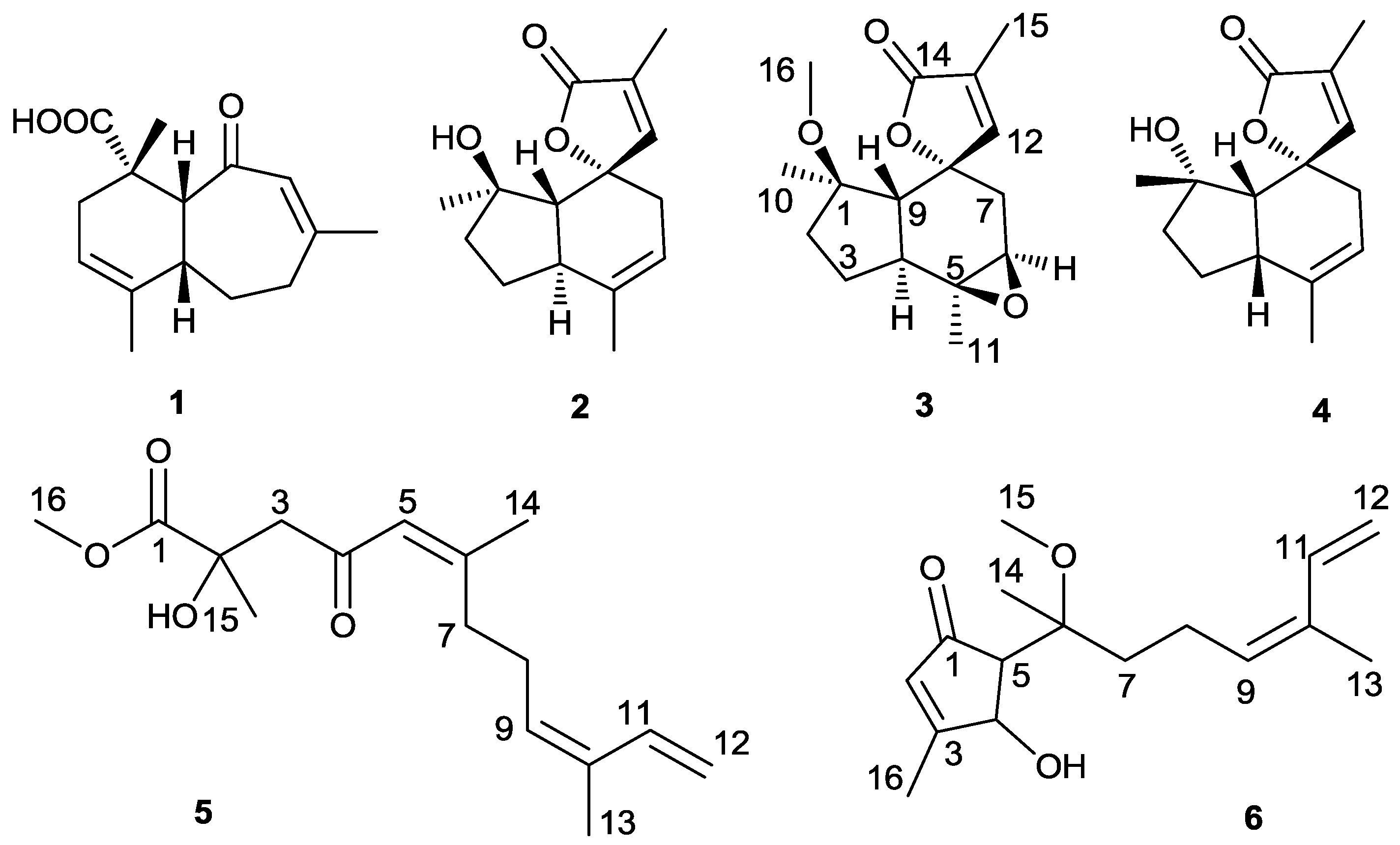
New Sinularianin Sesquiterpenes from Soft Coral Sinularia sp.
Abstract
:1. Introduction
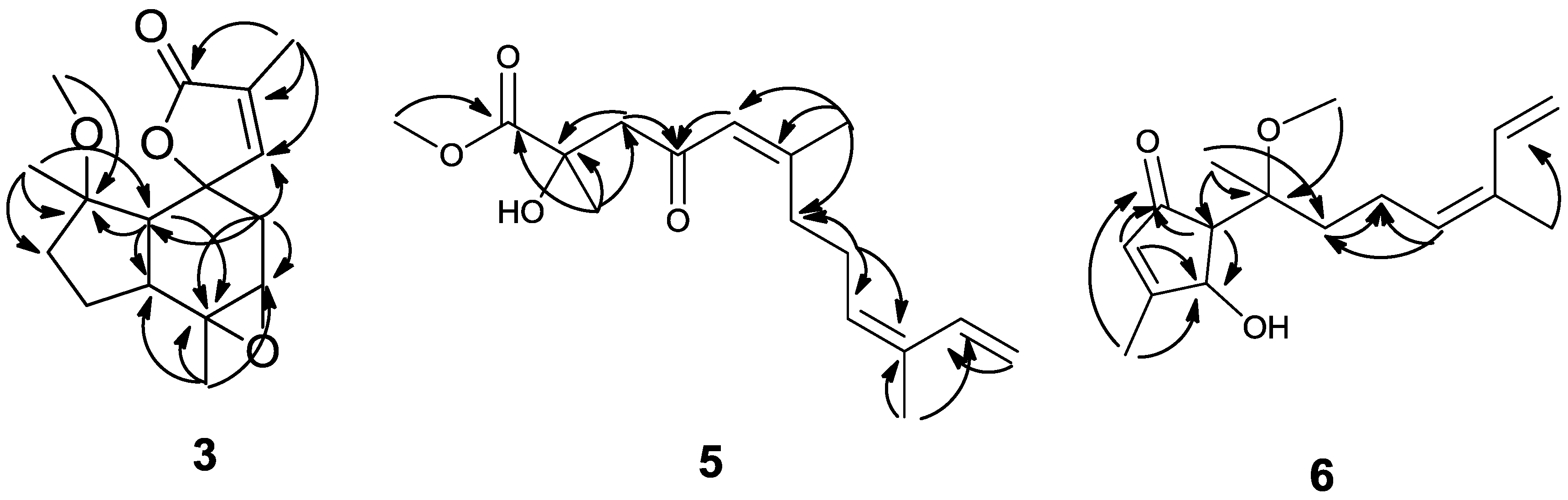
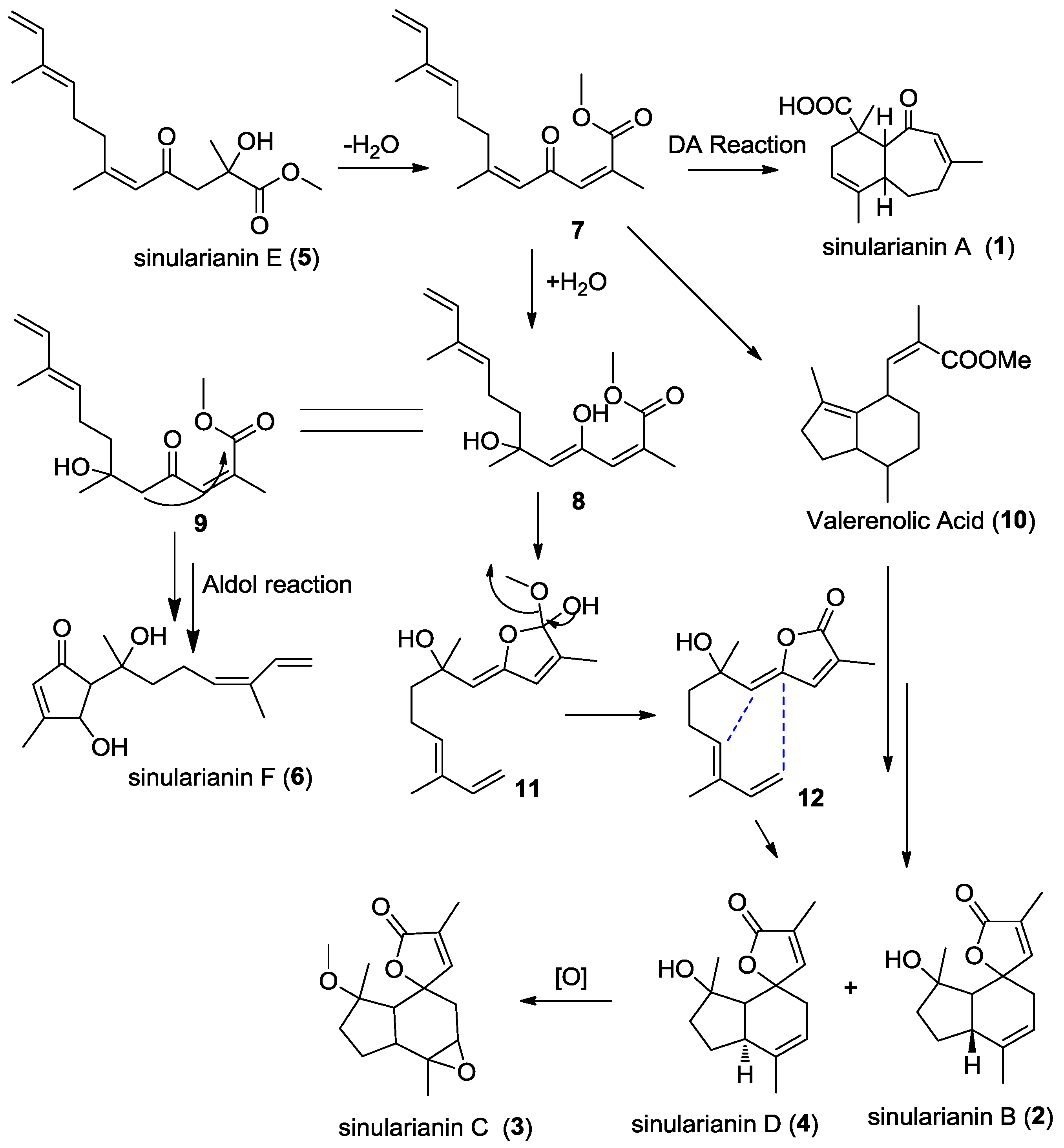
2. Results and Discussion
| Position | 3 | 4 | ||
|---|---|---|---|---|
| 1H | 13C | 1H | 13C | |
| 1 | 84.2 | 78.4 | ||
| 2 | 1.95 m | 35.7 | 1.92 m | 43.1 |
| 1.53 m | 1.79 m | |||
| 3 | 1.96 m | 23.6 | 2.01 m | 25.9 |
| 1.56 m | ||||
| 4 | 2.43 m | 41.1 | 2.90 m | 41.8 |
| 5 | 61.8 | 137.5 | ||
| 6 | 3.11 d (5.0) | 59.5 | 5.26 s | 117.2 |
| 7 | 2.26 d (16.0) | 38.7 | 2.53 m | 39.4 |
| 1.76 dd (16.0, 5.0) | 1.91 m | |||
| 8 | 86.5 | 85.6 | ||
| 9 | 2.00 d (13.0) | 50.4 | 1.55 d (12.5) | 55.5 |
| 10 | 1.00 s | 21.3 | 1.26 s | 28.5 |
| 11 | 1.41 s | 20.5 | 1.74 s | 20.5 |
| 12 | 7.17 d (1.5) | 154.7 | 7.03 d (1.5) | 150.9 |
| 13 | 129.5 | 129.7 | ||
| 14 | 175.6 | 174 | ||
| 15 | 1.86 s | 10.3 | 1.94 d (1.5) | 10.6 |
| 16 | 3.17 s | 50.8 | ||

| Position | 5 | 6 | ||
|---|---|---|---|---|
| 1H | 13C | 1H | 13C | |
| 1 | 176.5 | 203.2 | ||
| 2 | 72.9 | 5.86 s | 131.2 | |
| 3 | 3.15 d (17.5) | 52.9 | 174.9 | |
| 2.80 d (17.5) | ||||
| 4 | 199.3 | 4.79 s | 75.3 | |
| 5 | 6.01 s | 122.9 | 2.65 s | 61.5 |
| 6 | 160.3 | 78 | ||
| 7 | 2.22 m | 40.9 | 1.82 m | 34.1 |
| 2.12 m | ||||
| 8 | 2.34 m | 25.8 | 2.19 m | 22.1 |
| 2.20 m | ||||
| 9 | 5.42 t (7.0) | 130.8 | 5.52 m | 132.3 |
| 10 | 135 | 134.1 | ||
| 11 | 6.36 dd (17.5, 10.5) | 141 | 6.36 dd (17.0, 10.5) | 141.5 |
| 12 | 5.13 d (17.5) | 111.3 | 5.10 d (17.0) | 110.7 |
| 4.96 d (10.5) | 4.94 d (10.5) | |||
| 13 | 1.74 s | 11.7 | 1.76 s | 11.6 |
| 14 | 2.14 s | 19.7 | 1.03 s | 22.7 |
| 15 | 1.40 s | 26.2 | 3.21 s | 48.5 |
| 16 | 3.72 s | 52.7 | 2.16 s | 15.6 |
| Concentration | IR (%) | |||||
|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | |
| 10 µg/mL | 41.3 | 29.6 | 24.3 | 43.0 | 30.0 | 36.1 |
3. Experimental Section
3.1. General Experimental Procedures
3.2. Animal Material
3.3. Extraction and Isolation
 = −6.0 (c = 0.01, MeOH); UV (MeOH): λmax (log ε) = 204.2 (1.70); IR (KBr) νmax 3421, 2927, 2854, 1735, 1666 cm–1 (Supplementary Figure S21); 1H- and 13C-NMR (see Table 1); ESI-MS m/z 271 [M + Na]+, 519 [2M + Na]+.
= −6.0 (c = 0.01, MeOH); UV (MeOH): λmax (log ε) = 204.2 (1.70); IR (KBr) νmax 3421, 2927, 2854, 1735, 1666 cm–1 (Supplementary Figure S21); 1H- and 13C-NMR (see Table 1); ESI-MS m/z 271 [M + Na]+, 519 [2M + Na]+. 3.4. The Cell-Based HEK293 NF-κB Luciferase Reporter Gene Assay
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Yang, B.; Zhou, X.F.; Huang, H.; Yang, X.W.; Liu, J.; Lin, X.P.; Li, X.B.; Peng, Y.; Liu, Y.H. New cembrane diterpenoids from a Hainan soft coral Sinularia sp. Mar. Drugs 2012, 10, 2023–2032. [Google Scholar] [CrossRef]
- Chao, C.H.; Chou, K.J.; Huang, C.Y.; Wen, Z.H.; Hsu, C.H.; Wu, Y.C.; Dai, C.F.; Sheu, J.H. Steroids from the soft coral Sinularia crassa. Mar. Drugs 2012, 10, 439–450. [Google Scholar] [CrossRef]
- Cheng, S.Y.; Huang, K.J.; Wang, S.K.; Duh, C.Y. Capilloquinol: A novel farnesyl quinol from the Dongsha atoll soft coral Sinularia capillosa. Mar. Drugs 2011, 9, 1469–1476. [Google Scholar] [CrossRef]
- Li, R.; Shao, C.L.; Qi, X.; Li, X.B.; Li, J.; Sun, L.L.; Wang, C.Y. Polyoxygenated sterols from the South China Sea soft coral Sinularia sp. Mar. Drugs 2012, 10, 1422–1432. [Google Scholar] [CrossRef]
- Kamel, H.N.; Slattery, M. Terpenoids of Sinularia: Chemistry and biomedical applications. Pharm. Biol. 2005, 43, 253–269. [Google Scholar] [CrossRef]
- Su, J.H.; Wen, Z.H. Bioactive cembrane-based diterpenoids from the soft coral Sinularia triangular. Mar. Drugs 2011, 9, 944–951. [Google Scholar] [CrossRef]
- Tsai, T.C.; Wu, Y.J.; Su, J.H.; Lin, W.T.; Lin, Y.S. A new spatane diterpenoid from the cultured soft coral Sinularia leptoclados. Mar. Drugs 2013, 11, 114–123. [Google Scholar] [CrossRef]
- Tseng, Y.J.; Shen, K.P.; Lin, H.L.; Huang, C.Y.; Dai, C.F.; Sheu, J.H. Lochmolins A–G, new sesquiterpenoids from the soft coral Sinularia lochmodes. Mar. Drugs 2012, 10, 1572–1581. [Google Scholar] [CrossRef]
- Lai, D.W.; Li, Y.X.; Xu, M.J.; Deng, Z.W.; van Ofwegen, L.; Qian, P.Y.; Proksch, P.; Lin, W.H. Sinulariols A–S, 19-oxygenated cembranoids from the Chinese soft coral Sinularia rigida. Tetrahedron 2011, 67, 6018–6029. [Google Scholar]
- Chao, C.H.; Hsieh, C.H.; Chen, S.P.; Lu, C.K.; Dai, C.F.; Sheu, J.H. Sinularianins A and B, novel sesquiterpenoids from the Formosan soft coral Sinularia sp. Tetrahedron Lett. 2006, 47, 5889–5891. [Google Scholar]
- Lu, M.C.; Lee, N.L.; Tseng, S.W.; Su, J.H. Sinutriangulin A, a novel diterpenoid from the soft coral Sinularia triangula. Tetrahedron Lett. 2011, 52, 5869–5871. [Google Scholar]
- Putra, M.Y.; Ianaro, A.; Panza, E.; Bavestrello, G.; Cerrano, C.; Fattorusso, E.; Taglialatela-Scafati, O. Sinulasulfoxide and sinulasulfone, sulfur-containing alkaloids from the Indonesian soft coral Sinularia sp. Tetrahedron Lett. 2012, 53, 3937–3939. [Google Scholar] [CrossRef]
- Yamashita, T.; Nakao, Y.; Matsunaga, S.; Oikawa, T.; Imahara, Y.; Fusetani, N. A new antiangiogenic C-24 oxylipin from the soft coral Sinularia numerosa. Bioorg. Med. Chem. 2009, 17, 2181–2184. [Google Scholar] [CrossRef]
- Chai, M.C.; Wang, S.K.; Dai, C.F.; Duh, C.Y. A cytotoxic lobane diterpene from the Formosan soft coral Sinularia inelegans. J. Nat. Prod. 2000, 63, 843–844. [Google Scholar] [CrossRef]
- Sheu, J.H.; Chang, K.C.; Duh, C.Y. A cytotoxic 5α,8α-epidioxysterol from a soft coral Sinularia species. J. Nat. Prod. 2000, 63, 149–151. [Google Scholar] [CrossRef]
- Chao, C.H.; Chou, K.J.; Huang, C.Y.; Wen, Z.H.; Hsu, C.H.; Wu, Y.C.; Dai, C.F.; Sheu, J.H. Bioactive cembranoids from the soft coral Sinularia crassa. Mar. Drugs 2011, 9, 1955–1968. [Google Scholar] [CrossRef]
- Shi, H.Y.; Yu, S.J.; Liu, D.; van Ofwegen, L.; Proksch, P.; Lin, W.H. Sinularones A–I, new cyclopentenone and butenolide derivatives from a marine soft coral Sinularia sp. and their antifouling activity. Mar. Drugs 2012, 10, 1331–1344. [Google Scholar] [CrossRef]
- Wright, A.D.; Nielson, J.L.; Tapiolas, D.M.; Liptrot, C.H.; Motti, C.A. A great barrier reef Sinularia sp. yields two new cytotoxic diterpenes. Mar. Drugs 2012, 10, 1619–1630. [Google Scholar] [CrossRef]
- Jacobo-Herrera, N.J.; Vartiainen, N.; Bremner, P.; Gibbons, S.; Koistinaho, J.; Heinrich, M. NF-κB modulators from Valeriana officinalis. Phytother. Res. 2006, 20, 917–919. [Google Scholar] [CrossRef]
- Bos, R.; Hendriks, H.; Bruins, A.P.; Kloosterman, J.; Sipma, G. Isolation and identification of valerenane sesquiterpenoids from Valeriana officinalis. Phytochemistry 1986, 25, 133–135. [Google Scholar]
- Birnbaum, G.I.; Findlay, J.A.; Krepinsky, J.J. Stereochemistry of valerenane sesquiterpenoids-crystal-structure of valerenolic acid. J. Org. Chem. 1978, 43, 272–276. [Google Scholar] [CrossRef]
- Mao, S.C.; Guo, Y.W.; Shen, X. Two novel aromatic valerenane-type sesquiterpenes from the Chinese green alga Caulerpa taxifolia. Bioorg. Med. Chem. Lett. 2006, 16, 2947–2950. [Google Scholar]
- Kobayashi, M.; Yasuzawa, T.; Kyogoku, Y.; Kido, M.; Kitagawa, I. Three new ent-valerenane sesquiterpenes from an Okinawan soft coral. Chem. Pharm. Bull. 1982, 30, 3431–3434. [Google Scholar]
- Bowden, B.F.; Coll, J.C.; Desilva, E.D.; Decosta, M.S.L.; Djura, P.J.; Mahendran, M.; Tapiolas, D.M. Studies of Australian soft corals. XXXI. Novel furanosesquiterpenes from several Sinularian soft corals (Coelenterata, Octocorallia, Alcyonacea). Aust. J. Chem. 1983, 36, 371–376. [Google Scholar] [CrossRef]
- Yang, X.W.; Ding, Y.Q.; Li, X.C.; Ferreira, D.; Shen, Y.H.; Li, S.M.; Wang, N.; Zhang, W.D. Cycloabiesesquine A, a unique sesquiterpenoid from Abies delavayi. Chem. Commun. 2009, 25, 3771–3773. [Google Scholar]
- Peddibhotla, S.; Shi, R.X.; Khan, P.; Smith, L.H.; Mangravita-Novo, A.; Vicchiarelli, M.; Su, Y.; Okolotowicz, K.J.; Cashman, J.R.; Reed, J.C.; et al. Inhibition of protein kinase C-driven nuclear factor-kappa B activation: Synthesis, structure-activity relationship, and pharmacological profiling of pathway specific benzimidazole probe molecules. J. Med. Chem. 2010, 53, 4793–4797. [Google Scholar] [CrossRef]
Supplementary Files
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yang, B.; Liao, S.; Lin, X.; Wang, J.; Liu, J.; Zhou, X.; Yang, X.; Liu, Y. New Sinularianin Sesquiterpenes from Soft Coral Sinularia sp. Mar. Drugs 2013, 11, 4741-4750. https://doi.org/10.3390/md11124741
Yang B, Liao S, Lin X, Wang J, Liu J, Zhou X, Yang X, Liu Y. New Sinularianin Sesquiterpenes from Soft Coral Sinularia sp. Marine Drugs. 2013; 11(12):4741-4750. https://doi.org/10.3390/md11124741
Chicago/Turabian StyleYang, Bin, Shengrong Liao, Xiuping Lin, Junfeng Wang, Juan Liu, Xuefeng Zhou, Xianwen Yang, and Yonghong Liu. 2013. "New Sinularianin Sesquiterpenes from Soft Coral Sinularia sp." Marine Drugs 11, no. 12: 4741-4750. https://doi.org/10.3390/md11124741








