.2. Neuroprotective Activities of Starfish Polar Steroids in Oxygen-Glucose Deprivation Experiments in Vitro
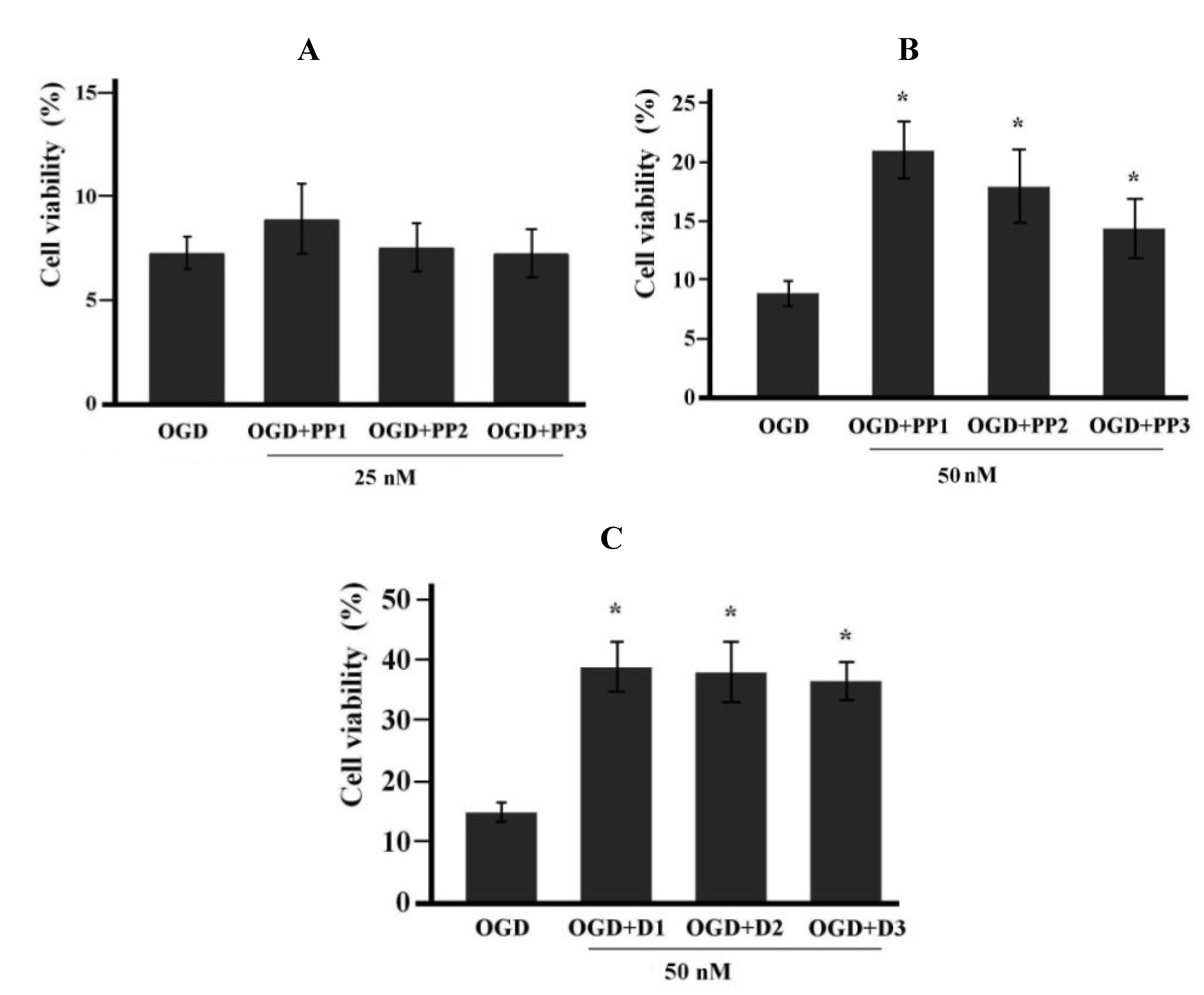
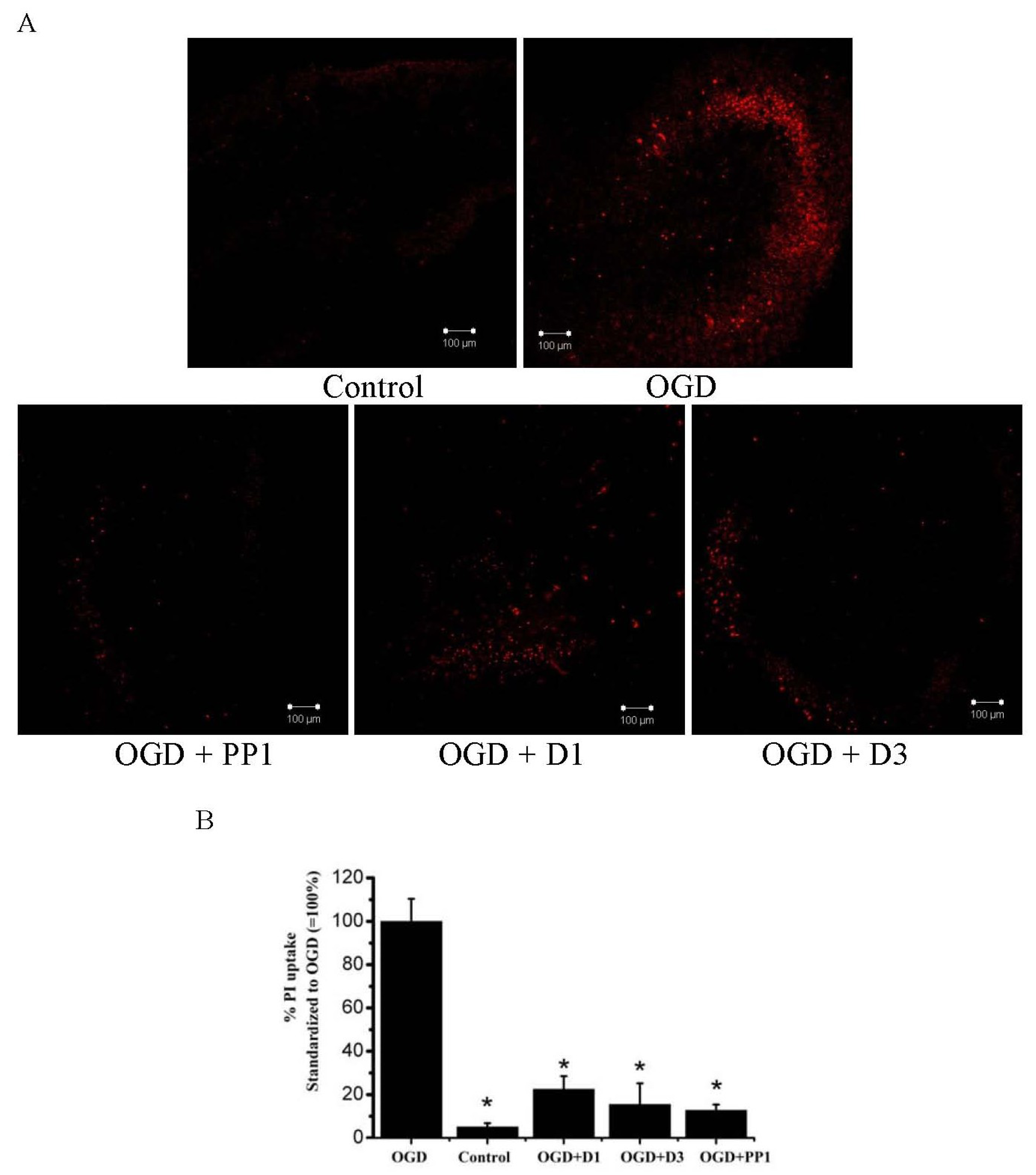
3. Experimental Section
3.1. Reagents and Tested Compounds
3.2. NB Cell Culture
3.3. Neuritogenic Effects in NB Cell Cultures
3.4. Oxygen-Glucose Deprivation in NB Cell Cultures
3.5. Organotypic Hippocampal Slice Culture
3.6. Oxygen-Glucose Deprivation Assay in OHSCs
4. Conclusions
Acknowledgments
References
- Bibel, M.; Barde, Y.A. Neurotrophins: Key regulators of cell fate and cell shape in the vertebrate nervous system. Genes Dev. 2000, 14, 2919–2937. [Google Scholar] [CrossRef]
- Huang, E.J.; Reichardt, L.F. Neurotrophins: Roles in neuronal development and function. Annu. Rev. Neurosci. 2001, 24, 677–736. [Google Scholar]
- Aloe, L.; Rocco, M.; Bianchi, P.; Manni, L. Nerve growth factor: From the early discoveries to the potential clinical use. J. Transl. Med. 2012, 10, 239. [Google Scholar] [CrossRef]
- Qi, J.; Luo, Y.; Gao, L. Structural diversity of neuritogenic substances and their application perspective. Mini Rev. Med. Chem. 2011, 11, 658–677. [Google Scholar] [CrossRef]
- More, S.V.; Koppula, S.; Kim, I.S.; Kumar, H.; Kim, B.W.; Choi, D.K. The role of bioactive compounds on the promotion of neurite outgrowth. Molecules 2012, 17, 6728–6753. [Google Scholar] [CrossRef]
- Ivanchina, N.V.; Kicha, A.A.; Stonik, V.A. Steroid glycosides from marine organisms. Steroids 2011, 76, 425–454. [Google Scholar] [CrossRef]
- Stonik, V.A.; Ivanchina, N.V.; Kicha, A.A. New polar steroids from starfish. Nat. Prod. Commun. 2008, 3, 1587–1610. [Google Scholar]
- Dong, G.; Xu, T.H.; Yang, B.; Lin, X.P.; Zhou, X.F.; Yang, X.W.; Liu, Y.H. Chemical constituents and bioactivities of starfish. Chem. Biodivers. 2011, 8, 740–791. [Google Scholar] [CrossRef]
- Han, C.G.; Qi, J.H.; Ojika, M.; Linkosides, M.-Q. Neuritogenic steroid glycosides from the Okinawan starfish Linkia laevigata. J. Nat. Med. 2007, 61, 138–145. [Google Scholar] [CrossRef]
- Qi, J.; Han, C.; Sasayama, Y.; Nakahara, H.; Shibata, T.; Uchida, K.; Ojika, M.; Granulatoside, A. A starfish steroid glycoside, enhances PC12 cell neuritogenesis induced by nerve growth factor through an activation of MAP kinase. Chem. Med. Chem. 2006, 1, 1351–1354. [Google Scholar]
- Pal’yanova, N.V.; Pankova, T.M.; Starostina, M.V.; Shtark, M.B.; Kicha, A.A.; Ivanchina, N.V.; Stonik, V.A. Neurotrophic effects of polyhydroxylated steroids and steroid glycosides in cultured neuroblastoma cells. Bull. Exp. Biol. Med. 2006, 141, 584–587. [Google Scholar] [CrossRef]
- Kicha, A.A.; Ivanchina, N.V.; Kalinovsky, A.I.; Dmitrenok, P.S.; Palyanova, N.V.; Pankova, T.M.; Starostina, M.V.; Gavagnin, M.; Stonik, V.A. New neuritogenic steroid glycosides from the Vietnamese starfish Linckia laevigata. Nat. Prod. Commun. 2007, 2, 41–46. [Google Scholar]
- Kicha, A.A.; Kapustina, I.I.; Ivanchina, N.V.; Kalinovsky, A.I.; Dmitrenok, P.S.; Stonik, V.A.; Pal’yanova, N.V.; Pankova, T.M.; Starostina, M.V. Polyhydroxylated steroid compounds from the Far Eastern starfish Distolasterias nipon. Russ. J. Bioorg. Chem. 2008, 34, 118–124. [Google Scholar] [CrossRef]
- Kicha, A.A.; Ivanchina, N.V.; Gorshkova, I.A.; Ponomarenko, L.P.; Likhatskaya, G.N.; Stonik, V.A. The distribution of free sterols, polyhydroxysteroids and steroid glycosides in various body components of the starfish Patiria (=Asterina) pectinifera. Comp. Biochem. Physiol. 2001, 128B, 43–52. [Google Scholar]
- Edsjo, A.; Holmquist, L.; Pahlman, S. Neuroblastoma as an experimental model for neuronal differentiation and hypoxia-induced tumor cell dedifferentiation. Semin. Cancer Biol. 2007, 17, 248–256. [Google Scholar] [CrossRef]
- Chen, P.C.; Pan, C.; Gharibani, P.M.; Prentice, H.; Wu, J.Y. Taurine exerts robust protection against hypoxia and oxygen/glucose deprivation in human neuroblastoma cell culture. Adv. Exp. Med. Biol. 2013, 775, 167–175. [Google Scholar]
- Gulaya, N.M.; Volkov, G.L.; Klimashevsky, V.M.; Govseeva, N.N.; Melnik, A.A. Changes in lipid composition of neuroblastoma C 1300 N18 cell during differentiation. Neuroscience 1989, 30, 153–164. [Google Scholar] [CrossRef]
- Kostenko, M.A.; Myakisheva, S.N.; Popov, V.I. Morphological differentiation of NIE-115 mouse neuroblastoma cells. Neurosci. Behav. Physiol. 1997, 27, 516–523. [Google Scholar] [CrossRef]
- Seidman, K.J.; Barsuk, J.H.; Johnson, R.F.; Weyhenmeyer, J.A. Differentiation of NG108-15 neuroblastoma cells by serum starvation or dimethyl sulfoxide results in marked differences in angiotensin II receptor subtype expression. J. Neurochem. 1996, 66, 1011–1018. [Google Scholar]
- Clejan, S.; Dotson, R.S.; Wolf, E.W.; Corb, M.P.; Ide, C.F. Morphological differentiation of N1E-115 neuroblastoma cells by dimethyl sulfoxide activation of lipid second messengers. Exp. Cell Res. 1996, 224, 16–27. [Google Scholar] [CrossRef]
- Falkenstein, E.; Tillmann, H.C.; Christ, M.; Feuring, M.; Wehling, M. Multiple actions of steroid hormones—A focus on rapid, nongenomic effects. Pharmacol. Rev. 2000, 52, 513–556. [Google Scholar]
- Numakawa, T.; Yokomaku, D.; Richards, M.; Hori, H.; Adachi, N.; Kunugi, H. Functional interactions between steroid hormones and neurotrophin BDNF. World J. Biol. Chem. 2010, 1, 133–143. [Google Scholar] [CrossRef]
- Fedorov, S.N.; Shubina, L.K.; Kicha, A.A.; Ivanchina, N.V.; Kwak, J.Y.; Jin, J.O.; Bode, A.M.; Dong, Z.; Stonik, V.A. Proapoptotic and anticarcinogenic activities of leviusculoside G from the starfish Henricia leviuscula and probable molecular mechanism. Nat. Prod. Commun. 2008, 3, 1575–1580. [Google Scholar]
- Zuo, Z.; Wang, Y.; Huang, Y. Isoflurane preconditioning protects human neuroblastoma SH-SY5Y cells against in vitro simulated ischemia-reperfusion through the activation of extracellular signal-regulated kinases pathway. Eur. J. Pharmacol. 2006, 542, 84–91. [Google Scholar] [CrossRef]
- Ahlgren, H.; Henjum, K.; Ottersen, O.P.; Runden-Pran, E. Validation of organotypical hippocampal slice cultures as an ex vivo model of brain ischemia: Different roles of NMDA receptors in cell death signalling after exposure to NMDA or oxygen and glucose deprivation. Cell Tissue Res. 2011, 345, 329–341. [Google Scholar] [CrossRef]
- Cimarosti, H.; Zamin, L.L.; Frozza, R.; Nassif, M.; Horn, A.P.; Tavares, A.; Netto, C.A.; Salbego, C. Estradiol protects against oxygen and glucose deprivation in rat hippocampal organotypic cultures and activates Akt and inactivates GSK-3β. Neurochem. Res. 2005, 30, 191–199. [Google Scholar] [CrossRef]
- Jin, K.L.; Mao, X.O.; Greenberg, D.A. Vascular endothelial growth factor: Direct neuroprotective effect in vitro ischemia. Proc. Natl. Acad. Sci. USA 2000, 97, 10242–10247. [Google Scholar]
- Hedborg, F.; Ulleras, E.; Grimelius, L.; Wassberg, E.; Maxwell, P.H.; Hero, B.; Berthold, F.; Schilling, F.; Harms, D.; Sandstedt, B.; et al. Evidence for hypoxia-induced neuronal-to-chromaffin metaplasia in neuroblastoma. FASEB J. 2003, 17, 598–609. [Google Scholar] [CrossRef]
- Smith, I.F.; Boyle, J.P.; Vaughan, P.F.T.; Pearson, H.A.; Peers, C. Effects of chronic hypoxia on Ca(2+) stores and capacitative Ca(2+) entry in human neuroblastoma (SH-SY5Y) cells. J. Neurochem. 2001, 79, 877–884. [Google Scholar]
- Axelson, H.; Fredlund, E.; Ovenberger, M.; Landberg, G.; Pahlman, S. Hypoxia-induced dedifferentiation of tumor cells—A mechanism behind heterogeneity and aggressiveness of solid tumors. Semin. Cell Dev. Biol. 2005, 16, 554–563. [Google Scholar] [CrossRef]
- Jogi, A.; Ora, I.; Nilsson, H.; Poellinger, L.; Axelson, H.; Pahlman, S. Hypoxia-induced dedifferentiation in neuroblastoma cells. Cancer Lett. 2003, 197, 145–150. [Google Scholar] [CrossRef]
- Pahlman, S.; Stockhausen, M.T.; Fredlund, E.; Axelson, H. Notch signaling in neuroblastoma. Semin. Cancer Biol. 2004, 14, 365–373. [Google Scholar] [CrossRef]
- Jogi, A.; Vallon-Christersson, J.; Holmquist, L.; Axelson, H.; Borg, A.; Pahlman, S. Human neuroblastoma cells exposed to hypoxia: Induction of genes associated with growth, survival, and aggressive behavior. Exp. Cell Res. 2004, 295, 469–487. [Google Scholar] [CrossRef]
- Holmquist, L.; Lofstedt, T.; Pahlman, S. Effect of hypoxia on the tumor phenotype: The neuroblastoma and breast cancer models. Adv. Exp. Med. Biol. 2006, 587, 179–193. [Google Scholar] [CrossRef]
- Nilsson, H.; Jogi, A.; Beckman, S.; Harris, A.L.; Poellinger, L.; Pahlman, S. HIF-2α expression in human fetal paraganglia and neuroblastoma: Relation to sympathetic differentiation, glucose deficiency, and hypoxia. Exp. Cell Res. 2005, 303, 447–456. [Google Scholar] [CrossRef]
- Hoehner, J.C.; Prabhakaran, K. Induced differentiation affords neuroblastoma cells protection from hypoxic injury. J. Pediatr. Surg. 2003, 38, 1069–1074. [Google Scholar] [CrossRef]
- Gerace, E.; Landucci, E.; Scartabelli, T.; Moroni, F.; Pellegrini-Giampietro, D.E. Rat hippocampal slice culture models for the evaluation of neuroprotective agents. Methods Mol. Biol. 2012, 846, 343–354. [Google Scholar] [CrossRef]
- Radley, E.; Akram, A.; Grubb, B.D.; Gibson, C.L. Investigation of the mechanisms of progesterone protection following oxygen-glucose deprivation in organotypic hippocampal slice cultures. Neurosci. Lett. 2012, 506, 131–135. [Google Scholar]
- Zamin, L.L.; Dillenburg-Pilla, P.; Argenta-Comiran, R.; Horn, A.P.; Simão, F.; Nassif, M.; Gerhardt, D.; Frozza, R.L.; Salbego, C. Protective effect of resveratrol against oxygen–glucose deprivation in organotypic hippocampal slice cultures: Involvement of PI3-K pathway. Neurobiol. Dis. 2006, 24, 170–182. [Google Scholar]
- Green, P.S.; Simpkins, J.W. Neuroprotective effects of estrogens: Potential mechanisms of action. Int. J. Dev. Neurosci. 2000, 18, 347–358. [Google Scholar] [CrossRef]
- Zhao, L.Q.; Brinton, R.D. Select estrogens within the complex formulation of conjugated equine estrogens (Premarin®) are protective against neurodegenerative insults: implications for a composition of estrogen therapy to promote neuronal function and prevent Alzheimer’s disease. BMC Neurosci. 2006, 7, 24–36. [Google Scholar] [CrossRef]
- Hurn, P.D.; Macrae, I.M. Estrogen as a neuroprotectant in stroke. J. Cereb. Blood Flow Metab. 2000, 20, 631–652. [Google Scholar] [CrossRef]
- Singh, M.; Su, C. Progesterone, brain-derived neurotrophic factor and neuroprotection. Neuroscience 2012, 239, 84–91. [Google Scholar] [CrossRef]
- Gahwiler, B.H.; Capogna, M.; Debanne, D.; McKinney, R.A.; Thompson, S.M. Organotypic slice cultures: A technique has come of age. Trends Neurosci. 1997, 20, 471–477. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Palyanova, N.V.; Pankova, T.M.; Starostina, M.V.; Kicha, A.A.; Ivanchina, N.V.; Stonik, V.A. Neuritogenic and Neuroprotective Effects of Polar Steroids from the Far East Starfishes Patiria pectinifera and Distolasterias nipon. Mar. Drugs 2013, 11, 1440-1455. https://doi.org/10.3390/md11051440
Palyanova NV, Pankova TM, Starostina MV, Kicha AA, Ivanchina NV, Stonik VA. Neuritogenic and Neuroprotective Effects of Polar Steroids from the Far East Starfishes Patiria pectinifera and Distolasterias nipon. Marine Drugs. 2013; 11(5):1440-1455. https://doi.org/10.3390/md11051440
Chicago/Turabian StylePalyanova, Natalia V., Tatyana M. Pankova, Marina V. Starostina, Alla A. Kicha, Natalia V. Ivanchina, and Valentin A. Stonik. 2013. "Neuritogenic and Neuroprotective Effects of Polar Steroids from the Far East Starfishes Patiria pectinifera and Distolasterias nipon" Marine Drugs 11, no. 5: 1440-1455. https://doi.org/10.3390/md11051440
APA StylePalyanova, N. V., Pankova, T. M., Starostina, M. V., Kicha, A. A., Ivanchina, N. V., & Stonik, V. A. (2013). Neuritogenic and Neuroprotective Effects of Polar Steroids from the Far East Starfishes Patiria pectinifera and Distolasterias nipon. Marine Drugs, 11(5), 1440-1455. https://doi.org/10.3390/md11051440






