Chemical Composition and Antioxidant/Antimicrobial Activities in Supercritical Carbon Dioxide Fluid Extract of Gloiopeltis tenax
Abstract
:1. Introduction
2. Results and Discussion
2.1. Gas Chromatography-Mass Spectrometry (GC-MS) Analysis of CO2-SFE Extracts from G. tenax

| NO. | RT a | Constituents | KI b | % c |
|---|---|---|---|---|
| 1 | 7.890 | p-hydroxybenzaldehyde | 1374.4 | 0.57 |
| 2 | 9.302 | (−)-thujopsene | 1437.1 | 4.68 |
| 3 | 10.484 | α-curcumene | 1484.0 | 1.54 |
| 4 | 10.833 | α-zingiberene | 1497.8 | 2.98 |
| 5 | 11.285 | (+)-cuparene | 1511.4 | 0.28 |
| 6 | 11.322 | (−)-β-bisabolene | 1512.5 | 1.00 |
| 7 | 14.703 | cedrol | 1607.3 | 3.91 |
| 8 | 16.431 | vanillylacetone | 1644.8 | 1.92 |
| 9 | 19.016 | n-heptadecane | 1700.7 | 10.30 |
| 10 | 23.014 | myristic acid | 1769.8 | 2.85 |
| 11 | 27.614 | fitone | 1842.1 | 2.53 |
| 12 | 33.382 | methhyl hexadecanoate | 1927.6 | 1.32 |
| 13 | 37.460 | palmitic acid | 1987.3 | 21.21 |
| 14 | 41.236 | linoleic acid | 2092.9 | 0.23 |
| 15 | 41.354 | hexadeca-1,4-lactone | 2096.5 | 0.57 |
| 16 | 42.602 | cis-9-octadecenoic acid | 2153.2 | 0.73 |
| 17 | 43.002 | stearic acid | 2172.0 | 0.93 |
| 18 | 46.128 | oleamide | 2361.5 | 0.24 |
| 19 | 46.948 | 2,2′-methylenebis(6-tert-butyl-4-methylphenol) | 2422.4 | 1.14 |
| 20 | 48.040 | 2-monopalmitin | 2511.2 | 1.83 |
| 21 | 52.476 | cholesta-4,6-dien-3β-ol | 2894.9 | 6.62 |
| 22 | 56.619 | cholesterol | 3122.3 | 5.74 |
| 23 | 58.639 | cholesta-3,5-dien-7-one | 3196.6 | 0.45 |
2.2. Antioxidant Activity Assays
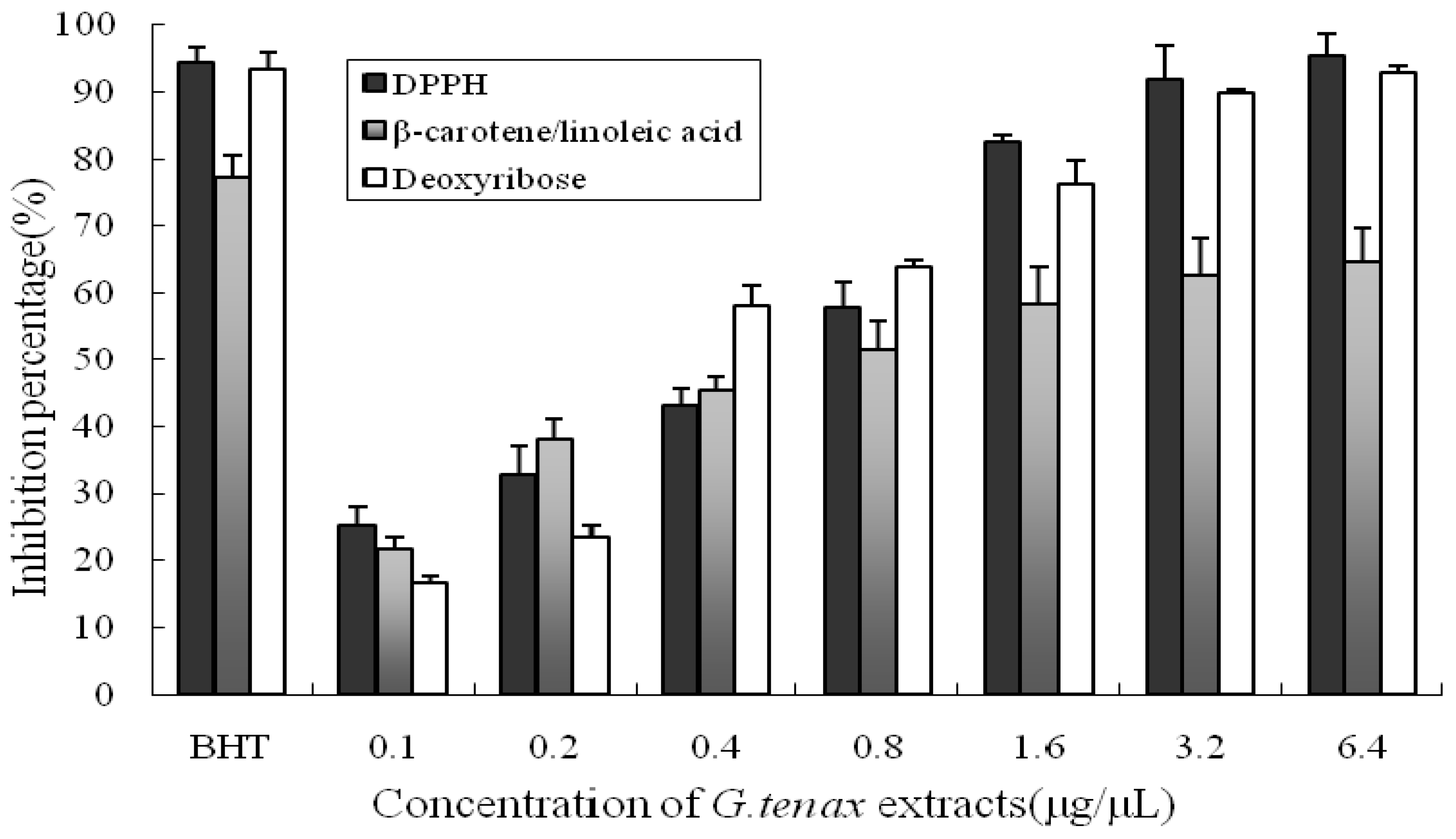
| Sample | DPPH | ∆S β-Carotene/Linoleic Acid | Deoxyribose |
|---|---|---|---|
| 0.1 μg/μL | 0.2206 ± 0.0079 * | 0.0154 ± 0.0003 * | 0.7544 ± 0.0091 * |
| 0.2 μg/μL | 0.1979 ± 0.0124 * | 0.0121 ± 0.0006 * | 0.6925 ± 0.0159 * |
| 0.4 μg/μL | 0.1680 ± 0.0078 * | 0.0107 ± 0.0004 * | 0.3792 ± 0.0277 * |
| 0.8 μg/μL | 0.1237 ± 0.0106 * | 0.0095 ± 0.0009 * | 0.3255 ± 0.0091 * |
| 1.6 μg/μL | 0.0514 ± 0.0030 * | 0.0082 ± 0.0011 * | 0.2136 ± 0.0299 * |
| 3.2 μg/μL | 0.0235 ± 0.0147 * | 0.0073 ± 0.0011 * | 0.0911 ± 0.0049 * |
| 6.4 μg/μL | 0.0134 ± 0.0097 * | 0.0069 ± 0.0010 * | 0.0628 ± 0.0070 * |
| BHT (0.05 mM) | 0.0162 ± 0.0175 * | 0.0044 ± 0.0037 * | 0.0623 ± 0.0103 * |
| Blank | 0.2947 ± 0.0074 | 0.0196 ± 0.0053 | 0.9365 ± 0.0325 |
2.3. Antimicrobial Susceptibility Testing
| Bacterial strains | G. tenax extracts dose (mg) | MIC (mg/mL) | ||||
|---|---|---|---|---|---|---|
| 0.3 | 0.6 | 1.2 | 2.5 | 5.0 | ||
| Staphyloccocus aureus | 24.3 | 25.1 | 26.4 | 27.7 | 28.9 | 3.9 |
| Enterococcus faecalis | 11.2 | 15.6 | 20.3 | 24.5 | 29.3 | 7.8 |
| Pseudomonas aeruginosa | 10.2 | 15.5 | 23.1 | 24.2 | 26.6 | 15.6 |
| Escherichia coli | 15.4 | 18.9 | 20.3 | 21.6 | 23.2 | 3.9 |
3. Experimental Section
3.1. Plant Materials
3.2. Chemicals and Reagents
3.3. Supercritical Fluid Extraction with Carbon Dioxide
3.4. Gas Chromatography-Mass Spectrometry Analysis
3.5. Antioxidant Assays of Extracts
3.5.1. DPPH Radical-Scavenging System
3.5.2. β-Carotene/Linoleic Acid-Coupled Oxidation Reaction
3.5.3. Deoxyribose Degradation by Iron-Dependent Hydroxyl Radical
3.6. Antimicrobial Susceptibility Testing and Determination of Minimum Inhibitory Concentration (MIC)
3.7. Statistical Analyses
4. Conclusions
Acknowledgments
References
- Lim, B.L.; Ryu, I.H. Purification, structural characterization, and antioxidant activity of antioxidant substance from the red seaweed Gloiopeltis tenax. J. Med. Food 2009, 12, 442–451. [Google Scholar] [CrossRef]
- Fang, Z.; Jeong, S.Y.; Jung, H.A.; Choi, J.S.; Min, B.S.; Woo, M.H. Anticholinesterase and antioxidant constituents from Gloiopeltis furcata. Chem. Pharm. Bull. 2010, 58, 1236–1239. [Google Scholar] [CrossRef]
- Kumar, K.S.; Ganesan, K.; Rao, P. Antioxidant potential of solvent extracts of Kappaphycus alvarezii (Doty) Doty—An edible seaweed. Food Chem. 2008, 107, 289–295. [Google Scholar] [CrossRef]
- Barja, G. Free radicals and aging. Trends Neurosci. 2004, 27, 595–600. [Google Scholar] [CrossRef]
- Giugliano, D.; Ceriello, A.; Paolisso, G. Oxidative stress and diabetic vascular complications. Diabetes Care 1996, 19, 257–267. [Google Scholar]
- Jenner, P. Oxidative damage in neurodegenerative disease. Lancet 1994, 344, 796–798. [Google Scholar] [CrossRef]
- Loft, S.; Poulsen, H. Cancer risk and oxidative DNA damage in man. J. Mol. Med. 1996, 74, 297–312. [Google Scholar] [CrossRef]
- Takahashi, O.; Hiraga, K. Dose-response study of hemorrhagic death by dietary butylated hydroxytoluene (BHT) in male rats. Toxicol. Appl. Pharmacol. 1978, 43, 399–406. [Google Scholar] [CrossRef]
- Nakagawa, Y.; Tayama, K. Nephrotoxicity of butylated hydroxytoluene in phenobarbital-pretreated male rats. Arch. Toxicol. 1988, 61, 359–365. [Google Scholar] [CrossRef]
- Eloff, J. Which extractant should be used for the screening and isolation of antimicrobial components from plants? J. Ethnopharmacol. 1998, 60, 1–8. [Google Scholar] [CrossRef]
- Rojas, R.; Bustamante, B.; Bauer, J.; Fernández, I.; Albán, J.; Lock, O. Antimicrobial activity of selected Peruvian medicinal plants. J. Ethnopharmacol. 2003, 88, 199–204. [Google Scholar] [CrossRef]
- Souza, B.W.; Cerqueira, M.A.; Martins, J.T.; Quintas, M.A.; Ferreira, A.C.; Teixeira, J.A.; Vicente, A.A. Antioxidant potential of two red seaweeds from the Brazilian coasts. J. Agric. Food Chem. 2011, 59, 5589–5594. [Google Scholar]
- Bae, S.J.; Choi, Y.H. Methanol extract of the seaweed Gloiopeltis furcata induces G2/M arrest and inhibits cyclooxygenase-2 activity in human hepatocarcinoma HepG2 cells. Phytother. Res. 2007, 21, 52–57. [Google Scholar] [CrossRef]
- Yang, E.J.; Moon, J.Y.; Kim, M.J.; Kim, D.S.; Kim, C.S.; Lee, W.J.; Lee, N.H.; Hyun, C.G. Inhibitory effect of Jeju endemic seaweeds on the production of pro-inflammatory mediators in mouse macrophage cell line RAW 264.7. J. Zhejiang Univ. Sci. B 2010, 11, 315–322. [Google Scholar]
- Kurihara, H.; Goto, Y.; Aida, M.; Hosokawa, M.; Takahashi, K. Antibacterial activity against cariogenic bacteria and inhibition of insoluble glucan production by free fatty acids obtained from dried Gloiopeltis furcata. Fish. Sci. 1999, 65, 129–132. [Google Scholar] [CrossRef]
- Saeki, Y.; Kato, T.; Naito, Y.; Takazoe, I.; Okuda, K. Inhibitory effects of funoran on the adherence and colonization of mutans streptococci. Caries Res. 1996, 30, 119–125. [Google Scholar] [CrossRef]
- Ren, D.L.; Wang, J.Z.; Noda, H.; Amano, H.; Ogawa, S. The effects of an algal polysaccharide from Gloiopeltis tenax on transplantable tumors and immune activities in mice. Planta Med. 1995, 61, 120–125. [Google Scholar] [CrossRef]
- Modzelewska, A.; Sur, S.; Kumar, S.K.; Khan, S.R. Sesquiterpenes: Natural products that decrease cancer growth. Curr. Med. Chem. Anticancer Agents 2005, 5, 477–499. [Google Scholar] [CrossRef]
- Abraham, W.R. Bioactive sesquiterpenes produced by fungi: Are they useful for humans as well? Curr. Med. Chem. 2001, 8, 583–606. [Google Scholar] [CrossRef]
- Repetto, M.G.; Boveris, A. Bioactivity of sesquiterpenes: Compounds that protect from alcohol-induced gastric mucosal lesions and oxidative damage. Mini Rev. Med. Chem. 2010, 10, 615–623. [Google Scholar] [CrossRef]
- Manter, D.K.; Kelsey, R.G.; Karchesy, J.J. Antimicrobial activity of extractable conifer heartwood compounds toward Phytophthora ramorum. J. Chem. Ecol. 2007, 33, 2133–2147. [Google Scholar] [CrossRef]
- Umeno, K.; Hori, E.; Tsubota, M.; Shojaku, H.; Miwa, T.; Nagashima, Y.; Yada, Y.; Suzuki, T.; Ono, T.; Nishijo, H. Effects of direct cedrol inhalation into the lower airway on autonomic nervous activity in totally laryngectomized subjects. Br. J. Clin. Pharmacol. 2008, 65, 188–196. [Google Scholar] [CrossRef]
- Lenfeld, J.; Motl, O.; Trka, A. Anti-inflammatory activity of extracts from Conyza canadensis. Pharmazie 1986, 41, 268–269. [Google Scholar]
- Nishikawa, K.; Aburai, N.; Yamada, K.; Koshino, H.; Tsuchiya, E.; Kimura, K. The bisabolane sesquiterpenoid endoperoxide, 3,6-epidioxy-1,10-bisaboladiene, isolated from Cacalia delphiniifolia inhibits the growth of human cancer cells and induces apoptosis. Biosci. Biotechnol. Biochem. 2008, 72, 2463–2466. [Google Scholar] [CrossRef]
- Kabuto, H.; Nishizawa, M.; Tada, M.; Higashio, C.; Shishibori, T.; Kohno, M. Zingerone [4-(4-hydroxy-3-methoxyphenyl)-2-butanone] prevents 6-hydroxydopamine-induced dopamine depression in mouse striatum and increases superoxide scavenging activity in serum. Neurochem. Res. 2005, 30, 325–332. [Google Scholar] [CrossRef]
- Rao, B.N.; Archana, P.R.; Aithal, B.K.; Rao, B.S. Protective effect of zingerone, a dietary compound against radiation induced genetic damage and apoptosis in human lymphocytes. Eur. J. Pharmacol. 2011, 657, 59–66. [Google Scholar] [CrossRef]
- Kim, M.K.; Chung, S.W.; Kim, D.H.; Kim, J.M.; Lee, E.K.; Kim, J.Y.; Ha, Y.M.; Kim, Y.H.; No, J.K.; Chung, H.S.; et al. Modulation of age-related NF-kappaB activation by dietary zingerone via MAPK pathway. Exp. Gerontol. 2010, 45, 419–426. [Google Scholar] [CrossRef]
- Farrugia, G.; Balzan, R. Oxidative stress and programmed cell death in yeast. Front. Oncol. 2012, 2, 64. [Google Scholar]
- Mensor, L.L.; Menezes, F.S.; Leitão, G.G.; Reis, A.S.; dos Santos, T.C.; Coube, C.S.; Lietão, S.G. Screening of Brazilian plant extracts for antioxidant activity by the use of DPPH free radical method. Phytother. Res. 2001, 15, 127–130. [Google Scholar] [CrossRef]
- Barzegar, A.; Moosavi-Movahedi, A.A. Intracellular ROS protection efficiency and free radical-scavenging activity of curcumin. PLoS One 2011, 6, e26012. [Google Scholar]
- 31. Niu, Y.; Wang, H.; Xie, Z.; Whent, M.; Gao, X.; Zhang, X.; Zou, S.; Yao, W.; Yu, L. Structural analysis and bioactivity of a polysaccharide from the roots of Astragalus membranaceus (Fisch) Bge. var. mongolicus (Bge.) Hsiao. Food Chem. 2011, 128, 620–626. [Google Scholar]
- Yakes, F.M.; Van Houten, B. Mitochondrial DNA damage is more extensive and persists longer than nuclear DNA damage in human cells following oxidative stress. Proc. Natl. Acad. Sci. USA 1997, 94, 514–519. [Google Scholar] [CrossRef]
- Cabiscol, E.; Piulats, E.; Echave, P.; Herrero, E.; Ros, J. Oxidative stress promotes specific protein damage in Saccharomyces cerevisiae. J. Biol. Chem. 2000, 275, 27393–27398. [Google Scholar]
- Bilinski, T.; Litwinska, J.; Blszczynski, M.; Bajus, A. Superoxide dismutase deficiency and the toxicity of the products of autooxidation of polyunsaturated fatty acids in yeast. Biochim. Biophys. Acta 1001, 102–106. [Google Scholar]
- Halliwell, B.; Gutteridge, J.; Aruoma, O.I. The deoxyribose method: A simple “test-tube” assay for determination of rate constants for reactions of hydroxyl radicals. Anal. Biochem. 1987, 165, 215–219. [Google Scholar]
- Pal, M.; Ghosh, M. Prophylactic effect of alpha-linolenic acid and alpha-eleostearic acid against MeHg induced oxidative stress, DNA damage and structural changes in RBC membrane. Food Chem. Toxicol. 2012, 50, 2811–2818. [Google Scholar] [CrossRef]
- Mantey, I.; Hill, R.; Foster, A.; Wilson, S.; Wade, J.; Edmonds, M. Infection of foot ulcers with Staphylococcus aureus associated with increased mortality in diabetic patients. Commun. Dis. Public Health 2000, 3, 288–290. [Google Scholar]
- Ishikawa, N.K.; Fukushi, Y.; Yamaji, K.; Tahara, S.; Takahashi, K. Antimicrobial cuparene-type sesquiterpenes, enokipodins C and D, from a mycelial culture of Flammulina velutipes. J. Nat. Prod. 2001, 64, 932–934. [Google Scholar] [CrossRef]
- Johnston, W.H.; Karchesy, J.J.; Constantine, G.H.; Craig, A.M. Antimicrobial activity of some Pacific Northwest woods against anaerobic bacteria and yeast. Phytother. Res. 2001, 15, 586–588. [Google Scholar] [CrossRef]
- Chen, J.C.; Huang, L.J.; Wu, S.L.; Kuo, S.C.; Ho, T.Y.; Hsiang, C.Y. Ginger and its bioactive component inhibit enterotoxigenic Escherichia coli heat-labile enterotoxin-induced diarrhea in mice. J. Agric. Food Chem. 2007, 55, 8390–8397. [Google Scholar] [CrossRef]
- Iwami, M.; Shiina, T.; Hirayama, H.; Shima, T.; Takewaki, T.; Shimizu, Y. Inhibitory effects of zingerone, a pungent component of Zingiber officinale Roscoe, on colonic motility in rats. J. Nat. Med. 2011, 65, 89–94. [Google Scholar] [CrossRef]
- Georgel, P.; Crozat, K.; Lauth, X.; Makrantonaki, E.; Seltmann, H.; Sovath, S.; Hoebe, K.; Du, X.; Rutschmann, S.; Jiang, Z.F.; et al. A toll-like receptor 2-responsive lipid effector pathway protects mammals against skin infections with gram-positive bacteria. Infect. Immun. 2005, 73, 4512–4521. [Google Scholar] [CrossRef]
- Skrivanova, E.; Marounek, M.; Dlouha, G.; Kanka, J. Susceptibility of Clostridium perfringens to CC fatty acids. Lett. Appl. Microbiol. 2005, 41, 77–81. [Google Scholar] [CrossRef]
- Farshori, N.N.; Banday, M.R.; Ahmad, A.; Khan, A.U.; Rauf, A. Synthesis, characterization, and in vitro antimicrobial activities of 5-alkenyl/hydroxyalkenyl-2-phenylamine-1,3,4-oxadiazoles and thiadiazoles. Bioorg. Med. Chem. Lett. 2010, 20, 1933–1938. [Google Scholar] [CrossRef]
- Huang, C.M.; Chen, C.H.; Pornpattananangkul, D.; Zhang, L.; Chan, M.; Hsieh, M.F. Eradication of drug resistant Staphylococcus aureus by liposomal oleic acids. Biomaterials 2011, 32, 214–221. [Google Scholar]
- Blois, M.S. Antioxidant determinations by the use of a stable free radical. Nature 1958, 181, 1199–1200. [Google Scholar] [CrossRef]
- Shon, M.Y.; Kim, T.H.; Sung, N.J. Antioxidants and free radical scavenging activity of Phellinus baumii (Phellinus of Hymenochaetaceae) extracts. Food Chem. 2003, 82, 593–597. [Google Scholar] [CrossRef]
- Gutteridge, J.M. Ferrous-salt-promoted damage to deoxyribose and benzoate. The increased effectiveness of hydroxyl-radical scavengers in the presence of EDTA. Biochem. J. 1987, 243, 709–714. [Google Scholar]
- Samples Availability: Available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zheng, J.; Chen, Y.; Yao, F.; Chen, W.; Shi, G. Chemical Composition and Antioxidant/Antimicrobial Activities in Supercritical Carbon Dioxide Fluid Extract of Gloiopeltis tenax. Mar. Drugs 2012, 10, 2634-2647. https://doi.org/10.3390/md10122634
Zheng J, Chen Y, Yao F, Chen W, Shi G. Chemical Composition and Antioxidant/Antimicrobial Activities in Supercritical Carbon Dioxide Fluid Extract of Gloiopeltis tenax. Marine Drugs. 2012; 10(12):2634-2647. https://doi.org/10.3390/md10122634
Chicago/Turabian StyleZheng, Jiaojiao, Yicun Chen, Fen Yao, Weizhou Chen, and Ganggang Shi. 2012. "Chemical Composition and Antioxidant/Antimicrobial Activities in Supercritical Carbon Dioxide Fluid Extract of Gloiopeltis tenax" Marine Drugs 10, no. 12: 2634-2647. https://doi.org/10.3390/md10122634
APA StyleZheng, J., Chen, Y., Yao, F., Chen, W., & Shi, G. (2012). Chemical Composition and Antioxidant/Antimicrobial Activities in Supercritical Carbon Dioxide Fluid Extract of Gloiopeltis tenax. Marine Drugs, 10(12), 2634-2647. https://doi.org/10.3390/md10122634





