Anti-Oxidative, Anti-Tumor-Promoting, and Anti-Carcinogensis Activities of Nitroastaxanthin and Nitrolutein, the Reaction Products of Astaxanthin and Lutein with Peroxynitrite
Abstract
:1. Introduction
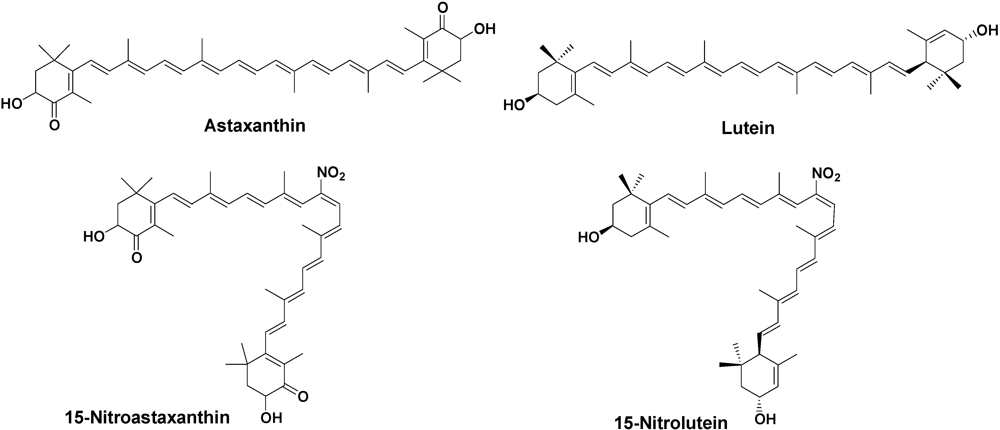
2. Results and Discussion
2.1. Inhibition of Tyrosine Nitration with Peroxynitrite by Astaxanthin and Lutein
2.2. Quenching Effects of 1O2 by 15-Nitroastaxanthin and 15-Nitrolutein
| Concentration\Compounds | 100 (μM) | 10 (μM) | 1 (μM) | IC50 (μM) |
|---|---|---|---|---|
| Astaxanthin | 93.8 | 65.5 | 30.8 | 7.0 |
| 15-Nitroastaxanthin | 91.8 | 48.5 | 5.0 | 20.0 |
| Lutein | 75.8 | 38.5 | 1.0 | 60.5 |
| 15-Nitrolutein | 72.2 | 36.5 | 0.8 | 71.5 |
2.3. Anti-Tumor-Promoting and Anti-Carcinogensis Activities of 15-Nitroastaxanthin and 15-Nitrolutein
| Concentration (mol ratio/TPA) b | 1000 | 500 | 100 | 10 | IC50 (nmol) |
|---|---|---|---|---|---|
| Compounds | values | ||||
| Astaxanthin | 5.0 (70) c | 29.0 | 78.0 | 100 | 307 |
| 15-Nitroastaxanthin | 4.1 (60) c | 28.5 | 76.9 | 100 | 300 |
| Lutein | 2.9 (70) c | 24.5 | 75.4 | 97.6 | 283 |
| 15-Nitrolutein | 1.6 (60) c | 23.3 | 74.1 | 95.4 | 277 |
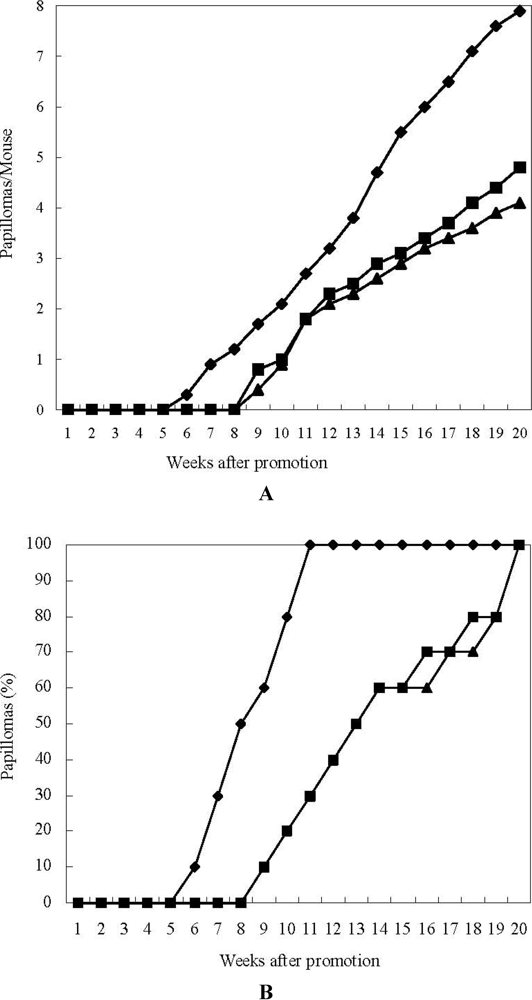
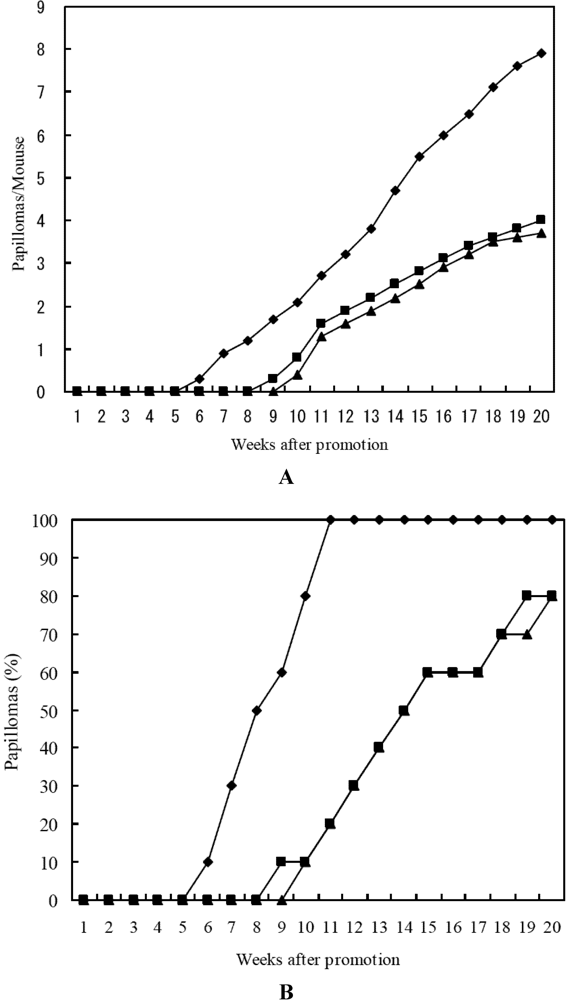
3. Experimental Section
3.1. Preparation of 15-Nitroastaxanthin and 15-Nitrolutein
3.2. Inhibition of Tyrosine Nitration with Peroxynitrite by Carotenoids
3.3. Measurement of 1O2 Quenching Activity
3.4. In Vitro Epstein-Barr Virus Early Antigen (EBV-EA) Activation Induction Effect
3.5. In Vivo Two-Stage Carcinogensis Assay on Mouse Skin Initiated by DMBA and Promoted by TPA
4. Conclusions
Acknowledgements
References
- Maoka, T.; Etoh, H. Some Biological Functions of Carotenoids in Japanese Food. In Functional Foods of the East; Shi, J., Ho, C.-T., Shahidi, F., Eds.; CRC Press: Boca Raton, FL, USA, 2010; pp. 85–97. [Google Scholar]
- Halliwell, B. Free radicals, antioxidants, and human disease: Curiosity, cause or consequence? Lancet 1994, 344, 721–724. [Google Scholar] [CrossRef]
- Lee, S.-J.; Bai, S.-K.; Lee, K.-S.; Namkoong, S.; Na, H.-J.; Ha, K.-S.; Han, J.-A.; Yim, S.-V.; Chang, K.; Kwon, Y.-G.; et al. Astaxanthin inhibits nitric oxide production and inflammatory gene expression by suppressing IκB kinase-dependent NF-κB activation. Mol. Cells 2003, 16, 97–105. [Google Scholar]
- Yoshioka, R.; Hayakawa, T.; Ishizuka, K.; Kulkarni, A.; Terada, Y.; Maoka, T.; Etoh, H. Nitration reaction of astaxanthin and β-carotene by peroxynitrite. Tetrahedron Lett. 2006, 47, 3637–3640. [Google Scholar]
- Hayakawa, T.; Kulkarni, A.; Terada, Y.; Maoka, T.; Etoh, H. Reaction of astaxanthin with peroxynitrite. Biosci. Biotechnol. Biochem. 2008, 72, 2716–2722. [Google Scholar]
- Tsuboi, M.; Etoh, H.; Yomoda, Y.; Kato, K.; Kato, H.; Kulkarni, A.; Terada, Y.; Maoka, T.; Mori, H.; Inakuma, T. Nitration reaction of lutein with peroxynitrite. Tetrahedron Lett. 2010, 51, 676–678. [Google Scholar]
- Niwa, T.; Doi, U.; Kato, Y.; Osawa, T. Antioxidative properties of phenolic antioxidants isolated from corn steep liquor. J. Agric. Food Chem. 2001, 49, 177–182. [Google Scholar]
- Kato, Y.; Ogino, Y.; Aoki, T.; Uchida, K.; Kawakishi, S.; Osawa, T. Phenolic antioxidants prevent peroxynitrite-derived collagen modification in vitro. J. Agric. Food. Chem. 1997, 45, 3004–3009. [Google Scholar]
- Tsuda, T.; Kato, Y.; Osawa, T. Mechanism for the peroxynitrite scavenging activity by anthocyanins. FEBS Lett. 2000, 484, 207–210. [Google Scholar]
- Goss, S.P.A.; Hogg, N.; Kalyanaraman, B. The effect of α-tocopherol on the nitration of γ-tocopherol by peroxynitrite. Arch. Biochem. Biophys. 1999, 363, 333–340. [Google Scholar]
- Tuboi, M.; Etoh, H.; Kato, K.; Nakatsugawa, H.; Kato, H.; Maejima, Y.; Natsumoto, G.; Mori, H.; Hosokawa, M.; Miyashita, K.; et al. Nitrocapsanthin and nitrofucoxanthin, respective products of capsanthin and fucoxanthin reaction with peroxynitrite. J. Agric. Food Chem. 2011, 59, 10572–10578. [Google Scholar]
- Groves, J.T.; Marla, S.S. Peroxynitrite-induced DNA stanrard scission mediated by a magnanese porphyrin. J. Am. Chem. Soc. 1995, 117, 9578–9579. [Google Scholar]
- Hirayama, O.; Nakamura, K.; Hamada, S.; Kobayashi, Y. Singlet oxygen quenching ability of naturally occurring carotenoids. Lipid 1994, 29, 149–150. [Google Scholar]
- Kobayashi, M.; Sakamoto, Y. Singlet oxygen quenching ability of astaxanthin esters from the green alga Haematococcus pluvialis. Biotechnol. Lett. 1999, 21, 265–269. [Google Scholar] [CrossRef]
- Osawa, A.; Ishii, Y.; Sasamura, N.; Morita, M.; Kasai, H.; Maoka, T.; Shindo, K. Characterization and antioxidative activities of rare C50 carotenoids-sarcinaxanthin, sarcinaxanthin monoglucoside, and sarcinaxanthin diglucoside-obtained from Micrococcus yunnanensis. J. Oleo Sci. 2010, 59, 653–659. [Google Scholar] [CrossRef]
- Miki, W. Biological functions and activities of animal carotenoids. Pure Appl. Chem. 1991, 63, 141–146. [Google Scholar]
- Shimidzu, N.; Goto, M.; Miki, W. Carotenoids as singlet oxygen quenchers in marine organisms. Fish. Sci. 1996, 62, 134–137. [Google Scholar]
- Tsushima, M.; Maoka, T.; Katsuyama, M.; Kozuka, M.; Matsuno, T.; Tokuda, H.; Nishino, H.; Iwashima, A. Inhibitory effect of natural carotenoids on Epstein-Barr virus activation activity of a tumor promoter in Raji cells. A screening study for anti-tumor promoters. Biol. Pharm. Bull. 1995, 18, 227–233. [Google Scholar] [CrossRef]
- Maoka, T.; Mochida, K.; Kozuka, M.; Ito, Y.; Fujiwara, Y.; Hashimoto, K.; Enjo, F.; Ogata, M.; Nobukuni, Y.; Tokuda, H.; et al. Cancer chemopreventive activity of carotenoids in the fruits of red paprika Capsicum annuum L. Cancer Lett. 2001, 172, 103–109. [Google Scholar] [CrossRef]
- Samples Availability: Available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Maoka, T.; Tokuda, H.; Suzuki, N.; Kato, H.; Etoh, H. Anti-Oxidative, Anti-Tumor-Promoting, and Anti-Carcinogensis Activities of Nitroastaxanthin and Nitrolutein, the Reaction Products of Astaxanthin and Lutein with Peroxynitrite. Mar. Drugs 2012, 10, 1391-1399. https://doi.org/10.3390/md10061391
Maoka T, Tokuda H, Suzuki N, Kato H, Etoh H. Anti-Oxidative, Anti-Tumor-Promoting, and Anti-Carcinogensis Activities of Nitroastaxanthin and Nitrolutein, the Reaction Products of Astaxanthin and Lutein with Peroxynitrite. Marine Drugs. 2012; 10(6):1391-1399. https://doi.org/10.3390/md10061391
Chicago/Turabian StyleMaoka, Takashi, Harukuni Tokuda, Nobutaka Suzuki, Hideaki Kato, and Hideo Etoh. 2012. "Anti-Oxidative, Anti-Tumor-Promoting, and Anti-Carcinogensis Activities of Nitroastaxanthin and Nitrolutein, the Reaction Products of Astaxanthin and Lutein with Peroxynitrite" Marine Drugs 10, no. 6: 1391-1399. https://doi.org/10.3390/md10061391




