Resiniferatoxin: The Evolution of the “Molecular Scalpel” for Chronic Pain Relief
Abstract
:1. Introduction
2. Resiniferatoxin Is a Mechanism Based Treatment for Chronic Pain
3. Preclinical Studies in Laboratory Animals
3.1. Corneal Application of Capsaicin
3.2. Intraplantar Capsaicin and Carrageenan
3.3. Noxious Thermal Stimulation
3.4. Operant Orofacial Assay
4. Preclinical Studies in Companion Dogs
4.1. Rationale
- Medical surveillance of dogs is second only to that of people and illnesses are managed by veterinary specialists using all of the diagnostic approaches of modern medicine [37].
- Dogs share the environment with people and thus the potential environmental risk factors for disease.
- Their large body size simplifies biologic sampling.
- The extended course of disease, compared to rodent models, allows for clinically relevant efficacy data collection, while the shorter overall lifespan of dogs, compared to humans, provides a time course of disease within a time-frame reasonable for efficient data collection.
- Outcome assessment instruments have been specifically developed to capture clinically and translationally relevant pain severity and pain impact data in these models [38].
- Dogs have significant intrabreed homogeneity coupled with marked interbreed heterogeneity, providing unique opportunities to understand the genetic underpinnings of disease [39].
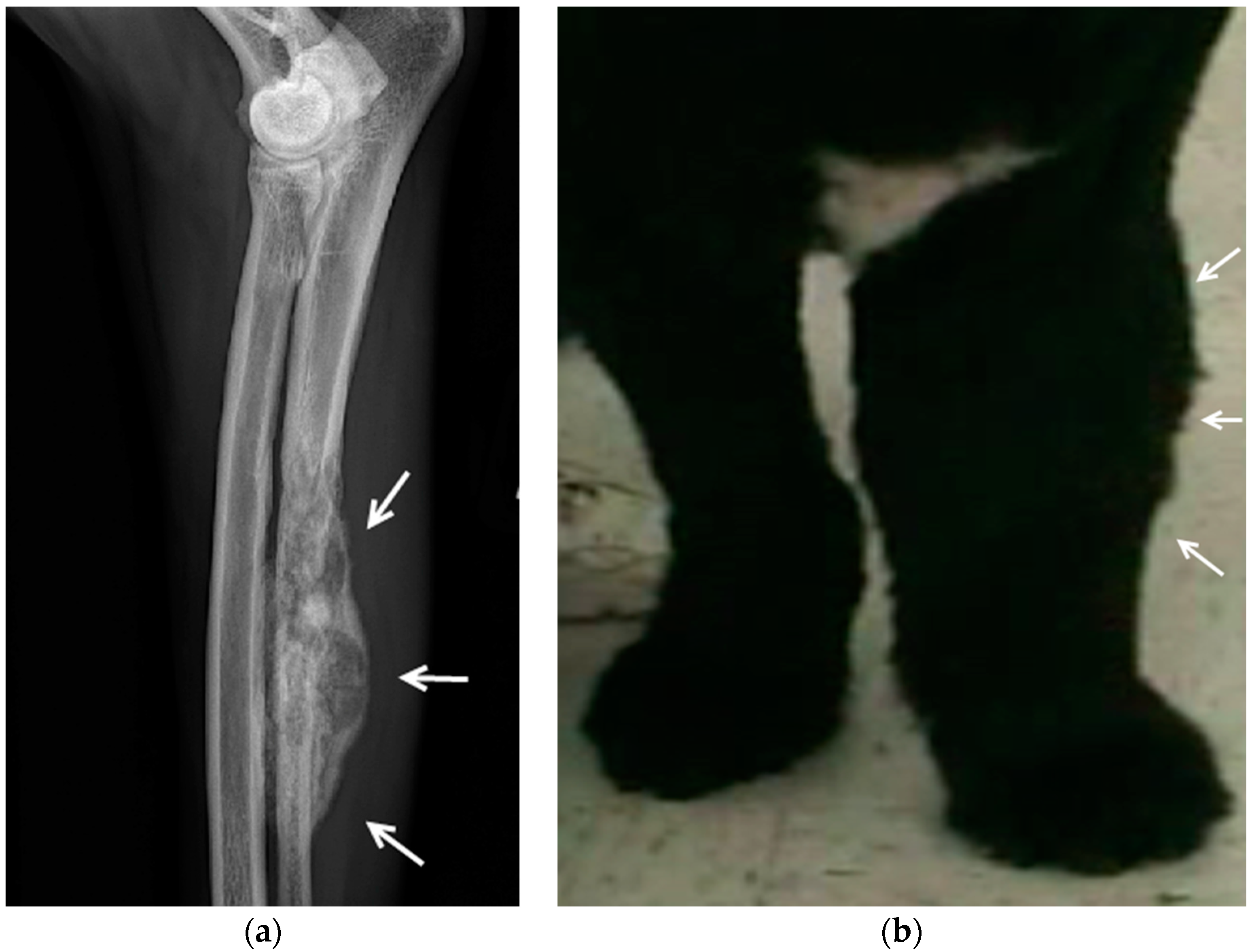
4.2. Canine Bone Cancer
4.2.1. Outcomes
4.2.2. Analgesic Efficacy of RTX
4.2.3. Adverse Events
5. The Clinical Trial
6. Conclusions
Acknowledgments
Conflicts of Interest
References
- Bevan, S.; Quallo, T.; Andersson, D.A. TRPV1. Handb. Exp. Pharmacol. 2014, 222, 207–245. [Google Scholar] [PubMed]
- Brederson, J.D.; Kym, P.R.; Szallasi, A. Targeting TRP channels for pain relief. Eur. J. Pharmacol. 2013, 716, 61–76. [Google Scholar] [CrossRef] [PubMed]
- Iadarola, M.J.; Mannes, A.J. The vanilloid agonist resiniferatoxin for interventional-based pain control. Curr. Top. Med. Chem. 2011, 11, 2171–2179. [Google Scholar] [CrossRef] [PubMed]
- Szallasi, A.; Sheta, M. Targeting TRPV1 for pain relief: Limits, losers and laurels. Expert Opin. Investig. Drugs 2012, 21, 1351–1369. [Google Scholar] [CrossRef] [PubMed]
- Wong, G.Y.; Gavva, N.R. Therapeutic potential of vanilloid receptor TRPV1 agonists and antagonists as analgesics: Recent advances and setbacks. Brain Res. Rev. 2009, 60, 267–277. [Google Scholar] [CrossRef] [PubMed]
- Helliwell, R.J.; McLatchie, L.M.; Clarke, M.; Winter, J.; Bevan, S.; McIntyre, P. Capsaicin sensitivity is associated with the expression of the vanilloid (capsaicin) receptor (VR1) mRNA in adult rat sensory ganglia. Neurosci. Lett. 1998, 250, 177–180. [Google Scholar] [CrossRef]
- Tominaga, M.; Caterina, M.J. Thermosensation and pain. J. Neurobiol. 2004, 61, 3–12. [Google Scholar] [CrossRef] [PubMed]
- Szallasi, A.; Blumberg, P.M. Vanilloid (Capsaicin) receptors and mechanisms. Pharmacol. Rev. 1999, 51, 159–212. [Google Scholar] [PubMed]
- Elokely, K.; Velisetty, P.; Delemotte, L.; Palovcak, E.; Klein, M.L.; Rohacs, T.; Carnevale, V. Understanding TRPV1 activation by ligands: Insights from the binding modes of capsaicin and resiniferatoxin. Proc. Natl. Acad. Sci. USA 2016, 113, E137–E145. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Cao, E.; Julius, D.; Cheng, Y. TRPV1 structures in nanodiscs reveal mechanisms of ligand and lipid action. Nature 2016, 534, 347–351. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Xiao, X.; Cheng, W.; Yang, W.; Yu, P.; Song, Z.; Yarov-Yarovoy, V.; Zheng, J. Structural mechanism underlying capsaicin binding and activation of the TRPV1 ion channel. Nat. Chem. Biol. 2015, 11, 518–524. [Google Scholar] [CrossRef] [PubMed]
- Darre, L.; Furini, S.; Domene, C. Permeation and dynamics of an open-activated TRPV1 channel. J. Mol. Biol. 2015, 427, 537–549. [Google Scholar] [CrossRef] [PubMed]
- Karai, L.J.; Russell, J.T.; Iadarola, M.J.; Oláh, Z. Vanilloid receptor 1 regulates multiple calcium compartments and contributes to Ca2+-induced Ca2+ release in sensory neurons. J. Biol. Chem. 2004, 279, 16377–16387. [Google Scholar] [CrossRef] [PubMed]
- Karai, L.; Brown, D.C.; Mannes, A.J.; Connelly, S.T.; Brown, J.; Gandal, M.; Wellisch, O.M.; Neubert, J.K.; Olah, Z.; Iadarola, M.J. Deletion of vanilloid receptor 1-expressing primary afferent neurons for pain control. J. Clin. Investig. 2004, 113, 1344–1352. [Google Scholar] [CrossRef] [PubMed]
- Caterina, M.J.; Rosen, T.A.; Tominaga, M.; Brake, A.J.; Julius, D. A capsaicin-receptor homologue with a high threshold for noxious heat. Nature 1999, 398, 436–441. [Google Scholar] [PubMed]
- Wiley, R.G.; Lappi, D.A. Targeted toxins. Curr. Protoc. Neurosci. 2001. [Google Scholar] [CrossRef]
- Olah, Z.; Karai, L.; Iadarola, M.J. Anandamide activates vanilloid receptor 1 (VR1) at acidic pH in dorsal root ganglia neurons and cells ectopically expressing VR1. J. Biol. Chem. 2001, 276, 31163–31170. [Google Scholar] [CrossRef] [PubMed]
- Olah, Z.; Szabo, T.; Karai, L.; Hough, C.; Fields, R.D.; Caudle, R.M.; Blumberg, P.M.; Iadarola, M.J. Ligand-induced dynamic membrane changes and cell deletion conferred by vanilloid receptor 1. J. Biol. Chem. 2001, 276, 11021–11030. [Google Scholar] [CrossRef] [PubMed]
- Caudle, R.M.; Karai, L.; Mena, N.; Cooper, B.Y.; Mannes, A.J.; Perez, F.M.; Iadarola, M.J.; Olah, Z. Resiniferatoxin-induced loss of plasma membrane in vanilloid receptor expressing cells. Neurotoxicology 2003, 24, 895–908. [Google Scholar] [CrossRef]
- Neubert, J.K.; Mannes, A.J.; Keller, J.; Wexel, M.; Iadarola, M.J.; Caudle, R.M. Peripheral targeting of the trigeminal ganglion via the infraorbital foramen as a therapeutic strategy. Brain Res. Protoc. 2005, 15, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Tender, G.C.; Walbridge, S.; Olah, Z.; Karai, L.; Iadarola, M.; Oldfield, E.H.; Lonser, R.R. Selective ablation of nociceptive neurons for elimination of hyperalgesia and neurogenic inflammation. J. Neurosurg. 2005, 102, 522–525. [Google Scholar] [CrossRef] [PubMed]
- Neubert, J.K.; King, C.; Malphurs, W.; Wong, F.; Weaver, J.P.; Jenkins, A.C.; Rossi, H.L.; Caudle, R.M. Characterization of mouse orofacial pain and the effects of lesioning TRPV1-expressing neurons on operant behavior. Mol. Pain 2008, 4, 43. [Google Scholar] [CrossRef] [PubMed]
- Jeffry, J.A.; Yu, S.Q.; Sikand, P.; Parihar, A.; Evans, M.S.; Premkumar, L.S. Selective targeting of TRPV1 expressing sensory nerve terminals in the spinal cord for long lasting analgesia. PLoS ONE 2009, 4, e7021. [Google Scholar] [CrossRef] [PubMed]
- Bishnoi, M.; Bosgraaf, C.A.; Premkumar, L.S. Preservation of acute pain and efferent functions following intrathecal resiniferatoxin-induced analgesia in rats. J. Pain 2011, 12, 991–1003. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.C.; Iadarola, M.J.; Perkowski, S.Z.; Erin, H.; Shofer, F.; Laszlo, K.J.; Olah, Z.; Mannes, A.J. Physiologic and antinociceptive effects of intrathecal resiniferatoxin in a canine bone cancer model. Anesthesiology 2005, 103, 1052–1059. [Google Scholar] [CrossRef] [PubMed]
- Vierck, C.J., Jr.; Hamilton, D.M.; Thornby, J.I. Pain reactivity of monkeys after lesions to the dorsal and lateral columns of the spinal cord. Exp. Brain Res. 1971, 13, 140–158. [Google Scholar] [CrossRef] [PubMed]
- Dubner, R.; Beitel, R.E.; Brown, F.J. A Behavioral Animal Model for the Study of Pain Mechanisms in Primates. In Pain: New Perspectives in Therapy and Research; Weisenberg, M., Tursky, B., Eds.; Springer US: Boston, MA, USA, 1976; pp. 155–170. [Google Scholar]
- Mogil, J.S. Animal models of pain: Progress and challenges. Nat. Rev. Neurosci. 2009, 10, 283–294. [Google Scholar] [CrossRef] [PubMed]
- Vierck, C.J.; Hansson, P.T.; Yezierski, R.P. Clinical and pre-clinical pain assessment: Are we measuring the same thing? Pain 2008, 135, 7–10. [Google Scholar] [CrossRef] [PubMed]
- Mogil, J.S.; Crager, S.E. What should we be measuring in behavioral studies of chronic pain in animals? Pain 2004, 112, 12–15. [Google Scholar] [CrossRef] [PubMed]
- Blackburn-Munro, G. Pain-like behaviours in animals—How human are they? Trends Pharmacol. Sci. 2004, 25, 299–305. [Google Scholar] [CrossRef] [PubMed]
- Rice, A.S.; Cimino-Brown, D.; Eisenach, J.C.; Kontinen, V.K.; Lacroix-Fralish, M.L.; Machin, I.; Mogil, J.S.; Stöhr, T. Animal models and the prediction of efficacy in clinical trials of analgesic drugs: A critical appraisal and call for uniform reporting standards. Pain 2008, 139, 243–247. [Google Scholar] [CrossRef] [PubMed]
- Khanna, C.; Lindblad-Toh, K.; Vail, D.; London, C.; Bergman, P.; Barber, L.; Breen, M.; Kitchell, B.; McNeil, E.; Modiano, J.F.; Niemi, S. The dog as a cancer model. Nat. Biotechnol. 2006, 24, 1065–1066. [Google Scholar] [CrossRef] [PubMed]
- Loscher, W. Animal models of intractable epilepsy. Prog. Neurobiol. 1997, 53, 239–258. [Google Scholar] [CrossRef]
- Nowend, K.L.; Starr-Moss, A.N.; Murphy, K.E. The function of dog models in developing gene therapy strategies for human health. Mamm. Genome 2011, 22, 476–485. [Google Scholar] [CrossRef] [PubMed]
- Vainio, O. Translational animal models using veterinary patients—An example of canine osteoarthritis (OA). Scand. J. Pain 2012, 3, 84–89. [Google Scholar] [CrossRef]
- Ostrander, E.A.; Galibert, F.; Patterson, D.F. Canine genetics comes of age. Trends Genet. 2000, 16, 117–124. [Google Scholar] [CrossRef]
- Brown, D.C.; Boston, R.; Coyne, J.C.; Farrar, J.T. A novel approach to the use of animals in studies of pain: Validation of the canine brief pain inventory in canine bone cancer. Pain Med. 2009, 10, 133–142. [Google Scholar] [CrossRef] [PubMed]
- Shearin, A.L.; Ostrander, E.A. Leading the way: Canine models of genomics and disease. Dis. Model. Mech. 2010, 3, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.C.; Agnello, K.; Iadarola, M.J. Intrathecal resiniferatoxin in a dog model: Efficacy in bone cancer pain. Pain 2015, 156, 1018–1024. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.C.; Agnello, K. Intrathecal substance P-saporin in the dog: Efficacy in bone cancer pain. Anesthesiology 2013, 119, 1178–1185. [Google Scholar] [CrossRef] [PubMed]
- Luger, N.M.; Mach, D.B.; Sevcik, M.A.; Mantyh, P.W. Bone cancer pain: From model to mechanism to therapy. J. Pain Symptom Manag. 2005, 29, S32–S46. [Google Scholar] [CrossRef] [PubMed]
- Mannes, A.J.; Brown, D.C.; Keller, J.; Cordes, L.; Eckenhoff, R.G.; Caudle, R.M.; Iadarola, M.J.; Meng, Q.C. Measurement of resiniferatoxin in serum samples by high-performance liquid chromatography. J. Chromatogr. B 2005, 823, 184–188. [Google Scholar] [CrossRef] [PubMed]
- Withrow, S.J.; Powers, B.E.; Straw, R.C.; Wilkins, R.M. Comparative aspects of osteosarcoma: Dog versus man. Clin. Orthop. Relat. Res. 1991, 270, 159–168. [Google Scholar] [CrossRef] [PubMed]
- MacEwen, E.G. Spontaneous tumors in dogs and cats: Models for the study of cancer biology and treatment. Cancer Metast. Rev. 1990, 9, 125–136. [Google Scholar] [CrossRef]
- Mueller, F.; Fuchs, B.; Kaser-Hotz, B. Comparative biology of human and canine osteosarcoma. Anticancer Res. 2007, 27, 155–164. [Google Scholar] [PubMed]
- Fan, T.M.; Charney, S.C.; De Lorimier, L.P.; Garrett, L.D.; Griffon, D.J.; Gordon-Evans, W.J.; Wypij, J.M. Double-blind placebo-controlled trial of adjuvant pamidronate with palliative radiotherapy and intravenous doxorubicin for canine appendicular osteosarcoma bone pain. J. Vet. Intern. Med. 2009, 23, 152–160. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.C.; Boston, R.C.; Farrar, J.T. Use of an activity monitor to detect response to treatment in dogs with osteoarthritis. J. Am. Vet. Med. Assoc. 2010, 237, 66–70. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.C.; Boston, R.C.; Coyne, J.C.; Farrar, J.T. Development and psychometric testing of an instrument designed to measure chronic pain in dogs with osteoarthritis. Am. J. Vet. Res. 2007, 68, 631–637. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.C.; Boston, R.C.; Coyne, J.C.; Farrar, J.T. Ability of the canine brief pain inventory to detect response to treatment in dogs with osteoarthritis. J. Am. Vet. Med. Assoc. 2008, 233, 1278–1283. [Google Scholar] [CrossRef] [PubMed]
- Wiseman-Orr, M.L.; Scott, E.M.; Reid, J.; Nolan, A.M. Validation of a structured questionnaire as an instrument to measure chronic pain in dogs on the basis of effects on health-related quality of life. Am. J. Vet. Res. 2006, 67, 1826–1836. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.C.; Iadarola, M.J. TRPV1 Agonist Cytotoxicity for Chronic Pain Relief: From Mechanistic Understanding to Clinical Application A2—Szallasi, Arpad. In TRP Channels as Therapeutic Targets; Academic Press: Boston, MA, USA, 2015; pp. 99–118. [Google Scholar]
- Davar, G.; Maciewicz, R.J. Deafferentation pain syndromes. Neurol. Clin. 1989, 7, 289–304. [Google Scholar] [PubMed]
- Whitworth, L.A.; Feler, C.A. Application of spinal ablative techniques for the treatment of benign chronic painful conditions: history, methods, and outcomes. Spine 2002, 27, 2607–2612. [Google Scholar] [CrossRef] [PubMed]
- Albe-Fessard, D. Neurophysical studies in rats deafferented by dorsal root sections. In Deafferentation Pain Syndromes: Pathophysiology and Treatments; Nashold, B., Ovelmen-Levitt, J., Eds.; Raven Press: New York, NY, USA, 1991; pp. 125–139. [Google Scholar]
- Forterre, F.; Jaggy, A.; Malik, Y.; Howard, J.; Rüfenacht, S.; Spreng, D. Non-selective cutaneous sensory neurectomy as an alternative treatment for auto-mutilation lesion following arthrodesis in three dogs. Vet. Comp. Orthop. Traumatol. 2009, 22, 233–237. [Google Scholar] [CrossRef] [PubMed]
- Kriz, N.; Yamamotova, A.; Tobias, J.; Rokyta, R. Tail-flick latency and self-mutilation following unilateral deafferentation in rats. Physiol. Res. 2006, 55, 213–220. [Google Scholar] [PubMed]
- Heiss, J.; Iadarola, M.; Cantor, F.; Oughourli, A.; Smith, R.; Mannes, A. A Phase I study of the intrathecal administration of resiniferatoxin for treating severe refractory pain associated with advanced cancer. J. Pain 2014, 15, S67. [Google Scholar] [CrossRef]
- Mannes, A.; Hughes, M.; Quezado, Z.; Berger, A.; Fojo, T.; Smith, R.; Butman, J.; Lonser, R.; Iadarola, M. Resiniferatoxin, a potent TRPV1 agonist: Intrathecal administration to treat severe pain associated with advanced cancer—Case report. J. Pain 2010, 11, S43. [Google Scholar] [CrossRef]
© 2016 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brown, D.C. Resiniferatoxin: The Evolution of the “Molecular Scalpel” for Chronic Pain Relief. Pharmaceuticals 2016, 9, 47. https://doi.org/10.3390/ph9030047
Brown DC. Resiniferatoxin: The Evolution of the “Molecular Scalpel” for Chronic Pain Relief. Pharmaceuticals. 2016; 9(3):47. https://doi.org/10.3390/ph9030047
Chicago/Turabian StyleBrown, Dorothy Cimino. 2016. "Resiniferatoxin: The Evolution of the “Molecular Scalpel” for Chronic Pain Relief" Pharmaceuticals 9, no. 3: 47. https://doi.org/10.3390/ph9030047




