N-Terminal Protease Gene Phylogeny Reveals the Potential for Novel Cyanobactin Diversity in Cyanobacteria
Abstract
:1. Introduction
2. Results
2.1. PCR Analyses
| Strain | Habitat a | Source b | Accession Number | |
|---|---|---|---|---|
| 16S rRNA | (A) gene | |||
| Cuspidothrix sp. LEGE 03284 | F | Montargil, Portugal | KC989703 | KF008260 |
| Microcystis aeruginosa LEGE 91347 | F | Reservoir, Bemposta dam, Portugal | KC989705 | KF008262 |
| Microcystis aeruginosa LEGE 91351 | F | Pond, Lagoa das Braças, Portugal | KC311966 | KF008265 |
| Microcystis aeruginosa LMECYA 1 | F | Reservoir, Montargil dam, Portugal | KC989706 | KF008263 |
| Microcystis sp. IZANCYA 45 | F | Pond, Lagoa da Vela, Portugal | KC311968 | KF008266 |
| Microcystis sp. LEGE 08328 | F | Lake Zumpango, Mexico | KC989704 | KF008261 |
| Microcystis sp. LEGE 08331 | F | Man-made channel, Cuemanco, Mexico | KC989707 | KF008264 |
| Phormidium sp. LEGE 06204 | F | WWTP, Febros river, Portugal | KC989699 | KF008256 |
| Phormidium sp. LEGE 06363 | F | WWTP, Febros river, Portugal | KC989700 | KF008257 |
| Phormidium sp. LEGE 07215 | F | WWTP, Febros river, Portugal | KC989698 | KF008255 |
| Rivularia sp. LEGE 07159 | M | Beach, Burgau, Portugal | KC989702 | KF008259 |
| Sphaerospermopsis sp. LEGE 00249 | F | Reservoir, Maranhão dam, Portugal | KC989701 | KF008258 |
2.2. Phylogenetic Analyses
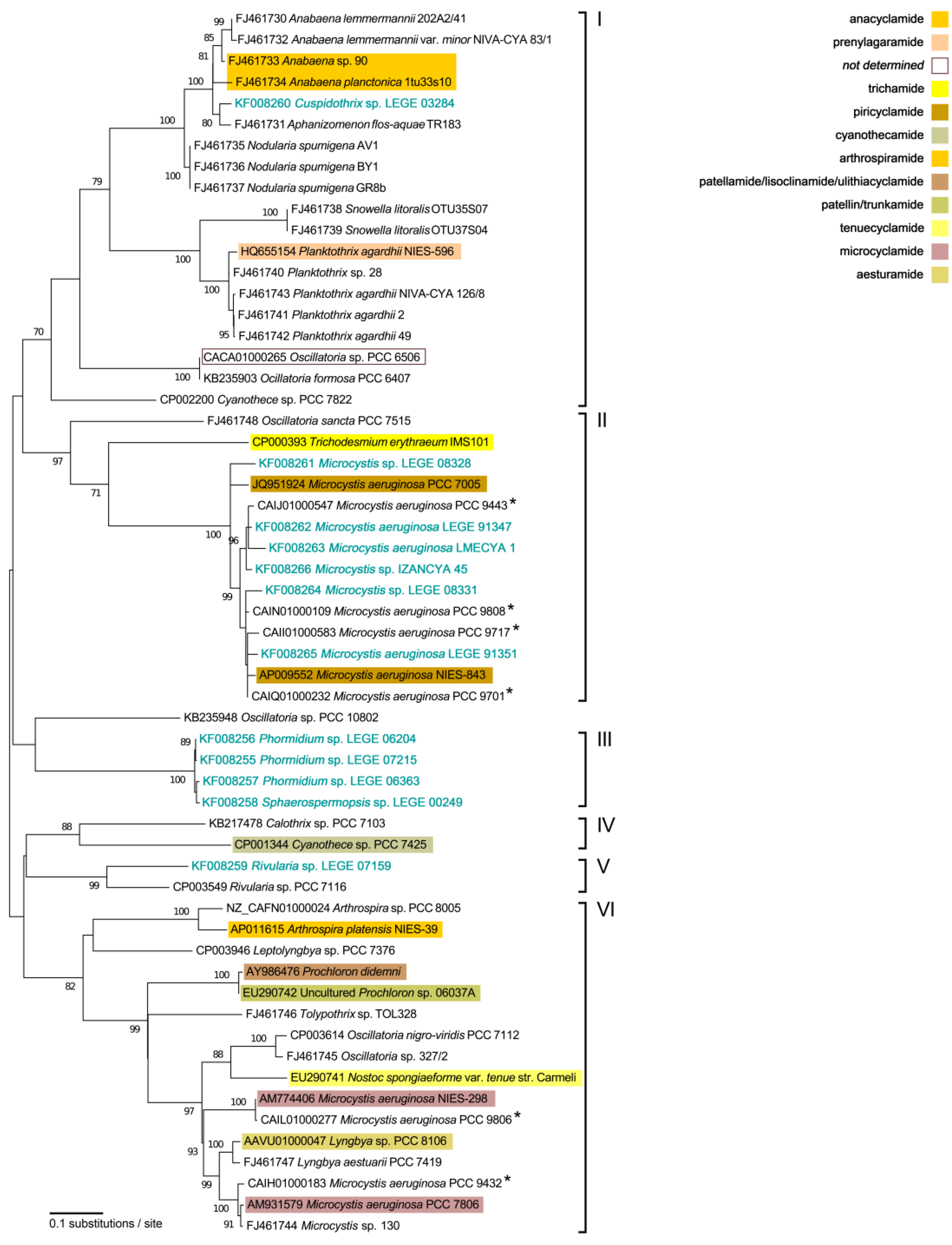

3. Discussion
4. Experimental Section
4.1. Cyanobacterial Strains
4.2. Degenerate Primers
| Order | Genus | Habitat(s) a | Number of tested strains | Number of positive strains |
|---|---|---|---|---|
| Chroococcales | Cyanobium | M | 9 | 0 |
| Gloeocapsa | M | 1 | 0 | |
| Microcystis | F | 15 | 5 | |
| Synechococcus | M | 5 | 0 | |
| Synechocystis | F; M | 3 | 0 | |
| Pleurocapsales | Chroococcopsis | M | 2 | 0 |
| Chroococcidiopsis | M | 2 | 0 | |
| Hyella | M | 1 | 0 | |
| Oscillatoriales | Leptolyngbya | F; M | 9 | 0 |
| Nodosilinea | M | 2 | 0 | |
| Oscillatoria | F | 3 | 0 | |
| Phormidium | F; E | 4 | 3 | |
| Planktothrix | F | 3 | 0 | |
| Plectonema | M | 1 | 0 | |
| Pseudanabaena | M | 4 | 0 | |
| Romeria | M | 1 | 0 | |
| Schizothrix | M | 1 | 0 | |
| Unidentified Pseudanabaenaceae | M | 1 | 0 | |
| Nostocales | Anabaena | F | 3 | 0 |
| Aphanizomenon | F | 4 | 0 | |
| Calothrix | M | 3 | 0 | |
| Cuspidothrix | F | 1 | 1 | |
| Cylindrospermopsis | F | 2 | 0 | |
| Nodularia | E | 1 | 0 | |
| Nostoc | M | 1 | 0 | |
| Rivularia | M | 1 | 1 | |
| Scytonema | M | 1 | 0 | |
| Sphaerospermopsis | F | 1 | 1 |
4.3. DNA Extraction, PCR Analyses and Sequencing
4.4. Nucleotide Sequence Accession Numbers
4.5. Phylogenetic Analyses
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Shih, P.M.; Wu, D.; Latifi, A.; Axen, S.D.; Fewer, D.P.; Talla, E.; Calteau, A.; Cai, F.; Tandeau de Marsac, N.; Rippka, R.; et al. Improving the coverage of the cyanobacterial phylum using diversity-driven genome sequencing. Proc. Natl. Acad. Sci. USA 2012, 110, 1053–1058. [Google Scholar]
- Tidgewell, K.; Clark, B.R.; Gerwick, W.H. The Natural Products Chemistry of Cyanobacteria. In Comprehensive Natural Products II Chemistry and Biology; Mander, L., Lui, H.-W., Eds.; Elsevier: Oxford, UK, 2010; Volume 2, pp. 141–188. [Google Scholar]
- Donia, M.S.; Ravel, J.; Schmidt, E.W. A global assembly line for cyanobactins. Nat. Chem. Biol. 2008, 4, 341–343. [Google Scholar] [CrossRef]
- Donia, M.S.; Schmidt, E.W. Cyanobactins—Ubiquitous Cyanobacterial Ribosomal Peptide Metabolites. In Comprehensive Natural Products II. Chemistry and Biology; Mander, L., Lui, H.-W., Eds.; Elsevier: Oxford, UK, 2010; Volume 2, pp. 539–558. [Google Scholar]
- Schmidt, E.W.; Donia, M.S. Chapter 23. Cyanobactin ribosomally synthesized peptides—A case of deep metagenome mining. Meth. Enzymol. 2009, 458, 575–596. [Google Scholar] [CrossRef]
- Donia, M.S.; Schmidt, E.W. Linking chemistry and genetics in the growing cyanobactin natural products family. Chem. Biol. 2011, 18, 508–519. [Google Scholar] [CrossRef]
- In, Y.; Doi, M.; Inoue, M.; Ishida, T.; Hamada, Y.; Shioiri, T. Patellamide A, a cytotoxic cyclic peptide from the ascidian Lissoclinum patella. Acta Crystallogr. C 1994, 50, 432–434. [Google Scholar] [CrossRef]
- Linington, R.G.; Gonzalez, J.; Urena, L.D.; Romero, L.I.; Ortega-Barria, E.; Gerwick, W.H. Venturamides A and B: Antimalarial constituents of the panamanian marine cyanobacterium Oscillatoria sp. J. Nat. Prod. 2007, 70, 397–401. [Google Scholar] [CrossRef]
- Portmann, C.; Blom, J.F.; Kaiser, M.; Brun, R.; Juttner, F.; Gademann, K. Isolation of aerucyclamides C and D and structure revision of microcyclamide 7806A: Heterocyclic ribosomal peptides from Microcystis aeruginosa PCC 7806 and their antiparasite evaluation. J. Nat. Prod. 2008, 71, 1891–1896. [Google Scholar] [CrossRef]
- Ogino, J.; Moore, R.E.; Patterson, G.M.; Smith, C.D. Dendroamides, new cyclic hexapeptides from a blue-green alga. Multidrug-resistance reversing activity of dendroamide A. J. Nat. Prod. 1996, 59, 581–586. [Google Scholar]
- Caba, J.M.; Rodriguez, I.M.; Manzanares, I.; Giralt, E.; Albericio, F. Solid-phase total synthesis of trunkamide A (1). J. Org. Chem. 2001, 66, 7568–7574. [Google Scholar]
- Leikoski, N.; Fewer, D.P.; Sivonen, K. Widespread occurrence and lateral transfer of the cyanobactin biosynthesis gene cluster in cyanobacteria. Appl. Environ. Microbiol. 2009, 75, 853–857. [Google Scholar] [CrossRef]
- Sivonen, K.; Leikoski, N.; Fewer, D.P.; Jokela, J. Cyanobactins—Ribosomal cyclic peptides produced by cyanobacteria. Appl. Microbiol. Biotechnol. 2010, 86, 1213–1225. [Google Scholar] [CrossRef]
- Leikoski, N.; Fewer, D.P.; Jokela, J.; Alakoski, P.; Wahlsten, M.; Sivonen, K. Analysis of an inactive cyanobactin biosynthetic gene cluster leads to discovery of new natural products from strains of the genus Microcystis. PLoS One 2012, 7, e43002. [Google Scholar]
- Houssen, W.E.; Koehnke, J.; Zollman, D.; Vendome, V.; Raab, A.; Smith, M.C.M.; Naismith, J.H.; Jaspars, M. The discovery of new cyanobactins from Cyanothece PCC 7425 defines a new signature for processing of patellamides. ChemBioChem 2012, 13, 2625–2776. [Google Scholar] [CrossRef]
- Murakami, M.; Itou, Y.; Ishida, K.; Shin, H.J. Prenylagaramides A and B, new cyclic peptides from two strains of Oscillatoria agardhii. J. Nat. Prod. 1999, 62, 752–755. [Google Scholar] [CrossRef]
- McIntosh, J.A.; Lin, Z.; Tianero, M.D.; Schmidt, E.W. Aesturamides, a natural library of cyanobactin cyclic peptides resulting from isoprene-derived Claisen rearrangements. ACS Chem. Biol. 2013. [Google Scholar] [CrossRef]
- Schmidt, E.W.; Nelson, J.T.; Rasko, D.A.; Sudek, S.; Eisen, J.A.; Haygood, M.G.; Ravel, J. Patellamide A and C biosynthesis by a microcin-like pathway in Prochloron didemni, the cyanobacterial symbiont of Lissoclinum patella. Proc. Natl. Acad. Sci. USA 2005, 102, 7315–7320. [Google Scholar] [CrossRef]
- Donia, M.S.; Hathaway, B.J.; Sudek, S.; Haygood, M.G.; Rosovitz, M.J.; Ravel, J.; Schmidt, E.W. Natural combinatorial peptide libraries in cyanobacterial symbionts of marine ascidians. Nat. Chem. Biol. 2006, 2, 729–735. [Google Scholar] [CrossRef]
- Arnison, P.G.; Bibb, M.J.; Bierbaum, G.; Bowers, A.A.; Bugni, T.S.; Bulaj, G.; Camarero, J.A.; Campopiano, D.J.; Challis, G.L.; Clardy, J.; et al. Ribosomally synthesized and post-translationally modified peptide natural products: Overview and recommendations for a universal nomenclature. Nat. Prod. Rep. 2012, 30, 108–160. [Google Scholar]
- Rajaniemi, P.; Hrouzek, P.; Kaštovská, K.; Willame, R.; Rantala, A.; Hoffmann, L.; Komárek, J.; Sivonen, K. Phylogenetic and morphological evaluation of the genera Anabaena, Aphanizomenon, Trichormus and Nostoc (Nostocales, Cyanobacteria). Int. J. Syst. Ecol. Microbiol. 2005, 55, 11–26. [Google Scholar] [CrossRef]
- Leikoski, N.; Fewer, D.P.; Jokela, J.; Wahlsten, M.; Rouhiainen, L.; Sivonen, K. Highly diverse cyanobactins in strains of the genus Anabaena. Appl. Environ. Microbiol. 2010, 76, 701–709. [Google Scholar] [CrossRef]
- Wacklin, P.; Hoffmann, L.; Komárek, J. Nomenclatural validation of the genetically revised cyanobacterial genus Dolichospermum (Ralfs ex Bornet et Flahault) comb. nova. Fottea 2009, 9, 59–64. [Google Scholar]
- Rajaniemi, P.; Komárek, J.; Hrouzek, P.; Willame, R.; Kaštovská, K.; Hoffmann, L.; Sivonen, K. Taxonomic consequences from the combined molecular and phenotype evaluation of selected Anabaena and Aphanizomenon strains. Arch. Hydrobiol. Algol. Stud. 2005, 117 (Suppl.), 371–391. [Google Scholar]
- Zapomělová, E.; Jezberová, J.; Hrouzek, P.; Hisem, D.; Rěháková, K.; Komárková, J. Polyphasic characterization of three strains of Anabaena reniformis and Aphanizomenon aphanizomenoides (cyanobacteria) and their reclassification to Sphaerospermum gen. nov. (incl. Anabaena kisseleviana). J. Phycol. 2009, 45, 1363–1373. [Google Scholar] [CrossRef]
- Gugger, M.F.; Hoffmann, L. Polyphyly of true branching cyanobacteria (Stigonematales). Int. J. Syst. Evol. Microbiol. 2004, 54, 349–357. [Google Scholar] [CrossRef]
- Ishida, T.; Watanabe, M.M.; Sugiyama, J.; Yokota, A. Evidence for polyphyletic origin of the members of the orders of Oscillatoriales and Pleurocapsales as determined by 16S rDNA analysis. FEMS Microbiol. Lett. 2001, 201, 79–82. [Google Scholar]
- Litvaitis, M.K. A molecular test of cyanobacterial phylogeny: Inferences from constraint analyses. Hydrobiologia 2002, 468, 135–145. [Google Scholar] [CrossRef]
- Honda, D.; Yokota, A.; Sugiyama, J. Detection of seven major evolutionary lineages in cyanobacteria based on the 16S rRNA gene sequence analysis with new sequences of five marine Synechococcus strains. J. Mol. Evol. 1999, 48, 723–739. [Google Scholar]
- Marquardt, J.; Palinska, K.A. Genotypic and phenotypic diversity of cyanobacteria assigned to the genus Phormidium (Oscillatoriales) from different habitats and geographical sites. Arch. Microbiol. 2007, 187, 397–413. [Google Scholar] [CrossRef]
- Henson, B.J.; Hesselbrock, S.M.; Watson, L.E.; Barnum, S.R. Molecular phylogeny of the heterocystous cyanobacteria (subsections IV and V) based on nifD. Int. J. Syst. Evol. Microbiol. 2004, 54, 493–497. [Google Scholar]
- Otsuka, S.; Suda, S.; Shibata, S.; Oyaizu, H.; Matsumoto, S.; Watanabe, M.M. A proposal for the unification of five species of the cyanobacterial genus Microcystis Kützing ex Lemmermann 1907 under the rules of the Bacteriological Code. Int. J. Syst. Evol. Microbiol. 2001, 51, 873–879. [Google Scholar] [CrossRef]
- Waterbury, J.B.; Stanier, R.Y. Patterns of growth and development in pleurocapsalean cyanobacteria. Microbiol. Mol. Biol. Rev. 1978, 42, 2–44. [Google Scholar]
- Komárek, J.; Anagnostidis, K. Modern approach to the classification system of Cyanophytes: 4-Nostocales. Arch. Hydrobiol. Suppl. 1989, 82, 247–345. [Google Scholar]
- Komárek, J.; Anagnostidis, K. Cyanoprokaryota, Part 1: Chroococcales. In Süsswasserflora von Mitteleuropa; Ettl, H., Gärtner, G., Heynig, H., Mollenhauer, D., Eds.; Spektrum Akademischer Verlag: Heidelberg, Germany, 1998; Volume 19/1, p. 548. [Google Scholar]
- Komárek, J.; Anagnostidis, K. Cyanoprokaryota, Part 2: Oscillatoriales. In Süsswasserflora von Mitteleuropa; Büdel, B., Gärtner, G., Krienitz, L., Schagerl, M., Eds.; Elsevier: Munich, Germany, 2005; Volume 19/2, p. 759. [Google Scholar]
- Paulino, S.; Sam-Bento, F.; Churro, C.; Alverca, E.; Dias, E.; Valério, E.; Pereira, P. The Estela Sousa e Silva Algal Culture Collection: A resource of biological and toxicological interest. Hydrobiologia 2009, 636, 489–492. [Google Scholar] [CrossRef]
- Thompson, J.D.; Higgins, D.G.; Gibson, T.J. CLUSTAL W: Improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res. 1994, 22, 4673–4680. [Google Scholar] [CrossRef]
- Tamura, K.; Peterson, D.; Peterson, N.; Stecher, G.; Nei, M.; Kumar, S. MEGA5: Molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol. Biol. Evol. 2011, 28, 2731–2739. [Google Scholar] [CrossRef]
- Neilan, B.A.; Jacobs, D.; del Dot, T.; Blackall, L.L.; Hawkins, P.R.; Cox, P.T.; Goodman, A.E. rRNA sequences and evolutionary relationships among toxic and non-toxic cyanobacteria of the genus Microcystis. Int. J. Syst. Bacteriol. 1997, 47, 693–697. [Google Scholar] [CrossRef]
- Nübel, U.; Garcia-Pichel, F.; Muyzer, G. PCR primers to amplify 16S rRNA genes from cyanobacteria. Appl. Environ. Microbiol. 1997, 63, 3327–3332. [Google Scholar]
- Blin, K.; Medema, M.H.; Kazempour, D.; Fischbach, M.A.; Breitling, R.; Takano, E.; Weber, T. antiSMASH 2.0—A versatile platform for genome mining of secondary metabolite producers. Nucleic Acids Res. 2013. [Google Scholar] [CrossRef]
- Edgar, R.C. MUSCLE: Multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 2004, 32, 1792–1797. [Google Scholar] [CrossRef]
- Felsenstein, J. Evolutionary trees from DNA sequences: A maximum likelihood approach. J. Mol. Evol. 1981, 17, 368–376. [Google Scholar] [CrossRef]
Supplementary Files
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Martins, J.; Leão, P.N.; Ramos, V.; Vasconcelos, V. N-Terminal Protease Gene Phylogeny Reveals the Potential for Novel Cyanobactin Diversity in Cyanobacteria. Mar. Drugs 2013, 11, 4902-4916. https://doi.org/10.3390/md11124902
Martins J, Leão PN, Ramos V, Vasconcelos V. N-Terminal Protease Gene Phylogeny Reveals the Potential for Novel Cyanobactin Diversity in Cyanobacteria. Marine Drugs. 2013; 11(12):4902-4916. https://doi.org/10.3390/md11124902
Chicago/Turabian StyleMartins, Joana, Pedro N. Leão, Vitor Ramos, and Vitor Vasconcelos. 2013. "N-Terminal Protease Gene Phylogeny Reveals the Potential for Novel Cyanobactin Diversity in Cyanobacteria" Marine Drugs 11, no. 12: 4902-4916. https://doi.org/10.3390/md11124902
APA StyleMartins, J., Leão, P. N., Ramos, V., & Vasconcelos, V. (2013). N-Terminal Protease Gene Phylogeny Reveals the Potential for Novel Cyanobactin Diversity in Cyanobacteria. Marine Drugs, 11(12), 4902-4916. https://doi.org/10.3390/md11124902






