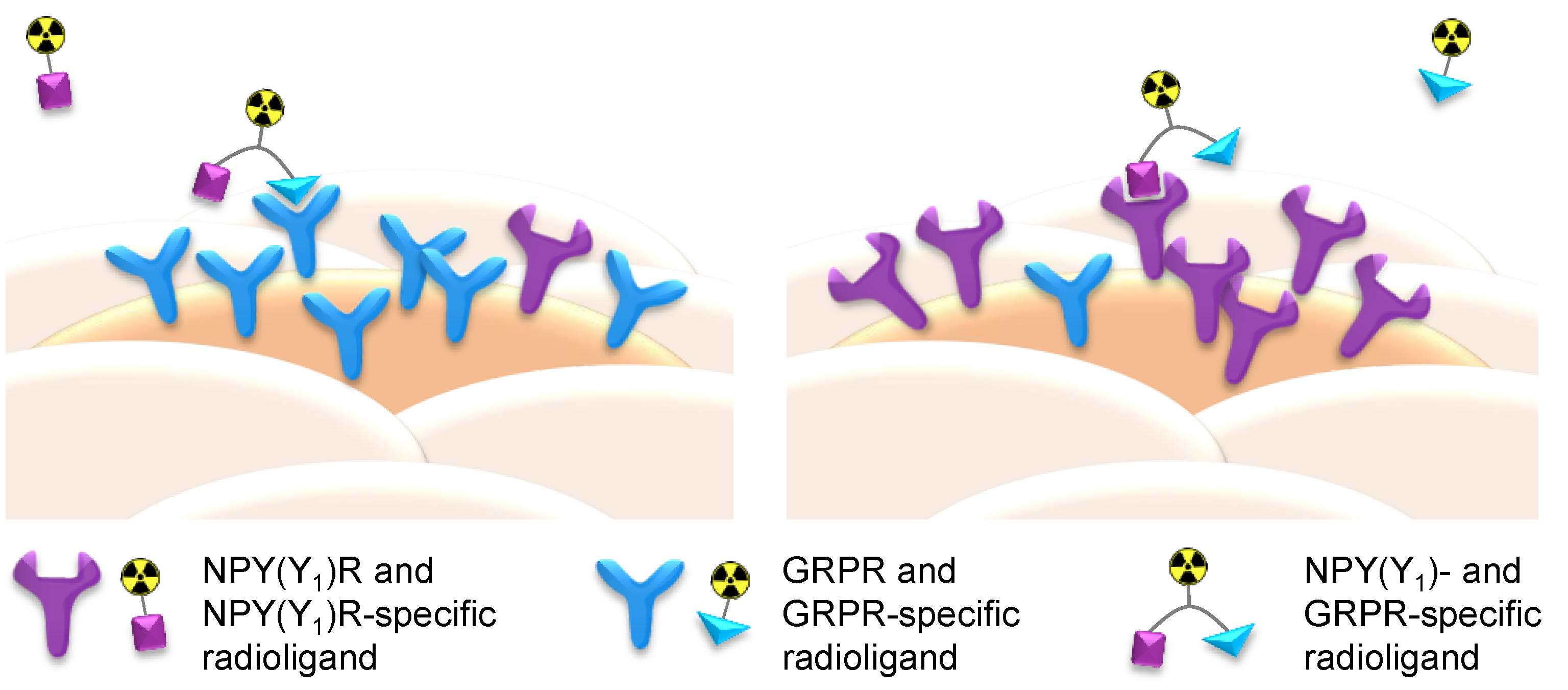
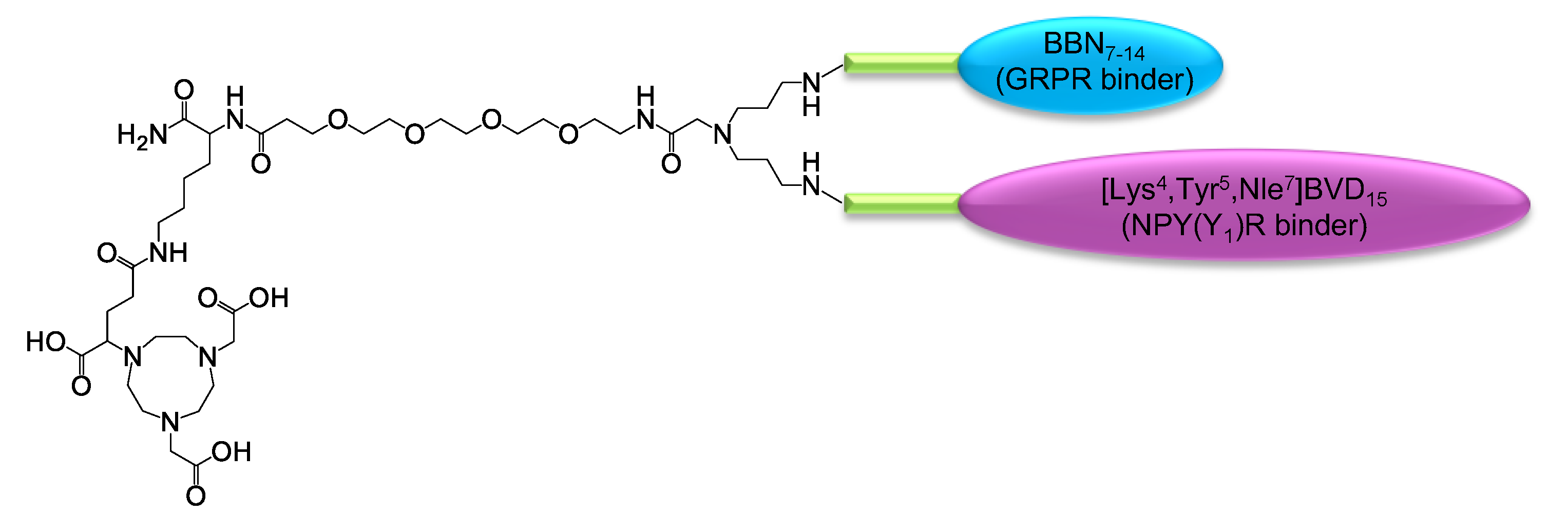
3. Materials and Methods
General. All commercially available chemicals were of analytical grade and were used without further purification. Resins for solid phase-based syntheses, PyBOP (Benzotriazole-1-yl-oxy-
tris-pyrrolidino-phosphonium hexafluorophosphate), Fmoc-protected standard amino acids as well as
bis-Boc-aminooxy acetic acid were purchased from NovaBiochem (Darmstadt, Germany). SFB (Succinimidyl-
p-formyl-benzoate) was synthesized according to a published procedure [
36]. Fmoc-ACMP-OH (Fmoc-4-amino-1-carboxymethyl-piperidine) was obtained from Iris Biotech (Marktredwitz, Germany), respectively. Fmoc-
l-Lys(Boc
2-Aoa)-OH, mono-Fmoc ethylene diamine hydrochloride, HBTU (
O-(Benzotriazol-1-yl)-
N,
N,
N′,
N′-tetramethyluronium hexafluorophosphate), Tracepur water and
N,
N-bis(
N′-Fmoc-3-aminopropyl)-glycine potassium hemisulfate ((Fmoc-NH-Propyl)
2-Gly-OH) were purchased from Iris Biotech, SigmaAldrich (Schnelldorf, Germany), Carl Roth (Karlsruhe, Germany), VWR (Bruchsal, Germany) and PolyPeptide (Strasbourg, France), respectively. NODA-GA-(
tBu)
3 and DOTA-(
tBu)
3 were obtained from CheMatech (Dijon, France).
Bis-amines
7–
11 and
bis-aldehydes
12–
16 were synthesized according to published procedures [
22] with minor modifications of the synthesis protocols. Details of these syntheses can be found in the
supplementary information.
Unless otherwise stated, the coupling reactions during solid phase-based syntheses were usually carried out in DMF for 30 min using 4 eq. of acid, 3.9 eq. of HBTU as coupling reagent and 4 eq. of DIPEA (N,N-Diisopropylethylamine) as base. Fmoc protecting groups were removed using 50% (v/v) piperidine in DMF.
For analytical and semipreparative HPLC chromatography, Dionex UltiMate 3000 systems equipped with a Chromolith Performance (RP-18e, 100-4.6 mm, Merck, Darmstadt, Germany) and a Chromolith SemiPrep (RP-18e, 100-10 mm, Merck) column were used, operated with a flow rate of 4 mL/min and H2O + 0.1% TFA and MeCN + 0.1% TFA as eluents. For radio-analytical HPLC chromatography, a Dionex UltiMate 3000 system equipped with a Chromolith Performance (RP-18e, 100-4.6 mm, Merck) column and a GabiStar radioactivity detector (Raytest, Straubenhardt, Germany) or an Agilent 1200 system equipped with a Chromolith Performance (RP-18e, 100-4.6 mm, Merck) column and a GabiStar radioactivity detector (Raytest) were used and operated with a flow rate of 4 mL/min and H2O + 0.1% TFA and MeCN + 0.1% TFA as eluents. MALDI (Matrix-Assisted Laser Desorption/Ionization) spectra were obtained with a Bruker Daltonics Microflex spectrometer (Bremen, Germany).
The human breast cancer cell lines T-47D, MDA-MB-231 and MCF-7 were purchased from SigmaAldrich (Schnelldorf, Germany), whereas the cell line BT-474 was obtained from the Leibniz-Institute DSMZ (Braunschweig, Germany). Dulbecco’s Modified Eagle Medium (DMEM), RPMI-1640 medium, 200 mM L-glutamine, 0.05% trypsin/EDTA and 0.25% trypsin/EDTA were purchased from Life Technologies. Fetal calf serum (FCS) was obtained from GE Healthcare Life Sciences and phosphate buffered saline (PBS) as well as β-estradiol were purchased from Sigma Aldrich. Bovine serum albumin (BSA) was purchased from CarlRoth (Karlsruhe, Germany).
The human 125I-labeled NPY(Y1)-binding peptide [125I]-Peptide-YY was obtained from PerkinElmer in a molar activity of 81.4 GBq/µmol. The γ-counter used was a 2480 WIZARD2 system (PerkinElmer, Rodgau, Germany).
For the in vivo evaluations, five week old female Fox Chase SCID mice were obtained from Janvier and implanted with estradiol pellets (0.36 mg/60 days; obtained from Innovative Research of America) one week prior to tumor cell inoculation. Tumor cells were inoculated using Matrigel basal membrane matrix with reduced growth factor (obtained from VWR). For PET/CT measurements, a small animal Albira II PET/SPECT/CT system (Bruker, Eggenstein-Leopoldshafen, Germany) was used.
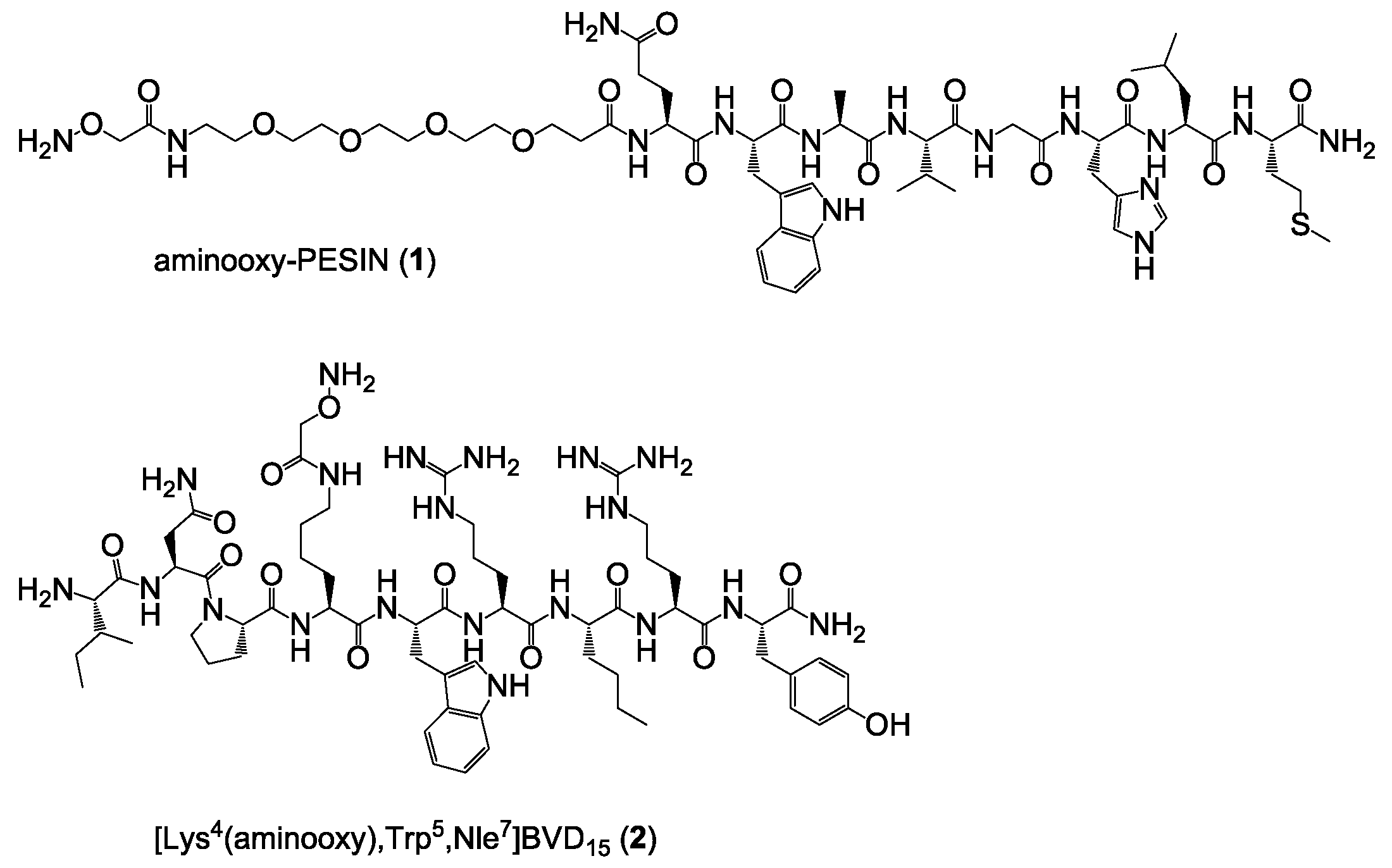
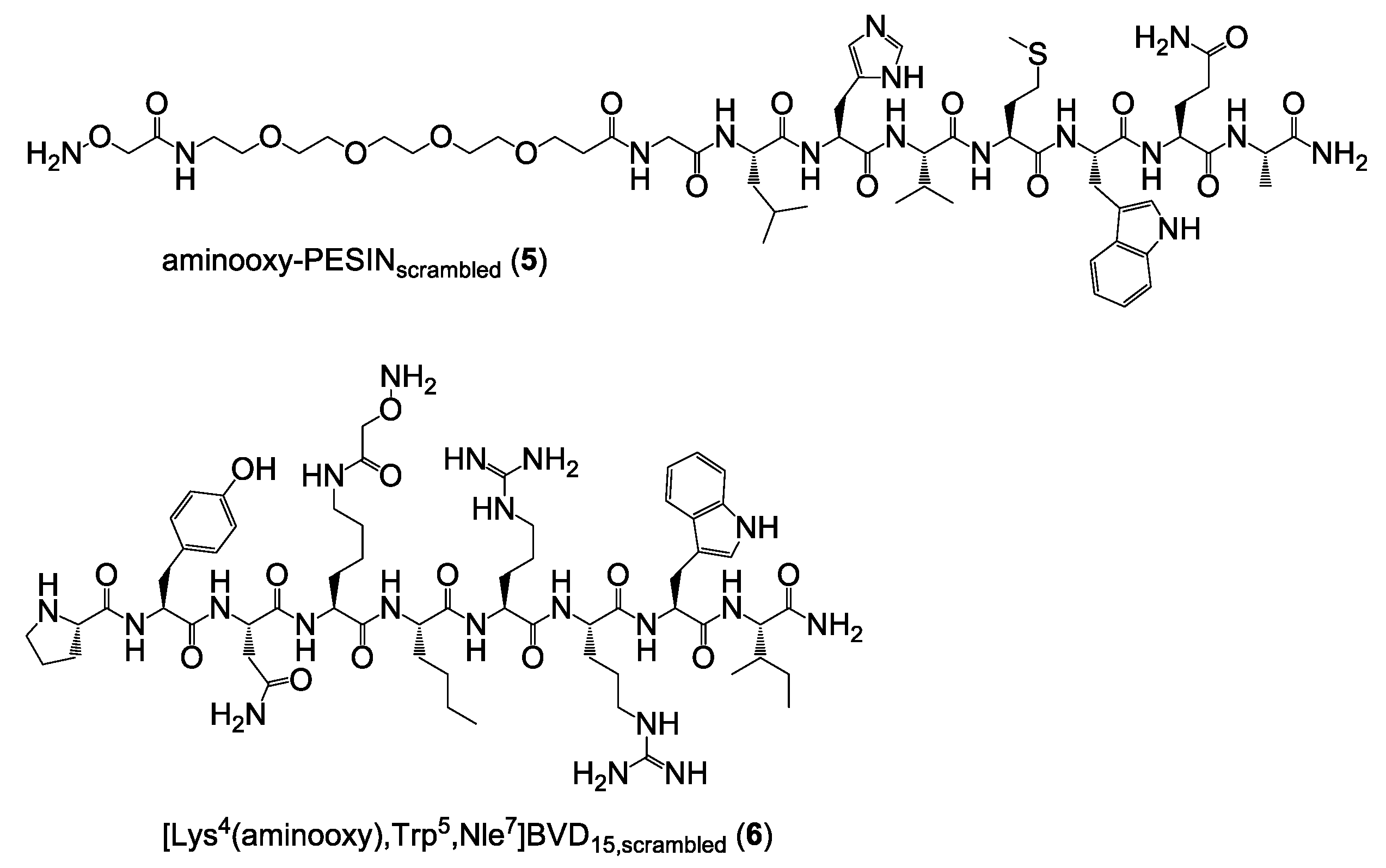
Synthesis of aminooxy-PESIN (1) and aminooxy-PESINscrambled (5). The peptides were synthesized on solid support by standard Fmoc solid-phase peptide synthesis using a commercially available standard Rink amide MBHA resin, HBTU as coupling reagent, standard Nα-Fmoc-amino acids, Nω-Fmoc-PEG4-OH and bis-Boc-aminooxy acetic acid. All amino acids (apart from bis-Boc-aminooxy acetic acid which was reacted for 60 min) were coupled within 30 min. The crude aminooxy-modified peptides were cleaved from the solid support using a mixture of TFA:TIS:H2O of 95:2.5:2.5 (v/v) for 60 min, suspended in diethyl ether and purified by semipreparative HPLC.
1 was purified using a gradient of 15–30% MeCN + 0.1% TFA in 8 min (Rt = 6.35 min) and isolated as white solid after lyophilization in yields of 27% (85.0 mg; 67.4 µmol). MALDI-MS (m/z) using 2,5-dihydroxybenzoic acid as matrix substance for [M + H+]+ (calculated): 1260.23 (1260.46); [M + Na+]+ (calculated): 1282.21 (1282.62); [M + K+]+ (calculated): 1298.16 (1298.60). MALDI-MS (m/z) using α-cyano-4-hydroxycinnamic acid as matrix substance for [M + H+]+ (calculated): 1260.63 (1260.46); [M + Na+]+ (calculated): 1282.56 (1282.62); [M + K+]+ (calculated): 1298.54 (1298.60).
5 was purified using a gradient of 10–60% MeCN + 0.1% TFA in 8 min (Rt = 5.42 min) and isolated as white solid after lyophilization in yields of 22% (28.1 mg, 22.2 µmol). MALDI-MS (m/z) using α-cyano-4-hydroxycinnamic acid as matrix substance for [M + H+]+ (calculated): 1260.81 (1260.46); [M + Na+]+ (calculated): 1282.84 (1282.62); [M + K+]+ (calculated): 1298.78 (1298.60).
Synthesis of [Lys4(aminooxy),Trp5,Nle7]BVD15 (2) and [Lys4(aminooxy),Trp5,Nle7]BVD15scrambled (6). The peptides were synthesized on solid support by standard Fmoc solid-phase peptide synthesis using a commercially available Rink amide MBHA resin, HBTU as coupling reagent, standard Nα-Fmoc-amino acids and Nα-Fmoc-l-Lys(Boc2-Aoa)-OH. The crude aminooxy-modified peptides were cleaved from the solid support using a mixture of TFA:TIS:H2O of 95:2.5:2.5 (v/v) for 90 min, suspended in diethyl ether and purified by semipreparative HPLC.
2 was purified using a gradient of 10–60% MeCN + 0.1% TFA in 8 min (Rt = 3.71 min) and isolated as white solid after lyophilization in yields of 35% (114.2 mg; 86.7 µmol). MALDI-MS (m/z) using 2,5-dihydroxybenzoic acid as matrix substance for [M + H+]+ (calculated): 1317.02 (1317.54); [M + Na+]+ (calculated): 1339.13 (1339.74); [M + K+]+ (calculated): 1355.03 (1355.71). MALDI-MS (m/z) using α-cyano-4-hydroxycinnamic acid as matrix substance for [M + H+]+ (calculated): 1317.89 (1317.54); [M + Na+]+ (calculated): 1339.89 (1339.74); [M + K+]+ (calculated): 1355.86 (1355.71).
6 was purified using a gradient of 10–50% MeCN + 0.1% TFA in 8 min (Rt = 4.31 min) and isolated as white solid after lyophilization in yields of 20% (26.0 mg; 19.7 µmol). MALDI-MS (m/z) using α-cyano-4-hydroxycinnamic acid as matrix substance for [M + H+]+ (calculated): 1317.37 (1317.54); [M + Na+]+ (calculated): 1339.38 (1339.74).
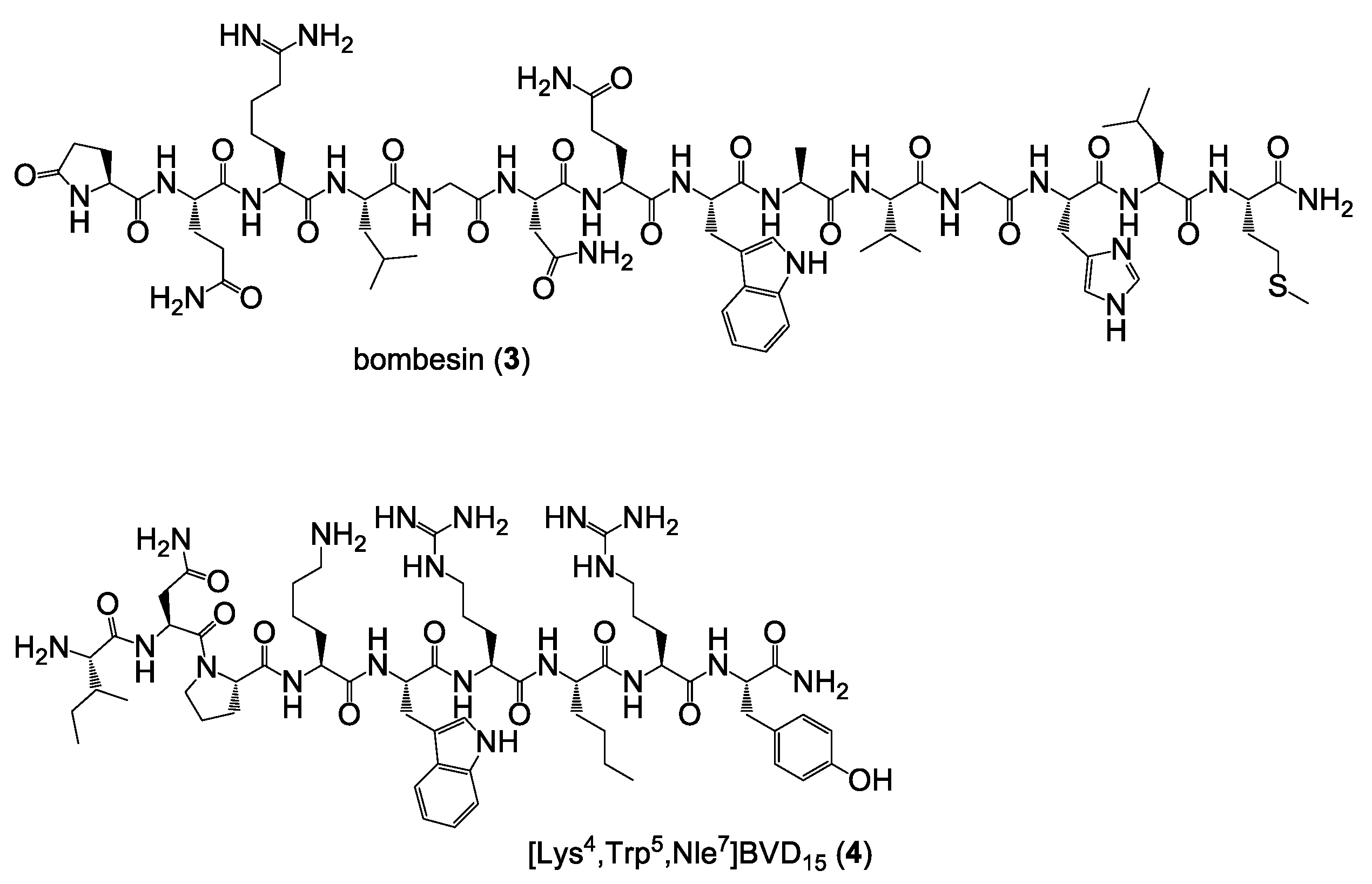
Synthesis of bombesin (3). The peptide was synthesized on solid support by standard Fmoc solid-phase peptide synthesis using a commercially available standard Rink amide resin, HBTU as coupling reagent and standard Nα-Fmoc-amino acids. All amino acids were coupled within 35 min. The crude peptide was cleaved from the solid support using a mixture of TFA:TIS:H2O of 95:2.5:2.5 (v/v) for 90 min, suspended in diethyl ether and purified by semipreparative HPLC using a gradient of 15–60% MeCN + 0.1% TFA in 5.5 min (Rt = 3.40 min) and isolated as white solid after lyophilization in yields of 22% (35.2 mg, 21.6 µmol). MALDI-MS (m/z) using α-cyano-4-hydroxycinnamic acid as matrix substance for [M + H+]+ (calculated): 1618.56 (1618.82); [M + Na+]+ (calculated): 1640.55 (1640.81); [M + K+]+ (calculated): 1656.57 (1656.78).
Synthesis of [Lys4,Trp5,Nle7]BVD15 (4). The peptide was synthesized on solid support by standard Fmoc solid-phase peptide synthesis using a commercially available standard Rink amide resin, HBTU as coupling reagent and standard Nα-Fmoc-amino acids. All amino acids were coupled within 30 min. The crude peptide was cleaved from the solid support using a mixture of TFA:TIS:H2O of 95:2.5:2.5 (v/v) for 120 min, suspended in diethyl ether and purified by semipreparative HPLC using a gradient of 10–60% MeCN + 0.1% TFA in 5.5 min (Rt = 3.05 min) and isolated as white solid after lyophilization in yields of 40% (49.2 mg; 39.5 µmol). MALDI-MS (m/z) using α-cyano-4-hydroxycinnamic acid as matrix substance for [M + H+]+ (calculated): 1244.63 (1244.73); [M + Na+]+ (calculated): 1266.65 (1266.72); [M + K+]+ (calculated): 1282.60 (1282.69).
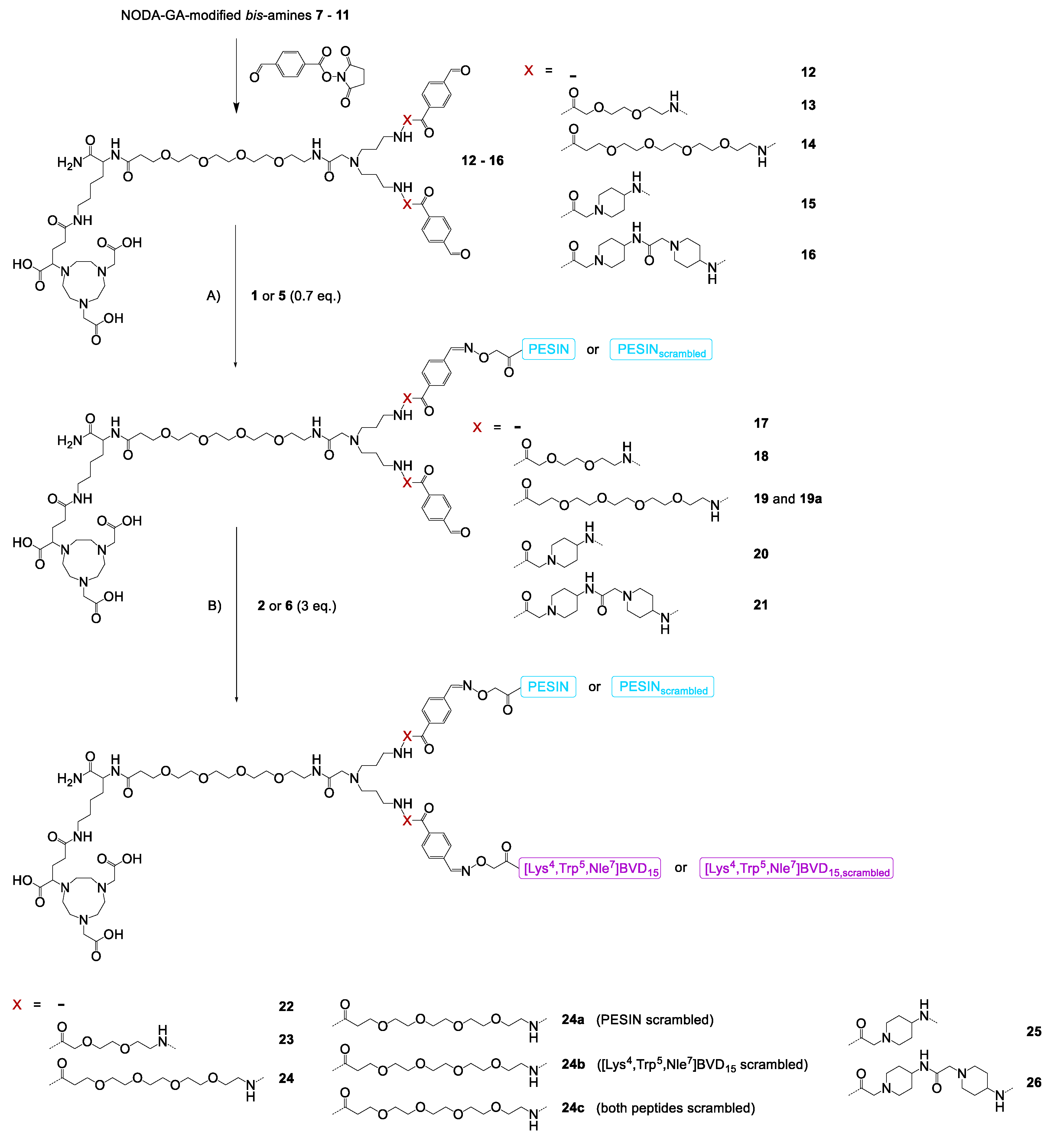
General synthesis of NODA-GA-PESIN-aldehydes (17−21) and NODA-GA-PESINscrambled-aldehyde (19a). To a solution of the respective branched NODA-GA-bis-aldehyde (12−16) in H2O + 0.1% TFA (250−500 µL) was added a solution of aminooxy-PESIN (1) or aminooxy-PESINscrambled (5) (0.7 eq.) in H2O + 0.1% TFA (250−500 µL). The pH of the solutions was adjusted to 4.0–4.6 by addition of phosphate buffer (0.1 M, pH 7.2, ~150 µL) and the reaction progress was monitored by analytical HPLC. The reactions were found to be finished within 5 min and the products were purified by semipreparative HPLC. The products were isolated as white solids after lyophilization. Gradients used for HPLC purification and synthesis yields for each compound are given below.
17: gradient: 20–45% MeCN + 0.1% TFA in 5 min (Rt = 4.45 min), yield: 47%. MALDI-MS (m/z) using α-cyano-4-hydroxycinnamic acid as matrix substance for [M + H+]+ (calculated): 2427.33 (2427.22); [M + Na+]+ (calculated): 2449.30 (2449.21); [M + K+]+ (calculated): 2465.49 (2465.18). MALDI-MS (m/z) using 2,5-dihydroxybenzoic acid as matrix substance for [M + H+]+ (calculated): 2426.69 (2427.22); [M + Na+]+ (calculated): 2448.99 (2449.21); [M + K+]+ (calculated): 2464.93 (2465.18). MALDI-MS (m/z) using sinapic acid as matrix substance for [M + H+]+ (calculated): 2427.38 (2427.22); [M + Na+]+ (calculated): 2449.25 (2449.21).
18: gradient: 25–45% MeCN + 0.1% TFA in 5 min (Rt = 3.76 min), yield: 51%. MALDI-MS (m/z) using α-cyano-4-hydroxycinnamic acid as matrix substance for [M + H+]+ (calculated): 2717.48 (2717.37); [M + Na+]+ (calculated): 2739.21 (2739.36); [M + K+]+ (calculated): 2755.49 (2755.33). MALDI-MS (m/z) using 2,5-dihydroxybenzoic acid as matrix substance for [M + H+]+ (calculated): 2717.18 (2717.37); [M + Na+]+ (calculated): 2739.60 (2739.36); [M + K+]+ (calculated): 2755.13 (2755.33).
19: gradient: 25–45% MeCN + 0.1% TFA in 5 min (Rt = 4.26 min), yield: 43%. MALDI-MS (m/z) using α-cyano-4-hydroxycinnamic acid as matrix substance for [M + H+]+ (calculated): 2921.63 (2921.50); [M + Na+]+ (calculated): 2943.64 (2943.49); [M + K+]+ (calculated): 2959.46 (2959.47). MALDI-MS (m/z) using 2,5-dihydroxybenzoic acid as matrix substance for [M + H+]+ (calculated): 2921.63 (2921.50); [M + Na+]+ (calculated): 2943.34 (2943.49); [M + K+]+ (calculated): 2959.57 (2959.47).
19a (PESIN scrambled): gradient: 25–45% MeCN + 0.1% TFA in 5.5 min (Rt = 4.18 min), yield: 44%. MALDI-MS (m/z) using α-cyano-4-hydroxycinnamic acid as matrix substance for [M + H+]+ (calculated): 2921.59 (2921.50); [M + Na+]+ (calculated): 2943.56 (2943.49); [M + K+]+ (calculated): 2959.52 (2959.47).
20: gradient: 20–45% MeCN + 0.1% TFA in 5 min (Rt = 3.93 min), yield: 58%. MALDI-MS (m/z) using α-cyano-4-hydroxycinnamic acid as matrix substance for [M + H+]+ (calculated): 2707.24 (2707.41); [M + Na+]+ (calculated): 2729.30 (2729.40); [M + K+]+ (calculated): 2745.58 (2745.37). MALDI-MS (m/z) using 2,5-dihydroxybenzoic acid as matrix substance for [M + H+]+ (calculated): 2706.98 (2707.41); [M + Na+]+ (calculated): 2728.88 (2729.40); [M + K+]+ (calculated): 2744.78 (2745.37).
21: gradient: 25–45% MeCN + 0.1% TFA in 5 min (Rt = 2.85 min), yield: 55%. MALDI-MS (m/z) using α-cyano-4-hydroxycinnamic acid as matrix substance for [M + H+]+ (calculated): 2987.39 (2987.60); [M + Na+]+ (calculated): 3009.40 (3009.59); [M + K+]+ (calculated): 3025.80 (3025.56). MALDI-MS (m/z) using 2,5-dihydroxybenzoic acid as matrix substance for [M + H+]+ (calculated): 2987.03 (2987.60). MALDI-MS (m/z) using sinapic acid as matrix substance for [M + H+]+ (calculated): 2987.81 (2987.60).
General synthesis of heterobivalent ligands 22−26 and scrambled analogs 24a–c. To a solution of the respective NODA-GA-PESIN-aldehyde (17−21 or 19a) in H2O + 0.1% TFA (250−500 µL) was added a solution of 2 or 6 (3 eq.) in H2O + 0.1% TFA (250−500 µL). The pH of the solutions was adjusted to 4.0–4.6 by addition of phosphate buffer (0.1 M, pH 7.2, ~150 µL) and the reaction progress was monitored by analytical HPLC. The reactions were found to be finished within 5 min and the products were purified by semipreparative HPLC. The products were isolated as white solids after lyophilization. Gradients used for HPLC purification and synthesis yields for each compound are given below.
22: gradient: 25–50% MeCN + 0.1% TFA in 5 min (Rt = 3.19 min), yield: 82%. MALDI-MS (m/z) using α-cyano-4-hydroxycinnamic acid as matrix substance for [M + H+]+ (calculated): 3728.21 (3728.29); [M + Na+]+ (calculated): 3750.49 (3750.28). MALDI-MS (m/z) using 2,5-dihydroxybenzoic acid as matrix substance for [M + H+]+ (calculated): 3728.71 (3728.29); [M + Na+]+ (calculated): 3750.76 (3750.28).
23: gradient: 25–45% MeCN + 0.1% TFA in 5 min (Rt = 3.65 min), yield: 79%. MALDI-MS (m/z) using α-cyano-4-hydroxycinnamic acid as matrix substance for [M + H+]+ (calculated): 4018.99 (4018.61); [M + Na+]+ (calculated): 4040.69 (4040.60). MALDI-MS (m/z) using 2,5-dihydroxybenzoic acid as matrix substance for [M + H+]+ (calculated): 4018.95 (4018.61); [M + Na+]+ (calculated): 4040.02 (4040.60).
24: gradient: 25–45% MeCN + 0.1% TFA in 6 min (Rt = 3.86 min), yield: 66%. MALDI-MS (m/z) using α-cyano-4-hydroxycinnamic acid as matrix substance for [M + H+]+ (calculated): 4222.98 (4222.87). MALDI-MS (m/z) using 2,5-dihydroxybenzoic acid as matrix substance for [M + H+]+ (calculated): 4222.48 (4222.87); [M + Na+]+ (calculated): 4244.96 (4244.86); [M + K+]+ (calculated): 4260.51 (4260.97). MALDI-MS (m/z) using sinapic acid as matrix substance for [M + H+]+ (calculated): 4222.87 (4222.87).
24a (PESIN scrambled): gradient: 25–45% MeCN + 0.1% TFA in 6 min (Rt = 3.55 min), yield: 49%. MALDI-MS (m/z) using α-cyano-4-hydroxycinnamic acid as matrix substance for [M + H+]+ (calculated): 4222.54 (4222.87). MALDI-MS (m/z) using 2,5-dihydroxybenzoic acid as matrix substance for [M + H+]+ (calculated): 4222.17 (4222.87); [M + Na+]+ (calculated): 4244.94 (4244.86).
24b ([Lys4,Trp5,Nle7]BVD15 scrambled): gradient: 25–40% MeCN + 0.1% TFA in 5.5 min (Rt = 4.58 min), yield: 63%. MALDI-MS (m/z) using α-cyano-4-hydroxycinnamic acid as matrix substance for [M + H+]+ (calculated): 4222.35 (4222.87). MALDI-MS (m/z) using 2,5-dihydroxybenzoic acid as matrix substance for [M + H+]+ (calculated): 4222.29 (4222.87); [M + Na+]+ (calculated): 4244.99 (4244.86); [M + K+]+ (calculated): 4260.49 (4260.97).
24c (PESIN and [Lys4,Trp5,Nle7]BVD15 scrambled): gradient: 25–45% MeCN + 0.1% TFA in 5.5 min (Rt = 4.36 min), yield: 61%. MALDI-MS (m/z) using α-cyano-4-hydroxycinnamic acid as matrix substance for [M + H+]+ (calculated): 4222.67 (4222.87); [M + K+]+ (calculated): 4260.98 (4260.97). MALDI-MS (m/z) using 2,5-dihydroxybenzoic acid as matrix substance for [M + H+]+ (calculated): 4222.48 (4222.87); [M + Na+]+ (calculated): 4244.90 (4244.86).
25: gradient: 25–50% MeCN + 0.1% TFA in 6 min (Rt = 2.76 min), yield: 75%. MALDI-MS (m/z) using α-cyano-4-hydroxycinnamic acid as matrix substance for [M + H+]+ (calculated): 4008.53 (4008.66); [M + Na+]+ (calculated): 4030.76 (4030.65); [M + K+]+ (calculated): 4046.33 (4046.76). MALDI-MS (m/z) using 2,5-dihydroxybenzoic acid as matrix substance for [M + H+]+ (calculated): 4009.12 (4008.66).
26: gradient: 25–45% MeCN + 0.1% TFA in 6 min (Rt = 2.83 min), yield: 73%. MALDI-MS (m/z) using 2,5-dihydroxybenzoic acid as matrix substance for [M + H+]+ (calculated): 4288.72 (4289.03). MALDI-MS (m/z) using sinapic acid as matrix substance for [M + H+]+ (calculated): 4289.59 (4289.03).
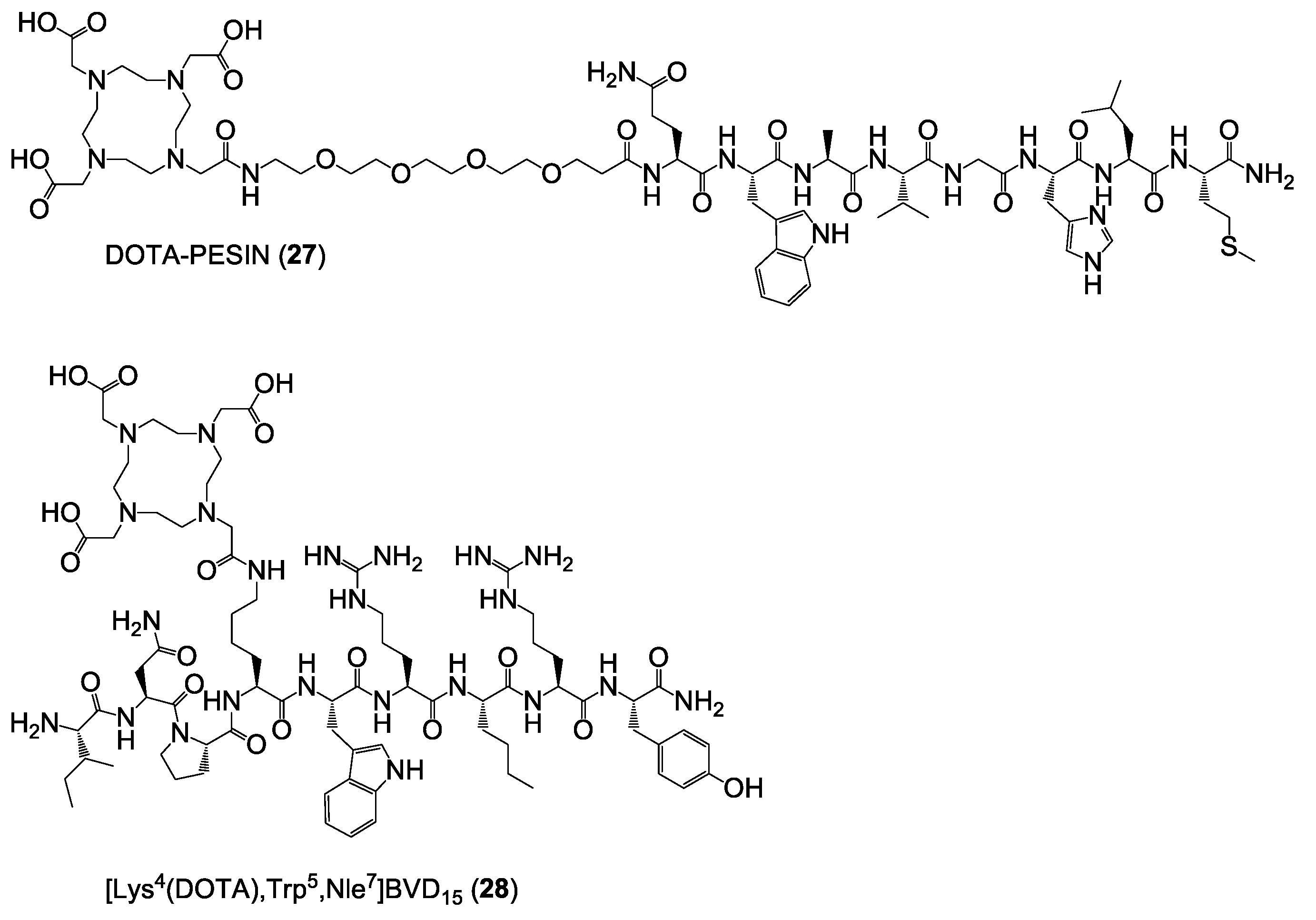
Synthesis of DOTA-PESIN (27). The peptide was synthesized on solid support by standard Fmoc solid-phase peptide synthesis using a commercially available standard Rink amide MBHA resin, HBTU as coupling reagent, standard Nα-Fmoc-amino acids, and Nω-Fmoc-PEG4-OH. After the conjugation of the PEG4 linker to the peptide sequence, DOTA-(tBu)3 was coupled within 120 min using an excess of the synthon of 2.7 eq. together with 2.6 eq. HBTU and 4 eq. DIPEA. The crude product was cleaved from the solid support using a mixture of TFA:TIS:H2O of 95:2.5:2.5 (v/v) for 3 h, suspended in diethyl ether and purified by semipreparative HPLC using a gradient of 20–35% MeCN + 0.1% TFA in 8 min (Rt = 4.34 min) and isolated as white solid after lyophilization in yields of 7% (10.7 mg; 6.8 µmol). MALDI-MS (m/z) using 2,5-dihydroxybenzoic acid as matrix substance for [M + H+]+ (calculated): 1573.02 (1573.80); [M + Na+]+ (calculated): 1595.06 (1595.79); [M + K+]+ (calculated): 1610.94 (1611.76). MALDI-MS (m/z) using α-cyano-4-hydroxycinnamic acid as matrix substance for [M + H+]+ (calculated): 1573.75 (1573.80); [M + Na+]+ (calculated): 1595.85 (1595.79).
Synthesis of [Lys4(DOTA),Trp5,Nle7]BVD15 (28). The peptide was synthesized on solid support by standard Fmoc solid-phase peptide synthesis using a commercially available standard Rink amide MBHA resin, HBTU as coupling reagent, standard Nα-Fmoc-amino acids and Fmoc-Lys(Mtt)-OH. After conjugation of the last amino acid, the lysine side chain Mtt-protecting group was removed with diluted TFA (TFA:DCM 1:99 (v/v)) within 2 h and DOTA-(tBu)3 was coupled in this position within 120 min using an excess of the synthon of 2.7 eq. together with 2.6 eq. HBTU and 4 eq. DIPEA. The crude DOTA-modified peptide was cleaved from the solid support using a mixture of TFA:TIS:H2O of 95:2.5:2.5 (v/v) for 3 h, suspended in diethyl ether and purified by semipreparative HPLC using a gradient of 20–25% MeCN + 0.1% TFA in 8 min (Rt = 2.58 min) and isolated as white solid after lyophilization in yields of 45% (73.4 mg; 45.0 µmol). MALDI-MS (m/z) using 2,5-dihydroxybenzoic acid as matrix substance for [M + H+]+ (calculated): 1630.38 (1630.91); [M + Na+]+ (calculated): 1652.59 (1652.90); [M + K+]+ (calculated): 1668.39 (1668.87). MALDI-MS (m/z) using α-cyano-4-hydroxycinnamic acid as matrix substance for [M + H+]+ (calculated): 1630.54 (1630.91). MALDI-MS (m/z) using sinapic acid as matrix substance for [M + H+]+ (calculated): 1630.53 (1630.91).
68Ga-radiolabeling of NODAGA-modified peptide heterodimers (22−26) and peptide monomers 27 and 28 for in vitro evaluations. The respective labeling precursor (10 nmol, dissolved in 10 µL of Tracepur water) was reacted with 110–150 MBq of 68Ga3+ obtained by an itG 68Ge/68Ga generator system (Garching, Germany). The generator was eluted with HCl (0.05 M, 3 mL) and the eluate was trapped on a cation exchange cartridge (Macherey-Nagel, Chromafix PS-H+). The 68Ga3+ was eluted from the cartridge using a NaCl solution (5 M, 1.5 mL) and the pH was adjusted to 3.5–4.0 by addition of sodium acetate solution (1.25 M, ~50 µL). After reaction for 10 min at 45 °C (22−26) or 99 °C (27 and 28), the reaction mixtures were analyzed by analytical radio-HPLC. The radiolabeled products were found to be 95–99% pure and obtained in molar activities of 10–15 GBq/µmol (non-optimized).
68Ga-radiolabeling of NODAGA-modified peptide heterodimers (24 and 24a–c) for in vivo evaluations. The respective labeling precursor (10 nmol, dissolved in 10 µL of Tracepur water) was reacted with 420–460 MBq of 68Ga3+ obtained by fractioned elution of an Eckert & Ziegler 68Ge/68Ga generator system (IGG100, Eckert & Ziegler, Berlin, Germany). The generator was eluted with HCl (0.1 M, 1.4 mL) and the pH was adjusted to 3.5–4.0 by addition of sodium acetate solution (1.25 M, 90–95 µL). After reaction for 10 min at 45 °C, the reaction mixtures were analyzed by analytical radio-HPLC. The radiolabeled products were found to be 95–99% pure and obtained in molar activities of 40–46 GBq/µmol (non-optimized). The pH of the radiotracer solution was adjusted to 6.0–7.0 using HEPES buffer (2.0 M, pH 8.0, 200 µL) and used for the in vivo studies.
Determination of radiotracer lipophilicity. The heterobivalent ligands (22–26) as well as the monomeric reference compounds (27 and 28) were radiolabeled with 68Ga as described before and 2 µL of the product solution (~65 pmol of the respective radioligand) were added to a mixture of phosphate buffer (0.05 M, pH 7.4, 800 µL) and 1-octanol (800 µL) and incubated for 5 min at ambient temperature under vigorous shaking. Both phases were separated by centrifugation and 100 µL of each phase were measured for radioactivity in a gamma-counter. From these data, the distribution coefficient logD was calculated from the following equation: logDo/w = log(cpmo/cpmw), where: cpmo = activity in the 1-octanol phase [cpm] (cpm = counts per minute), cpmw = activity in the aqueous phase [cpm]. These experiments were performed six times independently.
Determination of the stability of the ligands in human serum. The heterobivalent ligands (22–26) as well as the monomeric reference compounds (27 and 28) were radiolabeled with 68Ga as described before and 125 µL of the product solution were added to 500 µL of human serum and incubated at 37 °C. At defined time-points of 5, 15, 30, 60 and 90 min, aliquots of 75 µL of the mixture were added to 75 µL of ethanol and the precipitation of serum proteins was enhanced by ice-cooling for 2 min. After centrifugation, supernatant and precipitate were measured for radioactivity and the supernatant was analyzed by analytical radio-HPLC. These experiments were performed thrice.
Cell culture. All cell lines were grown in suitable culture medium at 37 °C in a humidified CO2 (5%) atmosphere. The human breast cancer cell lines T-47D, MDA-MB-231 and MCF-7 were grown in Dulbecco’s Modified Eagle Medium (DMEM), supplemented with 10% (v/v) fetal calf serum (FCS) and 1% (v/v) L-Glutamine. For a high expression of the NPY(Y1) receptor on T-47D cells, the medium for this cell line was further supplemented with 0.15% (w/v) β-estradiol. The BT-474 and PC-3 cell lines were grown in RPMI-1640 medium, also supplemented with 10% (v/v) fetal calf serum (FCS) and 1% (v/v) L-Glutamine.
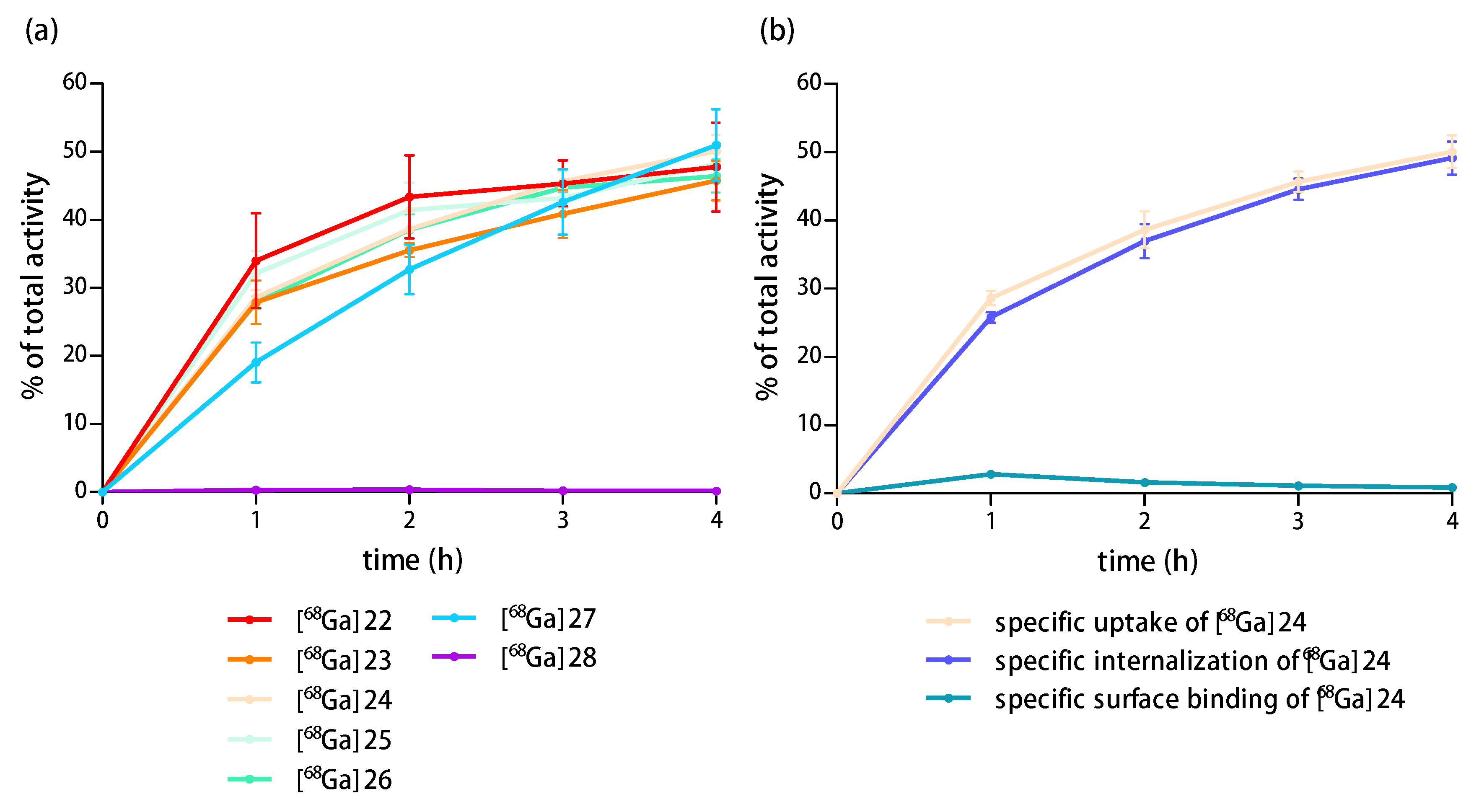
Internalization studies. Cells (T-47D, MDA-MB-231, MCF-7, BT-474 and PC-3, 1.5 × 106 cells per well) were seeded into 6-well plates and incubated overnight at 37 °C in a humidified CO2 (5%) atmosphere. The next day, the medium was removed and the cells were washed twice with the respective medium without supplements (ice-cold, 1 mL) and incubated with 3.7–4.0 kBq (0.37–4.0 pmol) of the respective 68Ga-radiolabeled ligand [68Ga]22−[68Ga]26, [68Ga]27 or [68Ga]28 (in 1.5 mL medium, containing 0.5% (w/v) BSA) for defined time-points of 1, 2, 3 or 4 h at 37 °C in a humidified CO2 (5%) atmosphere. A 1000-fold excess of the respective peptide (3 or 4) was used for blocking to determine the non-specific cell uptake. At each time point, the medium was removed and the cells were washed twice with the respective medium without supplements (ice-cold, 1 mL). Cells were treated twice with 1 mL glycine buffer (ice-cold, 50 mM glycine, 100 mM NaCl, pH 2.8) for 5 min at room temperature, followed by 2 mL NaOH solution (1 M) for 10 min at 37 °C. The supernatants were collected and the radioactivity measured in a gamma counter. The internalized and surface bound activity was expressed as percentage of measured to total added activity. Each data point was generated thrice in triplicates.
These internalization studies were performed accordingly on T-47D, MCF-7, BT-474 and PC-3 (negative control) cells with 125I-PYY (PerkinElmer, molar activity 81.4 GBq/µmol, 0.3 kBq, 0.0032 pmol). The cells were incubated for 1 h with the radioligand and additional blocking experiments were performed using a 1000-fold excess of 4, 28 and 24.
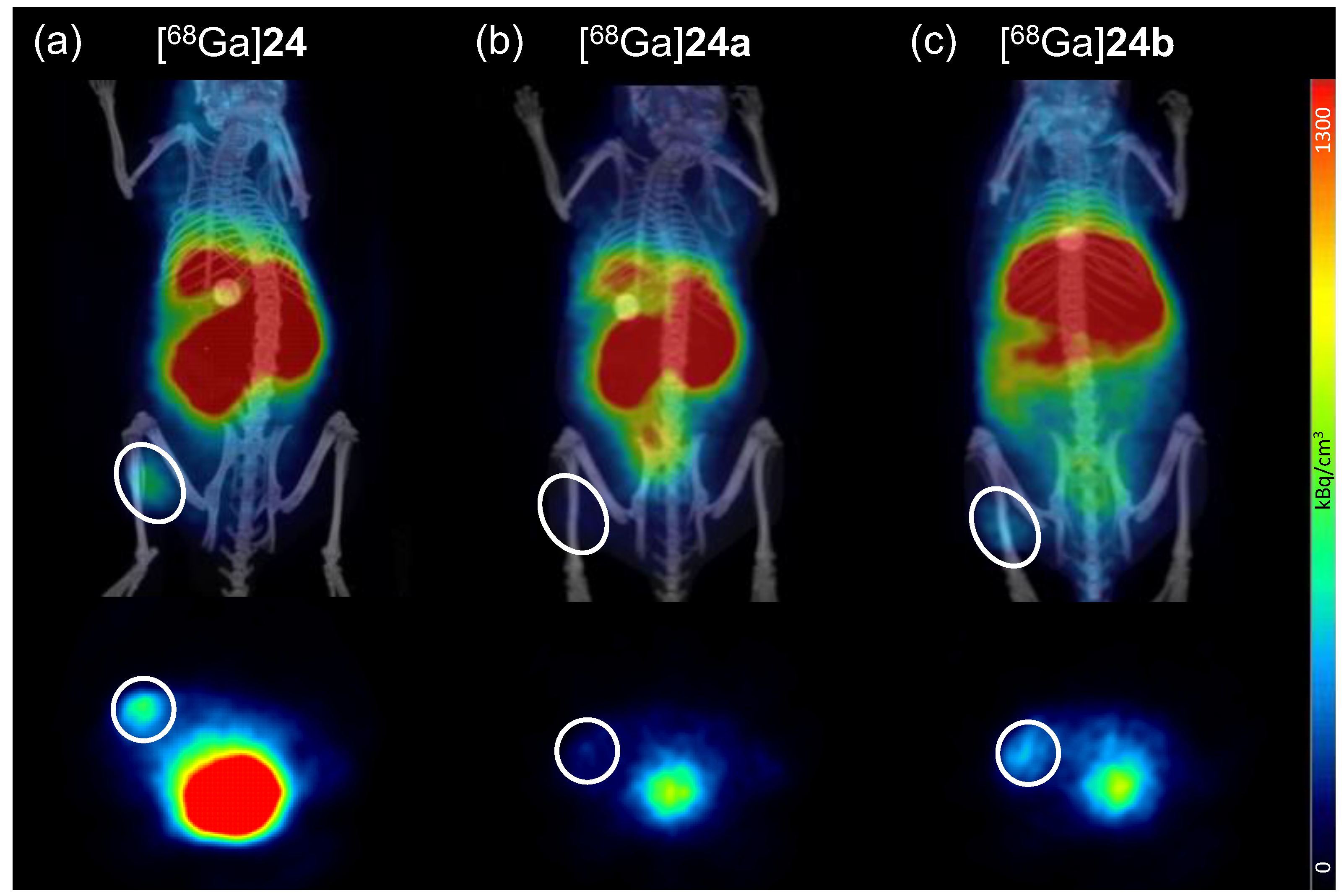
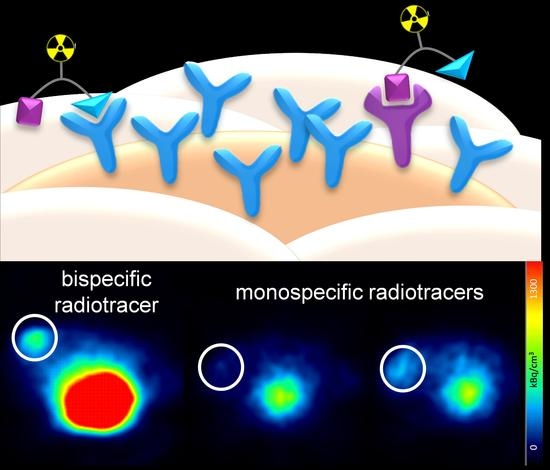
In vivo experiments. All animal experiments were performed in compliance with the German animal protection laws and protocols of the local committee (Regierungspräsidium Karlsruhe, approval number: 35-9185.81/G-206/15). 20, six week old female immunodeficient Fox Chase SCID (CB17/Icr-Prkdcscid/IcrIcoCrl) mice with an average weight of 20 g were subcutaneously implanted with 17β-estradiol pellets (0.36 mg/60 days). 4 days later, the tumors were induced by subcutaneous inoculation of 5 × 106 T-47D cells into the left flank of the approval number s. After induction, the tumors were allowed to grow for 8–10 weeks and reached a diameter of about 0.5 cm. For imaging, the animals were anaesthetized with isoflurane and injected with 5.5–8.0 MBq of the respective radioligand ([68Ga]24, [68Ga]24a or [68Ga]24b) into the lateral tail vein. Dynamic PET images were acquired over 90 min and CT images were obtained within further 30 min. After the end of the diagnostic scan, the animals were sacrificed, the organs were collected and measured in a gamma-counter.
The dynamic PET images were reconstructed using the Albira Suite Reconstructor (Bruker) with an iterative dynamic reconstruction with 12 iterations using an 2D-Maximum-Likelihood Expectation-Maximization (MLEM) algorithm and a cubic image voxel size of 0.5 mm after scatter and decay correction. Data were divided into time frames from 1 to 10 min (10 × 1 min, 10 × 2 min, 6 × 5 min and 3 × 10 min) for the assessment of temporal changes in regional tracer accumulation. The CT images were obtained at 45 kVp, with currents of 0.4 mA (high dose, good resolution). Acquisitions of 400 projections were taken and a 250 μm isotropic voxel size image was reconstructed via filtered back projection. The reconstructed PET data were manually fused with the CT images using PMOD 3.6.1.1. and analyzed. Volumes of interest (VOIs) were defined for the quantification of tracer accumulation in heart, liver, kidneys, tumor, and muscle. The results for each VOI were calculated as SUV (kBq/cm3) averaged for each time frame.