Antioxidant Activity of Hawaiian Marine Algae
Abstract
:1. Introduction
| Species | Collection Location, Island | Collection Date (day/month/year) | Depth (m) | Sample Number |
|---|---|---|---|---|
| Phaeophyta | ||||
| Colpomenia sinuosa | Leleiwi Beach Park, Hawaii | 7, November, 2009 | −0.5 | A0014 |
| Dictyopteris plagiogramma | Makai Pier, Oahu | 5, August, 2003 | −3.0 | A0023 |
| Hydroclathrus clathratus | Leleiwi Beach Park, Hawaii | 7, November, 2009 | −1.5 | A0016 |
| Sargassum echinocarpum | Richardson’s Ocean Park, Hawaii | 6, September, 2004 | −0.5 | A0030 |
| Sargassum obtusifolium | Richardson’s Ocean Park, Hawaii | 6, September, 2004 | −0.5 | A0033 |
| Spatoglossum macrodontum | Penguin Bank, off Molokai | 17 September, 2004 | −78.0 | A0021 |
| Spatoglossum macrodontum | Penguin Bank, off Molokai | March 2009 | −81.0 | A0042 |
| Turbinaria ornata | Richardson’s Ocean Park, Hawaii | 17 August, 2004 | −0.5 | A0031 |
| Turbinaria ornata | Waikoloa, Hawaii | 12, March, 2011 | −0.5 | A0050 |
| Chlorophyta | ||||
| Chaetomorpha antennina | Richardson’s Ocean Park, Hawaii | 26, October, 2009 | 0.25 | A0003 |
| Chaetomorpha antennina | Punaluu Beach Park, Hawaii | 15, July, 2004 | 0.25 | A0029 |
| Chlorodesmis caespitosa | Onekahakaha Beach Park, Hawaii | 29 April, 2010 | −1.0 | A0038 |
| Codium mamillosum | Penguin Bank, off Molokai | 22, September, 2004 | −78.0 | A0020 |
| Derbesia tenuissima | Fish tank, Marine Science Building | 1, March, 2010 | −0.5 | A0036 |
| Gayralia oxysperma | Richardson’s Ocean Park, Hawaii | 26, October, 2009 | 0.1 | A0004 |
| Gayralia oxysperma | Richardson’s Ocean Park, Hawaii | 6 September, 2004 | 0.1 | A0034 |
| Rhizoclonium africanum | Leleiwi Beach Park, Hawaii | 7, November, 2009 | 0.1 | A0013 |
| Ulva sp. | Penguin Bank, off Molokai | 26, March, 2009 | −104 | A0041 |
| Rhodophyta | ||||
| Acanthophora pacifica | Punaluu Beach Park, Hawaii | 15, July, 2004 | −2.0 | A0027 |
| Acanthophora spicifera | Richardson’s Ocean Park, Hawaii | 26, October, 2009 | −0.5 | A0002 |
| Ahnfeltiopsis concinna | Four-mile Beach Park, Hawaii | 2, November, 2009 | 0.2 | A0005 |
| Ahnfeltiopsis concinna | King’s Landing, Hawaii | 7, November, 2009 | 0.2 | A0007 |
| Ahnfeltiopsis concinna | Richardson’s Ocean Park, Hawaii | 26, October, 2009 | 0.2 | A0001 |
| Amansia glomerata | Kapoho, Hawaii | 9, November, 2009 | −1.0 | A0018 |
| Chondrus ocellatus | Leleiwi Beach Park, Hawaii | 7, November, 2009 | −0.5 | A0011 |
| Halymenia formosa | Mahaiula Bay, Hawaii | 1, August, 2003 | −2.5 | A0028 |
| Hypnea spinella | King’s Landing, Hawaii | 7, November, 2009 | 0.1 | A0008 |
| Rhodophyta | ||||
| Laurencia galtsoffii | Kapoho, Hawaii | 10, November, 2009 | −2.0 | A0019 |
| Laurencia mcdermidiae | Makapuu, Oahu | 5, August, 2003 | 0.1 | A0022 |
| Peyssonnelia inamoena | Penguin Bank, off Molokai | 25, March, 2009 | −109 | A0039 |
| Polyopes hakalauensis | King’s Landing, Hawaii | 7, November, 2009 | 0.1 | A0009 |
| Polysiphonia howei | Leleiwi Beach Park, Hawaii | 7, November, 2009 | 0.1 | A0012 |
| Portieria hornemanni | Richardson’s Ocean Park, Hawaii | 2, February, 2010 | −1.0 | A0037 |
| Pterocladiella capillacea | Four-mile Beach Park, Hawaii | 2, November, 2009 | 0.0 | A0006 |
| Pterocladiella capillacea | King’s Landing, Hawaii | 7, November, 2009 | 0.0 | A0010 |
| Stenopeltis gracilis | Leleiwi Beach Park, Hawaii | 7, November, 2009 | −1.0 | A0015 |
| Tricleocarpa cylindrica | Richardson’s Ocean Park, Hawaii | 6, September, 2004 | −1.0 | A0032 |
2. Results

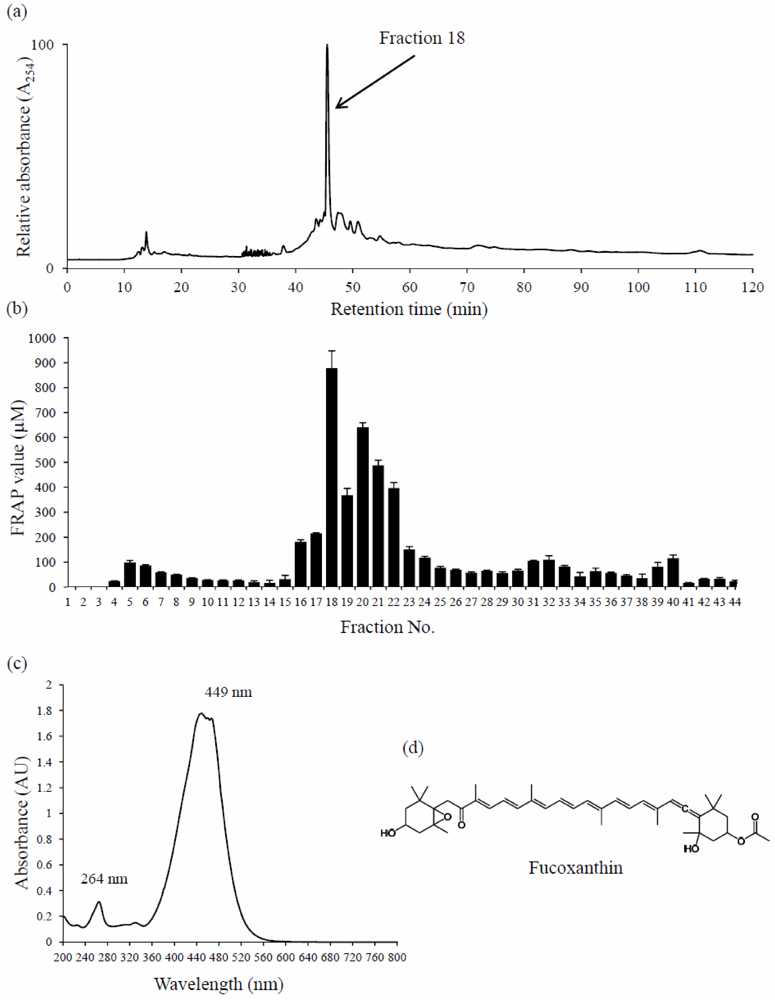
3. Discussion
3.1. Phaeophyta
3.2. Chlorophyta
3.3. Rhodophyta
4. Experimental Section
4.1. Macroalgae Collection
4.2. Sample Preparation
4.3. Extractions
4.4. Antioxidant Activity Assay
4.5. Bioassay-Guided Fractionation, Isolation, and Identification of the Active Principle
4.6. Statistics
5. Conclusions
Acknowledgments
References
- Cardozo, K.H.M.; Guaratini, T.; Barros, M.P.; Falcao, V.R.; Tonon, A.P.; Lopes, N.P.; Campos, S.; Torres, M.A.; Souza, A.O.; Colepicolo, P.; et al. Review: Metabolites from algae with economical impact. Comp. Biochem. Physiol. C 2007, 146, 60–78. [Google Scholar]
- Faulkner, D.J. Marine natural products. Nat. Prod. Rep. 2002, 19, 1–49. [Google Scholar]
- Blunt, J.W.; Copp, B.R.; Munro, M.H.; Northcote, P.T.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2011, 28, 196–268. [Google Scholar]
- Smit, A.J. Medicinal and pharmaceutical uses of seaweed natural products: A review. J. Appl. Phycol. 2004, 16, 245–262. [Google Scholar]
- Mallick, N.; Mohn, F.H. Reactive oxygen species: Response of algal cells. J. Plant Physiol. 2000, 157, 183–193. [Google Scholar]
- Matanjun, P.; Mohamed, S.; Mustapha, N.M.; Muhammad, K.; Ming, C.H. Antioxidant activities and phenolics content of eight species of seaweeds from north Borneo. J. Appl. Phycol. 2008, 20, 367–373. [Google Scholar]
- Halliwell, B. Antioxidant defence mechanisms: From the beginning to the end (of the beginning). Free Radic. Res. 1999, 31, 261–272. [Google Scholar]
- Ozben, T. Oxidative stress and apoptosis: Impact on cancer therapy. J. Pharm. Sci. 2007, 96, 2181–2196. [Google Scholar]
- Vijayavel, K.; Martinez, J.A. In vitro antioxidant and antimicrobial activities of two Hawaiian marine limu: Ulva fasciata (Chlorophyta) and Gracilaria salicornia (Rhodophyta). J. Med. Food 2010, 13, 1494–1499. [Google Scholar]
- Cornish, M.L.; Garbary, D.J. Antioxidants from macroalgae: Potential applications in human health and nutrition. Algae 2010, 25, 155–171. [Google Scholar]
- Zubia, M.; Robledo, D.; Freile-Pelegrin, Y. Antioxidant activities in tropical marine macroalgae from the Yucatan Peninsula, Mexico. J. Appl. Phycol. 2007, 19, 449–458. [Google Scholar]
- Abbott, I.A. Marine Red Algae of the Hawaiian Islands; Bishop Museum Press: Honolulu, HI, USA, 1999. [Google Scholar]
- Abbott, I.A.; Huisman, J.M. Marine Green and Brown Algae of the Hawaiian Islands; Bishop Museum Press: Honolulu, HI, USA, 2004. [Google Scholar]
- McDermid, K.J.; Stuercke, B. Nutritional composition of edible Hawaiian seaweeds. J. Appl. Phycol. 2003, 15, 513–524. [Google Scholar]
- Hardt, I.H.; Fenical, W.; Cronin, G.; Hay, M.E. Acutilols, potent herbivore feeding deterrents from the tropical brown alga, Dictyota acutiloba. Phytochemistry 1996, 43, 71–73. [Google Scholar]
- Chandini, S.K.; Ganesan, P.; Bhaskar, N. In vitro antioxidant activities of three selected brown seaweeds of India. Food Chem. 2008, 107, 707–713. [Google Scholar]
- Ananthi, S.; Raghavendran, H.R.B.; Sunil, A.G.; Gayathri, V.; Ramakrishnan, G.; Vasanthi, H.R. In vitro antioxidant and in vivo anti-inflammatory potential of crude polysaccharide from Turbinaria ornata (marine brown alga). Food Chem. Toxicol. 2010, 48, 187–192. [Google Scholar]
- Chattopadhyay, N.; Ghosh, T.; Sinha, S.; Chattopadhyay, K.; Karmakar, P.; Ray, B. Polysaccharides from Turbinaria conoides: Structural features and antioxidant capacity. Food Chem. 2010, 118, 823–829. [Google Scholar]
- Takaichi, S. Carotenoids in algae: Distribution, biosynthesis and functions. Mar. Drugs 2011, 9, 1101–1118. [Google Scholar]
- Peng, J.; Yuan, J.-P.; Wu, C.-F.; Wang, J.-H. Fucoxanthin, a marine carotenoid present in seaweeds and diatoms: Metabolism and bioactivities relevant to human health. Mar. Drugs 2011, 9, 1806–1828. [Google Scholar]
- Sugawara, T.; Matsubara, K.; Akagi, R.; Mori, M.; Hirata, T. Antiangiogenic activity of brown algae fucoxanthin and its deacetylated product, fucoxanthiol. J. Agric. Food Chem. 2006, 54, 9805–9810. [Google Scholar]
- Maeda, H.; Hosokawa, M.; Sashima, T.; Funayama, K.; Miyashita, K. Fucoxanthin from edible seaweed, Undaria pinnatifida, shows antiobesity effect through UCP1 expression in white adipose tissues. Biochem. Biophys. Res. Commun. 2005, 332, 392–397. [Google Scholar] [CrossRef]
- Maeda, H.; Tsukui, T.; Sashima, T.; Hosokawa, M.; Miyashita, K. Seaweed carotenoid, fucoxanthin, as a multi-functional nutrient. Asia Pac. J. Clin. Nutr. 2008, 17, 196–199. [Google Scholar]
- Nishino, H. Cancer chemoprevention by natural carotenoids and their related compounds. J. Cell. Biochem. Suppl. 1995, 22, 231–235. [Google Scholar]
- Sangeetha, R.K.; Bhaskar, N.; Baskaran, V. Comparative effects of β-carotene and fucoxanthin on retinol deficiency induced oxidative stress in rats. Mol. Cell. Biochem. 2009, 331, 59–67. [Google Scholar]
- Sachindra, N.M.; Sato, E.; Maeda, H.; Hosokawa, M.; Niwano, Y.; Kohno, M.; Miyashita, K. Radical scavenging and singlet oxygen quenching activity of marine carotenoid fucoxanthin and its metabolites. J. Agric. Food Chem. 2007, 55, 8516–8522. [Google Scholar]
- Kim, K.N.; Heo, S.J.; Kang, S.M.; Ahn, G.; Jeon, Y.J. Fucoxanthin induces apoptosis in human leukemia HL-60 cells through a ROS-mediated Bcl-xL pathway. Toxicol. in Vitro 2010, 24, 1648–1654. [Google Scholar]
- Heo, S.J.; Cha, S.H.; Lee, K.W.; Cho, S.K.; Jeon, Y.J. Antioxidant activities of Chlorophyta and Phaeophyta from Jeju Island. Algae 2005, 20, 251–260. [Google Scholar]
- Wang, H.; Chiu, L.C.M.; Ooi, V.E.C.; Ang, P.O. Seaweed polysaccharides with anticancer potential. Bot. Mar. 2008, 51, 313–319. [Google Scholar]
- Kang, K.; Park, Y.; Hwang, H.J.; Kim, S.H.; Lee, J.G.; Shin, H.-C. Antioxidative properties of brown algae polyphenolics and their perspectives as chemopreventive agents against vascular risk factors. Arch. Pharm. Res. 2003, 26, 286–293. [Google Scholar]
- Firdaus, M.; Astawan, M.; Muchtadi, D.; Wresdiyati, T.; Waspadji, S.; Karyono, S.S. Prevention of endothelial dysfunction in streptozotocin-induced diabetic rats by Sargassum echinocarpum extract. Med. J. Indones. 2010, 19, 32–35. [Google Scholar]
- Cassolato, J.E.F.; Noseda, M.D.; Pujol, C.A.; Pellizzari, F.M.; Damonte, E.B.; Duarte, M.E.R. Chemical structure and antiviral activity of the sulfated heterorhamnan isolated from the green seaweed Gayralia oxysperma. Carbohydr. Res. 2008, 343, 3085–3095. [Google Scholar]
- Li, H.; Mao, W.; Zhang, X.; Qi, X.; Chen, Y.; Chen, Y.; Xu, J.; Zhao, C.; Hou, Y.; Yang, Y.; et al. Structural characterization of an anticoagulant-active sulfated polysaccharide isolated from the green alga Monostroma latissimum. Carbohyd. Polym. 2011, 85, 394–400. [Google Scholar] [CrossRef]
- Premalatha, M.; Dhasarathan, P.; Theriappan, P. Phytochemical characterization and antimicrobial efficiency of seaweed samples, Ulva fasciata and Chaetomorpha antennina. Int. J. Pharm. Biol. Sci. 2011, 2, 288–293. [Google Scholar]
- Murcia, M.A.; Jimenez, A.M.; Martinez-Tome, M. Vegetables antioxidant losses during industrial processing and refrigerated storage. Food Res. Int. 2009, 42, 1046–1052. [Google Scholar]
- Fujimoto, K.; Ohmura, H.; Kaneda, T. Screening for antioxygenic compounds in marine algae and bromophenols as effective principles in a red alga Polysiphonia ulceolata. Bull. Jpn. Soc. Sci. Fish. 1985, 51, 1139–1143. [Google Scholar]
- Kim, Y.A.; Kong, C.S.; Um, Y.R.; Lee, J.I.; Nam, T.J.; Seo, Y. Antioxidant efficacy of extracts from a variety of seaweeds in a cellular system. Ocean Sci. J. 2008, 43, 31–37. [Google Scholar]
- De la Coba, F.; Aguilera, J.; Figueroa, F.L.; de Galvez, M.V.; Herrera, E. Antioxidant activity of mycosporine-like amino acids isolated from three red macroalgae and one marine lichen. J. Appl. Phycol. 2008, 21, 161–169. [Google Scholar]
- Huisman, J.M.; Abbott, I.A.; Smith, C.M. Hawaiian Reef Plants; University of Hawaii Sea Grant College Program: Honolulu, HI, USA, 2007. [Google Scholar]
- Benzie, I.F.F.; Strain, J.J. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar]
- Benzie, I.F.F.; Strain, J.J. Ferric reducing antioxidant power assay: Direct measure of total antioxidant activity of biological fluids and modified version for simultaneous measurement of total antioxidant power and ascorbic acid concentration. Meth. Enzymol. 1999, 299, 15–27. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kelman, D.; Posner, E.K.; McDermid, K.J.; Tabandera, N.K.; Wright, P.R.; Wright, A.D. Antioxidant Activity of Hawaiian Marine Algae. Mar. Drugs 2012, 10, 403-416. https://doi.org/10.3390/md10020403
Kelman D, Posner EK, McDermid KJ, Tabandera NK, Wright PR, Wright AD. Antioxidant Activity of Hawaiian Marine Algae. Marine Drugs. 2012; 10(2):403-416. https://doi.org/10.3390/md10020403
Chicago/Turabian StyleKelman, Dovi, Ellen Kromkowski Posner, Karla J. McDermid, Nicole K. Tabandera, Patrick R. Wright, and Anthony D. Wright. 2012. "Antioxidant Activity of Hawaiian Marine Algae" Marine Drugs 10, no. 2: 403-416. https://doi.org/10.3390/md10020403
APA StyleKelman, D., Posner, E. K., McDermid, K. J., Tabandera, N. K., Wright, P. R., & Wright, A. D. (2012). Antioxidant Activity of Hawaiian Marine Algae. Marine Drugs, 10(2), 403-416. https://doi.org/10.3390/md10020403




