Clinical Trial: Marine Lipid Suppositories as Laxatives
Abstract
:1. Introduction
2. Materials and Methods
3. Clinical Trial
| Study group week 1 (n) | Study group week 2 (n) | Control group week 1 (n) | Control group week 2 (n) | |
|---|---|---|---|---|
| Itching | ||||
| 1 (none) | 14 | 12 | 13 | 13 |
| 2 | 1 | 3 | 2 | 2 |
| 3 | 0 | 0 | 0 | 0 |
| 4 | 0 | 0 | 0 | 0 |
| 5 (severe) | 0 | 0 | 0 | 0 |
| Pain | ||||
| 1 (none) | 12 | 12 | 14 | 14 |
| 2 | 2 | 3 | 1 | 1 |
| 3 | 1 | 0 | 0 | 0 |
| 4 | 0 | 0 | 0 | 0 |
| 5 (severe) | 0 | 0 | 0 | 0 |
| Mucus | 2 | 2 | 1 | 0 |
| Blood | 2 | 1 | 0 | 0 |
| Urge for defecation | ||||
| None | 1 | 1 | 9 | 10 |
| 1–5 min | 7 | 7 | 1 | 1 |
| 5–10 min | 5 | 7 | 3 | 2 |
| 10–30 min | 1 | 0 | 2 | 2 |
| Longer | 1 | 0 | 0 | 0 |
| Bowel movement | ||||
| None | 1 | 2 | 10 | 10 |
| 1–5 min | 6 | 6 | 0 | 1 |
| 5–10 min | 4 | 4 | 1 | 1 |
| 10–30 min | 2 | 3 | 3 | 3 |
| Longer | 2 | 0 | 1 | 0 |
| Disturbing Smell | ||||
| 1 | 5 | 5 | 14 | 14 |
| 2 | 5 | 5 | 0 | 1 |
| 3 | 4 | 2 | 1 | 0 |
| 4 | 1 | 3 | 0 | 0 |
| 5 | 0 | 0 | 0 | 0 |
4. Results
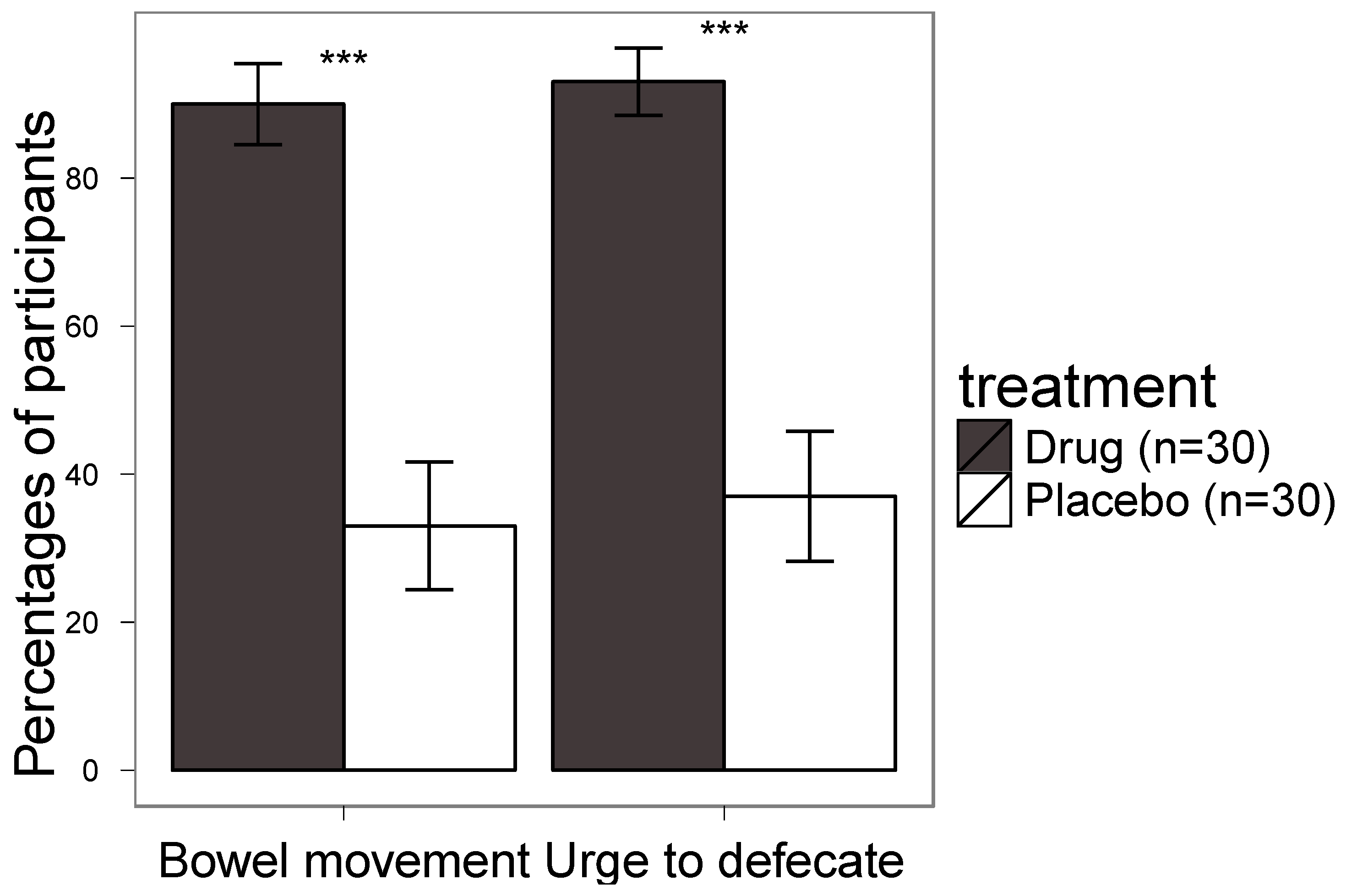
5. Discussion
6. Conclusion
Acknowledgements
Declare
References
- Loftsson, T.; Gudmundsdottir, T.K.; Fridriksdottir, H.; Sigurdardottir, A.M.; Thorkelsson, J.; Gudmundsson, G.; Hjaltason, B. Fatty acids from cod-liver oil as skin penetration enhancers. Pharmazie 1995, 50, 188–190. [Google Scholar]
- Genuis, S.J. A fishy recommendation: Omega-3 fatty acid intake in pregnancy. BJOG Int. J. Obstet. Gynaecol. 2008, 115, 1–4. [Google Scholar] [CrossRef]
- Moyad, M.A. An introduction to dietary/supplemental omega-3 fatty acids for general health and prevention: Part I. Urol. Oncol. Semin. Orig. Investig. 2005, 23, 28–35. [Google Scholar] [CrossRef]
- Ward, O.P.; Singh, A. Omega-3/6 fatty acids: Alternative sources of production. Process Biochem. 2005, 40, 3627–3652. [Google Scholar] [CrossRef]
- Riediger, N.D.; Othman, R.A.; Suh, M.; Moghadasian, M.H. A Systemic Review of the Roles of n-3 Fatty Acids in Health and Disease. J. Am. Diet. Assoc. 2009, 109, 668–679. [Google Scholar]
- Singer, P.; Shapiro, H.; Theilla, M.; Anbar, R.; Singer, J.; Cohen, J. Anti-inflammatory properties of omega-3 fatty acids in critical illness: Novel mechanisms and an integrative perspective. Intensive Care Med. 2008, 34, 1580–1592. [Google Scholar] [CrossRef]
- Goldberg, R.J.; Katz, J. A meta-analysis of the analgesic effects of omega-3 polyunsaturated fatty acid supplementation for inflammatory joint pain. Pain 2007, 129, 210–223. [Google Scholar] [CrossRef]
- Grenyer, B.F.S.; Crowe, T.; Meyer, B.; Owen, A.J.; Grigonis-Deane, E.M.; Caputi, P.; Howe, P.R.C. Fish oil supplementation in the treatment of major depression: A randomised double-blind placebo-controlled trial. Prog. Neuropsychopharmacol. Biol. Psychiatry 2007, 31, 1393–1396. [Google Scholar] [CrossRef]
- Heird, W.C. The Role of Polyunsaturated Fatty Acids in Term and Preterm Infants and Breastfeeding Mothers. Pediatr. Clin. N. Am. 2001, 48, 173–188. [Google Scholar] [CrossRef]
- Murphy, B.L.; Stoll, A.L.; Harris, P.Q.; Ravichandran, C.; Babb, S.M.; Carlezon, W.A., Jr.; Cohen, B.M. Omega-3 Fatty Acid Treatment, With or Without Cytidine, Fails to Show Therapeutic Properties in Bipolar Disorder: A Double-Blind, Randomized Add-on Clinical Trial. J. Clin. Psychopharmacol. 2012, 32, 699–703. [Google Scholar]
- Horrobin, D.F.; Bennett, C.N. Depression and bipolar disorder: Relationships to impaired fatty acid and phospholipid metabolism and to diabetes, cardiovascular disease, immunological abnormalities, cancer, ageing and osteoporosis—Possible candidate genes. Prostaglandins Leukot. Essent. Fat. Acids 1999, 60, 217–234. [Google Scholar] [CrossRef]
- Jacobson, J.L.; Jacobson, S.W.; Muckle, G.; Kaplan-Estrin, M.; Ayotte, P.; Dewailly, E. Beneficial Effects of a Polyunsaturated Fatty Acid on Infant Development: Evidence from the Inuit of Arctic Quebec. J. Pediatr. 2008, 152, 356–364.e1. [Google Scholar]
- Kris-Etherton, P.M.; Harris, W.S.; Appel, L.J.; Comm, N. Fish consumption, fish oil, omega-3 fatty acids, and cardiovascular disease. Circulation 2002, 106, 2747–2757. [Google Scholar] [CrossRef]
- Babu, U.; Wiesenfeld, P.; Gaines, D.; Raybourne, R.B. Effect of long chain fatty acids on Salmonella killing, superoxide and nitric oxide production by chicken macrophages. Int. J. Food Microbiol. 2009, 132, 67–72. [Google Scholar]
- Berquin, I.M.; Edwards, I.J.; Chen, Y.Q. Multi-targeted therapy of cancer by omega-3 fatty acids. Cancer Lett. 2008, 269, 363–377. [Google Scholar] [CrossRef]
- Shin, S.Y.; Bajpai, V.K.; Kim, H.R.; Kang, S.C. Antibacterial activity of eicosapentaenoic acid (EPA) against foodborne and food spoilage microorganisms. LWT Food Sci. Technol. 2007, 40, 1515–1519. [Google Scholar] [CrossRef]
- Shin, S.Y.; Bajpai, V.K.; Kim, H.R.; Kang, S.C. Antibacterial activity of bioconverted eicosapentaenoic (EPA) and docosahexaenoic acid (DHA) against foodborne pathogenic bacteria. Int. J. Food Microbiol. 2007, 113, 233–236. [Google Scholar] [CrossRef]
- Bajpai, V.K.; Shin, S.Y.; Kim, H.R.; Kang, S.C. Anti-fungal action of bioconverted eicosapentaenoic acid (bEPA) against plant pathogens. Ind. Crop. Prod. 2008, 27, 136–141. [Google Scholar] [CrossRef]
- Loftsson, T.; Thormar, H.; Ólafsson, J.H.; Gunnarsdóttir, T.M.; Hjaltason, B.; Guðmundsson, G. Fatty acid extract from cod-liver oil: Activity against herpes simplex virus and enhancement of transdermal delivery of acyclovir. Pharm. Pharmacol. Commun. 1998, 4, 287–291. [Google Scholar]
- Thompson, L.; Spiller, R.C. Impact of polyunsaturated fatty acids on human colonic bacterial metabolism: An in vitro and in vivo study. Br. J. Nutr. 1995, 74, 733–741. [Google Scholar] [CrossRef]
- Teitelbaum, J.E.; Allan Walker, W. Review: The role of omega-3 fatty acids in intestinal inflammation. J. Nutr. Biochem. 2001, 12, 21–32. [Google Scholar] [CrossRef]
- Johanson, J. Nonsurgical treatment of hemorrhoids. J. Gastrointest. Surg. 2002, 6, 290–294. [Google Scholar] [CrossRef]
- Kaidar-Person, O.; Person, B.; Wexner, S.D. Hemorrhoidal Disease: A Comprehensive Review. J. Am. Coll. Surg. 2007, 204, 102–117. [Google Scholar] [CrossRef]
- Madoff, R.D.; Fleshman, J.W. American gastroenterological association technical review on the diagnosis and treatment of hemorrhoids. Gastroenterology 2004, 126, 1463–1473. [Google Scholar] [CrossRef]
- Billingham, R.P.; Isler, J.T.; Kimmins, M.H.; Nelson, J.M.; Schweitzer, J.; Murphy, M.M. The diagnosis and management of common anorectal disorders. Curr. Probl. Surg. 2004, 41, 586–645. [Google Scholar] [CrossRef]
- Kroes, R.; Schaefer, E.J.; Squire, R.A.; Williams, G.M. A review of the safety of DHA45-oil. Food Chem. Toxicol. 2003, 41, 1433–1446. [Google Scholar] [CrossRef]
- Spiller, R.C.; Brown, M.L.; Phillips, S.F. Decreased fluid tolerance, accelerated transit, and abnormal motility of the human colon induced by oleic acid. Gastroenterology 1986, 91, 100–107. [Google Scholar]
- Klaschik, E.; Nauck, F.; Ostgathe, C. Constipation—modern laxative therapy. Support. Care Cancer 2003, 11, 679–685. [Google Scholar] [CrossRef]
- Evans, I.L. The use of bisacodyl suppositories in preparation for sigmoidoscopy. Gut 1964, 5, 271–273. [Google Scholar] [CrossRef]
- Samples Availability: Available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ormarsson, O.T.; Geirsson, T.; Bjornsson, E.S.; Jonsson, T.; Moller, P.H.; Loftsson, T.; Stefansson, E. Clinical Trial: Marine Lipid Suppositories as Laxatives. Mar. Drugs 2012, 10, 2047-2054. https://doi.org/10.3390/md10092047
Ormarsson OT, Geirsson T, Bjornsson ES, Jonsson T, Moller PH, Loftsson T, Stefansson E. Clinical Trial: Marine Lipid Suppositories as Laxatives. Marine Drugs. 2012; 10(9):2047-2054. https://doi.org/10.3390/md10092047
Chicago/Turabian StyleOrmarsson, Orri Thor, Thormodur Geirsson, Einar Stefan Bjornsson, Tomas Jonsson, Pall Hegli Moller, Thorsteinn Loftsson, and Einar Stefansson. 2012. "Clinical Trial: Marine Lipid Suppositories as Laxatives" Marine Drugs 10, no. 9: 2047-2054. https://doi.org/10.3390/md10092047
APA StyleOrmarsson, O. T., Geirsson, T., Bjornsson, E. S., Jonsson, T., Moller, P. H., Loftsson, T., & Stefansson, E. (2012). Clinical Trial: Marine Lipid Suppositories as Laxatives. Marine Drugs, 10(9), 2047-2054. https://doi.org/10.3390/md10092047





